User login
Achieving a ‘new sexual-health paradigm’ means expanding STI care
A vital aspect of expanding access and care for sexually transmitted infections (STIs) in the United States is broadening responsibility for this care across the health care system and other community resources, according to an article published online July 6 in Clinical Infectious Diseases. This expansion and decentralization of care are central to adopting the “new sexual health paradigm” recommended by a National Academies report that was published in March.
“STIs represent a sizable, longstanding, and growing public health challenge,” write Vincent Guilamo-Ramos, PhD, MPH, dean and professor at the Duke University School of Nursing and director of the Center for Latino Adolescent and Family Health (CLAFH) at Duke University, both in Durham, N.C., and his colleagues. Yet the limitations on the current STI workforce and limited federal funding and support for STI prevention and care mean it will take clinicians of all types from across the health care spectrum to meet the challenge, they explain.
“For too long, STI prevention and treatment has been perceived as the sole responsibility of a narrow workforce of specialized STI and HIV service providers,” Dr. Guilamo-Ramos and his coauthor, Marco Thimm-Kaiser, MPH, associate in research at Duke University and epidemiologist at CLAFH, wrote in an email.
“However, the resources allocated to this STI specialty workforce have diminished over time, along with decreasing investments in the broader U.S. public health infrastructure,” they continued. “At the same time – and in part due to this underinvestment – STI rates have soared, reaching a record high for the sixth year in a row in 2019.”
Those factors led to the National Academies report, which recommends moving “away from the traditional, disease-focused perspective on STIs in favor of a holistic perspective of sexual health as an integral component of overall health and well-being,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote to this news organization.
In their article, the authors review the limitations in the STI workforce, the implications of those limitations for the broader health care industry, and what it will take for STI and HIV specialists as well as regulators to ensure it’s possible to achieve the paradigm shift recommended by the National Academies.
Currently, the biggest limitation is access to care, said Laura Mercer, MD, MBA, of the department of obstetrics and gynecology and the ob.gyn. clerkship director at the University of Arizona, Phoenix. Dr. Mercer, who was not involved with the National Academies report or the analysis of it, said in an interview that it’s essential to emphasize “sexual health as a core element of routine primary and preventative care” to ensure it becomes more accessible to patients without the need to seek out specialty care.
Dr. Guilamo-Ramos and his colleagues drive home the importance of such a shift by noting that more than 200 million Americans live in counties with no practicing infectious disease physicians. The disparities are greatest in Southern states, which account for 40% of all reported STIs. The workforce shortage has continued to worsen alongside the deterioration of the clinical infrastructure supporting STI specialty services, the authors write.
Hence the need to expand accountability for care not only to primary-care physicians but also to nurses, pharmacists, physician assistants, nurse practitioners, and behavioral health practitioners. Doing so also requires normalizing sexual health services across health care professions.
“Prevention is a crucial first step” to this, Dr. Mercer said. “This is particularly important as we recall that almost half of new sexually transmitted infections occur in teenagers. Destigmatizing sexual health and sexual health education will also help encourage patients of all ages to request and accept testing.”
Further, with primary care practitioners managing most STI testing and treatment, subspecialists can focus primarily on complex or refractory cases, she added. Ways to help broaden care include developing point-of-care testing for STIs and improving the accuracy of existing testing, she said.
“The goal is to make routine sexual health services accessible in a wide range of settings, such as in primary care, at pharmacies, and in community-based settings, and to draw on a broader workforce for delivery of sexual health services,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview.
Kevin Ault, MD, professor of obstetrics and gynecology and director of clinical and translational research at the University of Kansas Medical Center in Kansas City, said that many medical organizations, such as the American College of Obstetricians and Gynecologists, have long advocated incorporating sexual health into routine preventive care. He also noted that pharmacists have already become proactive in preventing STIs and could continue to do so.
“Vaccines for hepatitis and human papillomavirus are commonly available at pharmacies,” Dr. Ault said. He was not involved in the article by Dr. Guilamo-Ramos and colleagues or the original report. “Pharmacists could also fill a gap by administering injectable medications such as penicillin. States would have to approve changes in policy, but many states have already done this for expedited partner therapy.”
Dr. Guilamo-Ramos and Mr. Thimm-Kaiser noted similar barriers that must be removed to broaden delivery of STI services.
“Unfortunately, too many highly trained health care providers who are well-positioned for the delivery of sexual health services face regulatory or administrative barriers to practice to the full scope of their training,” they wrote. “These barriers can have a particularly negative impact in medically underserved communities, where physician shortages are common and where novel, decentralized health care service delivery models that draw on nonphysician providers may hold the greatest promise.”
As more diverse health care practitioners take on these roles, ID and HIV specialists can provide their expertise in developing training and technical assistance to support generalists, Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They can also aid in aligning “clinical training curricula, licensing criteria, and practice guidelines with routine delivery of sexual health services.”
Dr. Guilamo-Ramos and his coauthors offer specific recommendations for professional training, licensing, and practice guidelines to help overcome the “insufficient knowledge, inadequate training, and absence of explicit protocols” that currently impede delivery of STI services in general practice settings.
Although the paradigm shift recommended by the National Academies is ambitious, it’s also necessary, and “none of the recommendations are out of reach,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview. They pointed out how the COVID-19 pandemic has highlighted how underresourced the health care workforce and infrastructure are and how great health care disparities are.
“There is momentum toward rebuilding the nation’s health and public health system in a more effective and efficient way,” they said, and many of the STI report’s recommendations “overlap with priorities for the broader health and public health system moving forward.”
Dr. Mercer also believes the recommendations are realistic, “but only the beginning,” she told this news organization. “Comprehensive sexual education to expand knowledge about STI prevention and public health campaigns to help destigmatize sexual health care in general will remain crucial,” she said.
Sexual education, expanded access, and destigmatizing sexual care are particularly important for reaching the populations most in need of care, such as adolescents and young adults, as well as ethnic, racial, sexual, and gender-minority youth.
“It cannot be overstated how important of a priority population adolescents and young adults are,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They noted that those aged 15-24 account for half of all STIs each year but represent only a quarter of the sexually active population. “Targeted efforts for STI prevention and treatment among adolescents and young adults are therefore essential for an overall successful strategy to address STIs and sexual health in the United States.”
The National Academies report was supported by the Centers for Disease Control and Prevention and the National Association of County and City Health Officials. Dr. Mercer, Dr. Ault, and Mr. Thimm-Kaiser have disclosed no relevant financial relationships. Dr. Guilamo-Ramos has received grants and personal fees from ViiV Health care.
A version of this article first appeared on Medscape.com.
A vital aspect of expanding access and care for sexually transmitted infections (STIs) in the United States is broadening responsibility for this care across the health care system and other community resources, according to an article published online July 6 in Clinical Infectious Diseases. This expansion and decentralization of care are central to adopting the “new sexual health paradigm” recommended by a National Academies report that was published in March.
“STIs represent a sizable, longstanding, and growing public health challenge,” write Vincent Guilamo-Ramos, PhD, MPH, dean and professor at the Duke University School of Nursing and director of the Center for Latino Adolescent and Family Health (CLAFH) at Duke University, both in Durham, N.C., and his colleagues. Yet the limitations on the current STI workforce and limited federal funding and support for STI prevention and care mean it will take clinicians of all types from across the health care spectrum to meet the challenge, they explain.
“For too long, STI prevention and treatment has been perceived as the sole responsibility of a narrow workforce of specialized STI and HIV service providers,” Dr. Guilamo-Ramos and his coauthor, Marco Thimm-Kaiser, MPH, associate in research at Duke University and epidemiologist at CLAFH, wrote in an email.
“However, the resources allocated to this STI specialty workforce have diminished over time, along with decreasing investments in the broader U.S. public health infrastructure,” they continued. “At the same time – and in part due to this underinvestment – STI rates have soared, reaching a record high for the sixth year in a row in 2019.”
Those factors led to the National Academies report, which recommends moving “away from the traditional, disease-focused perspective on STIs in favor of a holistic perspective of sexual health as an integral component of overall health and well-being,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote to this news organization.
In their article, the authors review the limitations in the STI workforce, the implications of those limitations for the broader health care industry, and what it will take for STI and HIV specialists as well as regulators to ensure it’s possible to achieve the paradigm shift recommended by the National Academies.
Currently, the biggest limitation is access to care, said Laura Mercer, MD, MBA, of the department of obstetrics and gynecology and the ob.gyn. clerkship director at the University of Arizona, Phoenix. Dr. Mercer, who was not involved with the National Academies report or the analysis of it, said in an interview that it’s essential to emphasize “sexual health as a core element of routine primary and preventative care” to ensure it becomes more accessible to patients without the need to seek out specialty care.
Dr. Guilamo-Ramos and his colleagues drive home the importance of such a shift by noting that more than 200 million Americans live in counties with no practicing infectious disease physicians. The disparities are greatest in Southern states, which account for 40% of all reported STIs. The workforce shortage has continued to worsen alongside the deterioration of the clinical infrastructure supporting STI specialty services, the authors write.
Hence the need to expand accountability for care not only to primary-care physicians but also to nurses, pharmacists, physician assistants, nurse practitioners, and behavioral health practitioners. Doing so also requires normalizing sexual health services across health care professions.
“Prevention is a crucial first step” to this, Dr. Mercer said. “This is particularly important as we recall that almost half of new sexually transmitted infections occur in teenagers. Destigmatizing sexual health and sexual health education will also help encourage patients of all ages to request and accept testing.”
Further, with primary care practitioners managing most STI testing and treatment, subspecialists can focus primarily on complex or refractory cases, she added. Ways to help broaden care include developing point-of-care testing for STIs and improving the accuracy of existing testing, she said.
“The goal is to make routine sexual health services accessible in a wide range of settings, such as in primary care, at pharmacies, and in community-based settings, and to draw on a broader workforce for delivery of sexual health services,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview.
Kevin Ault, MD, professor of obstetrics and gynecology and director of clinical and translational research at the University of Kansas Medical Center in Kansas City, said that many medical organizations, such as the American College of Obstetricians and Gynecologists, have long advocated incorporating sexual health into routine preventive care. He also noted that pharmacists have already become proactive in preventing STIs and could continue to do so.
“Vaccines for hepatitis and human papillomavirus are commonly available at pharmacies,” Dr. Ault said. He was not involved in the article by Dr. Guilamo-Ramos and colleagues or the original report. “Pharmacists could also fill a gap by administering injectable medications such as penicillin. States would have to approve changes in policy, but many states have already done this for expedited partner therapy.”
Dr. Guilamo-Ramos and Mr. Thimm-Kaiser noted similar barriers that must be removed to broaden delivery of STI services.
“Unfortunately, too many highly trained health care providers who are well-positioned for the delivery of sexual health services face regulatory or administrative barriers to practice to the full scope of their training,” they wrote. “These barriers can have a particularly negative impact in medically underserved communities, where physician shortages are common and where novel, decentralized health care service delivery models that draw on nonphysician providers may hold the greatest promise.”
As more diverse health care practitioners take on these roles, ID and HIV specialists can provide their expertise in developing training and technical assistance to support generalists, Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They can also aid in aligning “clinical training curricula, licensing criteria, and practice guidelines with routine delivery of sexual health services.”
Dr. Guilamo-Ramos and his coauthors offer specific recommendations for professional training, licensing, and practice guidelines to help overcome the “insufficient knowledge, inadequate training, and absence of explicit protocols” that currently impede delivery of STI services in general practice settings.
Although the paradigm shift recommended by the National Academies is ambitious, it’s also necessary, and “none of the recommendations are out of reach,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview. They pointed out how the COVID-19 pandemic has highlighted how underresourced the health care workforce and infrastructure are and how great health care disparities are.
“There is momentum toward rebuilding the nation’s health and public health system in a more effective and efficient way,” they said, and many of the STI report’s recommendations “overlap with priorities for the broader health and public health system moving forward.”
Dr. Mercer also believes the recommendations are realistic, “but only the beginning,” she told this news organization. “Comprehensive sexual education to expand knowledge about STI prevention and public health campaigns to help destigmatize sexual health care in general will remain crucial,” she said.
Sexual education, expanded access, and destigmatizing sexual care are particularly important for reaching the populations most in need of care, such as adolescents and young adults, as well as ethnic, racial, sexual, and gender-minority youth.
“It cannot be overstated how important of a priority population adolescents and young adults are,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They noted that those aged 15-24 account for half of all STIs each year but represent only a quarter of the sexually active population. “Targeted efforts for STI prevention and treatment among adolescents and young adults are therefore essential for an overall successful strategy to address STIs and sexual health in the United States.”
The National Academies report was supported by the Centers for Disease Control and Prevention and the National Association of County and City Health Officials. Dr. Mercer, Dr. Ault, and Mr. Thimm-Kaiser have disclosed no relevant financial relationships. Dr. Guilamo-Ramos has received grants and personal fees from ViiV Health care.
A version of this article first appeared on Medscape.com.
A vital aspect of expanding access and care for sexually transmitted infections (STIs) in the United States is broadening responsibility for this care across the health care system and other community resources, according to an article published online July 6 in Clinical Infectious Diseases. This expansion and decentralization of care are central to adopting the “new sexual health paradigm” recommended by a National Academies report that was published in March.
“STIs represent a sizable, longstanding, and growing public health challenge,” write Vincent Guilamo-Ramos, PhD, MPH, dean and professor at the Duke University School of Nursing and director of the Center for Latino Adolescent and Family Health (CLAFH) at Duke University, both in Durham, N.C., and his colleagues. Yet the limitations on the current STI workforce and limited federal funding and support for STI prevention and care mean it will take clinicians of all types from across the health care spectrum to meet the challenge, they explain.
“For too long, STI prevention and treatment has been perceived as the sole responsibility of a narrow workforce of specialized STI and HIV service providers,” Dr. Guilamo-Ramos and his coauthor, Marco Thimm-Kaiser, MPH, associate in research at Duke University and epidemiologist at CLAFH, wrote in an email.
“However, the resources allocated to this STI specialty workforce have diminished over time, along with decreasing investments in the broader U.S. public health infrastructure,” they continued. “At the same time – and in part due to this underinvestment – STI rates have soared, reaching a record high for the sixth year in a row in 2019.”
Those factors led to the National Academies report, which recommends moving “away from the traditional, disease-focused perspective on STIs in favor of a holistic perspective of sexual health as an integral component of overall health and well-being,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote to this news organization.
In their article, the authors review the limitations in the STI workforce, the implications of those limitations for the broader health care industry, and what it will take for STI and HIV specialists as well as regulators to ensure it’s possible to achieve the paradigm shift recommended by the National Academies.
Currently, the biggest limitation is access to care, said Laura Mercer, MD, MBA, of the department of obstetrics and gynecology and the ob.gyn. clerkship director at the University of Arizona, Phoenix. Dr. Mercer, who was not involved with the National Academies report or the analysis of it, said in an interview that it’s essential to emphasize “sexual health as a core element of routine primary and preventative care” to ensure it becomes more accessible to patients without the need to seek out specialty care.
Dr. Guilamo-Ramos and his colleagues drive home the importance of such a shift by noting that more than 200 million Americans live in counties with no practicing infectious disease physicians. The disparities are greatest in Southern states, which account for 40% of all reported STIs. The workforce shortage has continued to worsen alongside the deterioration of the clinical infrastructure supporting STI specialty services, the authors write.
Hence the need to expand accountability for care not only to primary-care physicians but also to nurses, pharmacists, physician assistants, nurse practitioners, and behavioral health practitioners. Doing so also requires normalizing sexual health services across health care professions.
“Prevention is a crucial first step” to this, Dr. Mercer said. “This is particularly important as we recall that almost half of new sexually transmitted infections occur in teenagers. Destigmatizing sexual health and sexual health education will also help encourage patients of all ages to request and accept testing.”
Further, with primary care practitioners managing most STI testing and treatment, subspecialists can focus primarily on complex or refractory cases, she added. Ways to help broaden care include developing point-of-care testing for STIs and improving the accuracy of existing testing, she said.
“The goal is to make routine sexual health services accessible in a wide range of settings, such as in primary care, at pharmacies, and in community-based settings, and to draw on a broader workforce for delivery of sexual health services,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview.
Kevin Ault, MD, professor of obstetrics and gynecology and director of clinical and translational research at the University of Kansas Medical Center in Kansas City, said that many medical organizations, such as the American College of Obstetricians and Gynecologists, have long advocated incorporating sexual health into routine preventive care. He also noted that pharmacists have already become proactive in preventing STIs and could continue to do so.
“Vaccines for hepatitis and human papillomavirus are commonly available at pharmacies,” Dr. Ault said. He was not involved in the article by Dr. Guilamo-Ramos and colleagues or the original report. “Pharmacists could also fill a gap by administering injectable medications such as penicillin. States would have to approve changes in policy, but many states have already done this for expedited partner therapy.”
Dr. Guilamo-Ramos and Mr. Thimm-Kaiser noted similar barriers that must be removed to broaden delivery of STI services.
“Unfortunately, too many highly trained health care providers who are well-positioned for the delivery of sexual health services face regulatory or administrative barriers to practice to the full scope of their training,” they wrote. “These barriers can have a particularly negative impact in medically underserved communities, where physician shortages are common and where novel, decentralized health care service delivery models that draw on nonphysician providers may hold the greatest promise.”
As more diverse health care practitioners take on these roles, ID and HIV specialists can provide their expertise in developing training and technical assistance to support generalists, Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They can also aid in aligning “clinical training curricula, licensing criteria, and practice guidelines with routine delivery of sexual health services.”
Dr. Guilamo-Ramos and his coauthors offer specific recommendations for professional training, licensing, and practice guidelines to help overcome the “insufficient knowledge, inadequate training, and absence of explicit protocols” that currently impede delivery of STI services in general practice settings.
Although the paradigm shift recommended by the National Academies is ambitious, it’s also necessary, and “none of the recommendations are out of reach,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser said in an interview. They pointed out how the COVID-19 pandemic has highlighted how underresourced the health care workforce and infrastructure are and how great health care disparities are.
“There is momentum toward rebuilding the nation’s health and public health system in a more effective and efficient way,” they said, and many of the STI report’s recommendations “overlap with priorities for the broader health and public health system moving forward.”
Dr. Mercer also believes the recommendations are realistic, “but only the beginning,” she told this news organization. “Comprehensive sexual education to expand knowledge about STI prevention and public health campaigns to help destigmatize sexual health care in general will remain crucial,” she said.
Sexual education, expanded access, and destigmatizing sexual care are particularly important for reaching the populations most in need of care, such as adolescents and young adults, as well as ethnic, racial, sexual, and gender-minority youth.
“It cannot be overstated how important of a priority population adolescents and young adults are,” Dr. Guilamo-Ramos and Mr. Thimm-Kaiser wrote. They noted that those aged 15-24 account for half of all STIs each year but represent only a quarter of the sexually active population. “Targeted efforts for STI prevention and treatment among adolescents and young adults are therefore essential for an overall successful strategy to address STIs and sexual health in the United States.”
The National Academies report was supported by the Centers for Disease Control and Prevention and the National Association of County and City Health Officials. Dr. Mercer, Dr. Ault, and Mr. Thimm-Kaiser have disclosed no relevant financial relationships. Dr. Guilamo-Ramos has received grants and personal fees from ViiV Health care.
A version of this article first appeared on Medscape.com.
C. diff eradication not necessary for clinical cure of recurrent infections with fecal transplant
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
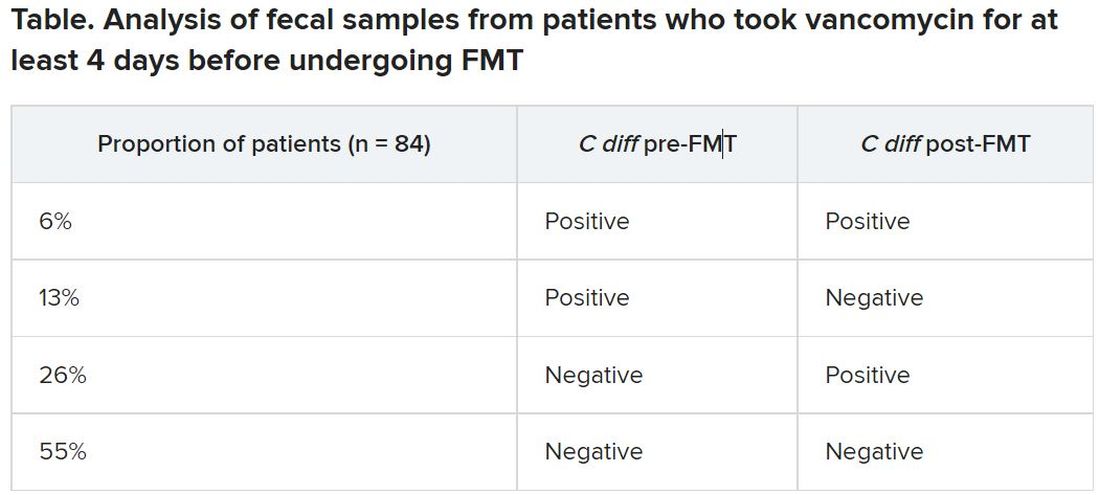
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
Help your patients understand their C. difficile diagnosis by sharing patient education from the AGA GI Patient Center: www.gastro.org/Cdiff.
A version of this article first appeared on Medscape.com.
Hyperimmune globulin fails to prevent congenital CMV infection
Administering hyperimmune globulin to pregnant women who tested positive for cytomegalovirus did not reduce CMV infections or deaths among their fetuses or newborns, according to a randomized controlled trial published online July 28 in the New England Journal of Medicine.
Up to 40,000 infants a year have congenital CMV infections, which can lead to stillbirth, neonatal death, deafness, and cognitive and motor delay. An estimated 35%-40% of fetuses of women with a primary CMV infection will develop an infection, write Brenna Hughes, MD, an associate professor of ob/gyn and chief of the division of maternal fetal medicine at Duke University, Durham, N.C., and colleagues.
Previous trials and observational studies have shown mixed results with hyperimmune globulin for the prevention of congenital CMV infection.
“It was surprising to us that none of the outcomes in this trial were in the direction of potential benefit,” Dr. Hughes told this news organization. “However, this is why it is important to do large trials in a diverse population.”
The study cohort comprised 206,082 pregnant women who were screened for CMV infection before 23 weeks’ gestation. Of those women, 712 (0.35%) tested positive for CMV. The researchers enrolled 399 women who had tested positive and randomly assigned them to receive either a monthly infusion of CMV hyperimmune globulin (100 mg/kg) or placebo until delivery. The researchers used a composite of CMV infection or, if no testing occurred, fetal/neonatal death as the primary endpoint.
The trial was stopped early for futility when data from 394 participants revealed that 22.7% of offspring in the hyperimmune globulin group and 19.4% of those in the placebo group had had a CMV infection or had died (relative risk = 1.17; P = .42).
When individual endpoints were examined, trends were detected in favor of the placebo, but they did not reach statistical significance. The incidence of death was higher in the hyperimmune globulin group (4.9%) than in the placebo group (2.6%). The rate of preterm birth was also higher in the intervention group (12.2%) than in the group that received placebo (8.3%). The incidence of birth weight below the fifth percentile was 10.3% in the intervention group and 5.4% in the placebo group.
One woman who received hyperimmune globulin experienced a severe allergic reaction to the first infusion. Additionally, more women in the hyperimmune globulin group experienced headaches and shaking chills during infusions than did those who received placebo. There were no differences in maternal outcomes between the groups. There were no thromboembolic or ischemic events in either group.
“These findings suggest CMV hyperimmune globulin should not be used for the prevention of congenital CMV in pregnant patients with primary CMV during pregnancy,” Dr. Hughes said in an interview.
“A CMV vaccine is likely to be the most effective public health measure that we can offer, and that should be at the forefront of research investments,” she said. “But some of the other medications that work against CMV should be tested on a large scale as well,” she said. For example, a small trial in Israel showed that high-dose valacyclovir in early pregnancy decreased congenital CMV, and thus the drug merits study in a larger trial, she said.
Other experts agree that developing a vaccine should be the priority.
“The ultimate goal for preventing the brain damage and birth defects caused by congenital CMV infection is a vaccine that is as effective as the rubella vaccine has been for eliminating congenital rubella syndrome and that can be given well before pregnancy,” said Sallie Permar, MD, PhD, chair of pediatrics at Weill Cornell Medicine and pediatrician-in-chief at New York–Presbyterian/Weill Cornell Medical Center and the New York–Presbyterian Komansky Children’s Hospital in New York.
“While trials of vaccines are ongoing, there is a need to have a therapeutic option, especially for the high-risk setting of a mother acquiring the virus for the first time during pregnancy,” Dr. Permar said in an interview.
Dr. Permar was not involved in this study but is involved in follow-up studies of this cohort and is conducting research on CMV maternal vaccines. She noted the need for safe, effective antiviral treatments and for research into newer immunoglobulin products, such as monoclonal antibodies.
Both Dr. Permar and Dr. Hughes highlighted the challenge of raising awareness about the danger of CMV infections during pregnancy.
“Pregnant women, and especially those who have or work with young children, who are frequently carriers of the infection, should be informed of this risk,” Dr. Permar said. She hopes universal testing of newborns will be implemented and that it enables people to recognize the frequency and burden of these infections. She remains optimistic about a vaccine.
“After 60 years of research into a CMV vaccine, I believe we are currently in a ‘golden age’ of CMV vaccine development,” she said. She noted that Moderna is about to launch a phase 3 mRNA vaccine trial for CMV. “Moreover, immune correlates of protection against CMV have been identified from previous partially effective vaccines, and animal models have improved for preclinical studies. Therefore, I believe we will have an effective and safe vaccine against this most common congenital infection in the coming years.”
The research was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Center for Advancing Translational Sciences. Dr. Hughes has served on Merck’s scientific advisory board. Various coauthors have received personal fees from Medela and nonfinancial support from Hologic; personal fees from Moderna and VBI vaccines, and grants from Novavax. Dr. Permar consults for Pfizer, Moderna, Merck, Sanofi, and Dynavax on their CMV vaccine programs, and she has a sponsored research program with Merck and Moderna on CMV vaccines.
A version of this article first appeared on Medscape.com.
Administering hyperimmune globulin to pregnant women who tested positive for cytomegalovirus did not reduce CMV infections or deaths among their fetuses or newborns, according to a randomized controlled trial published online July 28 in the New England Journal of Medicine.
Up to 40,000 infants a year have congenital CMV infections, which can lead to stillbirth, neonatal death, deafness, and cognitive and motor delay. An estimated 35%-40% of fetuses of women with a primary CMV infection will develop an infection, write Brenna Hughes, MD, an associate professor of ob/gyn and chief of the division of maternal fetal medicine at Duke University, Durham, N.C., and colleagues.
Previous trials and observational studies have shown mixed results with hyperimmune globulin for the prevention of congenital CMV infection.
“It was surprising to us that none of the outcomes in this trial were in the direction of potential benefit,” Dr. Hughes told this news organization. “However, this is why it is important to do large trials in a diverse population.”
The study cohort comprised 206,082 pregnant women who were screened for CMV infection before 23 weeks’ gestation. Of those women, 712 (0.35%) tested positive for CMV. The researchers enrolled 399 women who had tested positive and randomly assigned them to receive either a monthly infusion of CMV hyperimmune globulin (100 mg/kg) or placebo until delivery. The researchers used a composite of CMV infection or, if no testing occurred, fetal/neonatal death as the primary endpoint.
The trial was stopped early for futility when data from 394 participants revealed that 22.7% of offspring in the hyperimmune globulin group and 19.4% of those in the placebo group had had a CMV infection or had died (relative risk = 1.17; P = .42).
When individual endpoints were examined, trends were detected in favor of the placebo, but they did not reach statistical significance. The incidence of death was higher in the hyperimmune globulin group (4.9%) than in the placebo group (2.6%). The rate of preterm birth was also higher in the intervention group (12.2%) than in the group that received placebo (8.3%). The incidence of birth weight below the fifth percentile was 10.3% in the intervention group and 5.4% in the placebo group.
One woman who received hyperimmune globulin experienced a severe allergic reaction to the first infusion. Additionally, more women in the hyperimmune globulin group experienced headaches and shaking chills during infusions than did those who received placebo. There were no differences in maternal outcomes between the groups. There were no thromboembolic or ischemic events in either group.
“These findings suggest CMV hyperimmune globulin should not be used for the prevention of congenital CMV in pregnant patients with primary CMV during pregnancy,” Dr. Hughes said in an interview.
“A CMV vaccine is likely to be the most effective public health measure that we can offer, and that should be at the forefront of research investments,” she said. “But some of the other medications that work against CMV should be tested on a large scale as well,” she said. For example, a small trial in Israel showed that high-dose valacyclovir in early pregnancy decreased congenital CMV, and thus the drug merits study in a larger trial, she said.
Other experts agree that developing a vaccine should be the priority.
“The ultimate goal for preventing the brain damage and birth defects caused by congenital CMV infection is a vaccine that is as effective as the rubella vaccine has been for eliminating congenital rubella syndrome and that can be given well before pregnancy,” said Sallie Permar, MD, PhD, chair of pediatrics at Weill Cornell Medicine and pediatrician-in-chief at New York–Presbyterian/Weill Cornell Medical Center and the New York–Presbyterian Komansky Children’s Hospital in New York.
“While trials of vaccines are ongoing, there is a need to have a therapeutic option, especially for the high-risk setting of a mother acquiring the virus for the first time during pregnancy,” Dr. Permar said in an interview.
Dr. Permar was not involved in this study but is involved in follow-up studies of this cohort and is conducting research on CMV maternal vaccines. She noted the need for safe, effective antiviral treatments and for research into newer immunoglobulin products, such as monoclonal antibodies.
Both Dr. Permar and Dr. Hughes highlighted the challenge of raising awareness about the danger of CMV infections during pregnancy.
“Pregnant women, and especially those who have or work with young children, who are frequently carriers of the infection, should be informed of this risk,” Dr. Permar said. She hopes universal testing of newborns will be implemented and that it enables people to recognize the frequency and burden of these infections. She remains optimistic about a vaccine.
“After 60 years of research into a CMV vaccine, I believe we are currently in a ‘golden age’ of CMV vaccine development,” she said. She noted that Moderna is about to launch a phase 3 mRNA vaccine trial for CMV. “Moreover, immune correlates of protection against CMV have been identified from previous partially effective vaccines, and animal models have improved for preclinical studies. Therefore, I believe we will have an effective and safe vaccine against this most common congenital infection in the coming years.”
The research was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Center for Advancing Translational Sciences. Dr. Hughes has served on Merck’s scientific advisory board. Various coauthors have received personal fees from Medela and nonfinancial support from Hologic; personal fees from Moderna and VBI vaccines, and grants from Novavax. Dr. Permar consults for Pfizer, Moderna, Merck, Sanofi, and Dynavax on their CMV vaccine programs, and she has a sponsored research program with Merck and Moderna on CMV vaccines.
A version of this article first appeared on Medscape.com.
Administering hyperimmune globulin to pregnant women who tested positive for cytomegalovirus did not reduce CMV infections or deaths among their fetuses or newborns, according to a randomized controlled trial published online July 28 in the New England Journal of Medicine.
Up to 40,000 infants a year have congenital CMV infections, which can lead to stillbirth, neonatal death, deafness, and cognitive and motor delay. An estimated 35%-40% of fetuses of women with a primary CMV infection will develop an infection, write Brenna Hughes, MD, an associate professor of ob/gyn and chief of the division of maternal fetal medicine at Duke University, Durham, N.C., and colleagues.
Previous trials and observational studies have shown mixed results with hyperimmune globulin for the prevention of congenital CMV infection.
“It was surprising to us that none of the outcomes in this trial were in the direction of potential benefit,” Dr. Hughes told this news organization. “However, this is why it is important to do large trials in a diverse population.”
The study cohort comprised 206,082 pregnant women who were screened for CMV infection before 23 weeks’ gestation. Of those women, 712 (0.35%) tested positive for CMV. The researchers enrolled 399 women who had tested positive and randomly assigned them to receive either a monthly infusion of CMV hyperimmune globulin (100 mg/kg) or placebo until delivery. The researchers used a composite of CMV infection or, if no testing occurred, fetal/neonatal death as the primary endpoint.
The trial was stopped early for futility when data from 394 participants revealed that 22.7% of offspring in the hyperimmune globulin group and 19.4% of those in the placebo group had had a CMV infection or had died (relative risk = 1.17; P = .42).
When individual endpoints were examined, trends were detected in favor of the placebo, but they did not reach statistical significance. The incidence of death was higher in the hyperimmune globulin group (4.9%) than in the placebo group (2.6%). The rate of preterm birth was also higher in the intervention group (12.2%) than in the group that received placebo (8.3%). The incidence of birth weight below the fifth percentile was 10.3% in the intervention group and 5.4% in the placebo group.
One woman who received hyperimmune globulin experienced a severe allergic reaction to the first infusion. Additionally, more women in the hyperimmune globulin group experienced headaches and shaking chills during infusions than did those who received placebo. There were no differences in maternal outcomes between the groups. There were no thromboembolic or ischemic events in either group.
“These findings suggest CMV hyperimmune globulin should not be used for the prevention of congenital CMV in pregnant patients with primary CMV during pregnancy,” Dr. Hughes said in an interview.
“A CMV vaccine is likely to be the most effective public health measure that we can offer, and that should be at the forefront of research investments,” she said. “But some of the other medications that work against CMV should be tested on a large scale as well,” she said. For example, a small trial in Israel showed that high-dose valacyclovir in early pregnancy decreased congenital CMV, and thus the drug merits study in a larger trial, she said.
Other experts agree that developing a vaccine should be the priority.
“The ultimate goal for preventing the brain damage and birth defects caused by congenital CMV infection is a vaccine that is as effective as the rubella vaccine has been for eliminating congenital rubella syndrome and that can be given well before pregnancy,” said Sallie Permar, MD, PhD, chair of pediatrics at Weill Cornell Medicine and pediatrician-in-chief at New York–Presbyterian/Weill Cornell Medical Center and the New York–Presbyterian Komansky Children’s Hospital in New York.
“While trials of vaccines are ongoing, there is a need to have a therapeutic option, especially for the high-risk setting of a mother acquiring the virus for the first time during pregnancy,” Dr. Permar said in an interview.
Dr. Permar was not involved in this study but is involved in follow-up studies of this cohort and is conducting research on CMV maternal vaccines. She noted the need for safe, effective antiviral treatments and for research into newer immunoglobulin products, such as monoclonal antibodies.
Both Dr. Permar and Dr. Hughes highlighted the challenge of raising awareness about the danger of CMV infections during pregnancy.
“Pregnant women, and especially those who have or work with young children, who are frequently carriers of the infection, should be informed of this risk,” Dr. Permar said. She hopes universal testing of newborns will be implemented and that it enables people to recognize the frequency and burden of these infections. She remains optimistic about a vaccine.
“After 60 years of research into a CMV vaccine, I believe we are currently in a ‘golden age’ of CMV vaccine development,” she said. She noted that Moderna is about to launch a phase 3 mRNA vaccine trial for CMV. “Moreover, immune correlates of protection against CMV have been identified from previous partially effective vaccines, and animal models have improved for preclinical studies. Therefore, I believe we will have an effective and safe vaccine against this most common congenital infection in the coming years.”
The research was funded by the Eunice Kennedy Shriver National Institute of Child Health and Human Development and the National Center for Advancing Translational Sciences. Dr. Hughes has served on Merck’s scientific advisory board. Various coauthors have received personal fees from Medela and nonfinancial support from Hologic; personal fees from Moderna and VBI vaccines, and grants from Novavax. Dr. Permar consults for Pfizer, Moderna, Merck, Sanofi, and Dynavax on their CMV vaccine programs, and she has a sponsored research program with Merck and Moderna on CMV vaccines.
A version of this article first appeared on Medscape.com.
C. Diff eradication not necessary for clinical cure of recurrent infections with fecal transplant
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
A version of this article first appeared on Medscape.com.
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
A version of this article first appeared on Medscape.com.
It’s not necessary to completely eradicate all Clostridioides difficile to successfully treat recurrent C. difficile infections with fecal microbiota transplant (FMT), according to a study presented online July 12 at the European Congress of Clinical Microbiology & Infectious Diseases.
C. difficile colonization persisted for 3 weeks after FMT in about one-quarter of patients, but it’s not clear whether this is a persistent infection, a newly acquired infection, or partial persistence of a mixed infection, said Elisabeth Terveer, MD, a medical microbiologist at Leiden (the Netherlands) University Medical Center. In addition, “82% of patients with detectable C. diff do not relapse, so it’s absolutely not necessary for a cure,” she said.
Several mechanisms explain why FMT is a highly effective therapy for recurrent C. difficile infections, including restoration of bacterial metabolism in the gut, immune modulation, and direct competition between bacteria, Dr. Terveer said, but it’s less clear whether eradication of C. difficile spores is among these mechanisms.
Between May 2016 and April 2020, the researchers analyzed fecal samples from 84 patients who took vancomycin for at least 4 days before undergoing FMT. The researchers took fecal samples from patients before FMT and 3 weeks after FMT to culture them and the donor samples for presence of C. difficile, and they assessed clinical outcomes at 3 weeks and 6 months after FMT.
After antibiotic treatment but prior to FMT, 19% of patients (n = 16) still had a toxigenic C. difficile culture while the other 81% had a negative culture. None of the donor samples had a positive C. difficile culture. After FMT treatment, five patients who had a positive pre-FMT culture remained positive, and the other 11 were negative. Among the 81% of patients (n = 68) who had a negative culture just before FMT, 22 had a positive culture and 46 had a negative culture after FMT. Overall, 26% of patients post FMT had a positive C. difficile culture, a finding that was 10-fold higher than another study that assessed C. difficile with PCR testing, Dr. Terveer said.
The clinical cure rate after FMT was 94%, and five patients had relapses within 2 months of their FMT. These relapses were more prevalent in patients with a positive C. difficile culture prior to FMT (odds ratio [OR], 7.6; P = .045) and a positive C. difficile culture after FMT (OR, 13.6; P = .016). Still, 82% of patients who had a positive C. difficile culture post FMT remained clinically cured 2 months later.
It’s unclear why 19% of patients had a positive culture after their antibiotic pretreatment prior to FMT, Dr. Terveer said, but it may be because the pretreatment was of such a short duration.
“I think the advice should be: Give a full anti–C. diff antibiotic course to treat the C. diff infection, and then give FMT afterward to restore the microbiota and prevent further relapses,” Dr. Terveer told attendees.
Dimitri Drekonja, MD, chief of the Minneapolis VA Infectious Disease Section, said the findings were not necessarily surprising, but it would have been interesting for the researchers to have conducted DNA sequencing of the patients’ fecal samples post FMT to see what the biological diversity looked like.
“One school of thought has been that you have to repopulate the normal diverse microbiota of the colon” with FMT, and the other “is that you need to get rid of the C. diff that›s there,” Dr. Drekonja, who was not involved in the study, said in an interview. “I think more people think it’s the diverse microbiota because if it’s just getting rid of C. diff, we can get do that with antibiotics – but that gets rid of the other organisms.”
As long as you have a diverse microbiota post FMT, Dr. Drekonja said, then “having a few residual organisms, even if they get magnified in the culture process, is probably not that big a deal.”
But there’s a third school of thought that Dr. Drekonja said he himself falls into: “I don’t really care how it works, just that in well-done trials, it does work.” As long as large, robust, well-blinded trials show that FMT works, “I’m open to all sorts of ideas of what the mechanism is,” he said. “The main thing is that it does or doesn’t work.”
These findings basically reinforce current guidance not to test patients’ stools if they are asymptomatic, Dr. Drekonja said. In the past, clinicians sometimes tested patients’ stool after therapy to ensure the C. difficile was eradicated, regardless of whether the patient had symptoms of infection, he said.
“We’ve since become much more attuned that there are lots of people who have detectable C. diff in their stool without any symptoms,” whether detectable by culture or PCR, Dr. Drekonja said. “Generally, if you’re doing well and you’re not having diarrhea, don’t test, and if someone does test and finds it, pretend you didn’t see the test,” he advised. “This is a big part of diagnostic stewardship, which is: You don’t go testing people who are doing well.”
The Netherlands Donor Feces Bank used in the research is funded by a grant from Vedanta Biosciences. Dr. Drekonja had no disclosures.
A version of this article first appeared on Medscape.com.
Long COVID seen in patients with severe and mild disease
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.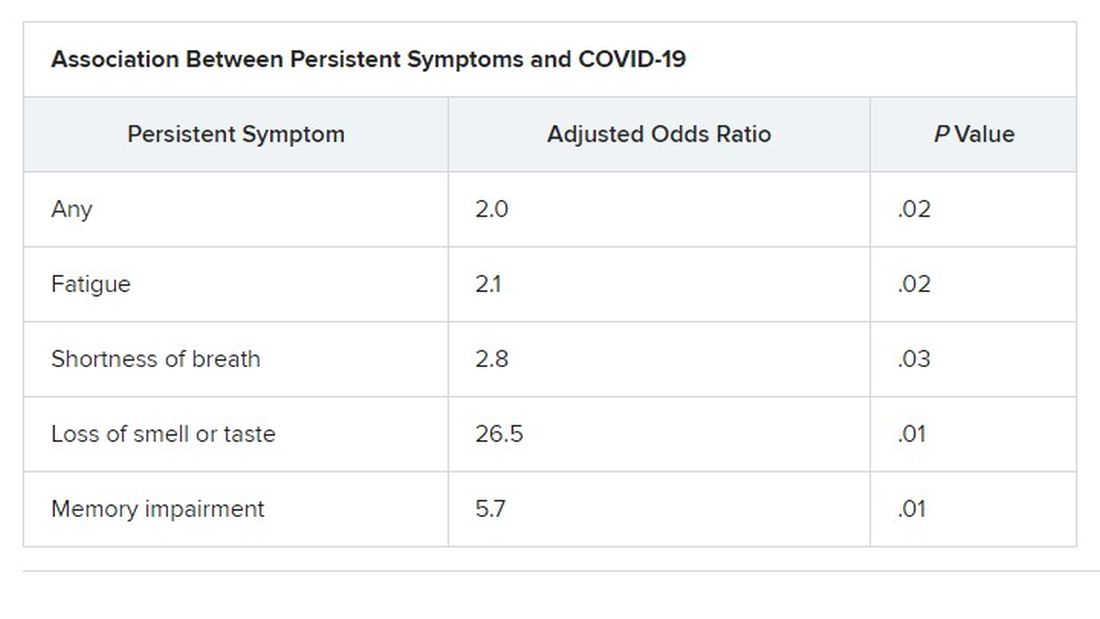
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Findings from the cohort, composed of 113 COVID-19 survivors who developed ARDS after admission to a single center before to April 16, 2020, were presented online at the 31st European Congress of Clinical Microbiology & Infectious Diseases by Judit Aranda, MD, from Complex Hospitalari Moisés Broggi in Barcelona.
Median age of the participants was 64 years, and 70% were male. At least one persistent symptom was experienced during follow-up by 81% of the cohort, with 45% reporting shortness of breath, 50% reporting muscle pain, 43% reporting memory impairment, and 46% reporting physical weakness of at least 5 on a 10-point scale.
Of the 104 participants who completed a 6-minute walk test, 30% had a decrease in oxygen saturation level of at least 4%, and 5% had an initial or final level below 88%. Of the 46 participants who underwent a pulmonary function test, 15% had a forced expiratory volume in 1 second below 70%.
And of the 49% of participants with pathologic findings on chest x-ray, most were bilateral interstitial infiltrates (88%).
In addition, more than 90% of participants developed depression, anxiety, or PTSD, Dr. Aranda reported.
Not the whole picture
This study shows that sicker people – “those in intensive care units with acute respiratory distress syndrome” – are “more likely to be struggling with more severe symptoms,” said Christopher Terndrup, MD, from the division of general internal medicine and geriatrics at Oregon Health & Science University, Portland.
But a Swiss study, also presented at the meeting, “shows how even mild COVID cases can lead to debilitating symptoms,” Dr. Terndrup said in an interview.
The investigation of long-term COVID symptoms in outpatients was presented online by Florian Desgranges, MD, from Lausanne (Switzerland) University Hospital. He and his colleagues found that more than half of those with a mild to moderate disease had persistent symptoms at least 3 months after diagnosis.
The prevalence of long COVID has varied in previous research, from 15% in a study of health care workers, to 46% in a study of patients with mild COVID, 52% in a study of young COVID outpatients, and 76% in a study of patients hospitalized with COVID.
Dr. Desgranges and colleagues evaluated patients seen in an ED or outpatient clinic from February to April 2020.
The 418 patients with a confirmed COVID-19 diagnosis were compared with a control group of 89 patients who presented to the same centers during the same time frame with similar symptoms – cough, shortness of breath, or fever – but had a negative SARS-CoV-2 test.
The number of patients with comorbidities was similar in the COVID and control groups (34% vs. 36%), as was median age (41 vs. 36 years) and the prevalence of women (62% vs 64%), but the proportion of health care workers was lower in the COVID group (64% vs 82%; P =.006).
Symptoms that persisted for at least 3 months were more common in the COVID than in the control group (53% vs. 37%). And patients in the COVID group reported more symptoms than those in the control group after adjustment for age, gender, smoking status, comorbidities, and timing of the survey phone call.
Levels of sleeping problems and headache were similar in the two groups.
“We have to remember that with COVID-19 came the psychosocial changes of the pandemic situation” Dr. Desgranges said.
This study suggests that some long-COVID symptoms – such as the fatigue, headache, and sleep disorders reported in the control group – could be related to the pandemic itself, which has caused psychosocial distress, Dr. Terndrup said.
Another study that looked at outpatients “has some fantastic long-term follow-up data, and shows that many patients are still engaging in rehabilitation programs nearly a year after their diagnosis,” he explained.
The COVID HOME study
That prospective longitudinal COVID HOME study, which assessed long-term symptoms in people who were never hospitalized for COVID, was presented online by Adriana Tami, MD, PhD, from the University Medical Center Groningen (the Netherlands).
The researchers visited the homes of patients to collect data, blood samples, and perform polymerase chain reaction (PCR) testing 1, 2, and 3 weeks after a diagnosis of COVID-19. If their PCR test was still positive, testing continued until week 6 or a negative test. In addition, participants completed questionnaires at week 2 and at months 3, 6 and 12 to assess fatigue, quality of life, and symptoms of depression and anxiety.
Three-month follow-up data were available for 134 of the 276 people initially enrolled in the study. Questionnaires were completed by 85 participants at 3 months, 62 participants at 6 months, and 10 participants at 12 months.
At least 40% of participants reported long-lasting symptoms at some point during follow-up, and at least 30% said they didn’t feel fully recovered at 12 months. The most common symptom was persistent fatigue, reported at 3, 6, and 12 months by at least 44% of participants. Other common symptoms – reported by at least 20% of respondents at 3, 6, and 12 months – were headache, mental or neurologic symptoms, and sleep disorders, shortness of breath, lack of smell or taste, and severe fatigue.
“We have a high proportion of nonhospitalized individuals who suffer from long COVID after more than 12 months,” Dr. Tami concluded, adding that the study is ongoing. “We have other variables that we want to look at, including duration viral shedding and serological results and variants.”
“These cohort studies are very helpful, but they can lead to inaccurate conclusions,” Dr. Terndrup cautioned.
They only provide pieces of the big picture, but they “do add to a growing body of knowledge about a significant portion of COVID patients still struggling with symptoms long after their initial infection. The symptoms can be quite variable but are dominated by both physical and mental fatigue, and tend to be worse in patients who were sicker at initial infection,” he said in an interview.
As a whole, these studies reinforce the need for treatment programs to help patients who suffer from long COVID, he added, but “I advise caution to folks suffering out there who seek ‘miracle cures’; across the world, we are collaborating to find solutions that are safe and effective.”
We are in desperate need of an equity lens in these studies.
“There is still a great deal to learn about long COVID,” said Dr. Terndrup. Data on underrepresented populations – such as Black, Indigenous, and people of color – are lacking from these and others studies, he explained. “We are in desperate need of an equity lens in these studies,” particularly in the United States, where there are “significant disparities” in the treatment of different populations.
However, “I do hope that this work can lead to a better understanding of how other viral infections can cause long-lasting symptoms,” said Dr. Terndrup.
“We have long proposed that after acute presentation, some microbes can cause chronic symptoms, like fatigue and widespread pain. Perhaps we can learn how to better care for these patients after learning from COVID’s significant impact on our societies across the globe.”
Dr. Aranda and Dr. Desgranges have disclosed no relevant financial relationships or study funding. The study by Dr. Tami’s team was funded by the University Medical Center Groningen Organization for Health Research and Development, and Connecting European Cohorts to Increase Common and Effective Response to SARS-CoV-2 Pandemic. Dr. Terndrup disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM EUROPEAN CONGRESS OF CLINICAL MICROBIOLOGY & INFECTIOUS DISEASES
Targeted outreach increases Black COVID-19 vaccination rates
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.
Thoughtful, targeted approaches are needed to increase COVID-19 vaccination rates in Black and Latinx communities, which often distrust the health care system and face barriers to vaccine access, new data show.
“Black, Latinx, and Native American individuals represent about a combined 60% of COVID-19 deaths, despite comprising significantly less of the United States population,” said Jacinda C. Abdul-Mutakabbir, PharmD, from Loma Linda (Calif.) University.
“To put this into perspective, Black individuals represent 13.4% of the United States population, while Native Americans represent 1.6%, clearly showing the disproportionate outcomes here,” she explained during her online presentation at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
The vaccine creates an opportunity to change the disproportionate way COVID-19 has affected racial and ethnic communities, said Dr. Abdul-Mutakabbir, but “a long history of mistreatment within the U.S. health care system decreases their trust for the system to use fair practices when delivering these vaccines.”
For people in minority communities, often “the fear of cost associated with health care keeps them from being vaccinated,” she said. “Also, there is a lack of vaccines actually allocated to these communities, or inconsistent computer-based sign-ups that make WiFi mandatory, which in turn has created additional barriers for vaccination access.”
Loma Linda University maintains the largest mass-vaccination site in San Bernardino County, the fourth-largest county in southern California. However, only 3.0% of the people vaccinated there have been Black. And although 8% of the state’s population is Black, only 2.7% of the Black population has been vaccinated.
In contrast, Black Californians have accounted for about 20% of COVID-19 cases in the state, and 20%-30% of COVID-19 deaths.
To promote equitable access to COVID-19 vaccines, Dr. Abdul-Mutakabbir and colleagues developed a “three-tiered approach.” First, they had local Black faith leaders hold summits ahead of the vaccination clinics. Next, at those summits, they had a Black pharmacist educate attendees about the vaccines. And finally, they held a low-barrier community vaccination clinic in a Black community, where the pharmacist oversaw the transport and preparation of the vaccines.
Because access, transportation, and internet are all barriers to vaccination, the clinic used paper-based registration and was held as a pop-up clinic at a local Black church. The team held two clinics for the first Moderna dose, one clinic for the second Moderna dose, and one clinic for the Johnson & Johnson one-dose vaccine.
During the Moderna first-dose clinics, 673 vaccinations were administered, and during the second-dose clinic, 366 were administered. Early data showed a return rate of 87%, but the team has yet to update the final numbers, Dr. Abdul-Mutakabbir reported. During the Johnson & Johnson clinic, 314 vaccinations were administered, nearly half to Black people. After the community vaccination clinics, the mass vaccination site saw a 0.6% increase in vaccinations for Black people.
Dr. Abdul-Mutakabbir’s team also held three community clinics in Latinx communities. During the first-dose Moderna pop-up clinic, 258 vaccinations were administered, and during the second-dose clinic, 253 were, for a 98% return rate. Approximately 92% of those vaccinated were Latinx.
The study findings are not surprising, said Rhea Boyd, MD, director of equity and justice for California Children’s Trust.
“The barriers to vaccination are known and clear,” she said in an interview. “Mobile clinics with paper appointments address a number of those barriers head on, like transportation, internet access, and accessibility. Having Black providers leading the effort and church leaders involved also has been shown to increase confidence in the operations and process.”
Information campaigns can help counter online disinformation. Ultimately, however, “the main barrier to vaccination is access,” Dr. Boyd said. “Address access and rates will increase.”
The health inequities seen in vaccination rates among Black and Latinx people “are a product of structural and systemic racism,” Dr. Abdul-Mutakabbir said. “To create equitable processes, it is essential that we evaluate how we approach each of these different minoritized groups.”
Dr. Abdul-Mutakabbir disclosed no relevant financial relationships. Dr. Boyd codeveloped THE CONVERSATION, a national campaign to bring credible information about the COVID vaccines to Black and Latinx communities in partnership with KFF, BCAC, and Unidos US.
A version of this article first appeared on Medscape.com.
Early heparin treatment linked to lower COVID-19 mortality
Early treatment with low-molecular-weight heparin (LMWH) reduces the risk for death in patients with COVID-19, a retrospective cohort study shows.
Heparin could reduce the risk for blood clots, Andrea De Vito, MD, of the unit of infectious diseases at the University of Sassari, Italy, said during his online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
“Several studies try to describe the role played by coagulopathies in COVID-19 death,” but the mechanism causing them is still unclear, Dr. De Vito explained.
Some guidelines have suggested heparin as a treatment for hospitalized COVID-19 patients, but few have looked at nonhospitalized patients. In fact, the National Institutes of Health discourages the use of heparin in nonhospitalized COVID-19 patients, and guidance for the home care of COVID-19 patients from the World Health Organization doesn’t mention heparin treatment at all, he said.
To examine the benefits of early heparin – whether administered at home or in the hospital – Dr. De Vito and colleagues looked at a cohort of older adults with COVID-19 who were evaluated or treated at an Italian university hospital.
“Some patients were hospitalized immediately after symptoms onset; other people preferred to call their general practitioner and started the treatment at home,” Dr. De Vito said in an interview. “Other people were hospitalized for worsening of symptoms later in the course of the disease.”
Of the 734 patients, 296 received heparin within 5 days of the onset of symptoms or a positive COVID-19 test. Of the remaining 438 patients, 196 received LMWH treatment later during the disease course, and the rest never received LMWH.
All patients who received early heparin were treated with LMWH 4,000 IU, or 6,000 IU if their body mass index was above 30 kg/m2. This was reduced to 2,000 IU if estimated glomerular filtration rate (eGFR) dropped below 30 mL/min. None of the patients had previously received heparin.
Median age was slightly younger for patients who received early heparin than for those who did not (76.8 vs. 78.5 years).
Other demographic characteristics, such as sex and BMI, were similar in the two groups, as were rates of comorbidities, such as hypertension, cardiovascular disease, diabetes, chronic obstructive pulmonary disease, kidney disease, and neurologic conditions. Also similar were the frequency of symptoms (such as fever, cough, and shortness of breath) and rates of treatment with remdesivir or steroids.
Rates of hospital admission were not significantly different between patients who received early heparin and those who did not (65% vs. 61%). There was also no significant difference in use of a venturi mask (35% vs. 28%), noninvasive ventilation (13% vs. 14%), or intubation (5% vs. 8%).
However, rates of death were significantly lower in patients who received early heparin than in those who did not (13% vs. 25%; P < .0001).
There was a trend toward shorter hospital stays for patients treated with early heparin, but the difference was not significant (median, 10 vs. 13 days; P = .08).
Researchers also conducted a separate analysis of 219 COVID-19 patients who received LMWH at home, regardless of when during their disease course they received it. These patients were significantly less likely to be hospitalized than were patients who did not receive LMWH at home (odds ratio, 0.2; P < .0001).
Comparatively, early heparin treatment had a greater effect on the risk for death and the risk for hospitalization than did other factors.
“Thromboemboli are a major complication of COVID. There is good consensus that hospitalized patients with COVID should receive anticoagulants prophylactically, although the best dose is being studied,” said Judy Stone, MD, an infectious disease physician and journalist who was not involved in the study.
“This study extends those findings of benefit from anticoagulants to nonhospitalized patients, with fewer deaths in those treated with low-molecular-weight heparin,” Dr. Stone told this news organization. “The major limitation is that the study is retrospective and observational. The next step would be to confirm these findings prospectively, randomizing a similar group to LMWH or no anticoagulation.”
Another limitation of the study is that some of the patients lived in nursing homes and might have received care from nurses that eliminated the need for hospitalization, Dr. De Vito added.
The study did not note any external funding. The authors have disclosed no relevant financial relationships. Dr. Stone is a member of the advisory committee for the C-Path CURE Drug Repurposing Collaboratory (CDRC) program and has written for Medscape.
A version of this article first appeared on Medscape.com.
Early treatment with low-molecular-weight heparin (LMWH) reduces the risk for death in patients with COVID-19, a retrospective cohort study shows.
Heparin could reduce the risk for blood clots, Andrea De Vito, MD, of the unit of infectious diseases at the University of Sassari, Italy, said during his online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
“Several studies try to describe the role played by coagulopathies in COVID-19 death,” but the mechanism causing them is still unclear, Dr. De Vito explained.
Some guidelines have suggested heparin as a treatment for hospitalized COVID-19 patients, but few have looked at nonhospitalized patients. In fact, the National Institutes of Health discourages the use of heparin in nonhospitalized COVID-19 patients, and guidance for the home care of COVID-19 patients from the World Health Organization doesn’t mention heparin treatment at all, he said.
To examine the benefits of early heparin – whether administered at home or in the hospital – Dr. De Vito and colleagues looked at a cohort of older adults with COVID-19 who were evaluated or treated at an Italian university hospital.
“Some patients were hospitalized immediately after symptoms onset; other people preferred to call their general practitioner and started the treatment at home,” Dr. De Vito said in an interview. “Other people were hospitalized for worsening of symptoms later in the course of the disease.”
Of the 734 patients, 296 received heparin within 5 days of the onset of symptoms or a positive COVID-19 test. Of the remaining 438 patients, 196 received LMWH treatment later during the disease course, and the rest never received LMWH.
All patients who received early heparin were treated with LMWH 4,000 IU, or 6,000 IU if their body mass index was above 30 kg/m2. This was reduced to 2,000 IU if estimated glomerular filtration rate (eGFR) dropped below 30 mL/min. None of the patients had previously received heparin.
Median age was slightly younger for patients who received early heparin than for those who did not (76.8 vs. 78.5 years).
Other demographic characteristics, such as sex and BMI, were similar in the two groups, as were rates of comorbidities, such as hypertension, cardiovascular disease, diabetes, chronic obstructive pulmonary disease, kidney disease, and neurologic conditions. Also similar were the frequency of symptoms (such as fever, cough, and shortness of breath) and rates of treatment with remdesivir or steroids.
Rates of hospital admission were not significantly different between patients who received early heparin and those who did not (65% vs. 61%). There was also no significant difference in use of a venturi mask (35% vs. 28%), noninvasive ventilation (13% vs. 14%), or intubation (5% vs. 8%).
However, rates of death were significantly lower in patients who received early heparin than in those who did not (13% vs. 25%; P < .0001).
There was a trend toward shorter hospital stays for patients treated with early heparin, but the difference was not significant (median, 10 vs. 13 days; P = .08).
Researchers also conducted a separate analysis of 219 COVID-19 patients who received LMWH at home, regardless of when during their disease course they received it. These patients were significantly less likely to be hospitalized than were patients who did not receive LMWH at home (odds ratio, 0.2; P < .0001).
Comparatively, early heparin treatment had a greater effect on the risk for death and the risk for hospitalization than did other factors.
“Thromboemboli are a major complication of COVID. There is good consensus that hospitalized patients with COVID should receive anticoagulants prophylactically, although the best dose is being studied,” said Judy Stone, MD, an infectious disease physician and journalist who was not involved in the study.
“This study extends those findings of benefit from anticoagulants to nonhospitalized patients, with fewer deaths in those treated with low-molecular-weight heparin,” Dr. Stone told this news organization. “The major limitation is that the study is retrospective and observational. The next step would be to confirm these findings prospectively, randomizing a similar group to LMWH or no anticoagulation.”
Another limitation of the study is that some of the patients lived in nursing homes and might have received care from nurses that eliminated the need for hospitalization, Dr. De Vito added.
The study did not note any external funding. The authors have disclosed no relevant financial relationships. Dr. Stone is a member of the advisory committee for the C-Path CURE Drug Repurposing Collaboratory (CDRC) program and has written for Medscape.
A version of this article first appeared on Medscape.com.
Early treatment with low-molecular-weight heparin (LMWH) reduces the risk for death in patients with COVID-19, a retrospective cohort study shows.
Heparin could reduce the risk for blood clots, Andrea De Vito, MD, of the unit of infectious diseases at the University of Sassari, Italy, said during his online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
“Several studies try to describe the role played by coagulopathies in COVID-19 death,” but the mechanism causing them is still unclear, Dr. De Vito explained.
Some guidelines have suggested heparin as a treatment for hospitalized COVID-19 patients, but few have looked at nonhospitalized patients. In fact, the National Institutes of Health discourages the use of heparin in nonhospitalized COVID-19 patients, and guidance for the home care of COVID-19 patients from the World Health Organization doesn’t mention heparin treatment at all, he said.
To examine the benefits of early heparin – whether administered at home or in the hospital – Dr. De Vito and colleagues looked at a cohort of older adults with COVID-19 who were evaluated or treated at an Italian university hospital.
“Some patients were hospitalized immediately after symptoms onset; other people preferred to call their general practitioner and started the treatment at home,” Dr. De Vito said in an interview. “Other people were hospitalized for worsening of symptoms later in the course of the disease.”
Of the 734 patients, 296 received heparin within 5 days of the onset of symptoms or a positive COVID-19 test. Of the remaining 438 patients, 196 received LMWH treatment later during the disease course, and the rest never received LMWH.
All patients who received early heparin were treated with LMWH 4,000 IU, or 6,000 IU if their body mass index was above 30 kg/m2. This was reduced to 2,000 IU if estimated glomerular filtration rate (eGFR) dropped below 30 mL/min. None of the patients had previously received heparin.
Median age was slightly younger for patients who received early heparin than for those who did not (76.8 vs. 78.5 years).
Other demographic characteristics, such as sex and BMI, were similar in the two groups, as were rates of comorbidities, such as hypertension, cardiovascular disease, diabetes, chronic obstructive pulmonary disease, kidney disease, and neurologic conditions. Also similar were the frequency of symptoms (such as fever, cough, and shortness of breath) and rates of treatment with remdesivir or steroids.
Rates of hospital admission were not significantly different between patients who received early heparin and those who did not (65% vs. 61%). There was also no significant difference in use of a venturi mask (35% vs. 28%), noninvasive ventilation (13% vs. 14%), or intubation (5% vs. 8%).
However, rates of death were significantly lower in patients who received early heparin than in those who did not (13% vs. 25%; P < .0001).
There was a trend toward shorter hospital stays for patients treated with early heparin, but the difference was not significant (median, 10 vs. 13 days; P = .08).
Researchers also conducted a separate analysis of 219 COVID-19 patients who received LMWH at home, regardless of when during their disease course they received it. These patients were significantly less likely to be hospitalized than were patients who did not receive LMWH at home (odds ratio, 0.2; P < .0001).
Comparatively, early heparin treatment had a greater effect on the risk for death and the risk for hospitalization than did other factors.
“Thromboemboli are a major complication of COVID. There is good consensus that hospitalized patients with COVID should receive anticoagulants prophylactically, although the best dose is being studied,” said Judy Stone, MD, an infectious disease physician and journalist who was not involved in the study.
“This study extends those findings of benefit from anticoagulants to nonhospitalized patients, with fewer deaths in those treated with low-molecular-weight heparin,” Dr. Stone told this news organization. “The major limitation is that the study is retrospective and observational. The next step would be to confirm these findings prospectively, randomizing a similar group to LMWH or no anticoagulation.”
Another limitation of the study is that some of the patients lived in nursing homes and might have received care from nurses that eliminated the need for hospitalization, Dr. De Vito added.
The study did not note any external funding. The authors have disclosed no relevant financial relationships. Dr. Stone is a member of the advisory committee for the C-Path CURE Drug Repurposing Collaboratory (CDRC) program and has written for Medscape.
A version of this article first appeared on Medscape.com.
Long COVID symptoms reported by 6% of pediatric patients
The prevalence of long COVID in children has been unclear, and is complicated by the lack of a consistent definition, said Anna Funk, PhD, an epidemiologist at the University of Calgary (Alba.), during her online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
In the several small studies conducted to date, rates range from 0% to 67% 2-4 months after infection, Dr. Funk reported.
To examine prevalence, she and her colleagues, as part of the Pediatric Emergency Research Network (PERN) global research consortium, assessed more than 10,500 children who were screened for SARS-CoV-2 when they presented to the ED at 1 of 41 study sites in 10 countries – Australia, Canada, Indonesia, the United States, plus three countries in Latin America and three in Western Europe – from March 2020 to June 15, 2021.
PERN researchers are following up with the more than 3,100 children who tested positive 14, 30, and 90 days after testing, tracking respiratory, neurologic, and psychobehavioral sequelae.
Dr. Funk presented data on the 1,884 children who tested positive for SARS-CoV-2 before Jan. 20, 2021, and who had completed 90-day follow-up; 447 of those children were hospitalized and 1,437 were not.
Symptoms were reported more often by children admitted to the hospital than not admitted (9.8% vs. 4.6%). Common persistent symptoms were respiratory in 2% of cases, systemic (such as fatigue and fever) in 2%, neurologic (such as headache, seizures, and continued loss of taste or smell) in 1%, and psychological (such as new-onset depression and anxiety) in 1%.
“This study provides the first good epidemiological data on persistent symptoms among SARS-CoV-2–infected children, regardless of severity,” said Kevin Messacar, MD, a pediatric infectious disease clinician and researcher at Children’s Hospital Colorado in Aurora, who was not involved in the study.
And the findings show that, although severe COVID and chronic symptoms are less common in children than in adults, they are “not nonexistent and need to be taken seriously,” he said in an interview.
After adjustment for country of enrollment, children aged 10-17 years were more likely to experience persistent symptoms than children younger than 1 year (odds ratio, 2.4; P = .002).
Hospitalized children were more than twice as likely to experience persistent symptoms as nonhospitalized children (OR, 2.5; P < .001). And children who presented to the ED with at least seven symptoms were four times more likely to have long-term symptoms than those who presented with fewer symptoms (OR, 4.02; P = .01).
‘Some reassurance’
“Given that COVID is new and is known to have acute cardiac and neurologic effects, particularly in children with [multisystem inflammatory syndrome], there were initially concerns about persistent cardiovascular and neurologic effects in any infected child,” Dr. Messacar explained. “These data provide some reassurance that this is uncommon among children with mild or moderate infections who are not hospitalized.”
But “the risk is not zero,” he added. “Getting children vaccinated when it is available to them and taking precautions to prevent unvaccinated children getting COVID is the best way to reduce the risk of severe disease or persistent symptoms.”
The study was limited by its lack of data on variants, reliance on self-reported symptoms, and a population drawn solely from EDs, Dr. Funk acknowledged.
No external funding source was noted. Dr. Messacar and Dr. Funk disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The prevalence of long COVID in children has been unclear, and is complicated by the lack of a consistent definition, said Anna Funk, PhD, an epidemiologist at the University of Calgary (Alba.), during her online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
In the several small studies conducted to date, rates range from 0% to 67% 2-4 months after infection, Dr. Funk reported.
To examine prevalence, she and her colleagues, as part of the Pediatric Emergency Research Network (PERN) global research consortium, assessed more than 10,500 children who were screened for SARS-CoV-2 when they presented to the ED at 1 of 41 study sites in 10 countries – Australia, Canada, Indonesia, the United States, plus three countries in Latin America and three in Western Europe – from March 2020 to June 15, 2021.
PERN researchers are following up with the more than 3,100 children who tested positive 14, 30, and 90 days after testing, tracking respiratory, neurologic, and psychobehavioral sequelae.
Dr. Funk presented data on the 1,884 children who tested positive for SARS-CoV-2 before Jan. 20, 2021, and who had completed 90-day follow-up; 447 of those children were hospitalized and 1,437 were not.
Symptoms were reported more often by children admitted to the hospital than not admitted (9.8% vs. 4.6%). Common persistent symptoms were respiratory in 2% of cases, systemic (such as fatigue and fever) in 2%, neurologic (such as headache, seizures, and continued loss of taste or smell) in 1%, and psychological (such as new-onset depression and anxiety) in 1%.
“This study provides the first good epidemiological data on persistent symptoms among SARS-CoV-2–infected children, regardless of severity,” said Kevin Messacar, MD, a pediatric infectious disease clinician and researcher at Children’s Hospital Colorado in Aurora, who was not involved in the study.
And the findings show that, although severe COVID and chronic symptoms are less common in children than in adults, they are “not nonexistent and need to be taken seriously,” he said in an interview.
After adjustment for country of enrollment, children aged 10-17 years were more likely to experience persistent symptoms than children younger than 1 year (odds ratio, 2.4; P = .002).
Hospitalized children were more than twice as likely to experience persistent symptoms as nonhospitalized children (OR, 2.5; P < .001). And children who presented to the ED with at least seven symptoms were four times more likely to have long-term symptoms than those who presented with fewer symptoms (OR, 4.02; P = .01).
‘Some reassurance’
“Given that COVID is new and is known to have acute cardiac and neurologic effects, particularly in children with [multisystem inflammatory syndrome], there were initially concerns about persistent cardiovascular and neurologic effects in any infected child,” Dr. Messacar explained. “These data provide some reassurance that this is uncommon among children with mild or moderate infections who are not hospitalized.”
But “the risk is not zero,” he added. “Getting children vaccinated when it is available to them and taking precautions to prevent unvaccinated children getting COVID is the best way to reduce the risk of severe disease or persistent symptoms.”
The study was limited by its lack of data on variants, reliance on self-reported symptoms, and a population drawn solely from EDs, Dr. Funk acknowledged.
No external funding source was noted. Dr. Messacar and Dr. Funk disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The prevalence of long COVID in children has been unclear, and is complicated by the lack of a consistent definition, said Anna Funk, PhD, an epidemiologist at the University of Calgary (Alba.), during her online presentation of the findings at the 31st European Congress of Clinical Microbiology & Infectious Diseases.
In the several small studies conducted to date, rates range from 0% to 67% 2-4 months after infection, Dr. Funk reported.
To examine prevalence, she and her colleagues, as part of the Pediatric Emergency Research Network (PERN) global research consortium, assessed more than 10,500 children who were screened for SARS-CoV-2 when they presented to the ED at 1 of 41 study sites in 10 countries – Australia, Canada, Indonesia, the United States, plus three countries in Latin America and three in Western Europe – from March 2020 to June 15, 2021.
PERN researchers are following up with the more than 3,100 children who tested positive 14, 30, and 90 days after testing, tracking respiratory, neurologic, and psychobehavioral sequelae.
Dr. Funk presented data on the 1,884 children who tested positive for SARS-CoV-2 before Jan. 20, 2021, and who had completed 90-day follow-up; 447 of those children were hospitalized and 1,437 were not.
Symptoms were reported more often by children admitted to the hospital than not admitted (9.8% vs. 4.6%). Common persistent symptoms were respiratory in 2% of cases, systemic (such as fatigue and fever) in 2%, neurologic (such as headache, seizures, and continued loss of taste or smell) in 1%, and psychological (such as new-onset depression and anxiety) in 1%.
“This study provides the first good epidemiological data on persistent symptoms among SARS-CoV-2–infected children, regardless of severity,” said Kevin Messacar, MD, a pediatric infectious disease clinician and researcher at Children’s Hospital Colorado in Aurora, who was not involved in the study.
And the findings show that, although severe COVID and chronic symptoms are less common in children than in adults, they are “not nonexistent and need to be taken seriously,” he said in an interview.
After adjustment for country of enrollment, children aged 10-17 years were more likely to experience persistent symptoms than children younger than 1 year (odds ratio, 2.4; P = .002).
Hospitalized children were more than twice as likely to experience persistent symptoms as nonhospitalized children (OR, 2.5; P < .001). And children who presented to the ED with at least seven symptoms were four times more likely to have long-term symptoms than those who presented with fewer symptoms (OR, 4.02; P = .01).
‘Some reassurance’
“Given that COVID is new and is known to have acute cardiac and neurologic effects, particularly in children with [multisystem inflammatory syndrome], there were initially concerns about persistent cardiovascular and neurologic effects in any infected child,” Dr. Messacar explained. “These data provide some reassurance that this is uncommon among children with mild or moderate infections who are not hospitalized.”
But “the risk is not zero,” he added. “Getting children vaccinated when it is available to them and taking precautions to prevent unvaccinated children getting COVID is the best way to reduce the risk of severe disease or persistent symptoms.”
The study was limited by its lack of data on variants, reliance on self-reported symptoms, and a population drawn solely from EDs, Dr. Funk acknowledged.
No external funding source was noted. Dr. Messacar and Dr. Funk disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Patients on methotrexate show T-cell response to Pfizer vaccine
People taking methotrexate had low antibody responses after the first dose of the Pfizer-BioNTech mRNA COVID-19 vaccine, but did show evidence of T-cell–mediated immune responses, findings from a small study show.
The common immunosuppressant has previously been linked to poor antibody responses to mRNA COVID-19 vaccines, but this appears to be the first study to look at T-cell responses in people taking methotrexate.
The study findings were presented online July 11 at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published in The Lancet Rheumatology.
“These findings indicate that seroconversion alone might not adequately reflect vaccine immunogenicity in individuals with immune-mediated inflammatory diseases receiving therapeutic immunosuppression, and caution against routine use of seroconversion data in isolation in clinical practice,” Satveer K. Mahil, MBBChir, PhD, from St. John’s Institute of Dermatology, Guy’s and St. Thomas’ NHS Foundation Trust, London, and colleagues wrote.
“When taking into account functional humoral immunity and T-cell responses, our data suggest that targeted biologics do not impair vaccine responses and provide some reassurance to this vulnerable population,” they wrote. “Notably, although methotrexate attenuated humoral immunity, cellular responses were preserved.”
Dr. Mahil and colleagues assessed 84 consecutive patients from a psoriasis specialist clinic that serves London and southeast England. Median age of the cohort was 43 years, and 85% were White. All had a confirmed psoriasis diagnosis, received the first dose of the Pfizer-BioNTech COVID-19 vaccine, and were taking either methotrexate (17 patients) or a targeted biologic (27 were taking a tumor necrosis factor inhibitor, 15 an interleukin-17 inhibitor, and 25 an IL-23 inhibitor). In addition, 17 healthy patients not receiving immunosuppression therapy who received the Pfizer-BioNTech vaccine served as the control group.
Four weeks after the study participants received their first dose of the vaccine, 78% of the immunosuppressed patients underwent seroconversion – producing measurable antibodies – as did 100% of the control group. Patients taking methotrexate had the lowest seroconversion rate at 47%, compared with 79% with TNF inhibitors, 83% with IL-23 inhibitors, and 100% with IL-17 inhibitors.
Participants taking methotrexate also had lower neutralizing activity against SARS-CoV-2 than control subjects and those taking a targeted biologic, who had similar levels of neutralizing activity.
All participants had low neutralizing titers against the alpha (B.1.1.7) variant.
The researchers also assessed cellular immunity, “defined as the presence of T cells secreting interferon-gamma, IL-2, or IL-21 in response to stimulation with two peptide pools spanning the entire length of the SARS-CoV-2 spike glycoprotein.”
A T-cell response was seen in 84% of participants taking immunosuppressants, including 93% of those in the methotrexate group and 69% of control subjects.
‘Some protection is better than none’
These findings regarding antibodies match what has been seen in other research, said Ignacio Sanz, MD, director of the Lowance Center for Human Immunology at Emory University, Atlanta.
It would be helpful to see antibody responses after the second doses, he added. Those data will be reported later, according to Dr. Mahil and colleagues.
“The authors make the valid point that T-cell immunity should also be measured. The information is meaningful and supports the idea that there could be protection still provided,” Dr. Sanz said in an interview, adding that it would have been helpful to see CD8 T-cell response as well.
“My message to patients, still, is that some protection is better than none, and that, indeed, protection may be afforded in different ways, including T-cell immunity, which, to the extent tested, seems to be induced,” he said. But discussion of B cells independent of their role in producing antibodies is missing.
“When it comes to B-cell responses, antibodies are the easier and more direct measurement. However, it is perfectly possible that the vaccine may fail to induce high antibody titers and still generate good B-cell immunity,” in the same way virus-specific memory B cells do, he explained. “They would not directly produce antibodies, yet they would be available for a good and quick response in the case of subsequent encounter with the virus and, incidentally, in the case of a booster dose. It is possible that the generation of antibody-producing plasma cells might be uncoupled from the generation of memory B cells.”
Temporarily stopping methotrexate
It is well known that methotrexate impairs humoral responses to influenza and pneumococcal vaccines, write Caoilfhionn M. Connolly, MD, and Julie J. Paik, MD, both from the Johns Hopkins University, Baltimore, in an accompanying comment.
Research has also shown that temporarily stopping methotrexate therapy for 2 weeks enhances response to the flu vaccine in patients with rheumatoid arthritis, which prompted the American College of Rheumatology to recommended temporary interruption of methotrexate for 1 week after each dose of the COVID-19 vaccine, the pair notes.
“Although it is encouraging that cellular responses appear to be preserved even in patients with poor humoral responses, these findings are not consistent across study groups,” Dr. Connolly and Dr. Paik explained. “During this period of clinical uncertainty, patients might remain vulnerable, especially after the first dose, and should engage in risk mitigation strategies.”
Mild adverse events after vaccination were reported by 75% of the immunosuppressed patients – most commonly injection-site pain, headache, and fatigue – and by 94% of control subjects. No participants reported moderate or severe adverse effects.
However, 11% of immunosuppressed patients reported a worsening of psoriasis symptoms after vaccination.
This research was funded by the U.K. National Institute for Health Research. Dr. Mahil has received departmental income from AbbVie, Celgene, Eli Lilly, Janssen-Cilag, Novartis, Sano, and UCB unrelated to this study. Seven other authors have relationships with a wide range of pharmaceutical and other companies. Dr. Sanz, Dr. Connolly, and Dr. Paik disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People taking methotrexate had low antibody responses after the first dose of the Pfizer-BioNTech mRNA COVID-19 vaccine, but did show evidence of T-cell–mediated immune responses, findings from a small study show.
The common immunosuppressant has previously been linked to poor antibody responses to mRNA COVID-19 vaccines, but this appears to be the first study to look at T-cell responses in people taking methotrexate.
The study findings were presented online July 11 at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published in The Lancet Rheumatology.
“These findings indicate that seroconversion alone might not adequately reflect vaccine immunogenicity in individuals with immune-mediated inflammatory diseases receiving therapeutic immunosuppression, and caution against routine use of seroconversion data in isolation in clinical practice,” Satveer K. Mahil, MBBChir, PhD, from St. John’s Institute of Dermatology, Guy’s and St. Thomas’ NHS Foundation Trust, London, and colleagues wrote.
“When taking into account functional humoral immunity and T-cell responses, our data suggest that targeted biologics do not impair vaccine responses and provide some reassurance to this vulnerable population,” they wrote. “Notably, although methotrexate attenuated humoral immunity, cellular responses were preserved.”
Dr. Mahil and colleagues assessed 84 consecutive patients from a psoriasis specialist clinic that serves London and southeast England. Median age of the cohort was 43 years, and 85% were White. All had a confirmed psoriasis diagnosis, received the first dose of the Pfizer-BioNTech COVID-19 vaccine, and were taking either methotrexate (17 patients) or a targeted biologic (27 were taking a tumor necrosis factor inhibitor, 15 an interleukin-17 inhibitor, and 25 an IL-23 inhibitor). In addition, 17 healthy patients not receiving immunosuppression therapy who received the Pfizer-BioNTech vaccine served as the control group.
Four weeks after the study participants received their first dose of the vaccine, 78% of the immunosuppressed patients underwent seroconversion – producing measurable antibodies – as did 100% of the control group. Patients taking methotrexate had the lowest seroconversion rate at 47%, compared with 79% with TNF inhibitors, 83% with IL-23 inhibitors, and 100% with IL-17 inhibitors.
Participants taking methotrexate also had lower neutralizing activity against SARS-CoV-2 than control subjects and those taking a targeted biologic, who had similar levels of neutralizing activity.
All participants had low neutralizing titers against the alpha (B.1.1.7) variant.
The researchers also assessed cellular immunity, “defined as the presence of T cells secreting interferon-gamma, IL-2, or IL-21 in response to stimulation with two peptide pools spanning the entire length of the SARS-CoV-2 spike glycoprotein.”
A T-cell response was seen in 84% of participants taking immunosuppressants, including 93% of those in the methotrexate group and 69% of control subjects.
‘Some protection is better than none’
These findings regarding antibodies match what has been seen in other research, said Ignacio Sanz, MD, director of the Lowance Center for Human Immunology at Emory University, Atlanta.
It would be helpful to see antibody responses after the second doses, he added. Those data will be reported later, according to Dr. Mahil and colleagues.
“The authors make the valid point that T-cell immunity should also be measured. The information is meaningful and supports the idea that there could be protection still provided,” Dr. Sanz said in an interview, adding that it would have been helpful to see CD8 T-cell response as well.
“My message to patients, still, is that some protection is better than none, and that, indeed, protection may be afforded in different ways, including T-cell immunity, which, to the extent tested, seems to be induced,” he said. But discussion of B cells independent of their role in producing antibodies is missing.
“When it comes to B-cell responses, antibodies are the easier and more direct measurement. However, it is perfectly possible that the vaccine may fail to induce high antibody titers and still generate good B-cell immunity,” in the same way virus-specific memory B cells do, he explained. “They would not directly produce antibodies, yet they would be available for a good and quick response in the case of subsequent encounter with the virus and, incidentally, in the case of a booster dose. It is possible that the generation of antibody-producing plasma cells might be uncoupled from the generation of memory B cells.”
Temporarily stopping methotrexate
It is well known that methotrexate impairs humoral responses to influenza and pneumococcal vaccines, write Caoilfhionn M. Connolly, MD, and Julie J. Paik, MD, both from the Johns Hopkins University, Baltimore, in an accompanying comment.
Research has also shown that temporarily stopping methotrexate therapy for 2 weeks enhances response to the flu vaccine in patients with rheumatoid arthritis, which prompted the American College of Rheumatology to recommended temporary interruption of methotrexate for 1 week after each dose of the COVID-19 vaccine, the pair notes.
“Although it is encouraging that cellular responses appear to be preserved even in patients with poor humoral responses, these findings are not consistent across study groups,” Dr. Connolly and Dr. Paik explained. “During this period of clinical uncertainty, patients might remain vulnerable, especially after the first dose, and should engage in risk mitigation strategies.”
Mild adverse events after vaccination were reported by 75% of the immunosuppressed patients – most commonly injection-site pain, headache, and fatigue – and by 94% of control subjects. No participants reported moderate or severe adverse effects.
However, 11% of immunosuppressed patients reported a worsening of psoriasis symptoms after vaccination.
This research was funded by the U.K. National Institute for Health Research. Dr. Mahil has received departmental income from AbbVie, Celgene, Eli Lilly, Janssen-Cilag, Novartis, Sano, and UCB unrelated to this study. Seven other authors have relationships with a wide range of pharmaceutical and other companies. Dr. Sanz, Dr. Connolly, and Dr. Paik disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
People taking methotrexate had low antibody responses after the first dose of the Pfizer-BioNTech mRNA COVID-19 vaccine, but did show evidence of T-cell–mediated immune responses, findings from a small study show.
The common immunosuppressant has previously been linked to poor antibody responses to mRNA COVID-19 vaccines, but this appears to be the first study to look at T-cell responses in people taking methotrexate.
The study findings were presented online July 11 at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published in The Lancet Rheumatology.
“These findings indicate that seroconversion alone might not adequately reflect vaccine immunogenicity in individuals with immune-mediated inflammatory diseases receiving therapeutic immunosuppression, and caution against routine use of seroconversion data in isolation in clinical practice,” Satveer K. Mahil, MBBChir, PhD, from St. John’s Institute of Dermatology, Guy’s and St. Thomas’ NHS Foundation Trust, London, and colleagues wrote.
“When taking into account functional humoral immunity and T-cell responses, our data suggest that targeted biologics do not impair vaccine responses and provide some reassurance to this vulnerable population,” they wrote. “Notably, although methotrexate attenuated humoral immunity, cellular responses were preserved.”
Dr. Mahil and colleagues assessed 84 consecutive patients from a psoriasis specialist clinic that serves London and southeast England. Median age of the cohort was 43 years, and 85% were White. All had a confirmed psoriasis diagnosis, received the first dose of the Pfizer-BioNTech COVID-19 vaccine, and were taking either methotrexate (17 patients) or a targeted biologic (27 were taking a tumor necrosis factor inhibitor, 15 an interleukin-17 inhibitor, and 25 an IL-23 inhibitor). In addition, 17 healthy patients not receiving immunosuppression therapy who received the Pfizer-BioNTech vaccine served as the control group.
Four weeks after the study participants received their first dose of the vaccine, 78% of the immunosuppressed patients underwent seroconversion – producing measurable antibodies – as did 100% of the control group. Patients taking methotrexate had the lowest seroconversion rate at 47%, compared with 79% with TNF inhibitors, 83% with IL-23 inhibitors, and 100% with IL-17 inhibitors.
Participants taking methotrexate also had lower neutralizing activity against SARS-CoV-2 than control subjects and those taking a targeted biologic, who had similar levels of neutralizing activity.
All participants had low neutralizing titers against the alpha (B.1.1.7) variant.
The researchers also assessed cellular immunity, “defined as the presence of T cells secreting interferon-gamma, IL-2, or IL-21 in response to stimulation with two peptide pools spanning the entire length of the SARS-CoV-2 spike glycoprotein.”
A T-cell response was seen in 84% of participants taking immunosuppressants, including 93% of those in the methotrexate group and 69% of control subjects.
‘Some protection is better than none’
These findings regarding antibodies match what has been seen in other research, said Ignacio Sanz, MD, director of the Lowance Center for Human Immunology at Emory University, Atlanta.
It would be helpful to see antibody responses after the second doses, he added. Those data will be reported later, according to Dr. Mahil and colleagues.
“The authors make the valid point that T-cell immunity should also be measured. The information is meaningful and supports the idea that there could be protection still provided,” Dr. Sanz said in an interview, adding that it would have been helpful to see CD8 T-cell response as well.
“My message to patients, still, is that some protection is better than none, and that, indeed, protection may be afforded in different ways, including T-cell immunity, which, to the extent tested, seems to be induced,” he said. But discussion of B cells independent of their role in producing antibodies is missing.
“When it comes to B-cell responses, antibodies are the easier and more direct measurement. However, it is perfectly possible that the vaccine may fail to induce high antibody titers and still generate good B-cell immunity,” in the same way virus-specific memory B cells do, he explained. “They would not directly produce antibodies, yet they would be available for a good and quick response in the case of subsequent encounter with the virus and, incidentally, in the case of a booster dose. It is possible that the generation of antibody-producing plasma cells might be uncoupled from the generation of memory B cells.”
Temporarily stopping methotrexate
It is well known that methotrexate impairs humoral responses to influenza and pneumococcal vaccines, write Caoilfhionn M. Connolly, MD, and Julie J. Paik, MD, both from the Johns Hopkins University, Baltimore, in an accompanying comment.
Research has also shown that temporarily stopping methotrexate therapy for 2 weeks enhances response to the flu vaccine in patients with rheumatoid arthritis, which prompted the American College of Rheumatology to recommended temporary interruption of methotrexate for 1 week after each dose of the COVID-19 vaccine, the pair notes.
“Although it is encouraging that cellular responses appear to be preserved even in patients with poor humoral responses, these findings are not consistent across study groups,” Dr. Connolly and Dr. Paik explained. “During this period of clinical uncertainty, patients might remain vulnerable, especially after the first dose, and should engage in risk mitigation strategies.”
Mild adverse events after vaccination were reported by 75% of the immunosuppressed patients – most commonly injection-site pain, headache, and fatigue – and by 94% of control subjects. No participants reported moderate or severe adverse effects.
However, 11% of immunosuppressed patients reported a worsening of psoriasis symptoms after vaccination.
This research was funded by the U.K. National Institute for Health Research. Dr. Mahil has received departmental income from AbbVie, Celgene, Eli Lilly, Janssen-Cilag, Novartis, Sano, and UCB unrelated to this study. Seven other authors have relationships with a wide range of pharmaceutical and other companies. Dr. Sanz, Dr. Connolly, and Dr. Paik disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Standard medical mask can protect wearer from aerosols
A standard medical face mask is more effective at preventing the wearer from inhaling aerosols without causing substantial breathing resistance than various cloth, medical, or respirator masks, new research shows.
“Medical face masks with good filtration efficacies can provide even better protective effects than KN95 respirators,” Christian Sterr, MD, from Philipps University of Marburg (Germany), and colleagues wrote. “FFP2 respirators, on the other hand, could be useful in high-risk situations but require greater breathing effort and therefore physical stress for users.”
Extensive evidence has shown that face masks are an excellent form of source control, preventing infectious people from spreading the SARS-CoV-2 virus into the environment. But evidence has been less clear about how well masks protect the wearer from inhaling particles containing the virus.
The researchers conducted three experiments to test 32 different face masks. The findings were presented at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published online in PLOS One .
First they tested pressure drop, which “relates to how easily air can pass through the material,” said Chris Cappa, PhD, professor of civil and environmental engineering at the University of California, Davis, who was not involved with the study.
“Higher pressure drops mean that there is greater resistance to the air passing through. A higher pressure drop will typically mean breathing through the material will be slightly more challenging, compared to a low pressure drop. There is no relationship between pressure drop and the mask effectiveness,” he said in an interview.
Pressure drop was lowest with type II medical face masks, the typical three-ply surgical masks designed to stop large particles expelled by the wearer from entering the environment, was highest with respirators, including KN95 and FFP2 masks, and varied with the different cloth masks tested.
Next the researchers compared filtration efficacy, which “refers to how well the material removes particles from the air that passes through the mask material,” Dr. Cappa explained. They did this by placing each mask over the opening to an air collector that measured how many particles got through. “A mask that has 100% filtration efficacy will remove all particles from the air that passes through it and 0% means that no particles are removed.”
Cloth masks had the lowest filtration efficacy, at 28%. Certified face masks that met European Standards had a relatively high efficacy, at 70%; for uncertified face masks, filtration efficacy was 63%. As expected, KN95 and FFP2 masks had the highest filtration efficacy, at 94% and 98%, respectively.Finally, the researchers tested as-worn filtration efficacies. They placed each mask on a dummy head with an artificial airway that collected airborne particles. They then pumped a mixture of aerosol particles – ranging in size from 0.3 to 2.0 mcm – and particle-free pressurized air into the air-proof acrylic chamber in which the head was placed.
In this experiment, cloth masks and noncertified face masks were least effective, filtering less than 20% of aerosols. Interestingly, the cloth face mask with the highest filtration on its own (84%) had the lowest filtration efficacy (9%), apparently because of its very high pressure drop (breathing resistance). When more effort is required to breathe through a mask, more air can bypass the filtration system.
Type II medical face masks, however, filtered 47% of aerosols, KN95 masks filtered 41%, and FFP2 masks filtered 65%. Face shields did not prevent the inhalation of any aerosols.
“We know that face shields will only be effective in stopping very large droplets, essentially visible spittle,” Dr. Cappa explained. “Most of the particles that we exhale will travel right around a face shield.”
The “optimal mask effect is a combination of high filter performance and low filter resistance,” which applies to most of the FFP2 and medical type II face masks tested, Dr. Sterr and colleagues wrote. “The type II medical masks in our random sample showed very good as-worn filtration performances with a low additional work of breathing at the same time.”
Although this study showed how well different masks filtered out particles, it could not assess how well different masks prevent actual infection.
“Like any virus, SARS-CoV-2 can only infect people as long as it is viable,” the researchers wrote. “Moreover, a certain number of viable virus particles need to be inhaled to trigger an infection. Thus, the assessed filtration efficacy may differ from the provided protection rate against SARS-CoV-2.”
In addition, particles containing the virus could dry out while going through the mask and become less infectious. “Even a small reduction in inhaled particles might prevent infection or at least lead to a less severe infection,” they noted.
In fact, filtration efficacy does not necessarily indicate how well the mask filters out particles while being worn. “This might be due to the combined effects of mask fit and pressure drop of the mask material and therefore tendency for mask leakage,” the team wrote. “High pressure drop results in higher breathing resistance and therefore supports leakage, especially if combined to a loosely fitting mask.”
These findings are “in line with what we already knew,” Dr. Cappa explained. “Even if the mask material filters out nearly all particles that pass through it, as is the case for high-efficiency masks such as N95 and FFP2, if the mask does not fit well, then it will only provide moderate protection for the wearer.”
Although the findings reaffirm the different levels of filtration provided by various cloth masks, they do not “provide any guidance on which types of cloth masks are better or worse,” he said. But they do show that “medical face masks will generally provide more protection to the wearer.”
It’s not surprising that face shields offer little protection from aerosols, Dr. Cappa said, but they can provide added protection when worn with a mask.
“A face shield could prevent large droplets that might shoot out when a person coughs or sneezes from depositing on a person’s eye,” he pointed out. And it can help “redirect the plume of particles that an infected person exhales, which could be useful in close quarters. However, even then those particles will keep moving around and could be inhaled. A mask can really help to decrease the amount inhaled.”
The study did not use external funding. The authors and Dr. Cappa disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A standard medical face mask is more effective at preventing the wearer from inhaling aerosols without causing substantial breathing resistance than various cloth, medical, or respirator masks, new research shows.
“Medical face masks with good filtration efficacies can provide even better protective effects than KN95 respirators,” Christian Sterr, MD, from Philipps University of Marburg (Germany), and colleagues wrote. “FFP2 respirators, on the other hand, could be useful in high-risk situations but require greater breathing effort and therefore physical stress for users.”
Extensive evidence has shown that face masks are an excellent form of source control, preventing infectious people from spreading the SARS-CoV-2 virus into the environment. But evidence has been less clear about how well masks protect the wearer from inhaling particles containing the virus.
The researchers conducted three experiments to test 32 different face masks. The findings were presented at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published online in PLOS One .
First they tested pressure drop, which “relates to how easily air can pass through the material,” said Chris Cappa, PhD, professor of civil and environmental engineering at the University of California, Davis, who was not involved with the study.
“Higher pressure drops mean that there is greater resistance to the air passing through. A higher pressure drop will typically mean breathing through the material will be slightly more challenging, compared to a low pressure drop. There is no relationship between pressure drop and the mask effectiveness,” he said in an interview.
Pressure drop was lowest with type II medical face masks, the typical three-ply surgical masks designed to stop large particles expelled by the wearer from entering the environment, was highest with respirators, including KN95 and FFP2 masks, and varied with the different cloth masks tested.
Next the researchers compared filtration efficacy, which “refers to how well the material removes particles from the air that passes through the mask material,” Dr. Cappa explained. They did this by placing each mask over the opening to an air collector that measured how many particles got through. “A mask that has 100% filtration efficacy will remove all particles from the air that passes through it and 0% means that no particles are removed.”
Cloth masks had the lowest filtration efficacy, at 28%. Certified face masks that met European Standards had a relatively high efficacy, at 70%; for uncertified face masks, filtration efficacy was 63%. As expected, KN95 and FFP2 masks had the highest filtration efficacy, at 94% and 98%, respectively.Finally, the researchers tested as-worn filtration efficacies. They placed each mask on a dummy head with an artificial airway that collected airborne particles. They then pumped a mixture of aerosol particles – ranging in size from 0.3 to 2.0 mcm – and particle-free pressurized air into the air-proof acrylic chamber in which the head was placed.
In this experiment, cloth masks and noncertified face masks were least effective, filtering less than 20% of aerosols. Interestingly, the cloth face mask with the highest filtration on its own (84%) had the lowest filtration efficacy (9%), apparently because of its very high pressure drop (breathing resistance). When more effort is required to breathe through a mask, more air can bypass the filtration system.
Type II medical face masks, however, filtered 47% of aerosols, KN95 masks filtered 41%, and FFP2 masks filtered 65%. Face shields did not prevent the inhalation of any aerosols.
“We know that face shields will only be effective in stopping very large droplets, essentially visible spittle,” Dr. Cappa explained. “Most of the particles that we exhale will travel right around a face shield.”
The “optimal mask effect is a combination of high filter performance and low filter resistance,” which applies to most of the FFP2 and medical type II face masks tested, Dr. Sterr and colleagues wrote. “The type II medical masks in our random sample showed very good as-worn filtration performances with a low additional work of breathing at the same time.”
Although this study showed how well different masks filtered out particles, it could not assess how well different masks prevent actual infection.
“Like any virus, SARS-CoV-2 can only infect people as long as it is viable,” the researchers wrote. “Moreover, a certain number of viable virus particles need to be inhaled to trigger an infection. Thus, the assessed filtration efficacy may differ from the provided protection rate against SARS-CoV-2.”
In addition, particles containing the virus could dry out while going through the mask and become less infectious. “Even a small reduction in inhaled particles might prevent infection or at least lead to a less severe infection,” they noted.
In fact, filtration efficacy does not necessarily indicate how well the mask filters out particles while being worn. “This might be due to the combined effects of mask fit and pressure drop of the mask material and therefore tendency for mask leakage,” the team wrote. “High pressure drop results in higher breathing resistance and therefore supports leakage, especially if combined to a loosely fitting mask.”
These findings are “in line with what we already knew,” Dr. Cappa explained. “Even if the mask material filters out nearly all particles that pass through it, as is the case for high-efficiency masks such as N95 and FFP2, if the mask does not fit well, then it will only provide moderate protection for the wearer.”
Although the findings reaffirm the different levels of filtration provided by various cloth masks, they do not “provide any guidance on which types of cloth masks are better or worse,” he said. But they do show that “medical face masks will generally provide more protection to the wearer.”
It’s not surprising that face shields offer little protection from aerosols, Dr. Cappa said, but they can provide added protection when worn with a mask.
“A face shield could prevent large droplets that might shoot out when a person coughs or sneezes from depositing on a person’s eye,” he pointed out. And it can help “redirect the plume of particles that an infected person exhales, which could be useful in close quarters. However, even then those particles will keep moving around and could be inhaled. A mask can really help to decrease the amount inhaled.”
The study did not use external funding. The authors and Dr. Cappa disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A standard medical face mask is more effective at preventing the wearer from inhaling aerosols without causing substantial breathing resistance than various cloth, medical, or respirator masks, new research shows.
“Medical face masks with good filtration efficacies can provide even better protective effects than KN95 respirators,” Christian Sterr, MD, from Philipps University of Marburg (Germany), and colleagues wrote. “FFP2 respirators, on the other hand, could be useful in high-risk situations but require greater breathing effort and therefore physical stress for users.”
Extensive evidence has shown that face masks are an excellent form of source control, preventing infectious people from spreading the SARS-CoV-2 virus into the environment. But evidence has been less clear about how well masks protect the wearer from inhaling particles containing the virus.
The researchers conducted three experiments to test 32 different face masks. The findings were presented at the 31st European Congress of Clinical Microbiology & Infectious Diseases and published online in PLOS One .
First they tested pressure drop, which “relates to how easily air can pass through the material,” said Chris Cappa, PhD, professor of civil and environmental engineering at the University of California, Davis, who was not involved with the study.
“Higher pressure drops mean that there is greater resistance to the air passing through. A higher pressure drop will typically mean breathing through the material will be slightly more challenging, compared to a low pressure drop. There is no relationship between pressure drop and the mask effectiveness,” he said in an interview.
Pressure drop was lowest with type II medical face masks, the typical three-ply surgical masks designed to stop large particles expelled by the wearer from entering the environment, was highest with respirators, including KN95 and FFP2 masks, and varied with the different cloth masks tested.
Next the researchers compared filtration efficacy, which “refers to how well the material removes particles from the air that passes through the mask material,” Dr. Cappa explained. They did this by placing each mask over the opening to an air collector that measured how many particles got through. “A mask that has 100% filtration efficacy will remove all particles from the air that passes through it and 0% means that no particles are removed.”
Cloth masks had the lowest filtration efficacy, at 28%. Certified face masks that met European Standards had a relatively high efficacy, at 70%; for uncertified face masks, filtration efficacy was 63%. As expected, KN95 and FFP2 masks had the highest filtration efficacy, at 94% and 98%, respectively.Finally, the researchers tested as-worn filtration efficacies. They placed each mask on a dummy head with an artificial airway that collected airborne particles. They then pumped a mixture of aerosol particles – ranging in size from 0.3 to 2.0 mcm – and particle-free pressurized air into the air-proof acrylic chamber in which the head was placed.
In this experiment, cloth masks and noncertified face masks were least effective, filtering less than 20% of aerosols. Interestingly, the cloth face mask with the highest filtration on its own (84%) had the lowest filtration efficacy (9%), apparently because of its very high pressure drop (breathing resistance). When more effort is required to breathe through a mask, more air can bypass the filtration system.
Type II medical face masks, however, filtered 47% of aerosols, KN95 masks filtered 41%, and FFP2 masks filtered 65%. Face shields did not prevent the inhalation of any aerosols.
“We know that face shields will only be effective in stopping very large droplets, essentially visible spittle,” Dr. Cappa explained. “Most of the particles that we exhale will travel right around a face shield.”
The “optimal mask effect is a combination of high filter performance and low filter resistance,” which applies to most of the FFP2 and medical type II face masks tested, Dr. Sterr and colleagues wrote. “The type II medical masks in our random sample showed very good as-worn filtration performances with a low additional work of breathing at the same time.”
Although this study showed how well different masks filtered out particles, it could not assess how well different masks prevent actual infection.
“Like any virus, SARS-CoV-2 can only infect people as long as it is viable,” the researchers wrote. “Moreover, a certain number of viable virus particles need to be inhaled to trigger an infection. Thus, the assessed filtration efficacy may differ from the provided protection rate against SARS-CoV-2.”
In addition, particles containing the virus could dry out while going through the mask and become less infectious. “Even a small reduction in inhaled particles might prevent infection or at least lead to a less severe infection,” they noted.
In fact, filtration efficacy does not necessarily indicate how well the mask filters out particles while being worn. “This might be due to the combined effects of mask fit and pressure drop of the mask material and therefore tendency for mask leakage,” the team wrote. “High pressure drop results in higher breathing resistance and therefore supports leakage, especially if combined to a loosely fitting mask.”
These findings are “in line with what we already knew,” Dr. Cappa explained. “Even if the mask material filters out nearly all particles that pass through it, as is the case for high-efficiency masks such as N95 and FFP2, if the mask does not fit well, then it will only provide moderate protection for the wearer.”
Although the findings reaffirm the different levels of filtration provided by various cloth masks, they do not “provide any guidance on which types of cloth masks are better or worse,” he said. But they do show that “medical face masks will generally provide more protection to the wearer.”
It’s not surprising that face shields offer little protection from aerosols, Dr. Cappa said, but they can provide added protection when worn with a mask.
“A face shield could prevent large droplets that might shoot out when a person coughs or sneezes from depositing on a person’s eye,” he pointed out. And it can help “redirect the plume of particles that an infected person exhales, which could be useful in close quarters. However, even then those particles will keep moving around and could be inhaled. A mask can really help to decrease the amount inhaled.”
The study did not use external funding. The authors and Dr. Cappa disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.