User login
Facial Temperature Can Reveal Age and Disease
This transcript has been edited for clarity.
My oldest daughter is at sleepaway camp for a couple of weeks, and the camp has a photographer who goes around all day taking pictures of the kids, which get uploaded to a private Facebook group. In the past, I would go online every day (or, okay, several times a day) and scroll through all those pictures looking for one that features my kid.
I don’t have to do that anymore. This year, I simply uploaded a picture of my daughter to an app and artificial intelligence (AI) takes care of the rest, recognizing her face amidst the sea of smiling children, and flagging just those photos for me to peruse. It’s amazing, really. And a bit scary.
The fact that facial recognition has penetrated the summer camp market should tell you that the tech is truly ubiquitous. But today we’re going to think a bit more about what AI can do with a picture of your face, because the power of facial recognition is not just skin deep.
What’s got me hot and bothered about facial images is this paper, appearing in Cell Metabolism, which adds a new layer to the standard facial-analysis playbook: facial temperature.
To understand this paper, you need to understand a whole field of research that is developing various different “clocks” for age.
It turns out that age really is just a number. Our cells, our proteins, our biochemistry can be analyzed to give different numbers. These “clocks,” as distinct from the calendar we usually use to measure our age, might have more predictive power than the number itself.
There are numerous molecular clocks, such as telomere length, that not only correlate with calendar age but are superior to calendar age in predicting age-related complications. Testing telomere length typically requires a blood sample — and remains costly. But we can use other sources to estimate age; how about a photo?
I mean, we do this all the time when we meet someone new or, as a physician, when we meet a new patient. I have often written that a patient “appears younger than their stated age,” and we’ve all had the experience of hearing how old someone is and being shocked. I mean, have you seen Sharon Stone recently? She’s 66 years old. Okay — to be fair, there might be some outside help there. But you get the point.
Back to the Cell Metabolism paper. Researchers report on multiple algorithms to obtain an “age” from a picture of an individual’s face.
The first algorithm is pretty straightforward. Researchers collected 2811 images, all of Han Chinese individuals ranging in age from 20 to 90 years, and reconstructed a 3D facial map from those. 
They then trained a convolutional neural network to predict the individuals’ ages from the pictures. It was quite accurate, as you can see here.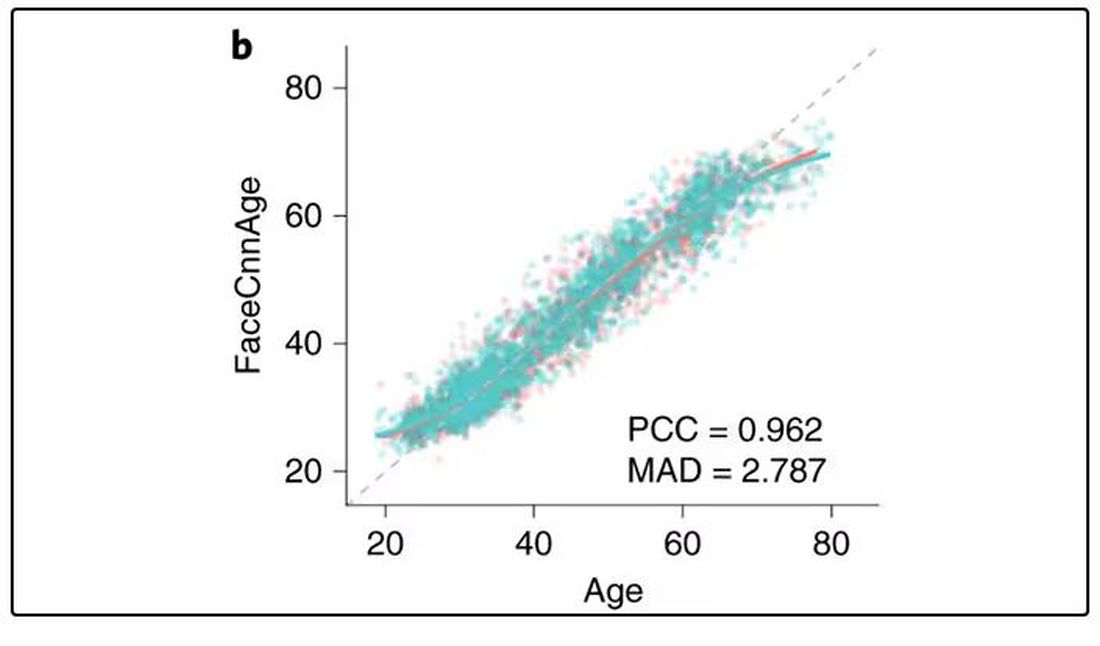
In the AI age, this may not seem that impressive. A brief search online turned up dozens of apps that promised to guess my age from a photo.
I sent this rather unflattering picture of myself to ChatGPT which, after initially demurring and saying it was not designed to guess ages, pegged me at somewhere between 35 and 45, which I am taking as a major victory.
But the Cell Metabolism paper goes deeper. Literally. 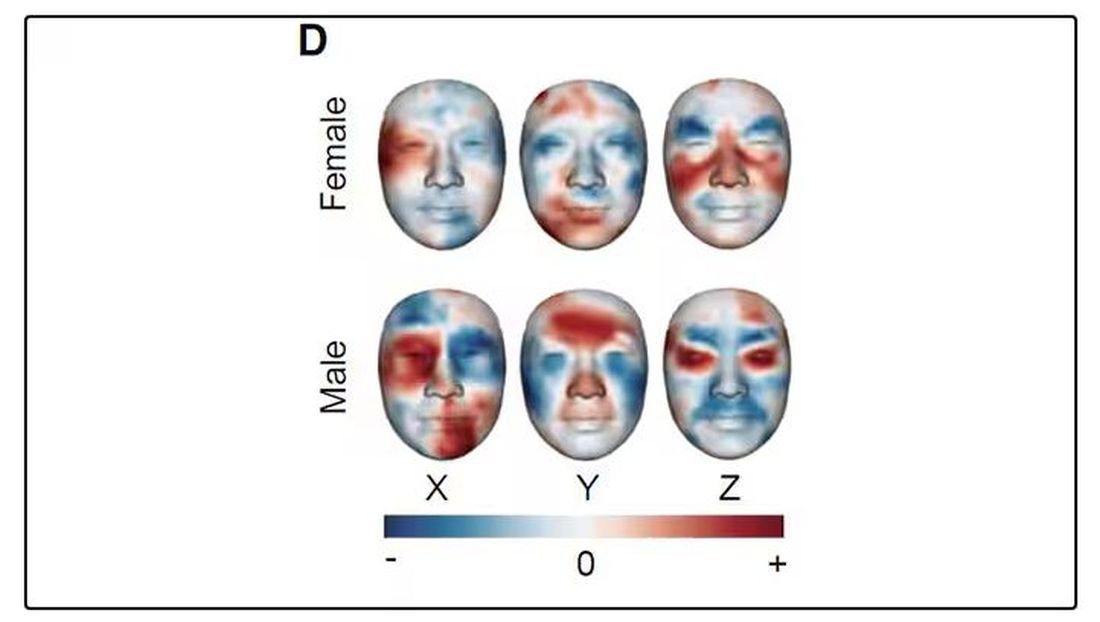
And this is where things start to get interesting. Because sure, the visible part of your face can change depending on makeup, expression, plastic surgery, and the like. But the temperature? That’s harder to fake.
It turns out that the temperature distribution in your face changes as you get older. There is a cooling of the nose and the cheeks, for example.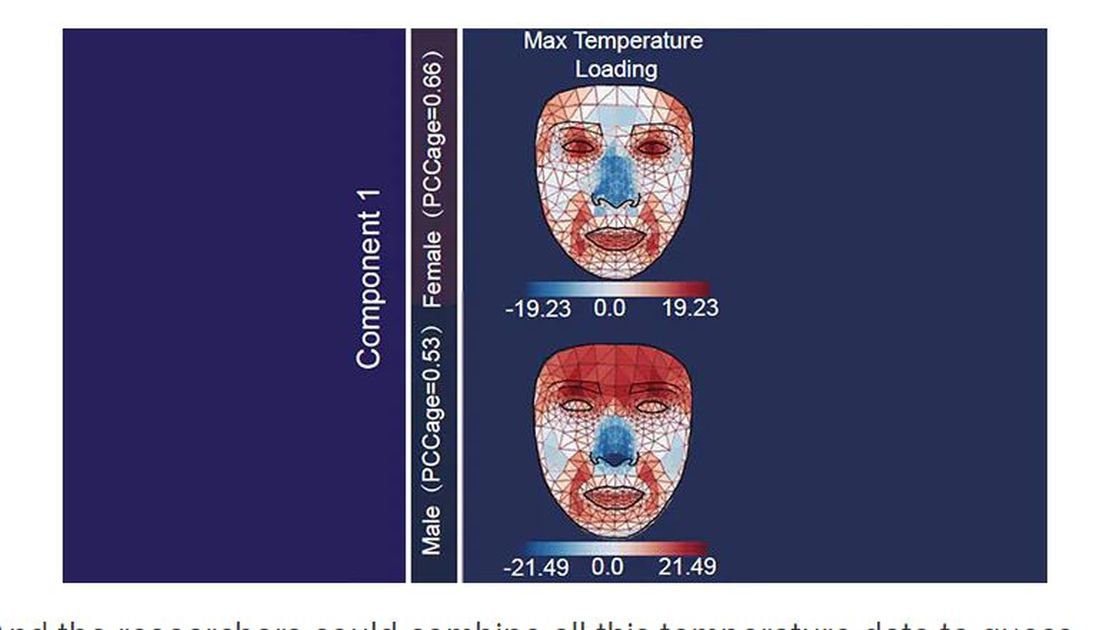
And the researchers could combine all this temperature data to guess someone’s calendar age fairly accurately, though notably not as accurately as the model that just looks at the pictures.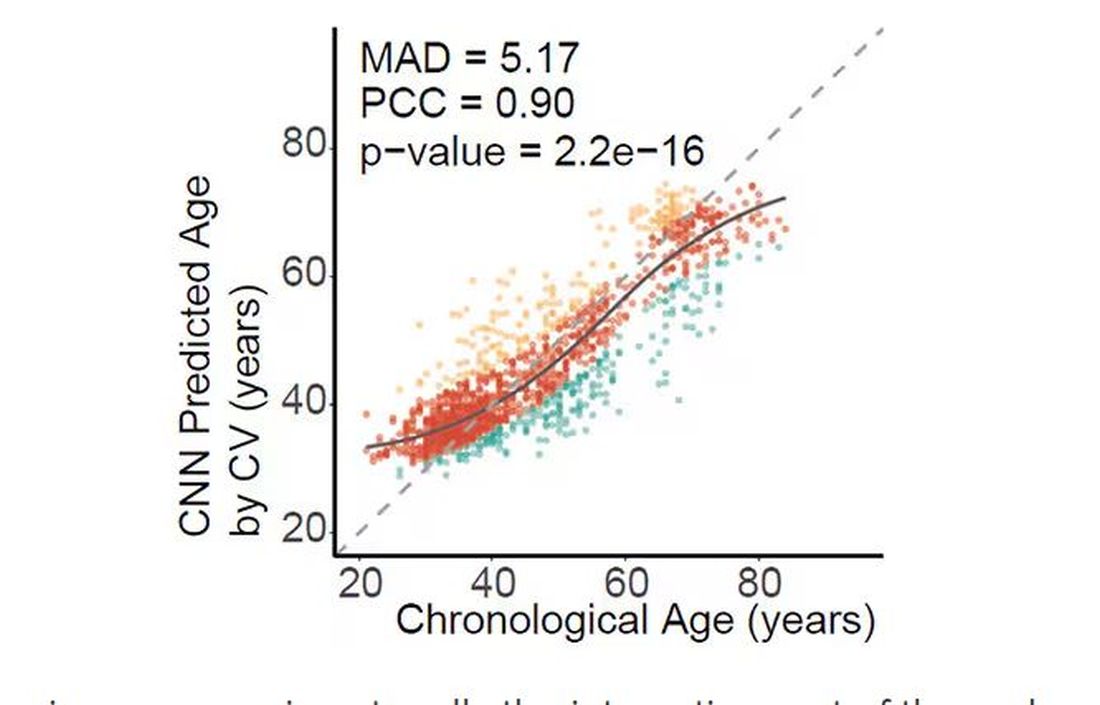
But guessing your age is not really the interesting part of thermal imaging of the face. It’s guessing — or, rather, predicting — the state of your metabolism. All these study participants had extensive metabolic testing performed, as well as detailed analysis of their lifestyle behaviors. And facial images could be used to predict those factors.
For example, the 3D reconstruction of the faces could predict who ate seafood (they tend to look younger than their actual age) compared with who ate poultry and meat (they tend to look older). The thermal imaging could predict who got more sleep (they look younger from a temperature perspective) and who ate more yogurt (also younger-appearing, temperature-wise). Facial temperature patterns could identify those with higher BMI, higher blood pressure, higher fasting glucose.
The researchers used the difference between actual and predicted age as a metric to measure illness as well. You can see here how, on average, individuals with hypertension, diabetes, and even liver cysts are “older,” at least by face temperature.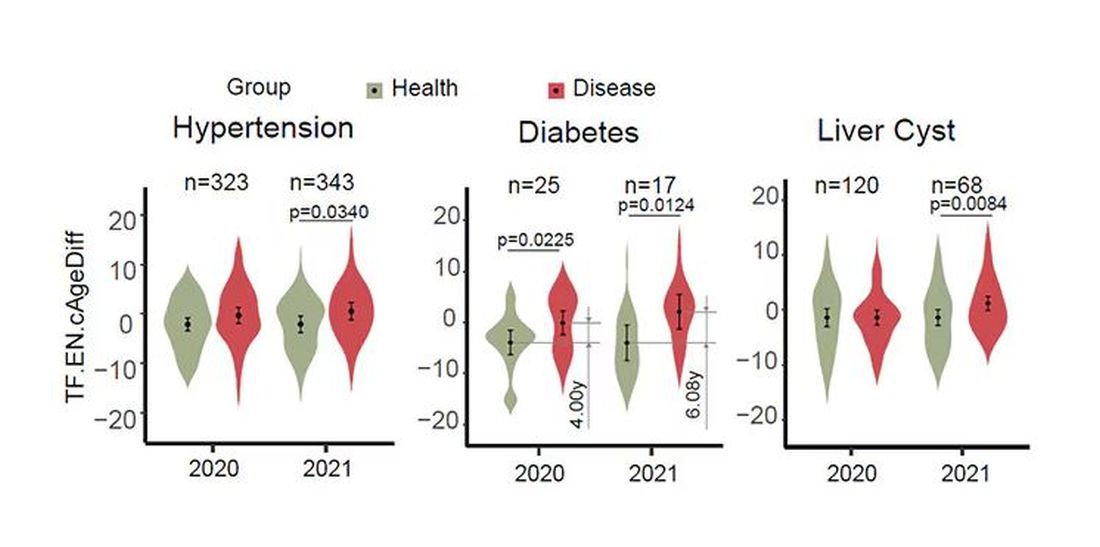
It may even be possible to use facial temperature as biofeedback. In a small study, the researchers measured the difference between facial temperature age and real age before and after 2 weeks of jump-roping. It turns out that 2 weeks of jump-roping can make you look about 5 years younger, at least as judged by a thermal camera. Or like the Predator.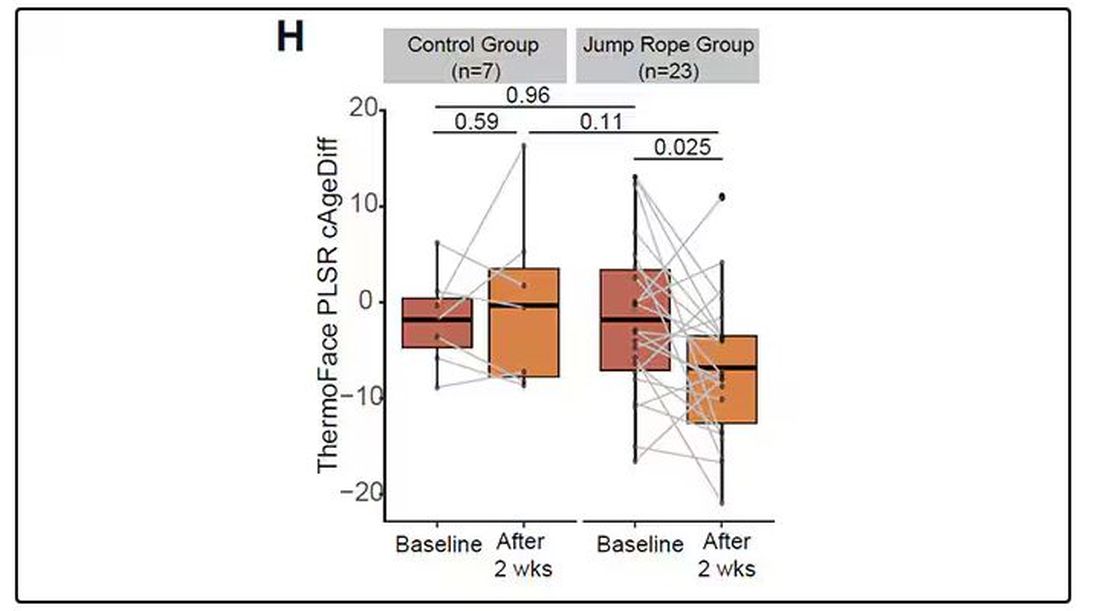
Okay, this is all very cool, but I’m not saying we’ll all be doing facial temperature tests in the near future. No; what this study highlights for me is how much information about ourselves is available to those who know how to decode it. Maybe those data come from the wrinkles in our faces, or the angles of our smiles, or the speed with which we type, or the temperature of our elbows. The data have always been there, actually, but we’ve never had the tools powerful enough to analyze them until now.
When I was a kid, I was obsessed with Star Trek — I know, you’re shocked — and, of course, the famous tricorder, a scanner that could tell everything about someone’s state of health in 5 seconds from 3 feet away. That’s how I thought medicine really would be in the future. Once I got to medical school, I was disabused of that notion. But the age of data, the age of AI, may mean the tricorder age is not actually that far away.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
My oldest daughter is at sleepaway camp for a couple of weeks, and the camp has a photographer who goes around all day taking pictures of the kids, which get uploaded to a private Facebook group. In the past, I would go online every day (or, okay, several times a day) and scroll through all those pictures looking for one that features my kid.
I don’t have to do that anymore. This year, I simply uploaded a picture of my daughter to an app and artificial intelligence (AI) takes care of the rest, recognizing her face amidst the sea of smiling children, and flagging just those photos for me to peruse. It’s amazing, really. And a bit scary.
The fact that facial recognition has penetrated the summer camp market should tell you that the tech is truly ubiquitous. But today we’re going to think a bit more about what AI can do with a picture of your face, because the power of facial recognition is not just skin deep.
What’s got me hot and bothered about facial images is this paper, appearing in Cell Metabolism, which adds a new layer to the standard facial-analysis playbook: facial temperature.
To understand this paper, you need to understand a whole field of research that is developing various different “clocks” for age.
It turns out that age really is just a number. Our cells, our proteins, our biochemistry can be analyzed to give different numbers. These “clocks,” as distinct from the calendar we usually use to measure our age, might have more predictive power than the number itself.
There are numerous molecular clocks, such as telomere length, that not only correlate with calendar age but are superior to calendar age in predicting age-related complications. Testing telomere length typically requires a blood sample — and remains costly. But we can use other sources to estimate age; how about a photo?
I mean, we do this all the time when we meet someone new or, as a physician, when we meet a new patient. I have often written that a patient “appears younger than their stated age,” and we’ve all had the experience of hearing how old someone is and being shocked. I mean, have you seen Sharon Stone recently? She’s 66 years old. Okay — to be fair, there might be some outside help there. But you get the point.
Back to the Cell Metabolism paper. Researchers report on multiple algorithms to obtain an “age” from a picture of an individual’s face.
The first algorithm is pretty straightforward. Researchers collected 2811 images, all of Han Chinese individuals ranging in age from 20 to 90 years, and reconstructed a 3D facial map from those. 
They then trained a convolutional neural network to predict the individuals’ ages from the pictures. It was quite accurate, as you can see here.
In the AI age, this may not seem that impressive. A brief search online turned up dozens of apps that promised to guess my age from a photo.
I sent this rather unflattering picture of myself to ChatGPT which, after initially demurring and saying it was not designed to guess ages, pegged me at somewhere between 35 and 45, which I am taking as a major victory.
But the Cell Metabolism paper goes deeper. Literally. 
And this is where things start to get interesting. Because sure, the visible part of your face can change depending on makeup, expression, plastic surgery, and the like. But the temperature? That’s harder to fake.
It turns out that the temperature distribution in your face changes as you get older. There is a cooling of the nose and the cheeks, for example.
And the researchers could combine all this temperature data to guess someone’s calendar age fairly accurately, though notably not as accurately as the model that just looks at the pictures.
But guessing your age is not really the interesting part of thermal imaging of the face. It’s guessing — or, rather, predicting — the state of your metabolism. All these study participants had extensive metabolic testing performed, as well as detailed analysis of their lifestyle behaviors. And facial images could be used to predict those factors.
For example, the 3D reconstruction of the faces could predict who ate seafood (they tend to look younger than their actual age) compared with who ate poultry and meat (they tend to look older). The thermal imaging could predict who got more sleep (they look younger from a temperature perspective) and who ate more yogurt (also younger-appearing, temperature-wise). Facial temperature patterns could identify those with higher BMI, higher blood pressure, higher fasting glucose.
The researchers used the difference between actual and predicted age as a metric to measure illness as well. You can see here how, on average, individuals with hypertension, diabetes, and even liver cysts are “older,” at least by face temperature.
It may even be possible to use facial temperature as biofeedback. In a small study, the researchers measured the difference between facial temperature age and real age before and after 2 weeks of jump-roping. It turns out that 2 weeks of jump-roping can make you look about 5 years younger, at least as judged by a thermal camera. Or like the Predator.
Okay, this is all very cool, but I’m not saying we’ll all be doing facial temperature tests in the near future. No; what this study highlights for me is how much information about ourselves is available to those who know how to decode it. Maybe those data come from the wrinkles in our faces, or the angles of our smiles, or the speed with which we type, or the temperature of our elbows. The data have always been there, actually, but we’ve never had the tools powerful enough to analyze them until now.
When I was a kid, I was obsessed with Star Trek — I know, you’re shocked — and, of course, the famous tricorder, a scanner that could tell everything about someone’s state of health in 5 seconds from 3 feet away. That’s how I thought medicine really would be in the future. Once I got to medical school, I was disabused of that notion. But the age of data, the age of AI, may mean the tricorder age is not actually that far away.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
My oldest daughter is at sleepaway camp for a couple of weeks, and the camp has a photographer who goes around all day taking pictures of the kids, which get uploaded to a private Facebook group. In the past, I would go online every day (or, okay, several times a day) and scroll through all those pictures looking for one that features my kid.
I don’t have to do that anymore. This year, I simply uploaded a picture of my daughter to an app and artificial intelligence (AI) takes care of the rest, recognizing her face amidst the sea of smiling children, and flagging just those photos for me to peruse. It’s amazing, really. And a bit scary.
The fact that facial recognition has penetrated the summer camp market should tell you that the tech is truly ubiquitous. But today we’re going to think a bit more about what AI can do with a picture of your face, because the power of facial recognition is not just skin deep.
What’s got me hot and bothered about facial images is this paper, appearing in Cell Metabolism, which adds a new layer to the standard facial-analysis playbook: facial temperature.
To understand this paper, you need to understand a whole field of research that is developing various different “clocks” for age.
It turns out that age really is just a number. Our cells, our proteins, our biochemistry can be analyzed to give different numbers. These “clocks,” as distinct from the calendar we usually use to measure our age, might have more predictive power than the number itself.
There are numerous molecular clocks, such as telomere length, that not only correlate with calendar age but are superior to calendar age in predicting age-related complications. Testing telomere length typically requires a blood sample — and remains costly. But we can use other sources to estimate age; how about a photo?
I mean, we do this all the time when we meet someone new or, as a physician, when we meet a new patient. I have often written that a patient “appears younger than their stated age,” and we’ve all had the experience of hearing how old someone is and being shocked. I mean, have you seen Sharon Stone recently? She’s 66 years old. Okay — to be fair, there might be some outside help there. But you get the point.
Back to the Cell Metabolism paper. Researchers report on multiple algorithms to obtain an “age” from a picture of an individual’s face.
The first algorithm is pretty straightforward. Researchers collected 2811 images, all of Han Chinese individuals ranging in age from 20 to 90 years, and reconstructed a 3D facial map from those. 
They then trained a convolutional neural network to predict the individuals’ ages from the pictures. It was quite accurate, as you can see here.
In the AI age, this may not seem that impressive. A brief search online turned up dozens of apps that promised to guess my age from a photo.
I sent this rather unflattering picture of myself to ChatGPT which, after initially demurring and saying it was not designed to guess ages, pegged me at somewhere between 35 and 45, which I am taking as a major victory.
But the Cell Metabolism paper goes deeper. Literally. 
And this is where things start to get interesting. Because sure, the visible part of your face can change depending on makeup, expression, plastic surgery, and the like. But the temperature? That’s harder to fake.
It turns out that the temperature distribution in your face changes as you get older. There is a cooling of the nose and the cheeks, for example.
And the researchers could combine all this temperature data to guess someone’s calendar age fairly accurately, though notably not as accurately as the model that just looks at the pictures.
But guessing your age is not really the interesting part of thermal imaging of the face. It’s guessing — or, rather, predicting — the state of your metabolism. All these study participants had extensive metabolic testing performed, as well as detailed analysis of their lifestyle behaviors. And facial images could be used to predict those factors.
For example, the 3D reconstruction of the faces could predict who ate seafood (they tend to look younger than their actual age) compared with who ate poultry and meat (they tend to look older). The thermal imaging could predict who got more sleep (they look younger from a temperature perspective) and who ate more yogurt (also younger-appearing, temperature-wise). Facial temperature patterns could identify those with higher BMI, higher blood pressure, higher fasting glucose.
The researchers used the difference between actual and predicted age as a metric to measure illness as well. You can see here how, on average, individuals with hypertension, diabetes, and even liver cysts are “older,” at least by face temperature.
It may even be possible to use facial temperature as biofeedback. In a small study, the researchers measured the difference between facial temperature age and real age before and after 2 weeks of jump-roping. It turns out that 2 weeks of jump-roping can make you look about 5 years younger, at least as judged by a thermal camera. Or like the Predator.
Okay, this is all very cool, but I’m not saying we’ll all be doing facial temperature tests in the near future. No; what this study highlights for me is how much information about ourselves is available to those who know how to decode it. Maybe those data come from the wrinkles in our faces, or the angles of our smiles, or the speed with which we type, or the temperature of our elbows. The data have always been there, actually, but we’ve never had the tools powerful enough to analyze them until now.
When I was a kid, I was obsessed with Star Trek — I know, you’re shocked — and, of course, the famous tricorder, a scanner that could tell everything about someone’s state of health in 5 seconds from 3 feet away. That’s how I thought medicine really would be in the future. Once I got to medical school, I was disabused of that notion. But the age of data, the age of AI, may mean the tricorder age is not actually that far away.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
What Should Be Prioritized in Managing Early Diabetes?
ORLANDO, FLORIDA — What to prioritize first in managing early diabetes? That was the question debated on an expert panel at the American Diabetes Association (ADA) 84th Scientific Sessions, with impassioned responses ranging from a plea to “treat obesity first,” to a James Carville–inspired counterpoint of “it’s the glucose, stupid.”
With a focus on preventing complications and inducing remission rounding out the four positions argued,
“In clinical decision-making [for early diabetes], we are faced with weighing each of these variables for the individual patient, and while all are good options, strong arguments can be made for prioritizing each — with the potential of each choice to influence or improve all of the others,” Dr. Retnakaran told this news organization.
Which to Prioritize First?
Making the obesity first argument, Ania M. Jastreboff, MD, PhD, associate professor and director of the Yale Obesity Research Center at Yale School of Medicine, New Haven, Connecticut, noted the striking statistic that nearly 90% of people with type 2 diabetes have overweight or obesity and discussed the ever-expanding data showing the benefits of drugs including glucagon-like peptide 1 (GLP-1) receptor agonists not just in weight loss but also in kidney, cardiovascular, and, as presented at the meeting, sleep apnea improvement.
She contrasted the experiences of two patients with obesity: One treated for the obesity upon type 2 diagnosis — who had a quick normalization of lipids and hypertension soon after the obesity treatment — and the other presenting after 10 years with type 2 diabetes — who was on therapy for hypertension and hyperlipidemia but not for obesity and whose diseases were not as easily treated by that point.
“Why are we treating all the downstream effects and we’re not treating the disease that is potentially the root cause of all these other diseases?” Dr. Jastreboff said.
Complications?
Arguing in favor of focusing on complications, Roopa Mehta, MD, PhD, with the department of endocrinology and metabolism at Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán (INCMNSZ), Mexico City, made the case that stakes don’t get any higher in diabetes than when it comes the looming threat of potentially fatal complications.
Acute myocardial infarction, stroke, amputation, and end-stage renal disease are all on the list of unwanted outcomes and need to be considered even in the earliest stages, as data show early onset type 2 diabetes is linked to life expectancy.
“The main goal of management has always been to prevent complications,” she noted. Citing ADA guidelines, Dr. Mehta underscored the benefits of first- and second-line therapy of metformin, sodium-glucose cotransporter 2 (SGLT2) inhibitors, and GLP-1 receptor agonists for most patients.
Remission?
Discussing the priority of putting patients into disease remission, Roy Taylor, MD, professor of medicine and metabolism at Newcastle University and Newcastle Hospitals NHS in Newcastle upon Tyne, England, and author of the book Life Without Diabetes, focused on an evidence-based alternative to achieving remission — a nonpharmacologic approach that avoids costly and sometimes inaccessible drugs.
In the intervention, described in the DiRECT randomized trial and subsequently in the UK National Health Service Type 2 Diabetes Path to Remission Program, patients with overweight or obesity were placed on a highly restrictive diet of just 800-900 calories a day for 12-20 weeks, followed by maintenance for 12 months, and they not only achieved weight loss but also achieved diabetes remission, in some cases long term.
Acknowledging that “this is not for everyone,” Dr. Taylor asserted that “we have to realize there is a substantial minority of people who want to be healthy but who don’t want to be medicalized,” he said.
“They want their health, and they can do extremely well.”
Glucose?
In taking his self-titled “it’s the glucose, stupid” stand, David M. Nathan, MD, of the Diabetes Center, Massachusetts General Hospital, Harvard Medical School, in Boston, cited extensive evidence showing that early intensive blood glucose control with treatment including sulfonylureas, insulin, or metformin significantly reduced the risk for complications in type 2 diabetes 15 or more years later, including renal failure, blindness, amputation, and myocardial infarctions, in addition to a reduction in diabetes-related death.
“In many of these studies, you saw the benefit even in the setting of weight-gain,” Dr. Nathan underscored.
He further noted the “sobering” findings of the Look AHEAD study, which had to be stopped due to futility when an intensive lifestyle/weight loss intervention showed no significant benefits in terms of cardiovascular disease in people with type 2 diabetes at a median follow-up of 9.6 years.
Ultimately, “diabetes, type 1 and type 2, remains a gluco-centric disease,” Dr. Nathan asserted. “Hyperglycemia is the only universal link between all forms of diabetes and mortality, and the long-term complications of diabetes are intimately associated with hyperglycemia.”
Tackling the Caveats
The ensuing panel discussion did not fail to deliver in delving into key areas of contention, particularly in terms of GLP-1 treatment.
Regarding a lack of data on the potential long-term effects of GLP-1s: “Yes, there are a huge number of studies [on GLP-1 receptor agonists], but they are, in general, over short periods of time and driven by pharma, who get in and get out as quickly as they can and have little in the way of interest to do comparative effectiveness studies,” Dr. Nathan argued.
“Meanwhile, this is like the crack cocaine of medications — patients have to stay on it for a lifetime or they will regain the weight — are you concerned at all about a lifetime of exposure to GLP-1 [drugs]?” he asked the panel.
Dr. Jastreboff responded that the first GLP-1 receptor agonist medications were approved in 2005, nearly 20 years ago, by the US Food and Drug Administration.
“Do I think we need long-term lifetime data? Absolutely,” she said. “We need to do our due diligence, we need to be careful, we need to monitor patients, and when and if there are signals, we need to follow them.”
What about the notorious gastrointestinal side effects of the drugs? “A majority of them are mitigated by slow up-titration,” Dr. Jastreboff noted.
“If patients have nausea, I do not go up [in dose]. I invite patients to tell me if they’re having vomiting because I don’t want anybody to have it, and I can count on one hand how many of my patients do.”
Dr. Mehta added the concern that as the drugs’ popularity soars, “a lot of doctors don’t know when they need to put the brakes on [weight coming off too quickly].”
She underscored that “we are not treating obesity for weight loss or for cosmetic reasons — this is about optimizing health.”
Dr. Jastreboff noted that in her practice, “I down-titrate if they’re losing weight too quickly.”
“If the patient is losing more than 1% per week of their body weight, then I slow down to make sure they’re getting the nutrients that they need, that they have enough energy to exercise, and that they’re prioritizing protein and fruits and vegetables in their diet.
“We just need to go slow, and yes, we need to follow them long term,” she said.
Chiming in from the audience, Julio Rosenstock, MD, a recognized thought leader in type 2 diabetes, offered his own take on the issues, describing Dr. Taylor’s very low–calorie diet suggestion as “not realistic” and Dr. Nathan’s glucose-first argument to be “stuck in the past.”
Based on modern-day evidence, “there is no reason on earth to start [diabetes treatment] with only metformin,” asserted Dr. Rosenstock, director of the Velocity Clinical Research center at Medical City and clinical professor of medicine at the University of Texas Southwestern Medical Center, Dallas.
“We need to start at the very least with metformin and a sodium-glucose cotransporter 2 (SGLT2) inhibitor from day 1, and then, if it’s affordable and there is access, with a GLP-1 receptor agonist,” he said.
“There is nothing better these days than those agents that consistently have shown a reduction of cardiovascular events and slowing of kidney disease progression.”
Overall, however, “I think you are all right,” he added, a sentiment shared by most.
Noting that the discussion as a whole represents a virtual sea change from the evidence-based options that would have been discussed only a decade ago, Dr. Retnakaran summed up his take-home message: “Stay tuned.
“You could easily see things changing in the next decade to come as we get more data and evidence to support what we ultimately should prioritize an early type 2 diabetes, so this is an exciting time.”
Dr. Retnakaran disclosed ties with Novo Nordisk, Boehringer Ingelheim, Novartis, Sanofi, and Eli Lilly. Dr. Jastreboff disclosed ties with Amgen, AstraZeneca, Boehringer Ingelheim, Biohaven, Eli Lilly, Intellihealth, Novo Nordisk, Pfizer, Regeneron, Scholar Rock, Structure Therapeutics, Terms Pharmaceutical, Weight Watchers, and Zealand Pharmaceuticals. Dr. Roopa had relationships with Novo Nordisk, Boehringer Ingelheim, Amgen, AstraZeneca, Eli Lilly, Silanes, and Sanofi. Dr. Taylor received lecture fees from Novartis, Lilly, Abbott, and Nestle Health and research funding from Diabetes UK and is an advisor to Fast800. Dr. Rosenstock reported relationships with Applied Therapeutics, AstraZeneca, Biomea Fusion, Boehringer Ingelheim, Eli Lilly and Company, Hanmi, Merck, Oramed, Structure Therapeutics, Novartis, Novo Nordisk, Pfizer, Ragor, and Sanofi. Dr. Nathan had no disclosures to report.
A version of this article first appeared on Medscape.com.
ORLANDO, FLORIDA — What to prioritize first in managing early diabetes? That was the question debated on an expert panel at the American Diabetes Association (ADA) 84th Scientific Sessions, with impassioned responses ranging from a plea to “treat obesity first,” to a James Carville–inspired counterpoint of “it’s the glucose, stupid.”
With a focus on preventing complications and inducing remission rounding out the four positions argued,
“In clinical decision-making [for early diabetes], we are faced with weighing each of these variables for the individual patient, and while all are good options, strong arguments can be made for prioritizing each — with the potential of each choice to influence or improve all of the others,” Dr. Retnakaran told this news organization.
Which to Prioritize First?
Making the obesity first argument, Ania M. Jastreboff, MD, PhD, associate professor and director of the Yale Obesity Research Center at Yale School of Medicine, New Haven, Connecticut, noted the striking statistic that nearly 90% of people with type 2 diabetes have overweight or obesity and discussed the ever-expanding data showing the benefits of drugs including glucagon-like peptide 1 (GLP-1) receptor agonists not just in weight loss but also in kidney, cardiovascular, and, as presented at the meeting, sleep apnea improvement.
She contrasted the experiences of two patients with obesity: One treated for the obesity upon type 2 diagnosis — who had a quick normalization of lipids and hypertension soon after the obesity treatment — and the other presenting after 10 years with type 2 diabetes — who was on therapy for hypertension and hyperlipidemia but not for obesity and whose diseases were not as easily treated by that point.
“Why are we treating all the downstream effects and we’re not treating the disease that is potentially the root cause of all these other diseases?” Dr. Jastreboff said.
Complications?
Arguing in favor of focusing on complications, Roopa Mehta, MD, PhD, with the department of endocrinology and metabolism at Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán (INCMNSZ), Mexico City, made the case that stakes don’t get any higher in diabetes than when it comes the looming threat of potentially fatal complications.
Acute myocardial infarction, stroke, amputation, and end-stage renal disease are all on the list of unwanted outcomes and need to be considered even in the earliest stages, as data show early onset type 2 diabetes is linked to life expectancy.
“The main goal of management has always been to prevent complications,” she noted. Citing ADA guidelines, Dr. Mehta underscored the benefits of first- and second-line therapy of metformin, sodium-glucose cotransporter 2 (SGLT2) inhibitors, and GLP-1 receptor agonists for most patients.
Remission?
Discussing the priority of putting patients into disease remission, Roy Taylor, MD, professor of medicine and metabolism at Newcastle University and Newcastle Hospitals NHS in Newcastle upon Tyne, England, and author of the book Life Without Diabetes, focused on an evidence-based alternative to achieving remission — a nonpharmacologic approach that avoids costly and sometimes inaccessible drugs.
In the intervention, described in the DiRECT randomized trial and subsequently in the UK National Health Service Type 2 Diabetes Path to Remission Program, patients with overweight or obesity were placed on a highly restrictive diet of just 800-900 calories a day for 12-20 weeks, followed by maintenance for 12 months, and they not only achieved weight loss but also achieved diabetes remission, in some cases long term.
Acknowledging that “this is not for everyone,” Dr. Taylor asserted that “we have to realize there is a substantial minority of people who want to be healthy but who don’t want to be medicalized,” he said.
“They want their health, and they can do extremely well.”
Glucose?
In taking his self-titled “it’s the glucose, stupid” stand, David M. Nathan, MD, of the Diabetes Center, Massachusetts General Hospital, Harvard Medical School, in Boston, cited extensive evidence showing that early intensive blood glucose control with treatment including sulfonylureas, insulin, or metformin significantly reduced the risk for complications in type 2 diabetes 15 or more years later, including renal failure, blindness, amputation, and myocardial infarctions, in addition to a reduction in diabetes-related death.
“In many of these studies, you saw the benefit even in the setting of weight-gain,” Dr. Nathan underscored.
He further noted the “sobering” findings of the Look AHEAD study, which had to be stopped due to futility when an intensive lifestyle/weight loss intervention showed no significant benefits in terms of cardiovascular disease in people with type 2 diabetes at a median follow-up of 9.6 years.
Ultimately, “diabetes, type 1 and type 2, remains a gluco-centric disease,” Dr. Nathan asserted. “Hyperglycemia is the only universal link between all forms of diabetes and mortality, and the long-term complications of diabetes are intimately associated with hyperglycemia.”
Tackling the Caveats
The ensuing panel discussion did not fail to deliver in delving into key areas of contention, particularly in terms of GLP-1 treatment.
Regarding a lack of data on the potential long-term effects of GLP-1s: “Yes, there are a huge number of studies [on GLP-1 receptor agonists], but they are, in general, over short periods of time and driven by pharma, who get in and get out as quickly as they can and have little in the way of interest to do comparative effectiveness studies,” Dr. Nathan argued.
“Meanwhile, this is like the crack cocaine of medications — patients have to stay on it for a lifetime or they will regain the weight — are you concerned at all about a lifetime of exposure to GLP-1 [drugs]?” he asked the panel.
Dr. Jastreboff responded that the first GLP-1 receptor agonist medications were approved in 2005, nearly 20 years ago, by the US Food and Drug Administration.
“Do I think we need long-term lifetime data? Absolutely,” she said. “We need to do our due diligence, we need to be careful, we need to monitor patients, and when and if there are signals, we need to follow them.”
What about the notorious gastrointestinal side effects of the drugs? “A majority of them are mitigated by slow up-titration,” Dr. Jastreboff noted.
“If patients have nausea, I do not go up [in dose]. I invite patients to tell me if they’re having vomiting because I don’t want anybody to have it, and I can count on one hand how many of my patients do.”
Dr. Mehta added the concern that as the drugs’ popularity soars, “a lot of doctors don’t know when they need to put the brakes on [weight coming off too quickly].”
She underscored that “we are not treating obesity for weight loss or for cosmetic reasons — this is about optimizing health.”
Dr. Jastreboff noted that in her practice, “I down-titrate if they’re losing weight too quickly.”
“If the patient is losing more than 1% per week of their body weight, then I slow down to make sure they’re getting the nutrients that they need, that they have enough energy to exercise, and that they’re prioritizing protein and fruits and vegetables in their diet.
“We just need to go slow, and yes, we need to follow them long term,” she said.
Chiming in from the audience, Julio Rosenstock, MD, a recognized thought leader in type 2 diabetes, offered his own take on the issues, describing Dr. Taylor’s very low–calorie diet suggestion as “not realistic” and Dr. Nathan’s glucose-first argument to be “stuck in the past.”
Based on modern-day evidence, “there is no reason on earth to start [diabetes treatment] with only metformin,” asserted Dr. Rosenstock, director of the Velocity Clinical Research center at Medical City and clinical professor of medicine at the University of Texas Southwestern Medical Center, Dallas.
“We need to start at the very least with metformin and a sodium-glucose cotransporter 2 (SGLT2) inhibitor from day 1, and then, if it’s affordable and there is access, with a GLP-1 receptor agonist,” he said.
“There is nothing better these days than those agents that consistently have shown a reduction of cardiovascular events and slowing of kidney disease progression.”
Overall, however, “I think you are all right,” he added, a sentiment shared by most.
Noting that the discussion as a whole represents a virtual sea change from the evidence-based options that would have been discussed only a decade ago, Dr. Retnakaran summed up his take-home message: “Stay tuned.
“You could easily see things changing in the next decade to come as we get more data and evidence to support what we ultimately should prioritize an early type 2 diabetes, so this is an exciting time.”
Dr. Retnakaran disclosed ties with Novo Nordisk, Boehringer Ingelheim, Novartis, Sanofi, and Eli Lilly. Dr. Jastreboff disclosed ties with Amgen, AstraZeneca, Boehringer Ingelheim, Biohaven, Eli Lilly, Intellihealth, Novo Nordisk, Pfizer, Regeneron, Scholar Rock, Structure Therapeutics, Terms Pharmaceutical, Weight Watchers, and Zealand Pharmaceuticals. Dr. Roopa had relationships with Novo Nordisk, Boehringer Ingelheim, Amgen, AstraZeneca, Eli Lilly, Silanes, and Sanofi. Dr. Taylor received lecture fees from Novartis, Lilly, Abbott, and Nestle Health and research funding from Diabetes UK and is an advisor to Fast800. Dr. Rosenstock reported relationships with Applied Therapeutics, AstraZeneca, Biomea Fusion, Boehringer Ingelheim, Eli Lilly and Company, Hanmi, Merck, Oramed, Structure Therapeutics, Novartis, Novo Nordisk, Pfizer, Ragor, and Sanofi. Dr. Nathan had no disclosures to report.
A version of this article first appeared on Medscape.com.
ORLANDO, FLORIDA — What to prioritize first in managing early diabetes? That was the question debated on an expert panel at the American Diabetes Association (ADA) 84th Scientific Sessions, with impassioned responses ranging from a plea to “treat obesity first,” to a James Carville–inspired counterpoint of “it’s the glucose, stupid.”
With a focus on preventing complications and inducing remission rounding out the four positions argued,
“In clinical decision-making [for early diabetes], we are faced with weighing each of these variables for the individual patient, and while all are good options, strong arguments can be made for prioritizing each — with the potential of each choice to influence or improve all of the others,” Dr. Retnakaran told this news organization.
Which to Prioritize First?
Making the obesity first argument, Ania M. Jastreboff, MD, PhD, associate professor and director of the Yale Obesity Research Center at Yale School of Medicine, New Haven, Connecticut, noted the striking statistic that nearly 90% of people with type 2 diabetes have overweight or obesity and discussed the ever-expanding data showing the benefits of drugs including glucagon-like peptide 1 (GLP-1) receptor agonists not just in weight loss but also in kidney, cardiovascular, and, as presented at the meeting, sleep apnea improvement.
She contrasted the experiences of two patients with obesity: One treated for the obesity upon type 2 diagnosis — who had a quick normalization of lipids and hypertension soon after the obesity treatment — and the other presenting after 10 years with type 2 diabetes — who was on therapy for hypertension and hyperlipidemia but not for obesity and whose diseases were not as easily treated by that point.
“Why are we treating all the downstream effects and we’re not treating the disease that is potentially the root cause of all these other diseases?” Dr. Jastreboff said.
Complications?
Arguing in favor of focusing on complications, Roopa Mehta, MD, PhD, with the department of endocrinology and metabolism at Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán (INCMNSZ), Mexico City, made the case that stakes don’t get any higher in diabetes than when it comes the looming threat of potentially fatal complications.
Acute myocardial infarction, stroke, amputation, and end-stage renal disease are all on the list of unwanted outcomes and need to be considered even in the earliest stages, as data show early onset type 2 diabetes is linked to life expectancy.
“The main goal of management has always been to prevent complications,” she noted. Citing ADA guidelines, Dr. Mehta underscored the benefits of first- and second-line therapy of metformin, sodium-glucose cotransporter 2 (SGLT2) inhibitors, and GLP-1 receptor agonists for most patients.
Remission?
Discussing the priority of putting patients into disease remission, Roy Taylor, MD, professor of medicine and metabolism at Newcastle University and Newcastle Hospitals NHS in Newcastle upon Tyne, England, and author of the book Life Without Diabetes, focused on an evidence-based alternative to achieving remission — a nonpharmacologic approach that avoids costly and sometimes inaccessible drugs.
In the intervention, described in the DiRECT randomized trial and subsequently in the UK National Health Service Type 2 Diabetes Path to Remission Program, patients with overweight or obesity were placed on a highly restrictive diet of just 800-900 calories a day for 12-20 weeks, followed by maintenance for 12 months, and they not only achieved weight loss but also achieved diabetes remission, in some cases long term.
Acknowledging that “this is not for everyone,” Dr. Taylor asserted that “we have to realize there is a substantial minority of people who want to be healthy but who don’t want to be medicalized,” he said.
“They want their health, and they can do extremely well.”
Glucose?
In taking his self-titled “it’s the glucose, stupid” stand, David M. Nathan, MD, of the Diabetes Center, Massachusetts General Hospital, Harvard Medical School, in Boston, cited extensive evidence showing that early intensive blood glucose control with treatment including sulfonylureas, insulin, or metformin significantly reduced the risk for complications in type 2 diabetes 15 or more years later, including renal failure, blindness, amputation, and myocardial infarctions, in addition to a reduction in diabetes-related death.
“In many of these studies, you saw the benefit even in the setting of weight-gain,” Dr. Nathan underscored.
He further noted the “sobering” findings of the Look AHEAD study, which had to be stopped due to futility when an intensive lifestyle/weight loss intervention showed no significant benefits in terms of cardiovascular disease in people with type 2 diabetes at a median follow-up of 9.6 years.
Ultimately, “diabetes, type 1 and type 2, remains a gluco-centric disease,” Dr. Nathan asserted. “Hyperglycemia is the only universal link between all forms of diabetes and mortality, and the long-term complications of diabetes are intimately associated with hyperglycemia.”
Tackling the Caveats
The ensuing panel discussion did not fail to deliver in delving into key areas of contention, particularly in terms of GLP-1 treatment.
Regarding a lack of data on the potential long-term effects of GLP-1s: “Yes, there are a huge number of studies [on GLP-1 receptor agonists], but they are, in general, over short periods of time and driven by pharma, who get in and get out as quickly as they can and have little in the way of interest to do comparative effectiveness studies,” Dr. Nathan argued.
“Meanwhile, this is like the crack cocaine of medications — patients have to stay on it for a lifetime or they will regain the weight — are you concerned at all about a lifetime of exposure to GLP-1 [drugs]?” he asked the panel.
Dr. Jastreboff responded that the first GLP-1 receptor agonist medications were approved in 2005, nearly 20 years ago, by the US Food and Drug Administration.
“Do I think we need long-term lifetime data? Absolutely,” she said. “We need to do our due diligence, we need to be careful, we need to monitor patients, and when and if there are signals, we need to follow them.”
What about the notorious gastrointestinal side effects of the drugs? “A majority of them are mitigated by slow up-titration,” Dr. Jastreboff noted.
“If patients have nausea, I do not go up [in dose]. I invite patients to tell me if they’re having vomiting because I don’t want anybody to have it, and I can count on one hand how many of my patients do.”
Dr. Mehta added the concern that as the drugs’ popularity soars, “a lot of doctors don’t know when they need to put the brakes on [weight coming off too quickly].”
She underscored that “we are not treating obesity for weight loss or for cosmetic reasons — this is about optimizing health.”
Dr. Jastreboff noted that in her practice, “I down-titrate if they’re losing weight too quickly.”
“If the patient is losing more than 1% per week of their body weight, then I slow down to make sure they’re getting the nutrients that they need, that they have enough energy to exercise, and that they’re prioritizing protein and fruits and vegetables in their diet.
“We just need to go slow, and yes, we need to follow them long term,” she said.
Chiming in from the audience, Julio Rosenstock, MD, a recognized thought leader in type 2 diabetes, offered his own take on the issues, describing Dr. Taylor’s very low–calorie diet suggestion as “not realistic” and Dr. Nathan’s glucose-first argument to be “stuck in the past.”
Based on modern-day evidence, “there is no reason on earth to start [diabetes treatment] with only metformin,” asserted Dr. Rosenstock, director of the Velocity Clinical Research center at Medical City and clinical professor of medicine at the University of Texas Southwestern Medical Center, Dallas.
“We need to start at the very least with metformin and a sodium-glucose cotransporter 2 (SGLT2) inhibitor from day 1, and then, if it’s affordable and there is access, with a GLP-1 receptor agonist,” he said.
“There is nothing better these days than those agents that consistently have shown a reduction of cardiovascular events and slowing of kidney disease progression.”
Overall, however, “I think you are all right,” he added, a sentiment shared by most.
Noting that the discussion as a whole represents a virtual sea change from the evidence-based options that would have been discussed only a decade ago, Dr. Retnakaran summed up his take-home message: “Stay tuned.
“You could easily see things changing in the next decade to come as we get more data and evidence to support what we ultimately should prioritize an early type 2 diabetes, so this is an exciting time.”
Dr. Retnakaran disclosed ties with Novo Nordisk, Boehringer Ingelheim, Novartis, Sanofi, and Eli Lilly. Dr. Jastreboff disclosed ties with Amgen, AstraZeneca, Boehringer Ingelheim, Biohaven, Eli Lilly, Intellihealth, Novo Nordisk, Pfizer, Regeneron, Scholar Rock, Structure Therapeutics, Terms Pharmaceutical, Weight Watchers, and Zealand Pharmaceuticals. Dr. Roopa had relationships with Novo Nordisk, Boehringer Ingelheim, Amgen, AstraZeneca, Eli Lilly, Silanes, and Sanofi. Dr. Taylor received lecture fees from Novartis, Lilly, Abbott, and Nestle Health and research funding from Diabetes UK and is an advisor to Fast800. Dr. Rosenstock reported relationships with Applied Therapeutics, AstraZeneca, Biomea Fusion, Boehringer Ingelheim, Eli Lilly and Company, Hanmi, Merck, Oramed, Structure Therapeutics, Novartis, Novo Nordisk, Pfizer, Ragor, and Sanofi. Dr. Nathan had no disclosures to report.
A version of this article first appeared on Medscape.com.
Triple Therapy May Be Effective in Drug-Naive T2D
TOPLINE:
A triple combination therapy (TCT) of metformin, dapagliflozin, and saxagliptin is an effective and safe treatment option for drug-naive patients with type 2 diabetes (T2D) compared with stepwise add-on therapy.
METHODOLOGY:
- Current guidelines recommend early combination therapy to extend the time to treatment failure, reduce the risk for diabetic complications, and prevent clinical inertia in patients with T2D.
- This randomized controlled open-label trial conducted at nine sites in South Korea included 105 drug-naive patients with T2D (mean age, 49.5 years; 32.4% women) who either received triple therapy (metformin, dapagliflozin, and saxagliptin) or stepwise add-on therapy (initiated with metformin, followed by glimepiride and sitagliptin for those with baseline hemoglobin A1c levels < 9.0% or with initial dual metformin and glimepiride in those with A1c levels ≥ 9.0% followed by sitagliptin).
- The primary outcome was the proportion of patients who achieved A1c levels < 6.5% without hypoglycemia, weight gain ≥ 5%, or discontinuation of drugs because of adverse events at week 104.
- The secondary outcomes were the proportion of patients whose A1c levels dropped to < 7.0% at weeks 56 and 104 and dropped to < 6.5% at week 56, all without hypoglycemia, weight gain, nor discontinuation due to adverse events.
TAKEAWAY:
- At week 104, a higher proportion of patients in the triple therapy group achieved the primary outcome than those in the stepwise add-on therapy group (39.0% vs 17.1%; P = .027).
- In both groups, a similar proportion of patients (46.3%) achieved A1c levels < 6.5% at week 104, but the proportion of patients without hypoglycemia, weight gain, or discontinuation because of adverse events was higher in the triple therapy group than those in the stepwise add-on therapy group (83.3% vs 38.0%; P < .001).
IN PRACTICE:
The authors wrote: “Although the glycemic efficacy of each drug in the TCT was modest, the combination of these drugs resulted in a 2-year durable glycemic efficacy, with greater than a 2.5% reduction in A1c levels from baseline. The overall results of this study suggest a novel strategy for initial combination therapy in newly diagnosed T2D patients.”
SOURCE:
The study was led by Nam Hoon Kim, MD, of the Department of Internal Medicine, Korea University College of Medicine, Seoul. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study had a relatively small sample size as compared with previous clinical trials. More people in the standard therapy group had A1c levels ≥ 9.0%, which resulted in more than double the number of people receiving dual combination therapy over monotherapy in that group. The trial duration was insufficient to evaluate the cardiovascular outcomes.
DISCLOSURES:
The study was funded by AstraZeneca. Some authors reported financial ties with AstraZeneca and other pharmaceutical and medical device companies as members of advisory boards or recipients of grants, consulting fees, honoraria, or lecture fees.
A version of this article appeared on Medscape.com.
TOPLINE:
A triple combination therapy (TCT) of metformin, dapagliflozin, and saxagliptin is an effective and safe treatment option for drug-naive patients with type 2 diabetes (T2D) compared with stepwise add-on therapy.
METHODOLOGY:
- Current guidelines recommend early combination therapy to extend the time to treatment failure, reduce the risk for diabetic complications, and prevent clinical inertia in patients with T2D.
- This randomized controlled open-label trial conducted at nine sites in South Korea included 105 drug-naive patients with T2D (mean age, 49.5 years; 32.4% women) who either received triple therapy (metformin, dapagliflozin, and saxagliptin) or stepwise add-on therapy (initiated with metformin, followed by glimepiride and sitagliptin for those with baseline hemoglobin A1c levels < 9.0% or with initial dual metformin and glimepiride in those with A1c levels ≥ 9.0% followed by sitagliptin).
- The primary outcome was the proportion of patients who achieved A1c levels < 6.5% without hypoglycemia, weight gain ≥ 5%, or discontinuation of drugs because of adverse events at week 104.
- The secondary outcomes were the proportion of patients whose A1c levels dropped to < 7.0% at weeks 56 and 104 and dropped to < 6.5% at week 56, all without hypoglycemia, weight gain, nor discontinuation due to adverse events.
TAKEAWAY:
- At week 104, a higher proportion of patients in the triple therapy group achieved the primary outcome than those in the stepwise add-on therapy group (39.0% vs 17.1%; P = .027).
- In both groups, a similar proportion of patients (46.3%) achieved A1c levels < 6.5% at week 104, but the proportion of patients without hypoglycemia, weight gain, or discontinuation because of adverse events was higher in the triple therapy group than those in the stepwise add-on therapy group (83.3% vs 38.0%; P < .001).
IN PRACTICE:
The authors wrote: “Although the glycemic efficacy of each drug in the TCT was modest, the combination of these drugs resulted in a 2-year durable glycemic efficacy, with greater than a 2.5% reduction in A1c levels from baseline. The overall results of this study suggest a novel strategy for initial combination therapy in newly diagnosed T2D patients.”
SOURCE:
The study was led by Nam Hoon Kim, MD, of the Department of Internal Medicine, Korea University College of Medicine, Seoul. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study had a relatively small sample size as compared with previous clinical trials. More people in the standard therapy group had A1c levels ≥ 9.0%, which resulted in more than double the number of people receiving dual combination therapy over monotherapy in that group. The trial duration was insufficient to evaluate the cardiovascular outcomes.
DISCLOSURES:
The study was funded by AstraZeneca. Some authors reported financial ties with AstraZeneca and other pharmaceutical and medical device companies as members of advisory boards or recipients of grants, consulting fees, honoraria, or lecture fees.
A version of this article appeared on Medscape.com.
TOPLINE:
A triple combination therapy (TCT) of metformin, dapagliflozin, and saxagliptin is an effective and safe treatment option for drug-naive patients with type 2 diabetes (T2D) compared with stepwise add-on therapy.
METHODOLOGY:
- Current guidelines recommend early combination therapy to extend the time to treatment failure, reduce the risk for diabetic complications, and prevent clinical inertia in patients with T2D.
- This randomized controlled open-label trial conducted at nine sites in South Korea included 105 drug-naive patients with T2D (mean age, 49.5 years; 32.4% women) who either received triple therapy (metformin, dapagliflozin, and saxagliptin) or stepwise add-on therapy (initiated with metformin, followed by glimepiride and sitagliptin for those with baseline hemoglobin A1c levels < 9.0% or with initial dual metformin and glimepiride in those with A1c levels ≥ 9.0% followed by sitagliptin).
- The primary outcome was the proportion of patients who achieved A1c levels < 6.5% without hypoglycemia, weight gain ≥ 5%, or discontinuation of drugs because of adverse events at week 104.
- The secondary outcomes were the proportion of patients whose A1c levels dropped to < 7.0% at weeks 56 and 104 and dropped to < 6.5% at week 56, all without hypoglycemia, weight gain, nor discontinuation due to adverse events.
TAKEAWAY:
- At week 104, a higher proportion of patients in the triple therapy group achieved the primary outcome than those in the stepwise add-on therapy group (39.0% vs 17.1%; P = .027).
- In both groups, a similar proportion of patients (46.3%) achieved A1c levels < 6.5% at week 104, but the proportion of patients without hypoglycemia, weight gain, or discontinuation because of adverse events was higher in the triple therapy group than those in the stepwise add-on therapy group (83.3% vs 38.0%; P < .001).
IN PRACTICE:
The authors wrote: “Although the glycemic efficacy of each drug in the TCT was modest, the combination of these drugs resulted in a 2-year durable glycemic efficacy, with greater than a 2.5% reduction in A1c levels from baseline. The overall results of this study suggest a novel strategy for initial combination therapy in newly diagnosed T2D patients.”
SOURCE:
The study was led by Nam Hoon Kim, MD, of the Department of Internal Medicine, Korea University College of Medicine, Seoul. It was published online in Diabetes, Obesity and Metabolism.
LIMITATIONS:
The study had a relatively small sample size as compared with previous clinical trials. More people in the standard therapy group had A1c levels ≥ 9.0%, which resulted in more than double the number of people receiving dual combination therapy over monotherapy in that group. The trial duration was insufficient to evaluate the cardiovascular outcomes.
DISCLOSURES:
The study was funded by AstraZeneca. Some authors reported financial ties with AstraZeneca and other pharmaceutical and medical device companies as members of advisory boards or recipients of grants, consulting fees, honoraria, or lecture fees.
A version of this article appeared on Medscape.com.
Exercise Plus GLP-1 RAs Upped Weight Loss, Bone Retention
TOPLINE:
People with obesity who exercise while taking glucagon-like peptide 1 receptor agonists (GLP-1 RAs; liraglutide) showed increased weight loss and preserved bone health, according to a study published in JAMA Network Open.
METHODOLOGY:
- Patients were placed on an initial diet that consisted of no more than 800 calories per day for 8 weeks. Those who lost at least 5% of their starting weight were then placed into a 1-year program.
- Participants included 195 adults aged between 18 and 65 years with obesity and no diabetes, 64% of whom were women.
- They were split into four groups of interventions: Exercise only (48 patients), liraglutide only (49 patients), a combination of both (49 participants), and placebo (49 participants), for a 1-year period.
- Patients received liraglutide or volume-matched placebo as daily injections starting at 0.6 mg/d with a weekly increase until 3 mg/d was reached; exercise entailed 30-minute sessions for 4 days a week.
- Researchers studied bone health at each patient’s hip, spine, and forearm after they lost weight, by measuring bone mineral density (BMD).
TAKEAWAY:
- The overall average change in weight loss over the course of 52 weeks was 7.03 kg in the placebo group, 11.19 kg in the exercise group, 13.74 kg in the liraglutide group, and 16.88 kg in the combination group.
- BMD did not change in the combination group in comparison to the placebo group at the hip (mean change, −0.006 g/cm2; 95% CI, −0.017 to 0.004 g/cm2; P = .24) or spine (−0.010 g/cm2; 95% CI, −0.025 to 0.005 g/cm2; P = .20).
- BMD of the spine in the liraglutide group decreased in comparison to the exercise group (mean change, −0.016 g/cm2; 95% CI, −0.032 to −0.001 g/cm2; P = .04) and the placebo group, in addition to decreases in the hip.
IN PRACTICE:
“Our results show that the combination of exercise and GLP-1 RA was the most effective weight loss strategy while preserving bone health,” study authors wrote.
SOURCE:
The study was led by Simon Birk Kjær Jensen, PhD, of the Department of Biomedical Sciences and Faculty of Health and Medical Sciences at the University of Copenhagen in Denmark, and published on June 25 in JAMA Network Open.
LIMITATIONS:
The study only included adults aged between 18 and 65 years without other chronic diseases and may not apply to patients who are older or have diabetes. The study sample was diverse but was conducted in Denmark, with a population of generally similar ancestry.
DISCLOSURES:
One study author reported serving on advisory boards for AstraZeneca, Boehringer Ingelheim, Bayer, and Amgen, among others. Other authors reported various financial interests, including grants, personal fees, and salaries, from Amgen, Novo Nordisk, and Abbott Lab, among others.
A version of this article first appeared on Medscape.com.
TOPLINE:
People with obesity who exercise while taking glucagon-like peptide 1 receptor agonists (GLP-1 RAs; liraglutide) showed increased weight loss and preserved bone health, according to a study published in JAMA Network Open.
METHODOLOGY:
- Patients were placed on an initial diet that consisted of no more than 800 calories per day for 8 weeks. Those who lost at least 5% of their starting weight were then placed into a 1-year program.
- Participants included 195 adults aged between 18 and 65 years with obesity and no diabetes, 64% of whom were women.
- They were split into four groups of interventions: Exercise only (48 patients), liraglutide only (49 patients), a combination of both (49 participants), and placebo (49 participants), for a 1-year period.
- Patients received liraglutide or volume-matched placebo as daily injections starting at 0.6 mg/d with a weekly increase until 3 mg/d was reached; exercise entailed 30-minute sessions for 4 days a week.
- Researchers studied bone health at each patient’s hip, spine, and forearm after they lost weight, by measuring bone mineral density (BMD).
TAKEAWAY:
- The overall average change in weight loss over the course of 52 weeks was 7.03 kg in the placebo group, 11.19 kg in the exercise group, 13.74 kg in the liraglutide group, and 16.88 kg in the combination group.
- BMD did not change in the combination group in comparison to the placebo group at the hip (mean change, −0.006 g/cm2; 95% CI, −0.017 to 0.004 g/cm2; P = .24) or spine (−0.010 g/cm2; 95% CI, −0.025 to 0.005 g/cm2; P = .20).
- BMD of the spine in the liraglutide group decreased in comparison to the exercise group (mean change, −0.016 g/cm2; 95% CI, −0.032 to −0.001 g/cm2; P = .04) and the placebo group, in addition to decreases in the hip.
IN PRACTICE:
“Our results show that the combination of exercise and GLP-1 RA was the most effective weight loss strategy while preserving bone health,” study authors wrote.
SOURCE:
The study was led by Simon Birk Kjær Jensen, PhD, of the Department of Biomedical Sciences and Faculty of Health and Medical Sciences at the University of Copenhagen in Denmark, and published on June 25 in JAMA Network Open.
LIMITATIONS:
The study only included adults aged between 18 and 65 years without other chronic diseases and may not apply to patients who are older or have diabetes. The study sample was diverse but was conducted in Denmark, with a population of generally similar ancestry.
DISCLOSURES:
One study author reported serving on advisory boards for AstraZeneca, Boehringer Ingelheim, Bayer, and Amgen, among others. Other authors reported various financial interests, including grants, personal fees, and salaries, from Amgen, Novo Nordisk, and Abbott Lab, among others.
A version of this article first appeared on Medscape.com.
TOPLINE:
People with obesity who exercise while taking glucagon-like peptide 1 receptor agonists (GLP-1 RAs; liraglutide) showed increased weight loss and preserved bone health, according to a study published in JAMA Network Open.
METHODOLOGY:
- Patients were placed on an initial diet that consisted of no more than 800 calories per day for 8 weeks. Those who lost at least 5% of their starting weight were then placed into a 1-year program.
- Participants included 195 adults aged between 18 and 65 years with obesity and no diabetes, 64% of whom were women.
- They were split into four groups of interventions: Exercise only (48 patients), liraglutide only (49 patients), a combination of both (49 participants), and placebo (49 participants), for a 1-year period.
- Patients received liraglutide or volume-matched placebo as daily injections starting at 0.6 mg/d with a weekly increase until 3 mg/d was reached; exercise entailed 30-minute sessions for 4 days a week.
- Researchers studied bone health at each patient’s hip, spine, and forearm after they lost weight, by measuring bone mineral density (BMD).
TAKEAWAY:
- The overall average change in weight loss over the course of 52 weeks was 7.03 kg in the placebo group, 11.19 kg in the exercise group, 13.74 kg in the liraglutide group, and 16.88 kg in the combination group.
- BMD did not change in the combination group in comparison to the placebo group at the hip (mean change, −0.006 g/cm2; 95% CI, −0.017 to 0.004 g/cm2; P = .24) or spine (−0.010 g/cm2; 95% CI, −0.025 to 0.005 g/cm2; P = .20).
- BMD of the spine in the liraglutide group decreased in comparison to the exercise group (mean change, −0.016 g/cm2; 95% CI, −0.032 to −0.001 g/cm2; P = .04) and the placebo group, in addition to decreases in the hip.
IN PRACTICE:
“Our results show that the combination of exercise and GLP-1 RA was the most effective weight loss strategy while preserving bone health,” study authors wrote.
SOURCE:
The study was led by Simon Birk Kjær Jensen, PhD, of the Department of Biomedical Sciences and Faculty of Health and Medical Sciences at the University of Copenhagen in Denmark, and published on June 25 in JAMA Network Open.
LIMITATIONS:
The study only included adults aged between 18 and 65 years without other chronic diseases and may not apply to patients who are older or have diabetes. The study sample was diverse but was conducted in Denmark, with a population of generally similar ancestry.
DISCLOSURES:
One study author reported serving on advisory boards for AstraZeneca, Boehringer Ingelheim, Bayer, and Amgen, among others. Other authors reported various financial interests, including grants, personal fees, and salaries, from Amgen, Novo Nordisk, and Abbott Lab, among others.
A version of this article first appeared on Medscape.com.
Does Semaglutide Reduce Inflammation?
LYON, FRANCE — The anti-obesity drug semaglutide is associated with significant reductions in the inflammatory marker high-sensitivity C-reactive protein (CRP), even in patients who do not lose substantial amounts of weight with the drug, according to data from the SELECT clinical trial.
The research, presented at the European Atherosclerosis Society 2024, involved over 17,600 patients with overweight or obesity and had established cardiovascular disease but not diabetes.
“Weight loss was associated with greater high-sensitivity CRP reduction in both treatment groups,” said study presenter Jorge Plutzky, MD, director of Preventive Cardiology at Brigham and Women’s Hospital, Boston, but “with increased high-sensitivity CRP reductions in those receiving semaglutide.”
The drug also “significantly reduced high-sensitivity CRP early,” he said, “prior to major weight loss and in those who did not lose significant amounts of weight.” The reductions reached approximately 12% at 4 weeks and around 20% at 8 weeks, when the weight loss “was still quite modest,” at 2% and 3% of body weight, respectively. Even among patients who achieved weight loss of less than 2% body weight, semaglutide was associated with a reduction in high-sensitivity CRP levels.
In the SELECT trial, semaglutide also resulted in a consistent reduction of around 20% vs placebo in major adverse cardiovascular events such as cardiovascular mortality, nonfatal myocardial infarction, or nonfatal stroke.
But Naveed Sattar, MD, PhD, professor of cardiometabolic medicine at the University of Glasgow, Scotland, said in an interview that body weight “is probably the major driver” of CRP levels in the population, accounting for between 20% and 30% of the variation.
Dr. Sattar, who was not involved in the study, said that because drugs like semaglutide lower weight but also have anti-inflammatory effects, the question becomes: “Could the anti-inflammatory effects be part of the mechanisms by which these drugs affect the risk of major adverse cardiovascular events?”
Reducing Cardiovascular Events
The current analysis, however, cannot answer the question, he said. “All it tells us is about associations.”
“What we do know is semaglutide, predominantly by lowering weight, is lowering CRP levels and equally, we know that when you lose weight, you improve blood pressure, you improve lipids, and you reduce the risk of diabetes,” he said.
Dr. Sattar also took issue with the researchers’ conclusion that the high-sensitivity CRP reductions seen in SELECT occurred prior to major weight loss because the “pattern of CRP reduction and weight reduction is almost identical.”
Dr. Sattar also pointed out in a recent editorial that the drug appears to have a direct effect on blood vessels and the heart, which may lead to improvements in systemic inflammation. Consequently, he said, any assertion that semaglutide is genuinely anti-inflammatory is, at this stage, “speculation.”
Dr. Plutzky said that “systemic, chronic inflammation is implicated as a potential mechanism and therapeutic target in atherosclerosis and major adverse cardiovascular events, as well as obesity,” and high-sensitivity CRP levels are an “established biomarker of inflammation and have been shown to predict cardiovascular risk.”
However, the relationship between high-sensitivity CRP, responses to glucagon-like peptide 1 receptor agonists like semaglutide, and cardiovascular outcomes in obesity “remains incompletely understood,” said Dr. Plutzky.
A version of this article appeared on Medscape.com.
LYON, FRANCE — The anti-obesity drug semaglutide is associated with significant reductions in the inflammatory marker high-sensitivity C-reactive protein (CRP), even in patients who do not lose substantial amounts of weight with the drug, according to data from the SELECT clinical trial.
The research, presented at the European Atherosclerosis Society 2024, involved over 17,600 patients with overweight or obesity and had established cardiovascular disease but not diabetes.
“Weight loss was associated with greater high-sensitivity CRP reduction in both treatment groups,” said study presenter Jorge Plutzky, MD, director of Preventive Cardiology at Brigham and Women’s Hospital, Boston, but “with increased high-sensitivity CRP reductions in those receiving semaglutide.”
The drug also “significantly reduced high-sensitivity CRP early,” he said, “prior to major weight loss and in those who did not lose significant amounts of weight.” The reductions reached approximately 12% at 4 weeks and around 20% at 8 weeks, when the weight loss “was still quite modest,” at 2% and 3% of body weight, respectively. Even among patients who achieved weight loss of less than 2% body weight, semaglutide was associated with a reduction in high-sensitivity CRP levels.
In the SELECT trial, semaglutide also resulted in a consistent reduction of around 20% vs placebo in major adverse cardiovascular events such as cardiovascular mortality, nonfatal myocardial infarction, or nonfatal stroke.
But Naveed Sattar, MD, PhD, professor of cardiometabolic medicine at the University of Glasgow, Scotland, said in an interview that body weight “is probably the major driver” of CRP levels in the population, accounting for between 20% and 30% of the variation.
Dr. Sattar, who was not involved in the study, said that because drugs like semaglutide lower weight but also have anti-inflammatory effects, the question becomes: “Could the anti-inflammatory effects be part of the mechanisms by which these drugs affect the risk of major adverse cardiovascular events?”
Reducing Cardiovascular Events
The current analysis, however, cannot answer the question, he said. “All it tells us is about associations.”
“What we do know is semaglutide, predominantly by lowering weight, is lowering CRP levels and equally, we know that when you lose weight, you improve blood pressure, you improve lipids, and you reduce the risk of diabetes,” he said.
Dr. Sattar also took issue with the researchers’ conclusion that the high-sensitivity CRP reductions seen in SELECT occurred prior to major weight loss because the “pattern of CRP reduction and weight reduction is almost identical.”
Dr. Sattar also pointed out in a recent editorial that the drug appears to have a direct effect on blood vessels and the heart, which may lead to improvements in systemic inflammation. Consequently, he said, any assertion that semaglutide is genuinely anti-inflammatory is, at this stage, “speculation.”
Dr. Plutzky said that “systemic, chronic inflammation is implicated as a potential mechanism and therapeutic target in atherosclerosis and major adverse cardiovascular events, as well as obesity,” and high-sensitivity CRP levels are an “established biomarker of inflammation and have been shown to predict cardiovascular risk.”
However, the relationship between high-sensitivity CRP, responses to glucagon-like peptide 1 receptor agonists like semaglutide, and cardiovascular outcomes in obesity “remains incompletely understood,” said Dr. Plutzky.
A version of this article appeared on Medscape.com.
LYON, FRANCE — The anti-obesity drug semaglutide is associated with significant reductions in the inflammatory marker high-sensitivity C-reactive protein (CRP), even in patients who do not lose substantial amounts of weight with the drug, according to data from the SELECT clinical trial.
The research, presented at the European Atherosclerosis Society 2024, involved over 17,600 patients with overweight or obesity and had established cardiovascular disease but not diabetes.
“Weight loss was associated with greater high-sensitivity CRP reduction in both treatment groups,” said study presenter Jorge Plutzky, MD, director of Preventive Cardiology at Brigham and Women’s Hospital, Boston, but “with increased high-sensitivity CRP reductions in those receiving semaglutide.”
The drug also “significantly reduced high-sensitivity CRP early,” he said, “prior to major weight loss and in those who did not lose significant amounts of weight.” The reductions reached approximately 12% at 4 weeks and around 20% at 8 weeks, when the weight loss “was still quite modest,” at 2% and 3% of body weight, respectively. Even among patients who achieved weight loss of less than 2% body weight, semaglutide was associated with a reduction in high-sensitivity CRP levels.
In the SELECT trial, semaglutide also resulted in a consistent reduction of around 20% vs placebo in major adverse cardiovascular events such as cardiovascular mortality, nonfatal myocardial infarction, or nonfatal stroke.
But Naveed Sattar, MD, PhD, professor of cardiometabolic medicine at the University of Glasgow, Scotland, said in an interview that body weight “is probably the major driver” of CRP levels in the population, accounting for between 20% and 30% of the variation.
Dr. Sattar, who was not involved in the study, said that because drugs like semaglutide lower weight but also have anti-inflammatory effects, the question becomes: “Could the anti-inflammatory effects be part of the mechanisms by which these drugs affect the risk of major adverse cardiovascular events?”
Reducing Cardiovascular Events
The current analysis, however, cannot answer the question, he said. “All it tells us is about associations.”
“What we do know is semaglutide, predominantly by lowering weight, is lowering CRP levels and equally, we know that when you lose weight, you improve blood pressure, you improve lipids, and you reduce the risk of diabetes,” he said.
Dr. Sattar also took issue with the researchers’ conclusion that the high-sensitivity CRP reductions seen in SELECT occurred prior to major weight loss because the “pattern of CRP reduction and weight reduction is almost identical.”
Dr. Sattar also pointed out in a recent editorial that the drug appears to have a direct effect on blood vessels and the heart, which may lead to improvements in systemic inflammation. Consequently, he said, any assertion that semaglutide is genuinely anti-inflammatory is, at this stage, “speculation.”
Dr. Plutzky said that “systemic, chronic inflammation is implicated as a potential mechanism and therapeutic target in atherosclerosis and major adverse cardiovascular events, as well as obesity,” and high-sensitivity CRP levels are an “established biomarker of inflammation and have been shown to predict cardiovascular risk.”
However, the relationship between high-sensitivity CRP, responses to glucagon-like peptide 1 receptor agonists like semaglutide, and cardiovascular outcomes in obesity “remains incompletely understood,” said Dr. Plutzky.
A version of this article appeared on Medscape.com.
Pancreatic Gene Therapy: A ‘One-and-Done’ GLP-1 Treatment?
TOPLINE:
An experimental pancreatic gene therapy given to a mouse model of obesity as a one-time, single-dose treatment showed improvements in body composition and fasting glucose comparable with those achieved with the glucagon-like peptide 1 (GLP-1) receptor agonist semaglutide, without the reversal of fat-loss and glycemia improvements that are a key concern with the withdrawal of GLP-1 receptor agonist drugs.
METHODOLOGY:
- With initial preclinical research showing benefits in Yucatan pigs, the authors tested the pancreatic gene therapy in mice representing a validated model of diet-induced obesity.
- The mice were randomized to receive either a single-dose administration of the pancreatic gene therapy (n = 10), daily subcutaneous semaglutide injections (n = 10; 10 nmol/kg/d for 4 weeks), pancreatic gene therapy placebo (n = 8), or a semaglutide placebo (n = 8).
- The gene therapy is designed to be delivered directly to the pancreas with a needle puncture, using a proprietary endoscopic delivery method that is similar to procedures commonly performed by gastrointestinal endoscopists, limiting systemic exposure.
- At 4 weeks, semaglutide was discontinued, and 5 of the 10 mice in that group were randomized to the gene therapy, while the other 5 received placebo.
TAKEAWAY:
- At week 4, the pancreatic gene therapy arm had a reduction in fat mass of 21%, compared with 16% with semaglutide (P < .05; both P < .0001 vs placebo)
- The pancreatic gene therapy and semaglutide groups each preserved lean mass, with a loss of only 5% of body weight (both P < .0001 vs placebo).
- At week 8, mice withdrawn from semaglutide had nearly a full reversal of the fat and lean mass losses observed at 4 weeks, returning to within 1% and 2% below baseline, respectively, while the semaglutide-withdrawn mice treated with gene therapy maintained a fat reduction of 17% (P < .01) and lean mass of 5% (P < .0001).
- Significant improvements in fasting glucose were observed in the gene therapy and semaglutide-treated mice at week 4 (both 18%; P < .0001).
- While semaglutide-withdrawal resulted in a rebound of fasting glucose to baseline at week 8, those who had initially received gene therapy or were switched over to the therapy maintained fasting glucose reductions of 21% and 22% at 8 weeks (P < .0001 and P < .001), respectively.
- No indications of pancreatic inflammation or injury were observed in any of the groups.
IN PRACTICE:
The results suggest the therapy could represent “a reliable, ‘off ramp’ from chronic GLP-1 drugs that allows people to maintain the weight loss and blood sugar benefits, even as they stop taking these medicines,” said first author Harith Rajagopalan, MD, PhD, cofounder and chief executive officer of Fractyl Health, which is developing the gene therapy, in a press statement issued by the company.
The therapy is being developed as a candidate for the treatment of type 2 diabetes and plans are underway for the first in-human study in type 2 diabetes in 2025, Dr. Rajagopalan noted while presenting the results at the American Diabetes Association (ADA)’s 84th scientific sessions.
SOURCE:
The study was presented on June 23, 2024, at the annual meeting of the ADA’s 84th scientific sessions (Abstract #261-OR).
LIMITATIONS:
The pancreatic gene therapy is in early development and has not been assessed by any regulatory body for investigational or commercial use.
Asked by an audience member at the ADA presentation if the therapy would be reversible if complications were to arise, Dr. Rajagopalan responded that “there are ways to tune this effect in order to prevent complications from occurring, which we will discuss in due course.”
Also asked about the potential for a positive feedback loop with GLP-1 signaling and insulin signaling, Dr. Rajagopalan noted that “I don’t believe that we have seen any evidence of that risk so far. One could hypothesize, but we have not seen anything [in that regard] that would be a cause for concern.”
DISCLOSURES:
The study was funded by Fractyl Health, and Dr. Rajagopalan and the authors declared being employees and stockholders/shareholders of the company.
A version of this article first appeared on Medscape.com.
TOPLINE:
An experimental pancreatic gene therapy given to a mouse model of obesity as a one-time, single-dose treatment showed improvements in body composition and fasting glucose comparable with those achieved with the glucagon-like peptide 1 (GLP-1) receptor agonist semaglutide, without the reversal of fat-loss and glycemia improvements that are a key concern with the withdrawal of GLP-1 receptor agonist drugs.
METHODOLOGY:
- With initial preclinical research showing benefits in Yucatan pigs, the authors tested the pancreatic gene therapy in mice representing a validated model of diet-induced obesity.
- The mice were randomized to receive either a single-dose administration of the pancreatic gene therapy (n = 10), daily subcutaneous semaglutide injections (n = 10; 10 nmol/kg/d for 4 weeks), pancreatic gene therapy placebo (n = 8), or a semaglutide placebo (n = 8).
- The gene therapy is designed to be delivered directly to the pancreas with a needle puncture, using a proprietary endoscopic delivery method that is similar to procedures commonly performed by gastrointestinal endoscopists, limiting systemic exposure.
- At 4 weeks, semaglutide was discontinued, and 5 of the 10 mice in that group were randomized to the gene therapy, while the other 5 received placebo.
TAKEAWAY:
- At week 4, the pancreatic gene therapy arm had a reduction in fat mass of 21%, compared with 16% with semaglutide (P < .05; both P < .0001 vs placebo)
- The pancreatic gene therapy and semaglutide groups each preserved lean mass, with a loss of only 5% of body weight (both P < .0001 vs placebo).
- At week 8, mice withdrawn from semaglutide had nearly a full reversal of the fat and lean mass losses observed at 4 weeks, returning to within 1% and 2% below baseline, respectively, while the semaglutide-withdrawn mice treated with gene therapy maintained a fat reduction of 17% (P < .01) and lean mass of 5% (P < .0001).
- Significant improvements in fasting glucose were observed in the gene therapy and semaglutide-treated mice at week 4 (both 18%; P < .0001).
- While semaglutide-withdrawal resulted in a rebound of fasting glucose to baseline at week 8, those who had initially received gene therapy or were switched over to the therapy maintained fasting glucose reductions of 21% and 22% at 8 weeks (P < .0001 and P < .001), respectively.
- No indications of pancreatic inflammation or injury were observed in any of the groups.
IN PRACTICE:
The results suggest the therapy could represent “a reliable, ‘off ramp’ from chronic GLP-1 drugs that allows people to maintain the weight loss and blood sugar benefits, even as they stop taking these medicines,” said first author Harith Rajagopalan, MD, PhD, cofounder and chief executive officer of Fractyl Health, which is developing the gene therapy, in a press statement issued by the company.
The therapy is being developed as a candidate for the treatment of type 2 diabetes and plans are underway for the first in-human study in type 2 diabetes in 2025, Dr. Rajagopalan noted while presenting the results at the American Diabetes Association (ADA)’s 84th scientific sessions.
SOURCE:
The study was presented on June 23, 2024, at the annual meeting of the ADA’s 84th scientific sessions (Abstract #261-OR).
LIMITATIONS:
The pancreatic gene therapy is in early development and has not been assessed by any regulatory body for investigational or commercial use.
Asked by an audience member at the ADA presentation if the therapy would be reversible if complications were to arise, Dr. Rajagopalan responded that “there are ways to tune this effect in order to prevent complications from occurring, which we will discuss in due course.”
Also asked about the potential for a positive feedback loop with GLP-1 signaling and insulin signaling, Dr. Rajagopalan noted that “I don’t believe that we have seen any evidence of that risk so far. One could hypothesize, but we have not seen anything [in that regard] that would be a cause for concern.”
DISCLOSURES:
The study was funded by Fractyl Health, and Dr. Rajagopalan and the authors declared being employees and stockholders/shareholders of the company.
A version of this article first appeared on Medscape.com.
TOPLINE:
An experimental pancreatic gene therapy given to a mouse model of obesity as a one-time, single-dose treatment showed improvements in body composition and fasting glucose comparable with those achieved with the glucagon-like peptide 1 (GLP-1) receptor agonist semaglutide, without the reversal of fat-loss and glycemia improvements that are a key concern with the withdrawal of GLP-1 receptor agonist drugs.
METHODOLOGY:
- With initial preclinical research showing benefits in Yucatan pigs, the authors tested the pancreatic gene therapy in mice representing a validated model of diet-induced obesity.
- The mice were randomized to receive either a single-dose administration of the pancreatic gene therapy (n = 10), daily subcutaneous semaglutide injections (n = 10; 10 nmol/kg/d for 4 weeks), pancreatic gene therapy placebo (n = 8), or a semaglutide placebo (n = 8).
- The gene therapy is designed to be delivered directly to the pancreas with a needle puncture, using a proprietary endoscopic delivery method that is similar to procedures commonly performed by gastrointestinal endoscopists, limiting systemic exposure.
- At 4 weeks, semaglutide was discontinued, and 5 of the 10 mice in that group were randomized to the gene therapy, while the other 5 received placebo.
TAKEAWAY:
- At week 4, the pancreatic gene therapy arm had a reduction in fat mass of 21%, compared with 16% with semaglutide (P < .05; both P < .0001 vs placebo)
- The pancreatic gene therapy and semaglutide groups each preserved lean mass, with a loss of only 5% of body weight (both P < .0001 vs placebo).
- At week 8, mice withdrawn from semaglutide had nearly a full reversal of the fat and lean mass losses observed at 4 weeks, returning to within 1% and 2% below baseline, respectively, while the semaglutide-withdrawn mice treated with gene therapy maintained a fat reduction of 17% (P < .01) and lean mass of 5% (P < .0001).
- Significant improvements in fasting glucose were observed in the gene therapy and semaglutide-treated mice at week 4 (both 18%; P < .0001).
- While semaglutide-withdrawal resulted in a rebound of fasting glucose to baseline at week 8, those who had initially received gene therapy or were switched over to the therapy maintained fasting glucose reductions of 21% and 22% at 8 weeks (P < .0001 and P < .001), respectively.
- No indications of pancreatic inflammation or injury were observed in any of the groups.
IN PRACTICE:
The results suggest the therapy could represent “a reliable, ‘off ramp’ from chronic GLP-1 drugs that allows people to maintain the weight loss and blood sugar benefits, even as they stop taking these medicines,” said first author Harith Rajagopalan, MD, PhD, cofounder and chief executive officer of Fractyl Health, which is developing the gene therapy, in a press statement issued by the company.
The therapy is being developed as a candidate for the treatment of type 2 diabetes and plans are underway for the first in-human study in type 2 diabetes in 2025, Dr. Rajagopalan noted while presenting the results at the American Diabetes Association (ADA)’s 84th scientific sessions.
SOURCE:
The study was presented on June 23, 2024, at the annual meeting of the ADA’s 84th scientific sessions (Abstract #261-OR).
LIMITATIONS:
The pancreatic gene therapy is in early development and has not been assessed by any regulatory body for investigational or commercial use.
Asked by an audience member at the ADA presentation if the therapy would be reversible if complications were to arise, Dr. Rajagopalan responded that “there are ways to tune this effect in order to prevent complications from occurring, which we will discuss in due course.”
Also asked about the potential for a positive feedback loop with GLP-1 signaling and insulin signaling, Dr. Rajagopalan noted that “I don’t believe that we have seen any evidence of that risk so far. One could hypothesize, but we have not seen anything [in that regard] that would be a cause for concern.”
DISCLOSURES:
The study was funded by Fractyl Health, and Dr. Rajagopalan and the authors declared being employees and stockholders/shareholders of the company.
A version of this article first appeared on Medscape.com.
Compounded Semaglutide: How to Better Ensure Its Safety
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) receptor agonists such as semaglutide (marketed as Ozempic and Rybelsus for type 2 diabetes and as Wegovy for obesity) slow down digestion and curb hunger by working on the brain’s dopamine reward center. They are prescribed to promote weight loss, metabolic health in type 2 diabetes, and heart health in coronary artery disease.
Semaglutide can be prescribed in two forms: the brand-name version, which is approved and confirmed as safe and effective by the US Food and Drug Administration (FDA), and the versions that can be obtained from a compounding pharmacy. Compounding pharmacies are permitted by the FDA to produce what is “ essentially a copy” of approved medications when there’s an official shortage, which is currently the case with semaglutide and other GLP-1 receptor agonists.
Patients are often drawn to compounding pharmacies for pricing-related reasons. If semaglutide is prescribed for a clear indication like diabetes and is covered by insurance, the brand-name version is commonly dispensed. However, if it’s not covered, patients need to pay out of pocket for branded versions, which carry a monthly cost of $1000 or more. Alternatively, their doctors can prescribe compounded semaglutide, which some telehealth companies advertise at costs of approximately $150-$300 per month.
Potential Issues With Compounded Semaglutide
Compounding pharmacies produce drugs from raw materials containing active pharmaceutical ingredients (APIs). Although compounders use many of the same ingredients found in brand-name medications, for drugs like semaglutide, they may opt for specific salts that are not identical to those involved in the production of the standard versions. These salts are typically reserved for research purposes and may not be suitable for general use.
In late 2023, the FDA issued a letter asking the public to exercise caution when using compounded products containing semaglutide or semaglutide salts. This was followed in January 2024 by an FDA communication citing adverse events reported with the use of compounded semaglutide and advising patients to avoid these versions if an approved form of the drug is available.
Compound Pharmacies: A Closer Look
Compounding pharmacies have exploded in popularity in the past several decades. The compounding pharmacy market is expected to grow at 7.8% per year over the next decade.
Historically, compounding pharmacies have filled a niche for specialty vitamins for intravenous administration as well as chemotherapy medications. They also offer controlled substances, such as ketamine lozenges and nasal sprays, which are unavailable or are in short supply from traditional manufacturers.
Compounding pharmacies fall into two categories. First are compounding pharmacies covered under Section 503A of the Federal Food, Drug and Cosmetic Act; these drugs are neither tested nor monitored. Such facilities do not have to report adverse events to the FDA. The second category is Section 503B outsourcing facilities. These pharmacies choose to be tested by, to be inspected by, and to report adverse events to the FDA.
The FDA’s Latest Update on This Issue
This news organization contacted the FDA for an update on the adverse events reported about compounded semaglutide. From August 8, 2021, to March 31, 2024, they received more than 20,000 adverse events reports for FDA-approved semaglutide. Comparatively, there were 210 adverse events reported on compounded semaglutide products.
The FDA went on to describe that many of the adverse events reported were consistent with known reactions in the labeling, like nausea, diarrhea, and headache. Yet, they added that, “the FDA is unable to determine how, or if, other factors may have contributed to these adverse events, such as differences in ingredients and formulation between FDA-approved and compounded semaglutide products.” They also noted there was variation in the data quality in the reports they have received, which came only from 503B compounding pharmacies.
In conclusion, given the concerns about compounded semaglutide, it is prudent for the prescribing physicians as well as the patients taking the medication to know that risks are “higher” according to the FDA. We eagerly await more specific information from the FDA to better understand reported adverse events.
How to Help Patients Receive Safe Compounded Semaglutide
For clinicians considering prescribing semaglutide from compounding pharmacies, there are several questions worth asking, according to the Alliance for Pharmacy Compounding. First, find out whether the pharmacy complies with United States Pharmacopeia compounding standards and whether they source their APIs from FDA-registered facilities, the latter being required by federal law. It’s also important to ensure that these facilities undergo periodic third-party testing to verify medication purity and dosing.
Ask whether the pharmacy is accredited by the Pharmacy Compounding Accreditation Board (PCAB). Accreditation from the PCAB means that pharmacies have been assessed for processes related to continuous quality improvement. In addition, ask whether the pharmacy is designated as a 503B compounder and if not, why.
Finally, interviewing the pharmacist themselves can provide useful information about staffing, training, and their methods of preparing medications. For example, if they are preparing a sterile eye drop, it is important to ask about sterility testing.
Jesse M. Pines, MD, MBA, MSCE, is a clinical professor of emergency medicine at George Washington University in Washington, and a professor in the department of emergency medicine at Drexel University College of Medicine in Philadelphia, Pennsylvania. Dr. Pines is also the chief of clinical innovation at US Acute Care Solutions in Canton, Ohio. Robert D. Glatter, MD, is an assistant professor of emergency medicine at Zucker School of Medicine at Hofstra/Northwell in Hempstead, New York. Dr. Pines reported conflicts of interest with CSL Behring and Abbott Point-of-Care. Dr. Glatter reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Diabetic Foot Infections: A Peptide’s Potential Promise
At the recent American Diabetes Association (ADA) Scientific Sessions, researchers unveiled promising data on a novel antimicrobial peptide PL-5 spray. This innovative treatment shows significant promise for managing mild to moderate infected diabetic foot ulcers.
Of the 1.6 million people with diabetes in the United States and the tens of millions of similar people worldwide, 50% will require antimicrobials at some time during their life cycle. Diabetic foot infections are difficult to treat because of their resistance to conventional therapies, often leading to severe complications, including amputations.
To address this issue, the antimicrobial peptide PL-5 spray was developed with a novel mechanism of action to potentially improve treatment outcomes. The study aimed to assess the clinical efficacy and safety of the PL-5 spray combined with standard debridement procedures in treating mild to moderate diabetic foot ulcers.
This multicenter, randomized, double-blind, placebo-controlled clinical trial was conducted in four hospitals across China. Participants with mild to moderate diabetic foot ulcers were randomly assigned in a 2:1 ratio to either the PL-5 group or the placebo group, both receiving standard debridement. The primary endpoint was clinical efficacy at day 1 after the end of treatment (EOT1). Secondary endpoints included clinical efficacy at day 7 (EOT7), microbiological efficacy, drug-resistant bacteria clearance rate, wound healing rate, and safety outcomes evaluated at both EOT1 and EOT7.
The study included 47 participants, with 32 in the PL-5 group and 15 in the placebo group. Both groups had statistically comparable demographic and clinical characteristics. The primary endpoint showed a higher clinical efficacy (cure/improvement ratio) in the PL-5 group, compared with the control group (1.33 vs 0.55; P =.0764), suggesting a positive trend but not reaching statistical significance in this population.
Among the secondary endpoints, clinical efficacy at EOT7 was significantly higher in the PL-5 group than in the control group (1.6 vs 0.86). Microbial eradication rates were notably better in the PL-5 group at both EOT1 (57.89% vs 33.33%) and EOT7 (64.71% vs 40.00%). The clearance rates of drug-resistant bacteria were also higher in the PL-5 group at EOT1 (71.43% vs 50%).
Of importance, safety parameters showed no significant differences between the two groups (24.24% vs 33.33%), highlighting the favorable safety profile of PL-5 spray.
The study presented at the ADA Scientific Sessions provides a glint of promising evidence supporting the potential efficacy and safety of PL-5 spray in treating mild to moderate diabetic foot infections. Despite the limited sample size, the results suggest that PL-5 spray may enhance the recovery speed of diabetic foot wounds, particularly in clearing drug-resistant bacterial infections. These findings justify further investigation with larger sample sizes to confirm or refute the efficacy and potentially establish PL-5 spray as a standard treatment option in diabetic foot care.
The novel antimicrobial peptide PL-5 spray shows potential in addressing the challenging issue of diabetic foot infections. This recent ADA presentation sparked significant interest and discussions about the future of diabetic foot ulcer treatments, emphasizing the importance of innovative approaches in managing complex diabetic complications.
Dr. Armstrong is a professor of surgery and director of limb preservation at the University of Southern California, Los Angeles. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
At the recent American Diabetes Association (ADA) Scientific Sessions, researchers unveiled promising data on a novel antimicrobial peptide PL-5 spray. This innovative treatment shows significant promise for managing mild to moderate infected diabetic foot ulcers.
Of the 1.6 million people with diabetes in the United States and the tens of millions of similar people worldwide, 50% will require antimicrobials at some time during their life cycle. Diabetic foot infections are difficult to treat because of their resistance to conventional therapies, often leading to severe complications, including amputations.
To address this issue, the antimicrobial peptide PL-5 spray was developed with a novel mechanism of action to potentially improve treatment outcomes. The study aimed to assess the clinical efficacy and safety of the PL-5 spray combined with standard debridement procedures in treating mild to moderate diabetic foot ulcers.
This multicenter, randomized, double-blind, placebo-controlled clinical trial was conducted in four hospitals across China. Participants with mild to moderate diabetic foot ulcers were randomly assigned in a 2:1 ratio to either the PL-5 group or the placebo group, both receiving standard debridement. The primary endpoint was clinical efficacy at day 1 after the end of treatment (EOT1). Secondary endpoints included clinical efficacy at day 7 (EOT7), microbiological efficacy, drug-resistant bacteria clearance rate, wound healing rate, and safety outcomes evaluated at both EOT1 and EOT7.
The study included 47 participants, with 32 in the PL-5 group and 15 in the placebo group. Both groups had statistically comparable demographic and clinical characteristics. The primary endpoint showed a higher clinical efficacy (cure/improvement ratio) in the PL-5 group, compared with the control group (1.33 vs 0.55; P =.0764), suggesting a positive trend but not reaching statistical significance in this population.
Among the secondary endpoints, clinical efficacy at EOT7 was significantly higher in the PL-5 group than in the control group (1.6 vs 0.86). Microbial eradication rates were notably better in the PL-5 group at both EOT1 (57.89% vs 33.33%) and EOT7 (64.71% vs 40.00%). The clearance rates of drug-resistant bacteria were also higher in the PL-5 group at EOT1 (71.43% vs 50%).
Of importance, safety parameters showed no significant differences between the two groups (24.24% vs 33.33%), highlighting the favorable safety profile of PL-5 spray.
The study presented at the ADA Scientific Sessions provides a glint of promising evidence supporting the potential efficacy and safety of PL-5 spray in treating mild to moderate diabetic foot infections. Despite the limited sample size, the results suggest that PL-5 spray may enhance the recovery speed of diabetic foot wounds, particularly in clearing drug-resistant bacterial infections. These findings justify further investigation with larger sample sizes to confirm or refute the efficacy and potentially establish PL-5 spray as a standard treatment option in diabetic foot care.
The novel antimicrobial peptide PL-5 spray shows potential in addressing the challenging issue of diabetic foot infections. This recent ADA presentation sparked significant interest and discussions about the future of diabetic foot ulcer treatments, emphasizing the importance of innovative approaches in managing complex diabetic complications.
Dr. Armstrong is a professor of surgery and director of limb preservation at the University of Southern California, Los Angeles. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
At the recent American Diabetes Association (ADA) Scientific Sessions, researchers unveiled promising data on a novel antimicrobial peptide PL-5 spray. This innovative treatment shows significant promise for managing mild to moderate infected diabetic foot ulcers.
Of the 1.6 million people with diabetes in the United States and the tens of millions of similar people worldwide, 50% will require antimicrobials at some time during their life cycle. Diabetic foot infections are difficult to treat because of their resistance to conventional therapies, often leading to severe complications, including amputations.
To address this issue, the antimicrobial peptide PL-5 spray was developed with a novel mechanism of action to potentially improve treatment outcomes. The study aimed to assess the clinical efficacy and safety of the PL-5 spray combined with standard debridement procedures in treating mild to moderate diabetic foot ulcers.
This multicenter, randomized, double-blind, placebo-controlled clinical trial was conducted in four hospitals across China. Participants with mild to moderate diabetic foot ulcers were randomly assigned in a 2:1 ratio to either the PL-5 group or the placebo group, both receiving standard debridement. The primary endpoint was clinical efficacy at day 1 after the end of treatment (EOT1). Secondary endpoints included clinical efficacy at day 7 (EOT7), microbiological efficacy, drug-resistant bacteria clearance rate, wound healing rate, and safety outcomes evaluated at both EOT1 and EOT7.
The study included 47 participants, with 32 in the PL-5 group and 15 in the placebo group. Both groups had statistically comparable demographic and clinical characteristics. The primary endpoint showed a higher clinical efficacy (cure/improvement ratio) in the PL-5 group, compared with the control group (1.33 vs 0.55; P =.0764), suggesting a positive trend but not reaching statistical significance in this population.
Among the secondary endpoints, clinical efficacy at EOT7 was significantly higher in the PL-5 group than in the control group (1.6 vs 0.86). Microbial eradication rates were notably better in the PL-5 group at both EOT1 (57.89% vs 33.33%) and EOT7 (64.71% vs 40.00%). The clearance rates of drug-resistant bacteria were also higher in the PL-5 group at EOT1 (71.43% vs 50%).
Of importance, safety parameters showed no significant differences between the two groups (24.24% vs 33.33%), highlighting the favorable safety profile of PL-5 spray.
The study presented at the ADA Scientific Sessions provides a glint of promising evidence supporting the potential efficacy and safety of PL-5 spray in treating mild to moderate diabetic foot infections. Despite the limited sample size, the results suggest that PL-5 spray may enhance the recovery speed of diabetic foot wounds, particularly in clearing drug-resistant bacterial infections. These findings justify further investigation with larger sample sizes to confirm or refute the efficacy and potentially establish PL-5 spray as a standard treatment option in diabetic foot care.
The novel antimicrobial peptide PL-5 spray shows potential in addressing the challenging issue of diabetic foot infections. This recent ADA presentation sparked significant interest and discussions about the future of diabetic foot ulcer treatments, emphasizing the importance of innovative approaches in managing complex diabetic complications.
Dr. Armstrong is a professor of surgery and director of limb preservation at the University of Southern California, Los Angeles. He reported no relevant conflicts of interest.
A version of this article first appeared on Medscape.com.
Ghrelin Paradox: Unlocking New Avenues in Obesity Management
Despite their best efforts, 80% of people who lose weight regain it and many end up heavier within 5 years. Why? Our bodies fight back, revving up hunger while slowing metabolism after weight loss. In ongoing obesity discussions, ghrelin is in the spotlight as the “hunger hormone” playing a crucial role in driving appetite and facilitating weight gain.
Weight loss interventions, such as diet or gastric bypass surgery, may trigger an increase in ghrelin levels, potentially fueling long-term weight gain. Consequently, ghrelin remains a focal point of research into innovative antiobesity treatments.
Ghrelin, a hormone produced in the stomach, is often called the “hunger hormone.” Ghrelin is a circulating orexigenic gut hormone with growth hormone–releasing activity.
Since the discovery of ghrelin, in 1999, research in mice and people has focused on its effect on regulating appetite and implications for long-term weight control. When hunger strikes, ghrelin levels surge, sending signals to the brain that ramp up the appetite. Following a meal, ghrelin decreases, indicating fullness.
Studies have found that people who were injected with subcutaneous ghrelin experienced a 46% increase in hunger and ate 28% more at their next meal than those who didn’t receive a ghrelin injection.
We might expect high levels of ghrelin in individuals with obesity, but this is not the case. In fact, ghrelin levels are typically lower in individuals with obesity than in leaner individuals. This finding might seem to contradict the idea that obesity is due to high levels of the hunger hormone.
Excess weight could increase sensitivity to ghrelin, where more receptors lead to higher hunger stimulation with less ghrelin. Beyond hunger, ghrelin can also lead us to eat for comfort, as when stressed or anxious. Ghrelin and synthetic ghrelin mimetics increase body weight and fat mass by activating receptors in the arcuate nucleus of the hypothalamus (Müller et al.; Bany Bakar et al.). There, it also activates the brain’s reward pathways, making us crave food even when we are not hungry. This connection between ghrelin and emotional eating can contribute to stress-induced obesity.
In my clinical practice, I have seen individuals gain maximum weight when they are under more stress and are sleep-deprived. This is because ghrelin levels increased in these scenarios. This elevation of ghrelin in high-stress, low-sleep situations affects weight gain in women during the postpartum period and menopause.
Evidence also suggests that certain foods affect ghrelin levels. After a person eats carbohydrates, their ghrelin levels initially decrease quickly, but this is followed by a rise in ghrelin, leading them to become hungry again. In contrast, protein intake helps suppress ghrelin levels for longer. Hence, we advise patients to increase protein intake while reducing their carb intake, or to always eat protein along with carbs.
It makes sense that when individuals with obesity lose weight by fasting or caloric restriction and try to maintain that weight loss, their bodies tend to produce more ghrelin. This effect might explain why people who lose weight often find it hard to keep it off: Rising ghrelin levels after weight loss might drive them to eat more and regain weight.
Two prominent weight loss surgeries, sleeve gastrectomy (SG) and Roux-en-Y gastric bypass (RYGB), have opposite effects on ghrelin levels, reflecting their distinct mechanisms for weight loss. SG involves removal of the gastric fundus, where ghrelin is produced, resulting in a significant decrease in ghrelin levels; RYGB operates through malabsorption without directly affecting ghrelin production. Despite these differing approaches, both techniques demonstrate remarkable weight loss efficacy. Research comparing the two procedures reveals that SG leads to decreased fasting plasma ghrelin levels, whereas RYGB prompts an increase, highlighting the additional appetite-reducing mechanism of SG through ghrelin suppression. This contrast underscores the intricate role of ghrelin in appetite regulation and suggests that its manipulation can significantly influence weight loss outcomes.
With the effect of ghrelin in stimulating appetite being established, other studies have explored the relationship between ghrelin and insulin resistance. A meta-analysis by researchers at Qingdao University, Qingdao, China, found that circulating ghrelin levels were negatively correlated with insulin resistance in individuals with obesity and normal fasting glucose levels. The findings suggest that the role of ghrelin in obesity might extend beyond appetite regulation to influence metabolic pathways and that ghrelin may be a marker for predicting obesity.
Researchers are exploring potential therapeutic targets focusing on ghrelin modulation. Although selective neutralization of ghrelin has not yielded consistent results in rodent models, the interplay between ghrelin and LEAP2— a hormone that attaches to the same brain receptors — could be an area of interest for future obesity treatments.
Could ghrelin be the key to tackling obesity? Blocking ghrelin pharmacologically might be a strategy to keep weight off after weight loss, and it could help prevent the typical rebound effect seen with diets and withdrawal of medications. Considering the high rates of weight regain after diet-induced weight loss and withdrawal of weight loss medications, targeting ghrelin might be the missing link in long-term obesity treatment. It could be a valuable approach to improving long-term outcomes for obesity. However, these blockers might have significant side effects, given that ghrelin affects not only hunger but also the brain’s reward and pleasure centers. Therefore, caution will be needed in developing such medications owing to their potential impact on mood and mental health.
With ghrelin playing roles in hunger, reward pathways, and energy regulation, understanding this hormone is crucial in the fight against obesity. Stay tuned for future research that could shed light on the underlying mechanisms at play and hopefully results in clinical action steps.
Dimpi Desai, MD, is a professor in the Department of Medicine, Division of Endocrinology, Gerontology, and Metabolism, Stanford University, Stanford, California, and has disclosed no relevant financial relationships. Ashni Dharia, MD, is a resident in the Department of Internal Medicine, Allegheny General Hospital, Pittsburgh, Pennsylvania.
A version of this article appeared on Medscape.com.
Despite their best efforts, 80% of people who lose weight regain it and many end up heavier within 5 years. Why? Our bodies fight back, revving up hunger while slowing metabolism after weight loss. In ongoing obesity discussions, ghrelin is in the spotlight as the “hunger hormone” playing a crucial role in driving appetite and facilitating weight gain.
Weight loss interventions, such as diet or gastric bypass surgery, may trigger an increase in ghrelin levels, potentially fueling long-term weight gain. Consequently, ghrelin remains a focal point of research into innovative antiobesity treatments.
Ghrelin, a hormone produced in the stomach, is often called the “hunger hormone.” Ghrelin is a circulating orexigenic gut hormone with growth hormone–releasing activity.
Since the discovery of ghrelin, in 1999, research in mice and people has focused on its effect on regulating appetite and implications for long-term weight control. When hunger strikes, ghrelin levels surge, sending signals to the brain that ramp up the appetite. Following a meal, ghrelin decreases, indicating fullness.
Studies have found that people who were injected with subcutaneous ghrelin experienced a 46% increase in hunger and ate 28% more at their next meal than those who didn’t receive a ghrelin injection.
We might expect high levels of ghrelin in individuals with obesity, but this is not the case. In fact, ghrelin levels are typically lower in individuals with obesity than in leaner individuals. This finding might seem to contradict the idea that obesity is due to high levels of the hunger hormone.
Excess weight could increase sensitivity to ghrelin, where more receptors lead to higher hunger stimulation with less ghrelin. Beyond hunger, ghrelin can also lead us to eat for comfort, as when stressed or anxious. Ghrelin and synthetic ghrelin mimetics increase body weight and fat mass by activating receptors in the arcuate nucleus of the hypothalamus (Müller et al.; Bany Bakar et al.). There, it also activates the brain’s reward pathways, making us crave food even when we are not hungry. This connection between ghrelin and emotional eating can contribute to stress-induced obesity.
In my clinical practice, I have seen individuals gain maximum weight when they are under more stress and are sleep-deprived. This is because ghrelin levels increased in these scenarios. This elevation of ghrelin in high-stress, low-sleep situations affects weight gain in women during the postpartum period and menopause.
Evidence also suggests that certain foods affect ghrelin levels. After a person eats carbohydrates, their ghrelin levels initially decrease quickly, but this is followed by a rise in ghrelin, leading them to become hungry again. In contrast, protein intake helps suppress ghrelin levels for longer. Hence, we advise patients to increase protein intake while reducing their carb intake, or to always eat protein along with carbs.
It makes sense that when individuals with obesity lose weight by fasting or caloric restriction and try to maintain that weight loss, their bodies tend to produce more ghrelin. This effect might explain why people who lose weight often find it hard to keep it off: Rising ghrelin levels after weight loss might drive them to eat more and regain weight.
Two prominent weight loss surgeries, sleeve gastrectomy (SG) and Roux-en-Y gastric bypass (RYGB), have opposite effects on ghrelin levels, reflecting their distinct mechanisms for weight loss. SG involves removal of the gastric fundus, where ghrelin is produced, resulting in a significant decrease in ghrelin levels; RYGB operates through malabsorption without directly affecting ghrelin production. Despite these differing approaches, both techniques demonstrate remarkable weight loss efficacy. Research comparing the two procedures reveals that SG leads to decreased fasting plasma ghrelin levels, whereas RYGB prompts an increase, highlighting the additional appetite-reducing mechanism of SG through ghrelin suppression. This contrast underscores the intricate role of ghrelin in appetite regulation and suggests that its manipulation can significantly influence weight loss outcomes.
With the effect of ghrelin in stimulating appetite being established, other studies have explored the relationship between ghrelin and insulin resistance. A meta-analysis by researchers at Qingdao University, Qingdao, China, found that circulating ghrelin levels were negatively correlated with insulin resistance in individuals with obesity and normal fasting glucose levels. The findings suggest that the role of ghrelin in obesity might extend beyond appetite regulation to influence metabolic pathways and that ghrelin may be a marker for predicting obesity.
Researchers are exploring potential therapeutic targets focusing on ghrelin modulation. Although selective neutralization of ghrelin has not yielded consistent results in rodent models, the interplay between ghrelin and LEAP2— a hormone that attaches to the same brain receptors — could be an area of interest for future obesity treatments.
Could ghrelin be the key to tackling obesity? Blocking ghrelin pharmacologically might be a strategy to keep weight off after weight loss, and it could help prevent the typical rebound effect seen with diets and withdrawal of medications. Considering the high rates of weight regain after diet-induced weight loss and withdrawal of weight loss medications, targeting ghrelin might be the missing link in long-term obesity treatment. It could be a valuable approach to improving long-term outcomes for obesity. However, these blockers might have significant side effects, given that ghrelin affects not only hunger but also the brain’s reward and pleasure centers. Therefore, caution will be needed in developing such medications owing to their potential impact on mood and mental health.
With ghrelin playing roles in hunger, reward pathways, and energy regulation, understanding this hormone is crucial in the fight against obesity. Stay tuned for future research that could shed light on the underlying mechanisms at play and hopefully results in clinical action steps.
Dimpi Desai, MD, is a professor in the Department of Medicine, Division of Endocrinology, Gerontology, and Metabolism, Stanford University, Stanford, California, and has disclosed no relevant financial relationships. Ashni Dharia, MD, is a resident in the Department of Internal Medicine, Allegheny General Hospital, Pittsburgh, Pennsylvania.
A version of this article appeared on Medscape.com.
Despite their best efforts, 80% of people who lose weight regain it and many end up heavier within 5 years. Why? Our bodies fight back, revving up hunger while slowing metabolism after weight loss. In ongoing obesity discussions, ghrelin is in the spotlight as the “hunger hormone” playing a crucial role in driving appetite and facilitating weight gain.
Weight loss interventions, such as diet or gastric bypass surgery, may trigger an increase in ghrelin levels, potentially fueling long-term weight gain. Consequently, ghrelin remains a focal point of research into innovative antiobesity treatments.
Ghrelin, a hormone produced in the stomach, is often called the “hunger hormone.” Ghrelin is a circulating orexigenic gut hormone with growth hormone–releasing activity.
Since the discovery of ghrelin, in 1999, research in mice and people has focused on its effect on regulating appetite and implications for long-term weight control. When hunger strikes, ghrelin levels surge, sending signals to the brain that ramp up the appetite. Following a meal, ghrelin decreases, indicating fullness.
Studies have found that people who were injected with subcutaneous ghrelin experienced a 46% increase in hunger and ate 28% more at their next meal than those who didn’t receive a ghrelin injection.
We might expect high levels of ghrelin in individuals with obesity, but this is not the case. In fact, ghrelin levels are typically lower in individuals with obesity than in leaner individuals. This finding might seem to contradict the idea that obesity is due to high levels of the hunger hormone.
Excess weight could increase sensitivity to ghrelin, where more receptors lead to higher hunger stimulation with less ghrelin. Beyond hunger, ghrelin can also lead us to eat for comfort, as when stressed or anxious. Ghrelin and synthetic ghrelin mimetics increase body weight and fat mass by activating receptors in the arcuate nucleus of the hypothalamus (Müller et al.; Bany Bakar et al.). There, it also activates the brain’s reward pathways, making us crave food even when we are not hungry. This connection between ghrelin and emotional eating can contribute to stress-induced obesity.
In my clinical practice, I have seen individuals gain maximum weight when they are under more stress and are sleep-deprived. This is because ghrelin levels increased in these scenarios. This elevation of ghrelin in high-stress, low-sleep situations affects weight gain in women during the postpartum period and menopause.
Evidence also suggests that certain foods affect ghrelin levels. After a person eats carbohydrates, their ghrelin levels initially decrease quickly, but this is followed by a rise in ghrelin, leading them to become hungry again. In contrast, protein intake helps suppress ghrelin levels for longer. Hence, we advise patients to increase protein intake while reducing their carb intake, or to always eat protein along with carbs.
It makes sense that when individuals with obesity lose weight by fasting or caloric restriction and try to maintain that weight loss, their bodies tend to produce more ghrelin. This effect might explain why people who lose weight often find it hard to keep it off: Rising ghrelin levels after weight loss might drive them to eat more and regain weight.
Two prominent weight loss surgeries, sleeve gastrectomy (SG) and Roux-en-Y gastric bypass (RYGB), have opposite effects on ghrelin levels, reflecting their distinct mechanisms for weight loss. SG involves removal of the gastric fundus, where ghrelin is produced, resulting in a significant decrease in ghrelin levels; RYGB operates through malabsorption without directly affecting ghrelin production. Despite these differing approaches, both techniques demonstrate remarkable weight loss efficacy. Research comparing the two procedures reveals that SG leads to decreased fasting plasma ghrelin levels, whereas RYGB prompts an increase, highlighting the additional appetite-reducing mechanism of SG through ghrelin suppression. This contrast underscores the intricate role of ghrelin in appetite regulation and suggests that its manipulation can significantly influence weight loss outcomes.
With the effect of ghrelin in stimulating appetite being established, other studies have explored the relationship between ghrelin and insulin resistance. A meta-analysis by researchers at Qingdao University, Qingdao, China, found that circulating ghrelin levels were negatively correlated with insulin resistance in individuals with obesity and normal fasting glucose levels. The findings suggest that the role of ghrelin in obesity might extend beyond appetite regulation to influence metabolic pathways and that ghrelin may be a marker for predicting obesity.
Researchers are exploring potential therapeutic targets focusing on ghrelin modulation. Although selective neutralization of ghrelin has not yielded consistent results in rodent models, the interplay between ghrelin and LEAP2— a hormone that attaches to the same brain receptors — could be an area of interest for future obesity treatments.
Could ghrelin be the key to tackling obesity? Blocking ghrelin pharmacologically might be a strategy to keep weight off after weight loss, and it could help prevent the typical rebound effect seen with diets and withdrawal of medications. Considering the high rates of weight regain after diet-induced weight loss and withdrawal of weight loss medications, targeting ghrelin might be the missing link in long-term obesity treatment. It could be a valuable approach to improving long-term outcomes for obesity. However, these blockers might have significant side effects, given that ghrelin affects not only hunger but also the brain’s reward and pleasure centers. Therefore, caution will be needed in developing such medications owing to their potential impact on mood and mental health.
With ghrelin playing roles in hunger, reward pathways, and energy regulation, understanding this hormone is crucial in the fight against obesity. Stay tuned for future research that could shed light on the underlying mechanisms at play and hopefully results in clinical action steps.
Dimpi Desai, MD, is a professor in the Department of Medicine, Division of Endocrinology, Gerontology, and Metabolism, Stanford University, Stanford, California, and has disclosed no relevant financial relationships. Ashni Dharia, MD, is a resident in the Department of Internal Medicine, Allegheny General Hospital, Pittsburgh, Pennsylvania.
A version of this article appeared on Medscape.com.
Tirzepatide Reduces Sleep Interruptions, Halting Almost Half of CPAP Use
ORLANDO, FLA. — The diabetes and weight loss drug tirzepatide (Mounjaro for type 2 diabetes; Zepbound for obesity) was so effective at reducing sleep disruptions in patients with obesity and obstructive sleep apnea (OSA) that 40%-50% no longer needed to use a continuous positive airway pressure (CPAP) device, according to two new studies.
Tirzepatide, a long-acting glucose-dependent insulinotropic polypeptide (GIP) receptor agonist and glucagon-like peptide 1 (GLP-1) receptor agonist, also lowered C-reactive protein levels and systolic blood pressure. And patients taking the medication lost 18%-20% of their body weight.
said lead author Atul Malhotra, MD, professor of medicine at the University of California, San Diego, and director of sleep medicine at UC San Diego Health.
The two double-blind, randomized, controlled trials in patients with obesity and moderate to severe OSA were conducted at 60 sites in nine countries. The results were presented at the American Diabetes Association (ADA) 84th Scientific Sessions and simultaneously published online in the New England Journal of Medicine.
OSA affects 1 billion people worldwide and 30 million American adults, many of whom are undiagnosed. Obesity is a common risk factor. According to the ADA, 40% of those with obesity have OSA and 70% of those with OSA have obesity.
CPAP is an effective and the most-used intervention for OSA, but many patients refuse to use the device, stop using it, or cannot use it. Should tirzepatide eventually gain Food and Drug Administration approval for OSA, it would be the first drug approved for the condition.
“This new drug treatment offers a more accessible alternative for individuals who cannot tolerate or adhere to existing therapies,” said Dr. Malhotra.
Huge Reduction in Episodes, Severity
For the two studies, patients were enrolled who had moderate to severe OSA, defined as more than 15 events per hour (using the apnea-hypopnea index [AHI]) and a body mass index of 30 kg/m2 or greater. Those not using a CPAP device were enrolled in study 1, and those using a CPAP device were enrolled in study 2.
Participants received either the maximum tolerated dose of tirzepatide (10 or 15 mg by once-weekly injection) or placebo for 1 year. In study 1, 114 individuals received tirzepatide and 120 received placebo. For study 2, 119 patients received tirzepatide and 114 received placebo. All participants received regular lifestyle counseling sessions about nutrition and were instructed to reduce food intake by 500 kcal/day and to engage in at least 150 min/week of physical activity.
Enrollment was limited to 70% men to ensure adequate representation of women.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/hour on the AHI scale and a mean of 51.5 events/hour.
By 1 year, patients taking tirzepatide had 27-30 fewer events/hour, compared with 4-6 fewer events/hour for those taking placebo.
Up to half of those who received tirzepatide in both trials had less than 5 events/hour or 5-14 AHI events/hour and an Epworth Sleepiness Scale score of 10 or less. Those thresholds “represent a level at which CPAP therapy may not be recommended,” wrote the authors.
Patients in the tirzepatide group also had a decrease in systolic blood pressure from baseline of 9.7 mm Hg in study 1 and 7.6 mm Hg in study 2 at week 48.
The most common adverse events were diarrhea, nausea, and vomiting, which occurred in approximately a quarter of patients taking tirzepatide. There were two adjudicated-confirmed cases of acute pancreatitis in those taking tirzepatide in study 2.
Patients who received tirzepatide also reported fewer daytime and nighttime disturbances, as measured using the Patient-Reported Outcomes Measurement Information System Short Form scale for Sleep-Related Impairment and Sleep Disturbance.
Tirzepatide Plus CPAP Are Best
Writing in an accompanying editorial, Sanjay R. Patel, MD, noted that, although clinical guidelines have recommended that weight loss strategies be incorporated as part of OSA treatment, “the integration of obesity management into the approaches to care for obstructive sleep apnea has lagged.”
As many as half of patients abandon CPAP therapy within 3 years, wrote Dr. Patel, who is professor of medicine and epidemiology at the University of Pittsburgh, Pittsburgh, Pennsylvania, and medical director of the UPMC Comprehensive Sleep Disorders program. “An effective medication to treat obesity is thus an obvious avenue to pursue.”
Dr. Patel noted the large reductions in the number of events on the AHI scale. He wrote that the improvement in systolic blood pressure “was substantially larger than effects seen with CPAP therapy alone and indicate that tirzepatide may be an attractive option for those patients who seek to reduce their cardiovascular risk.”
Dr. Patel raised concerns about whether patients outside of a trial would stick with therapy, noting studies have shown high rates of discontinuation of GLP-1 receptor agonists.
And, he wrote, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Commenting on the study during the presentation of the results, Louis Aronne, MD, said he believes the trials demonstrate “the treatment of obesity with tirzepatide plus CPAP is really the optimal treatment for obstructive sleep apnea and obesity-related cardiometabolic risks.” Dr. Aronne is the Sanford I. Weill professor of metabolic research at Weill Cornell Medical College, New York City.
Dr. Aronne added there is still much to learn. It is still not clear whether tirzepatide had an independent effect in the OSA trial — as has been seen in other studies where the drug clearly reduced cardiovascular risk — or whether the positive results were primarily caused by weight loss.
“I believe that over time we’ll see that this particular effect in sleep apnea is related to weight,” he said.
The study was supported by Eli Lilly. Dr. Malhotra has reported being a paid consultant for Lilly and ZOLL Medical and a cofounder of Healcisio.
A version of this article appeared on Medscape.com.
ORLANDO, FLA. — The diabetes and weight loss drug tirzepatide (Mounjaro for type 2 diabetes; Zepbound for obesity) was so effective at reducing sleep disruptions in patients with obesity and obstructive sleep apnea (OSA) that 40%-50% no longer needed to use a continuous positive airway pressure (CPAP) device, according to two new studies.
Tirzepatide, a long-acting glucose-dependent insulinotropic polypeptide (GIP) receptor agonist and glucagon-like peptide 1 (GLP-1) receptor agonist, also lowered C-reactive protein levels and systolic blood pressure. And patients taking the medication lost 18%-20% of their body weight.
said lead author Atul Malhotra, MD, professor of medicine at the University of California, San Diego, and director of sleep medicine at UC San Diego Health.
The two double-blind, randomized, controlled trials in patients with obesity and moderate to severe OSA were conducted at 60 sites in nine countries. The results were presented at the American Diabetes Association (ADA) 84th Scientific Sessions and simultaneously published online in the New England Journal of Medicine.
OSA affects 1 billion people worldwide and 30 million American adults, many of whom are undiagnosed. Obesity is a common risk factor. According to the ADA, 40% of those with obesity have OSA and 70% of those with OSA have obesity.
CPAP is an effective and the most-used intervention for OSA, but many patients refuse to use the device, stop using it, or cannot use it. Should tirzepatide eventually gain Food and Drug Administration approval for OSA, it would be the first drug approved for the condition.
“This new drug treatment offers a more accessible alternative for individuals who cannot tolerate or adhere to existing therapies,” said Dr. Malhotra.
Huge Reduction in Episodes, Severity
For the two studies, patients were enrolled who had moderate to severe OSA, defined as more than 15 events per hour (using the apnea-hypopnea index [AHI]) and a body mass index of 30 kg/m2 or greater. Those not using a CPAP device were enrolled in study 1, and those using a CPAP device were enrolled in study 2.
Participants received either the maximum tolerated dose of tirzepatide (10 or 15 mg by once-weekly injection) or placebo for 1 year. In study 1, 114 individuals received tirzepatide and 120 received placebo. For study 2, 119 patients received tirzepatide and 114 received placebo. All participants received regular lifestyle counseling sessions about nutrition and were instructed to reduce food intake by 500 kcal/day and to engage in at least 150 min/week of physical activity.
Enrollment was limited to 70% men to ensure adequate representation of women.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/hour on the AHI scale and a mean of 51.5 events/hour.
By 1 year, patients taking tirzepatide had 27-30 fewer events/hour, compared with 4-6 fewer events/hour for those taking placebo.
Up to half of those who received tirzepatide in both trials had less than 5 events/hour or 5-14 AHI events/hour and an Epworth Sleepiness Scale score of 10 or less. Those thresholds “represent a level at which CPAP therapy may not be recommended,” wrote the authors.
Patients in the tirzepatide group also had a decrease in systolic blood pressure from baseline of 9.7 mm Hg in study 1 and 7.6 mm Hg in study 2 at week 48.
The most common adverse events were diarrhea, nausea, and vomiting, which occurred in approximately a quarter of patients taking tirzepatide. There were two adjudicated-confirmed cases of acute pancreatitis in those taking tirzepatide in study 2.
Patients who received tirzepatide also reported fewer daytime and nighttime disturbances, as measured using the Patient-Reported Outcomes Measurement Information System Short Form scale for Sleep-Related Impairment and Sleep Disturbance.
Tirzepatide Plus CPAP Are Best
Writing in an accompanying editorial, Sanjay R. Patel, MD, noted that, although clinical guidelines have recommended that weight loss strategies be incorporated as part of OSA treatment, “the integration of obesity management into the approaches to care for obstructive sleep apnea has lagged.”
As many as half of patients abandon CPAP therapy within 3 years, wrote Dr. Patel, who is professor of medicine and epidemiology at the University of Pittsburgh, Pittsburgh, Pennsylvania, and medical director of the UPMC Comprehensive Sleep Disorders program. “An effective medication to treat obesity is thus an obvious avenue to pursue.”
Dr. Patel noted the large reductions in the number of events on the AHI scale. He wrote that the improvement in systolic blood pressure “was substantially larger than effects seen with CPAP therapy alone and indicate that tirzepatide may be an attractive option for those patients who seek to reduce their cardiovascular risk.”
Dr. Patel raised concerns about whether patients outside of a trial would stick with therapy, noting studies have shown high rates of discontinuation of GLP-1 receptor agonists.
And, he wrote, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Commenting on the study during the presentation of the results, Louis Aronne, MD, said he believes the trials demonstrate “the treatment of obesity with tirzepatide plus CPAP is really the optimal treatment for obstructive sleep apnea and obesity-related cardiometabolic risks.” Dr. Aronne is the Sanford I. Weill professor of metabolic research at Weill Cornell Medical College, New York City.
Dr. Aronne added there is still much to learn. It is still not clear whether tirzepatide had an independent effect in the OSA trial — as has been seen in other studies where the drug clearly reduced cardiovascular risk — or whether the positive results were primarily caused by weight loss.
“I believe that over time we’ll see that this particular effect in sleep apnea is related to weight,” he said.
The study was supported by Eli Lilly. Dr. Malhotra has reported being a paid consultant for Lilly and ZOLL Medical and a cofounder of Healcisio.
A version of this article appeared on Medscape.com.
ORLANDO, FLA. — The diabetes and weight loss drug tirzepatide (Mounjaro for type 2 diabetes; Zepbound for obesity) was so effective at reducing sleep disruptions in patients with obesity and obstructive sleep apnea (OSA) that 40%-50% no longer needed to use a continuous positive airway pressure (CPAP) device, according to two new studies.
Tirzepatide, a long-acting glucose-dependent insulinotropic polypeptide (GIP) receptor agonist and glucagon-like peptide 1 (GLP-1) receptor agonist, also lowered C-reactive protein levels and systolic blood pressure. And patients taking the medication lost 18%-20% of their body weight.
said lead author Atul Malhotra, MD, professor of medicine at the University of California, San Diego, and director of sleep medicine at UC San Diego Health.
The two double-blind, randomized, controlled trials in patients with obesity and moderate to severe OSA were conducted at 60 sites in nine countries. The results were presented at the American Diabetes Association (ADA) 84th Scientific Sessions and simultaneously published online in the New England Journal of Medicine.
OSA affects 1 billion people worldwide and 30 million American adults, many of whom are undiagnosed. Obesity is a common risk factor. According to the ADA, 40% of those with obesity have OSA and 70% of those with OSA have obesity.
CPAP is an effective and the most-used intervention for OSA, but many patients refuse to use the device, stop using it, or cannot use it. Should tirzepatide eventually gain Food and Drug Administration approval for OSA, it would be the first drug approved for the condition.
“This new drug treatment offers a more accessible alternative for individuals who cannot tolerate or adhere to existing therapies,” said Dr. Malhotra.
Huge Reduction in Episodes, Severity
For the two studies, patients were enrolled who had moderate to severe OSA, defined as more than 15 events per hour (using the apnea-hypopnea index [AHI]) and a body mass index of 30 kg/m2 or greater. Those not using a CPAP device were enrolled in study 1, and those using a CPAP device were enrolled in study 2.
Participants received either the maximum tolerated dose of tirzepatide (10 or 15 mg by once-weekly injection) or placebo for 1 year. In study 1, 114 individuals received tirzepatide and 120 received placebo. For study 2, 119 patients received tirzepatide and 114 received placebo. All participants received regular lifestyle counseling sessions about nutrition and were instructed to reduce food intake by 500 kcal/day and to engage in at least 150 min/week of physical activity.
Enrollment was limited to 70% men to ensure adequate representation of women.
At baseline, 65%-70% of participants had severe OSA, with more than 30 events/hour on the AHI scale and a mean of 51.5 events/hour.
By 1 year, patients taking tirzepatide had 27-30 fewer events/hour, compared with 4-6 fewer events/hour for those taking placebo.
Up to half of those who received tirzepatide in both trials had less than 5 events/hour or 5-14 AHI events/hour and an Epworth Sleepiness Scale score of 10 or less. Those thresholds “represent a level at which CPAP therapy may not be recommended,” wrote the authors.
Patients in the tirzepatide group also had a decrease in systolic blood pressure from baseline of 9.7 mm Hg in study 1 and 7.6 mm Hg in study 2 at week 48.
The most common adverse events were diarrhea, nausea, and vomiting, which occurred in approximately a quarter of patients taking tirzepatide. There were two adjudicated-confirmed cases of acute pancreatitis in those taking tirzepatide in study 2.
Patients who received tirzepatide also reported fewer daytime and nighttime disturbances, as measured using the Patient-Reported Outcomes Measurement Information System Short Form scale for Sleep-Related Impairment and Sleep Disturbance.
Tirzepatide Plus CPAP Are Best
Writing in an accompanying editorial, Sanjay R. Patel, MD, noted that, although clinical guidelines have recommended that weight loss strategies be incorporated as part of OSA treatment, “the integration of obesity management into the approaches to care for obstructive sleep apnea has lagged.”
As many as half of patients abandon CPAP therapy within 3 years, wrote Dr. Patel, who is professor of medicine and epidemiology at the University of Pittsburgh, Pittsburgh, Pennsylvania, and medical director of the UPMC Comprehensive Sleep Disorders program. “An effective medication to treat obesity is thus an obvious avenue to pursue.”
Dr. Patel noted the large reductions in the number of events on the AHI scale. He wrote that the improvement in systolic blood pressure “was substantially larger than effects seen with CPAP therapy alone and indicate that tirzepatide may be an attractive option for those patients who seek to reduce their cardiovascular risk.”
Dr. Patel raised concerns about whether patients outside of a trial would stick with therapy, noting studies have shown high rates of discontinuation of GLP-1 receptor agonists.
And, he wrote, “racial disparities in the use of GLP-1 receptor agonists among patients with diabetes arouse concern that the addition of tirzepatide as a treatment option for obstructive sleep apnea without directly addressing policies relative to coverage of care will only further exacerbate already pervasive disparities in clinical care for obstructive sleep apnea.”
Commenting on the study during the presentation of the results, Louis Aronne, MD, said he believes the trials demonstrate “the treatment of obesity with tirzepatide plus CPAP is really the optimal treatment for obstructive sleep apnea and obesity-related cardiometabolic risks.” Dr. Aronne is the Sanford I. Weill professor of metabolic research at Weill Cornell Medical College, New York City.
Dr. Aronne added there is still much to learn. It is still not clear whether tirzepatide had an independent effect in the OSA trial — as has been seen in other studies where the drug clearly reduced cardiovascular risk — or whether the positive results were primarily caused by weight loss.
“I believe that over time we’ll see that this particular effect in sleep apnea is related to weight,” he said.
The study was supported by Eli Lilly. Dr. Malhotra has reported being a paid consultant for Lilly and ZOLL Medical and a cofounder of Healcisio.
A version of this article appeared on Medscape.com.
FROM ADA 2024
