User login
Smoking Cessation Offers Benefits at Any Age
This transcript has been edited for clarity.
I would like to briefly talk about a very interesting paper and one that probably has about as much to inform the doctor-patient relationship as any paper you can think of.
The title itself gives you a little bit of that answer before I even discuss the outcome. The paper is “The Benefits of Quitting Smoking at Different Ages,” recently published in The American Journal of Preventive Medicine.
I’m not going to even begin to attempt to explore the statistics of the analysis, but I think the conclusions are both fascinating and important. I will read the first sentence of the results and then just comment on some of the others because there’s just so much data here and I really want to focus on the punchline.
The results section said that, compared with people who never smoked, those who smoke currently, aged 35, 45, 55, 65, or 75, (those were all the groups they looked at), and who have smoked throughout adulthood until that age will lose an average of 9.1, 8.3, 7.3, 5.9, and 4.4 years of life, respectively — obviously, it’s a lot — if they continue to smoke for the rest of their lives.
We know that. It’s terrible. That’s why people should never smoke. Period. End of story. There’s no social value. There’s no health value of smoking. It’s a deadly recreational activity for multiple illnesses, and obviously, cancer is prominent among them.
Here’s the conclusion of the paper that I think is interesting. That doctor, whether it’s a primary care doctor, an oncologist, an ob/gyn, or a family doctor, is seeing Mr Smith or Mrs Jones in the office today, whether they know that patient well or not very well, and they’re still smoking. However, if the person we’re describing here quits smoking at these ages, how much life do they add back, compared with if they continued?
They may say: “Oh, I’ve been smoking all my life. What difference does it make? The die is cast.” Wrong! If you’ve been smoking your whole adult life — so let’s just say that you started at age 18, age 20, age 15, or even age 12 — but you quit smoking at the age of 35, you’re going to add 8 years of life on average. If you quit smoking when you’re 65, having smoked your whole adult life, you will add 1.7 years of life. That’s 1.7 years to be with your family, to be with your grandchildren, and enjoy life. If you ask, “Oh, what difference does it make?” It makes a big difference.
I’ll share another statistic and I’ll be done. I think this is really an interesting one. The chances of gaining at least a year of life among those who quit smoking at the age of 65 was 23.4%. There is a 1 out of 4 chance that you’re going to live an additional year if you stop at age 65. Even if you stop smoking at age 75, you have a 14% chance of living at least an additional year longer than you would have if you didn’t stop smoking.
There is much to think about here, much to consider, and much to discuss potentially with patients.
Dr. Markman is Professor of Medical Oncology and Therapeutics Research, City of Hope Comprehensive Cancer Center; President, Medicine & Science, City of Hope Atlanta, Chicago, Phoenix. He reported conflicts of interest with GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I would like to briefly talk about a very interesting paper and one that probably has about as much to inform the doctor-patient relationship as any paper you can think of.
The title itself gives you a little bit of that answer before I even discuss the outcome. The paper is “The Benefits of Quitting Smoking at Different Ages,” recently published in The American Journal of Preventive Medicine.
I’m not going to even begin to attempt to explore the statistics of the analysis, but I think the conclusions are both fascinating and important. I will read the first sentence of the results and then just comment on some of the others because there’s just so much data here and I really want to focus on the punchline.
The results section said that, compared with people who never smoked, those who smoke currently, aged 35, 45, 55, 65, or 75, (those were all the groups they looked at), and who have smoked throughout adulthood until that age will lose an average of 9.1, 8.3, 7.3, 5.9, and 4.4 years of life, respectively — obviously, it’s a lot — if they continue to smoke for the rest of their lives.
We know that. It’s terrible. That’s why people should never smoke. Period. End of story. There’s no social value. There’s no health value of smoking. It’s a deadly recreational activity for multiple illnesses, and obviously, cancer is prominent among them.
Here’s the conclusion of the paper that I think is interesting. That doctor, whether it’s a primary care doctor, an oncologist, an ob/gyn, or a family doctor, is seeing Mr Smith or Mrs Jones in the office today, whether they know that patient well or not very well, and they’re still smoking. However, if the person we’re describing here quits smoking at these ages, how much life do they add back, compared with if they continued?
They may say: “Oh, I’ve been smoking all my life. What difference does it make? The die is cast.” Wrong! If you’ve been smoking your whole adult life — so let’s just say that you started at age 18, age 20, age 15, or even age 12 — but you quit smoking at the age of 35, you’re going to add 8 years of life on average. If you quit smoking when you’re 65, having smoked your whole adult life, you will add 1.7 years of life. That’s 1.7 years to be with your family, to be with your grandchildren, and enjoy life. If you ask, “Oh, what difference does it make?” It makes a big difference.
I’ll share another statistic and I’ll be done. I think this is really an interesting one. The chances of gaining at least a year of life among those who quit smoking at the age of 65 was 23.4%. There is a 1 out of 4 chance that you’re going to live an additional year if you stop at age 65. Even if you stop smoking at age 75, you have a 14% chance of living at least an additional year longer than you would have if you didn’t stop smoking.
There is much to think about here, much to consider, and much to discuss potentially with patients.
Dr. Markman is Professor of Medical Oncology and Therapeutics Research, City of Hope Comprehensive Cancer Center; President, Medicine & Science, City of Hope Atlanta, Chicago, Phoenix. He reported conflicts of interest with GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I would like to briefly talk about a very interesting paper and one that probably has about as much to inform the doctor-patient relationship as any paper you can think of.
The title itself gives you a little bit of that answer before I even discuss the outcome. The paper is “The Benefits of Quitting Smoking at Different Ages,” recently published in The American Journal of Preventive Medicine.
I’m not going to even begin to attempt to explore the statistics of the analysis, but I think the conclusions are both fascinating and important. I will read the first sentence of the results and then just comment on some of the others because there’s just so much data here and I really want to focus on the punchline.
The results section said that, compared with people who never smoked, those who smoke currently, aged 35, 45, 55, 65, or 75, (those were all the groups they looked at), and who have smoked throughout adulthood until that age will lose an average of 9.1, 8.3, 7.3, 5.9, and 4.4 years of life, respectively — obviously, it’s a lot — if they continue to smoke for the rest of their lives.
We know that. It’s terrible. That’s why people should never smoke. Period. End of story. There’s no social value. There’s no health value of smoking. It’s a deadly recreational activity for multiple illnesses, and obviously, cancer is prominent among them.
Here’s the conclusion of the paper that I think is interesting. That doctor, whether it’s a primary care doctor, an oncologist, an ob/gyn, or a family doctor, is seeing Mr Smith or Mrs Jones in the office today, whether they know that patient well or not very well, and they’re still smoking. However, if the person we’re describing here quits smoking at these ages, how much life do they add back, compared with if they continued?
They may say: “Oh, I’ve been smoking all my life. What difference does it make? The die is cast.” Wrong! If you’ve been smoking your whole adult life — so let’s just say that you started at age 18, age 20, age 15, or even age 12 — but you quit smoking at the age of 35, you’re going to add 8 years of life on average. If you quit smoking when you’re 65, having smoked your whole adult life, you will add 1.7 years of life. That’s 1.7 years to be with your family, to be with your grandchildren, and enjoy life. If you ask, “Oh, what difference does it make?” It makes a big difference.
I’ll share another statistic and I’ll be done. I think this is really an interesting one. The chances of gaining at least a year of life among those who quit smoking at the age of 65 was 23.4%. There is a 1 out of 4 chance that you’re going to live an additional year if you stop at age 65. Even if you stop smoking at age 75, you have a 14% chance of living at least an additional year longer than you would have if you didn’t stop smoking.
There is much to think about here, much to consider, and much to discuss potentially with patients.
Dr. Markman is Professor of Medical Oncology and Therapeutics Research, City of Hope Comprehensive Cancer Center; President, Medicine & Science, City of Hope Atlanta, Chicago, Phoenix. He reported conflicts of interest with GlaxoSmithKline and AstraZeneca.
A version of this article first appeared on Medscape.com.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
Introduction
Large cell neuroendocrine carcinomas (LCNEC) of the lung are sufficiently rare that large trials to establish a standard of care are impractical. Treatment strategies effective for related malignancies, particularly small-cell lung cancer (SCLC), have been commonly applied to LCNEC of the lung, but it is important to recognize the unique features of LCNEC in order to make a diagnosis and to individualize treatment. As current long-term survival in patients with LCNEC of the lung remains poor, participation in clinical trials should be encouraged. Therapies under investigation include those targeted at the delta-like ligand 3 (DLL3), an antigen highly expressed in neuroendocrine (NE) tumors, and Seneca Valley oncolytic viral (SVV) therapy. Early introduction of palliative care should also be offered to optimize quality of life. High-quality data for LCNEC of the lung and novel breakthrough drugs are much needed.
Background
NE tumors can develop from NE cells in almost any organ.1 After the gastrointestinal tract, the lung is the most common site of NE malignancies. They account for only about 2% of all lung cancers but 25% of NE tumors.2 Criteria for differentiating NE tumors from other tumors in the lung were first proposed in 1991.3 In 2022, the World Health Organization described 5 major subtypes of NE lung malignancies.4 On a spectrum ranging from best to worst outcome among lung cancers, LCNEC has a significantly more aggressive course compared with typical carcinoids (TC) and atypical carcinoids (AC), approaching that of SCLC, which arguably has the worst outcome (Table).5
Table. Comparing NSCLC, SCLC, and LCNEC of the Lung
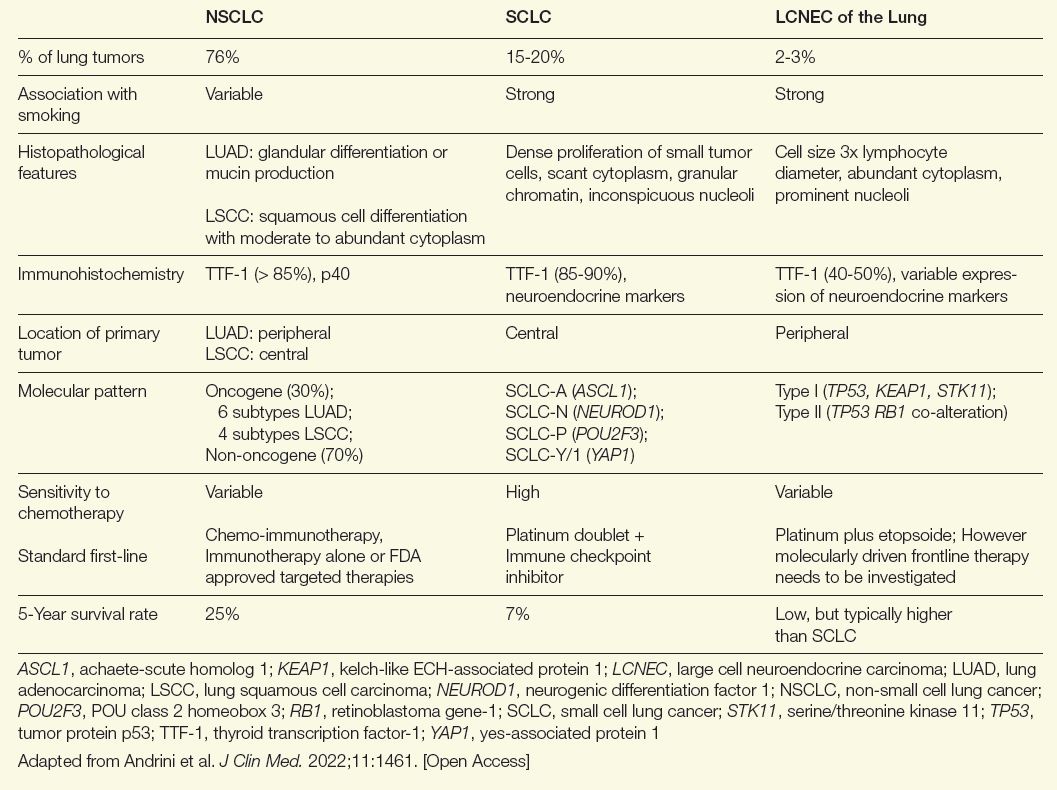
Similarities exist between LCNEC of the lung and other non-small cell lung cancer (NSCLC) types, but there are more parallels with SCLC. Both are more common in male patients and both are associated with a history of smoking.6 They also share a poor prognosis. If diagnosed at an advanced stage, 5-year survival rates for LCNEC of the lung and SCLC have been reported to be as low as 5% to 15%.6
The risk of a delay in establishing the correct diagnosis of LCNEC of the lung, even by experienced pathologists, is considerable. The key diagnostic criteria include expression of at least 1 NE marker, such as chromogranin-A or synaptophysin, a high proliferation rate (> 10 mitoses per high-power field), extensive necrosis, and NE morphology features, such as trabeculae and palisading and rosette formations.7 However, other lung cancers can also express NE markers and some features might be missed without relatively large tissue specimens.7
To improve diagnostic accuracy, additional criteria, such as absence of squamous or adenocarcinoma features or the demonstration of 2 or more NE markers are now being advocated in some reports,8 while others have advocated that terms such as “combined NSCLC/SCLC” should not be accepted as a substitute for differentiating and finalizing a diagnosis of LCNEC of the lung.7 Excisional or resection biopsies, as opposed to needle biopsies, might be required to obtain an adequate tissue sample to reach a definitive diagnosis.
Illustrating the potential for confusion with other lung cancers, LCNEC of the lung can be characterized by 2 subtypes.9 Type 1 is characterized by TP53 and STK11/KEAP1 alternations—similar to adenocarcinomas and squamous cell lung cancers—and it is associated with a higher expression of NE markers, such as ASCL1 and DLL3. Type 2 is typically characterized by inactivation of TP53 and RB1. Ultimately, type I LCNEC of the lung has a mutational pattern similar to NSCLC and type II has a pattern similar to SCLC. While LCNEC is typically located in the periphery of the lung, SCLC is typically centrally located and NSCLC can be found in either location. Complicated further by the fact that a proportion of these tumors have features shared with SCLC and rarer cancers, such as spindle-cell carcinoma and giant cell carcinoma, LCNEC should be considered in the differential diagnosis of any lung cancer with ambiguous features.7
For these reasons, a pathology review should be performed at a high-volume center whenever possible. As part of the diagnostic process, molecular testing should be gathered for all patients whether or not it is required to make or confirm the diagnosis. This information will be informative for guiding treatment, particularly second- and third-line interventions. Rather than being unique and definitive, the individual features of LCNEC of the lung—including the genetic, molecular, histologic, and morphologic characteristics—cumulatively support the diagnosis. After establishing a pathological diagnosis, staging of LCNEC of the lung is paramount. In addition, distinctions between the grades of LCNEC of the lung are relative. For example, tumors with a better relative prognosis typically have fewer gene mutations than tumors with a worse relative prognosis, but mutations are generally found in both.
Bronchoscopy with endobronchial ultrasound can be considered for both diagnosis and staging of locally advanced tumors, but a surgical specimen might still be required for a definitive diagnosis. Differentiating local LCNEC, which has been reported in about 25% of cases, from locally advanced and metastatic disease is critical for planning treatment. Fluorodeoxyglucose F18 (FDG) positron emission tomography (PET) plays an important role in staging LCNEC of the lung. Unlike TC and AC, for LCNEC of the lung there is a very limited role of somatostatin receptor agonist-based imaging or tetraazacyclododecanetetraacetic acid-DPhel-Tyr3-octreotate (DOTATATE) PET during diagnostic workup.
Therapeutic Strategies
In early stages, resection followed by adjuvant chemotherapy has long been used for LCNEC of the lung. Studies evaluating this approach, such as one that combined cisplatin and etoposide,10 suggest doublet chemotherapy after surgery offers a benefit in LCNEC of the lung comparable to that seen in SCLC. There is limited support for adjunctive radiotherapy in early-stage LCNEC of the lung,5 even if radiotherapy has shown benefit for patients ineligible for surgery.11
In locally advanced and advanced LCNEC (≥ stage III-B) ineligible for resection, chemoradiation has been associated with a survival advantage over chemotherapy alone,12 but due to the high rates of relapse and limited survival, efforts to move to novel therapies have been expanding for both LCNEC of the lung and SCLC. This includes immunotherapies used before or after chemoradiation. Again, much of the interest in immunotherapies has been derived from studies in SCLC, but several small studies have associated checkpoint inhibitors with substantial antitumor activity in patients with LCNEC.13,14 There are no large scale prospective trials to determine the optimal treatment in the first line setting for LCNEC of the lung and most data is extrapolated from treatment of ES-SCLC. In a retrospective study, however, comparing survival of palliative chemotherapy with a SCLC versus a NSCLC regimen, the SCLC regimen was favored.15
Following a pivotal trial of tarlatamab-dlle, that led to an accelerated approval for extensive-stage SCLC in May 2024,16 this drug has also been evaluated in a small group of patients with LCNEC of the lung. The parallels between LCNEC and SCLC have raised hope that this drug, which is a bispecific T-cell engager (BiTE) that binds to the DLL3 ligand and CD3, may provide benefit in LCNEC of the lung that is commensurate with the benefit seen in SCLC. A recently published LCNEC case study supports this potential.17 A high-grade NE-carcinoma-specific oncolytic virus called Seneca Valley virus holds promise. Preclinical data suggest encouraging anticancer activity when SVV is combined with immune checkpoint inhibitor therapy.18 SVV seems to attack cancer cells that express tumor endothelial marker 8 (TEM-8), making it an interesting target in future efforts for screening and tailoring treatment.19 Human studies are in development.
Due to the high frequency of relapse regardless of frontline therapies, there is also growing interest in maintenance strategies to extend disease control. Maintenance regimens that have been evaluated or are being considered include immunotherapies, even if the optimal sequence of treatment modalities remains unknown. The high rate of relapse also encourages early planning of sequential therapies based on molecular testing. Numerous studies of LCNEC of the lung have now identified activating mutations in targetable pathways, such as P13K/AKT/mTOR, KRAS, and FGFR1.18 Patients may also harbor a high tumor mutation burden, a characteristic that might favor treatment with immunotherapy. Each mutation is relevant to only a small proportion of patients with LCNEC. However, when all potentially targetable mutations are considered together, the proportion of patients with LCNEC who would benefit from an individualized therapy is substantial, thus supporting trials of individualized therapy, particularly in the second line.
The high rate of relapse with currently available therapies encourages enrollment in clinical trials, particularly among patients who have already failed a first-line strategy. In the United States, studies are enrolling patients with LCNEC of the lung for checkpoint inhibitors with or without combination chemotherapy, novel BiTE therapies, and novel therapies targeting specific activating pathways. Many of these trials offer enrollment to patients with either SCLC or LCNEC.
Due to poor survival, patients with advancing LCNEC of the lung should be considered for palliative care. Although no guideline protocol exists for palliative care, the American Society of Clinical Oncology recommends palliative care for all individuals with advanced cancer based on evidence of improved quality of life and, in some cases, survival.20
Summary
LCNEC is an uncommon lung malignancy with a poor prognosis in the advanced stages at which it is most often recognized. The risk of overlooking this cancer in the initial diagnosis emphasizes the need for an adequate index of suspicion and familiarity with its distinguishing characteristics. Treatments of LCNEC of the lung have been largely based on those used for SCLC, but there has been an evolution in the understanding of this disease, which includes a greater appreciation for heterogeneity among driving mutations, a growing interest in maintenance therapies to extend the time to relapse, and trials of a growing array of novel therapies, including immunotherapies and BiTEs. Early intervention with these novel therapies and an emphasis on palliative care is needed because LCNEC has such an aggressive course.
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
- Sultana Q, Kar J, Verma A, et al. A comprehensive review on neuroendocrine neoplasms: presentation, pathophysiology and management. J Clin Med. 2023;12(15):5138. doi:10.3390/jcm12155138
- Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2008;113(1):5-21. doi:10.1002/cncr.23542
- Travis WD, Linnoila RI, Tsokos MG, et al. Neuroendocrine tumors of the lung with proposed criteria for large-cell neuroendocrine carcinoma. An ultrastructural, immunohistochemical, and flow cytometric study of 35 cases. Am J Surg Pathol. 1991;15(6):529-553. doi:10.1097/00000478-199106000-00003
- Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG. Introduction to the 2015 World Health Organization classification of tumors of the lung, pleura, thymus, and heart. J Thorac Oncol. 2015;10(9):1240-1242. doi:10.1097/JTO.0000000000000663
- Andrini E, Marchese PV, De Biase D, et al. Large cell neuroendocrine carcinoma of the lung: current understanding and challenges. J Clin Med. 2022;11(5):1461. doi:10.3390/jcm11051461
- Lantuejoul S, Fernandez-Cuesta L, Damiola F, Girard N, McLeer A. New molecular classification of large cell neuroendocrine carcinoma and small cell lung carcinoma with potential therapeutic impacts. Transl Lung Cancer Res. 2020;9(5):2233-2244. doi:10.21037/tlcr-20-269
- Lindsay CR, Shaw EC, Moore DA, et al. Large cell neuroendocrine lung carcinoma: consensus statement from The British Thoracic Oncology Group and the Association of Pulmonary Pathologists. Br J Cancer. 2021;125(9):1210-1216. doi:10.1038/s41416-021-01407-9
- Derks JL, Dingemans AC, van Suylen RJ, et al. Is the sum of positive neuroendocrine immunohistochemical stains useful for diagnosis of large cell neuroendocrine carcinoma (LCNEC) on biopsy specimens? Histopathology. 2019;74(4):555-566. doi:10.1111/his.13800
- George J, Walter V, Peifer M, et al. Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. Nat Commun. 2018;9(1):1048. doi:10.1038/s41467-018-03099-x
- Iyoda A, Hiroshima K, Moriya Y, et al. Prospective study of adjuvant chemotherapy for pulmonary large cell neuroendocrine carcinoma. Ann Thorac Surg. 2006;82(5):1802-1807. doi:10.1016/j.athoracsur.2006.05.109
- Cao L, Wu HF, Zhao L, et al. The role of radiotherapy in pulmonary large cell neuroendocrine carcinoma: propensity score matching analysis. J Radiat Res. 2020;61(4):594-601. doi:10.1093/jrr/rraa036
- Limonnik V, Abel S, Finley GG, Long GS, Wegner RE. Factors associated with treatment receipt and overall survival for patients with locally advanced large cell neuroendocrine carcinoma of the lung: a National Cancer Database analysis. Lung Cancer. 2020;150:107-113. doi:10.1016/j.lungcan.2020.10.001
- Shi Z, Wei J, Xu M, Song Z. Efficacy and safety of immune checkpoint inhibitors in lung large-cell neuroendocrine carcinoma. J Thorac Dis. 2023;15(8):4172-4181. doi:10.21037/jtd-23-348
- Chauhan A, Arnold SM, Kolesar J, Thomas HE, Evers M, Anthony L. Immune checkpoint inhibitors in large cell neuroendocrine carcinoma: current status. Oncotarget. 2018;9(18):14738-14740. doi:10.18632/oncotarget.24553
- Chen H, Ishihara M, Horita N, et al. Effect of adjuvant and palliative chemotherapy in large cell neuroendocrine carcinoma of the lung: a systematic review and metaanalysis. Cancers (Basel). 2021;13(23):5948. doi:10.3390/cancers13235948
- Ahn MJ, Cho BC, Felip E, et al. Tarlatamab for patients with previously treated small-cell lung cancer. N Engl J Med. 2023;389(22):2063-2075. doi:10.1056/NEJMoa2307980
- Patel SA, Whang Y, Medley C, et al. Tartalamab for large-cell neuroendocrine carcinoma in a young adult: a case report. JTO Clin Res Rep. 2024;5(10):100712. doi:10.1016/j.jtocrr.2024.100712
- Corbett V, Hallenbeck P, Rychahou P, Chauhan A. Evolving role of Seneca Valley virus and its biomarker TEM8/ANTXR1 in cancer therapeutics. Front Mol Biosci. 2022;9:930207. doi:10.3389/fmolb.2022.930207
- Kareff SA, Corbett V, Hallenbeck P, Chauhan A. TEM8 in oncogenesis: protein biology, pre-clinical agents, and clinical rationale. Cells. 2023;12(22):2623. doi:10.3390/cells12222623
- Sanders JJ, Temin S, Ghoshal A, et al. Palliative care for patients with cancer: ASCO guideline update. J Clin Oncol. 2024;42(19):2336-2357. doi:10.1200/JCO.24.00542
Introduction
Large cell neuroendocrine carcinomas (LCNEC) of the lung are sufficiently rare that large trials to establish a standard of care are impractical. Treatment strategies effective for related malignancies, particularly small-cell lung cancer (SCLC), have been commonly applied to LCNEC of the lung, but it is important to recognize the unique features of LCNEC in order to make a diagnosis and to individualize treatment. As current long-term survival in patients with LCNEC of the lung remains poor, participation in clinical trials should be encouraged. Therapies under investigation include those targeted at the delta-like ligand 3 (DLL3), an antigen highly expressed in neuroendocrine (NE) tumors, and Seneca Valley oncolytic viral (SVV) therapy. Early introduction of palliative care should also be offered to optimize quality of life. High-quality data for LCNEC of the lung and novel breakthrough drugs are much needed.
Background
NE tumors can develop from NE cells in almost any organ.1 After the gastrointestinal tract, the lung is the most common site of NE malignancies. They account for only about 2% of all lung cancers but 25% of NE tumors.2 Criteria for differentiating NE tumors from other tumors in the lung were first proposed in 1991.3 In 2022, the World Health Organization described 5 major subtypes of NE lung malignancies.4 On a spectrum ranging from best to worst outcome among lung cancers, LCNEC has a significantly more aggressive course compared with typical carcinoids (TC) and atypical carcinoids (AC), approaching that of SCLC, which arguably has the worst outcome (Table).5
Table. Comparing NSCLC, SCLC, and LCNEC of the Lung

Similarities exist between LCNEC of the lung and other non-small cell lung cancer (NSCLC) types, but there are more parallels with SCLC. Both are more common in male patients and both are associated with a history of smoking.6 They also share a poor prognosis. If diagnosed at an advanced stage, 5-year survival rates for LCNEC of the lung and SCLC have been reported to be as low as 5% to 15%.6
The risk of a delay in establishing the correct diagnosis of LCNEC of the lung, even by experienced pathologists, is considerable. The key diagnostic criteria include expression of at least 1 NE marker, such as chromogranin-A or synaptophysin, a high proliferation rate (> 10 mitoses per high-power field), extensive necrosis, and NE morphology features, such as trabeculae and palisading and rosette formations.7 However, other lung cancers can also express NE markers and some features might be missed without relatively large tissue specimens.7
To improve diagnostic accuracy, additional criteria, such as absence of squamous or adenocarcinoma features or the demonstration of 2 or more NE markers are now being advocated in some reports,8 while others have advocated that terms such as “combined NSCLC/SCLC” should not be accepted as a substitute for differentiating and finalizing a diagnosis of LCNEC of the lung.7 Excisional or resection biopsies, as opposed to needle biopsies, might be required to obtain an adequate tissue sample to reach a definitive diagnosis.
Illustrating the potential for confusion with other lung cancers, LCNEC of the lung can be characterized by 2 subtypes.9 Type 1 is characterized by TP53 and STK11/KEAP1 alternations—similar to adenocarcinomas and squamous cell lung cancers—and it is associated with a higher expression of NE markers, such as ASCL1 and DLL3. Type 2 is typically characterized by inactivation of TP53 and RB1. Ultimately, type I LCNEC of the lung has a mutational pattern similar to NSCLC and type II has a pattern similar to SCLC. While LCNEC is typically located in the periphery of the lung, SCLC is typically centrally located and NSCLC can be found in either location. Complicated further by the fact that a proportion of these tumors have features shared with SCLC and rarer cancers, such as spindle-cell carcinoma and giant cell carcinoma, LCNEC should be considered in the differential diagnosis of any lung cancer with ambiguous features.7
For these reasons, a pathology review should be performed at a high-volume center whenever possible. As part of the diagnostic process, molecular testing should be gathered for all patients whether or not it is required to make or confirm the diagnosis. This information will be informative for guiding treatment, particularly second- and third-line interventions. Rather than being unique and definitive, the individual features of LCNEC of the lung—including the genetic, molecular, histologic, and morphologic characteristics—cumulatively support the diagnosis. After establishing a pathological diagnosis, staging of LCNEC of the lung is paramount. In addition, distinctions between the grades of LCNEC of the lung are relative. For example, tumors with a better relative prognosis typically have fewer gene mutations than tumors with a worse relative prognosis, but mutations are generally found in both.
Bronchoscopy with endobronchial ultrasound can be considered for both diagnosis and staging of locally advanced tumors, but a surgical specimen might still be required for a definitive diagnosis. Differentiating local LCNEC, which has been reported in about 25% of cases, from locally advanced and metastatic disease is critical for planning treatment. Fluorodeoxyglucose F18 (FDG) positron emission tomography (PET) plays an important role in staging LCNEC of the lung. Unlike TC and AC, for LCNEC of the lung there is a very limited role of somatostatin receptor agonist-based imaging or tetraazacyclododecanetetraacetic acid-DPhel-Tyr3-octreotate (DOTATATE) PET during diagnostic workup.
Therapeutic Strategies
In early stages, resection followed by adjuvant chemotherapy has long been used for LCNEC of the lung. Studies evaluating this approach, such as one that combined cisplatin and etoposide,10 suggest doublet chemotherapy after surgery offers a benefit in LCNEC of the lung comparable to that seen in SCLC. There is limited support for adjunctive radiotherapy in early-stage LCNEC of the lung,5 even if radiotherapy has shown benefit for patients ineligible for surgery.11
In locally advanced and advanced LCNEC (≥ stage III-B) ineligible for resection, chemoradiation has been associated with a survival advantage over chemotherapy alone,12 but due to the high rates of relapse and limited survival, efforts to move to novel therapies have been expanding for both LCNEC of the lung and SCLC. This includes immunotherapies used before or after chemoradiation. Again, much of the interest in immunotherapies has been derived from studies in SCLC, but several small studies have associated checkpoint inhibitors with substantial antitumor activity in patients with LCNEC.13,14 There are no large scale prospective trials to determine the optimal treatment in the first line setting for LCNEC of the lung and most data is extrapolated from treatment of ES-SCLC. In a retrospective study, however, comparing survival of palliative chemotherapy with a SCLC versus a NSCLC regimen, the SCLC regimen was favored.15
Following a pivotal trial of tarlatamab-dlle, that led to an accelerated approval for extensive-stage SCLC in May 2024,16 this drug has also been evaluated in a small group of patients with LCNEC of the lung. The parallels between LCNEC and SCLC have raised hope that this drug, which is a bispecific T-cell engager (BiTE) that binds to the DLL3 ligand and CD3, may provide benefit in LCNEC of the lung that is commensurate with the benefit seen in SCLC. A recently published LCNEC case study supports this potential.17 A high-grade NE-carcinoma-specific oncolytic virus called Seneca Valley virus holds promise. Preclinical data suggest encouraging anticancer activity when SVV is combined with immune checkpoint inhibitor therapy.18 SVV seems to attack cancer cells that express tumor endothelial marker 8 (TEM-8), making it an interesting target in future efforts for screening and tailoring treatment.19 Human studies are in development.
Due to the high frequency of relapse regardless of frontline therapies, there is also growing interest in maintenance strategies to extend disease control. Maintenance regimens that have been evaluated or are being considered include immunotherapies, even if the optimal sequence of treatment modalities remains unknown. The high rate of relapse also encourages early planning of sequential therapies based on molecular testing. Numerous studies of LCNEC of the lung have now identified activating mutations in targetable pathways, such as P13K/AKT/mTOR, KRAS, and FGFR1.18 Patients may also harbor a high tumor mutation burden, a characteristic that might favor treatment with immunotherapy. Each mutation is relevant to only a small proportion of patients with LCNEC. However, when all potentially targetable mutations are considered together, the proportion of patients with LCNEC who would benefit from an individualized therapy is substantial, thus supporting trials of individualized therapy, particularly in the second line.
The high rate of relapse with currently available therapies encourages enrollment in clinical trials, particularly among patients who have already failed a first-line strategy. In the United States, studies are enrolling patients with LCNEC of the lung for checkpoint inhibitors with or without combination chemotherapy, novel BiTE therapies, and novel therapies targeting specific activating pathways. Many of these trials offer enrollment to patients with either SCLC or LCNEC.
Due to poor survival, patients with advancing LCNEC of the lung should be considered for palliative care. Although no guideline protocol exists for palliative care, the American Society of Clinical Oncology recommends palliative care for all individuals with advanced cancer based on evidence of improved quality of life and, in some cases, survival.20
Summary
LCNEC is an uncommon lung malignancy with a poor prognosis in the advanced stages at which it is most often recognized. The risk of overlooking this cancer in the initial diagnosis emphasizes the need for an adequate index of suspicion and familiarity with its distinguishing characteristics. Treatments of LCNEC of the lung have been largely based on those used for SCLC, but there has been an evolution in the understanding of this disease, which includes a greater appreciation for heterogeneity among driving mutations, a growing interest in maintenance therapies to extend the time to relapse, and trials of a growing array of novel therapies, including immunotherapies and BiTEs. Early intervention with these novel therapies and an emphasis on palliative care is needed because LCNEC has such an aggressive course.
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
Introduction
Large cell neuroendocrine carcinomas (LCNEC) of the lung are sufficiently rare that large trials to establish a standard of care are impractical. Treatment strategies effective for related malignancies, particularly small-cell lung cancer (SCLC), have been commonly applied to LCNEC of the lung, but it is important to recognize the unique features of LCNEC in order to make a diagnosis and to individualize treatment. As current long-term survival in patients with LCNEC of the lung remains poor, participation in clinical trials should be encouraged. Therapies under investigation include those targeted at the delta-like ligand 3 (DLL3), an antigen highly expressed in neuroendocrine (NE) tumors, and Seneca Valley oncolytic viral (SVV) therapy. Early introduction of palliative care should also be offered to optimize quality of life. High-quality data for LCNEC of the lung and novel breakthrough drugs are much needed.
Background
NE tumors can develop from NE cells in almost any organ.1 After the gastrointestinal tract, the lung is the most common site of NE malignancies. They account for only about 2% of all lung cancers but 25% of NE tumors.2 Criteria for differentiating NE tumors from other tumors in the lung were first proposed in 1991.3 In 2022, the World Health Organization described 5 major subtypes of NE lung malignancies.4 On a spectrum ranging from best to worst outcome among lung cancers, LCNEC has a significantly more aggressive course compared with typical carcinoids (TC) and atypical carcinoids (AC), approaching that of SCLC, which arguably has the worst outcome (Table).5
Table. Comparing NSCLC, SCLC, and LCNEC of the Lung

Similarities exist between LCNEC of the lung and other non-small cell lung cancer (NSCLC) types, but there are more parallels with SCLC. Both are more common in male patients and both are associated with a history of smoking.6 They also share a poor prognosis. If diagnosed at an advanced stage, 5-year survival rates for LCNEC of the lung and SCLC have been reported to be as low as 5% to 15%.6
The risk of a delay in establishing the correct diagnosis of LCNEC of the lung, even by experienced pathologists, is considerable. The key diagnostic criteria include expression of at least 1 NE marker, such as chromogranin-A or synaptophysin, a high proliferation rate (> 10 mitoses per high-power field), extensive necrosis, and NE morphology features, such as trabeculae and palisading and rosette formations.7 However, other lung cancers can also express NE markers and some features might be missed without relatively large tissue specimens.7
To improve diagnostic accuracy, additional criteria, such as absence of squamous or adenocarcinoma features or the demonstration of 2 or more NE markers are now being advocated in some reports,8 while others have advocated that terms such as “combined NSCLC/SCLC” should not be accepted as a substitute for differentiating and finalizing a diagnosis of LCNEC of the lung.7 Excisional or resection biopsies, as opposed to needle biopsies, might be required to obtain an adequate tissue sample to reach a definitive diagnosis.
Illustrating the potential for confusion with other lung cancers, LCNEC of the lung can be characterized by 2 subtypes.9 Type 1 is characterized by TP53 and STK11/KEAP1 alternations—similar to adenocarcinomas and squamous cell lung cancers—and it is associated with a higher expression of NE markers, such as ASCL1 and DLL3. Type 2 is typically characterized by inactivation of TP53 and RB1. Ultimately, type I LCNEC of the lung has a mutational pattern similar to NSCLC and type II has a pattern similar to SCLC. While LCNEC is typically located in the periphery of the lung, SCLC is typically centrally located and NSCLC can be found in either location. Complicated further by the fact that a proportion of these tumors have features shared with SCLC and rarer cancers, such as spindle-cell carcinoma and giant cell carcinoma, LCNEC should be considered in the differential diagnosis of any lung cancer with ambiguous features.7
For these reasons, a pathology review should be performed at a high-volume center whenever possible. As part of the diagnostic process, molecular testing should be gathered for all patients whether or not it is required to make or confirm the diagnosis. This information will be informative for guiding treatment, particularly second- and third-line interventions. Rather than being unique and definitive, the individual features of LCNEC of the lung—including the genetic, molecular, histologic, and morphologic characteristics—cumulatively support the diagnosis. After establishing a pathological diagnosis, staging of LCNEC of the lung is paramount. In addition, distinctions between the grades of LCNEC of the lung are relative. For example, tumors with a better relative prognosis typically have fewer gene mutations than tumors with a worse relative prognosis, but mutations are generally found in both.
Bronchoscopy with endobronchial ultrasound can be considered for both diagnosis and staging of locally advanced tumors, but a surgical specimen might still be required for a definitive diagnosis. Differentiating local LCNEC, which has been reported in about 25% of cases, from locally advanced and metastatic disease is critical for planning treatment. Fluorodeoxyglucose F18 (FDG) positron emission tomography (PET) plays an important role in staging LCNEC of the lung. Unlike TC and AC, for LCNEC of the lung there is a very limited role of somatostatin receptor agonist-based imaging or tetraazacyclododecanetetraacetic acid-DPhel-Tyr3-octreotate (DOTATATE) PET during diagnostic workup.
Therapeutic Strategies
In early stages, resection followed by adjuvant chemotherapy has long been used for LCNEC of the lung. Studies evaluating this approach, such as one that combined cisplatin and etoposide,10 suggest doublet chemotherapy after surgery offers a benefit in LCNEC of the lung comparable to that seen in SCLC. There is limited support for adjunctive radiotherapy in early-stage LCNEC of the lung,5 even if radiotherapy has shown benefit for patients ineligible for surgery.11
In locally advanced and advanced LCNEC (≥ stage III-B) ineligible for resection, chemoradiation has been associated with a survival advantage over chemotherapy alone,12 but due to the high rates of relapse and limited survival, efforts to move to novel therapies have been expanding for both LCNEC of the lung and SCLC. This includes immunotherapies used before or after chemoradiation. Again, much of the interest in immunotherapies has been derived from studies in SCLC, but several small studies have associated checkpoint inhibitors with substantial antitumor activity in patients with LCNEC.13,14 There are no large scale prospective trials to determine the optimal treatment in the first line setting for LCNEC of the lung and most data is extrapolated from treatment of ES-SCLC. In a retrospective study, however, comparing survival of palliative chemotherapy with a SCLC versus a NSCLC regimen, the SCLC regimen was favored.15
Following a pivotal trial of tarlatamab-dlle, that led to an accelerated approval for extensive-stage SCLC in May 2024,16 this drug has also been evaluated in a small group of patients with LCNEC of the lung. The parallels between LCNEC and SCLC have raised hope that this drug, which is a bispecific T-cell engager (BiTE) that binds to the DLL3 ligand and CD3, may provide benefit in LCNEC of the lung that is commensurate with the benefit seen in SCLC. A recently published LCNEC case study supports this potential.17 A high-grade NE-carcinoma-specific oncolytic virus called Seneca Valley virus holds promise. Preclinical data suggest encouraging anticancer activity when SVV is combined with immune checkpoint inhibitor therapy.18 SVV seems to attack cancer cells that express tumor endothelial marker 8 (TEM-8), making it an interesting target in future efforts for screening and tailoring treatment.19 Human studies are in development.
Due to the high frequency of relapse regardless of frontline therapies, there is also growing interest in maintenance strategies to extend disease control. Maintenance regimens that have been evaluated or are being considered include immunotherapies, even if the optimal sequence of treatment modalities remains unknown. The high rate of relapse also encourages early planning of sequential therapies based on molecular testing. Numerous studies of LCNEC of the lung have now identified activating mutations in targetable pathways, such as P13K/AKT/mTOR, KRAS, and FGFR1.18 Patients may also harbor a high tumor mutation burden, a characteristic that might favor treatment with immunotherapy. Each mutation is relevant to only a small proportion of patients with LCNEC. However, when all potentially targetable mutations are considered together, the proportion of patients with LCNEC who would benefit from an individualized therapy is substantial, thus supporting trials of individualized therapy, particularly in the second line.
The high rate of relapse with currently available therapies encourages enrollment in clinical trials, particularly among patients who have already failed a first-line strategy. In the United States, studies are enrolling patients with LCNEC of the lung for checkpoint inhibitors with or without combination chemotherapy, novel BiTE therapies, and novel therapies targeting specific activating pathways. Many of these trials offer enrollment to patients with either SCLC or LCNEC.
Due to poor survival, patients with advancing LCNEC of the lung should be considered for palliative care. Although no guideline protocol exists for palliative care, the American Society of Clinical Oncology recommends palliative care for all individuals with advanced cancer based on evidence of improved quality of life and, in some cases, survival.20
Summary
LCNEC is an uncommon lung malignancy with a poor prognosis in the advanced stages at which it is most often recognized. The risk of overlooking this cancer in the initial diagnosis emphasizes the need for an adequate index of suspicion and familiarity with its distinguishing characteristics. Treatments of LCNEC of the lung have been largely based on those used for SCLC, but there has been an evolution in the understanding of this disease, which includes a greater appreciation for heterogeneity among driving mutations, a growing interest in maintenance therapies to extend the time to relapse, and trials of a growing array of novel therapies, including immunotherapies and BiTEs. Early intervention with these novel therapies and an emphasis on palliative care is needed because LCNEC has such an aggressive course.
Read more from the 2024 Rare Diseases Report: Hematology and Oncology.
- Sultana Q, Kar J, Verma A, et al. A comprehensive review on neuroendocrine neoplasms: presentation, pathophysiology and management. J Clin Med. 2023;12(15):5138. doi:10.3390/jcm12155138
- Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2008;113(1):5-21. doi:10.1002/cncr.23542
- Travis WD, Linnoila RI, Tsokos MG, et al. Neuroendocrine tumors of the lung with proposed criteria for large-cell neuroendocrine carcinoma. An ultrastructural, immunohistochemical, and flow cytometric study of 35 cases. Am J Surg Pathol. 1991;15(6):529-553. doi:10.1097/00000478-199106000-00003
- Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG. Introduction to the 2015 World Health Organization classification of tumors of the lung, pleura, thymus, and heart. J Thorac Oncol. 2015;10(9):1240-1242. doi:10.1097/JTO.0000000000000663
- Andrini E, Marchese PV, De Biase D, et al. Large cell neuroendocrine carcinoma of the lung: current understanding and challenges. J Clin Med. 2022;11(5):1461. doi:10.3390/jcm11051461
- Lantuejoul S, Fernandez-Cuesta L, Damiola F, Girard N, McLeer A. New molecular classification of large cell neuroendocrine carcinoma and small cell lung carcinoma with potential therapeutic impacts. Transl Lung Cancer Res. 2020;9(5):2233-2244. doi:10.21037/tlcr-20-269
- Lindsay CR, Shaw EC, Moore DA, et al. Large cell neuroendocrine lung carcinoma: consensus statement from The British Thoracic Oncology Group and the Association of Pulmonary Pathologists. Br J Cancer. 2021;125(9):1210-1216. doi:10.1038/s41416-021-01407-9
- Derks JL, Dingemans AC, van Suylen RJ, et al. Is the sum of positive neuroendocrine immunohistochemical stains useful for diagnosis of large cell neuroendocrine carcinoma (LCNEC) on biopsy specimens? Histopathology. 2019;74(4):555-566. doi:10.1111/his.13800
- George J, Walter V, Peifer M, et al. Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. Nat Commun. 2018;9(1):1048. doi:10.1038/s41467-018-03099-x
- Iyoda A, Hiroshima K, Moriya Y, et al. Prospective study of adjuvant chemotherapy for pulmonary large cell neuroendocrine carcinoma. Ann Thorac Surg. 2006;82(5):1802-1807. doi:10.1016/j.athoracsur.2006.05.109
- Cao L, Wu HF, Zhao L, et al. The role of radiotherapy in pulmonary large cell neuroendocrine carcinoma: propensity score matching analysis. J Radiat Res. 2020;61(4):594-601. doi:10.1093/jrr/rraa036
- Limonnik V, Abel S, Finley GG, Long GS, Wegner RE. Factors associated with treatment receipt and overall survival for patients with locally advanced large cell neuroendocrine carcinoma of the lung: a National Cancer Database analysis. Lung Cancer. 2020;150:107-113. doi:10.1016/j.lungcan.2020.10.001
- Shi Z, Wei J, Xu M, Song Z. Efficacy and safety of immune checkpoint inhibitors in lung large-cell neuroendocrine carcinoma. J Thorac Dis. 2023;15(8):4172-4181. doi:10.21037/jtd-23-348
- Chauhan A, Arnold SM, Kolesar J, Thomas HE, Evers M, Anthony L. Immune checkpoint inhibitors in large cell neuroendocrine carcinoma: current status. Oncotarget. 2018;9(18):14738-14740. doi:10.18632/oncotarget.24553
- Chen H, Ishihara M, Horita N, et al. Effect of adjuvant and palliative chemotherapy in large cell neuroendocrine carcinoma of the lung: a systematic review and metaanalysis. Cancers (Basel). 2021;13(23):5948. doi:10.3390/cancers13235948
- Ahn MJ, Cho BC, Felip E, et al. Tarlatamab for patients with previously treated small-cell lung cancer. N Engl J Med. 2023;389(22):2063-2075. doi:10.1056/NEJMoa2307980
- Patel SA, Whang Y, Medley C, et al. Tartalamab for large-cell neuroendocrine carcinoma in a young adult: a case report. JTO Clin Res Rep. 2024;5(10):100712. doi:10.1016/j.jtocrr.2024.100712
- Corbett V, Hallenbeck P, Rychahou P, Chauhan A. Evolving role of Seneca Valley virus and its biomarker TEM8/ANTXR1 in cancer therapeutics. Front Mol Biosci. 2022;9:930207. doi:10.3389/fmolb.2022.930207
- Kareff SA, Corbett V, Hallenbeck P, Chauhan A. TEM8 in oncogenesis: protein biology, pre-clinical agents, and clinical rationale. Cells. 2023;12(22):2623. doi:10.3390/cells12222623
- Sanders JJ, Temin S, Ghoshal A, et al. Palliative care for patients with cancer: ASCO guideline update. J Clin Oncol. 2024;42(19):2336-2357. doi:10.1200/JCO.24.00542
- Sultana Q, Kar J, Verma A, et al. A comprehensive review on neuroendocrine neoplasms: presentation, pathophysiology and management. J Clin Med. 2023;12(15):5138. doi:10.3390/jcm12155138
- Gustafsson BI, Kidd M, Chan A, Malfertheiner MV, Modlin IM. Bronchopulmonary neuroendocrine tumors. Cancer. 2008;113(1):5-21. doi:10.1002/cncr.23542
- Travis WD, Linnoila RI, Tsokos MG, et al. Neuroendocrine tumors of the lung with proposed criteria for large-cell neuroendocrine carcinoma. An ultrastructural, immunohistochemical, and flow cytometric study of 35 cases. Am J Surg Pathol. 1991;15(6):529-553. doi:10.1097/00000478-199106000-00003
- Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG. Introduction to the 2015 World Health Organization classification of tumors of the lung, pleura, thymus, and heart. J Thorac Oncol. 2015;10(9):1240-1242. doi:10.1097/JTO.0000000000000663
- Andrini E, Marchese PV, De Biase D, et al. Large cell neuroendocrine carcinoma of the lung: current understanding and challenges. J Clin Med. 2022;11(5):1461. doi:10.3390/jcm11051461
- Lantuejoul S, Fernandez-Cuesta L, Damiola F, Girard N, McLeer A. New molecular classification of large cell neuroendocrine carcinoma and small cell lung carcinoma with potential therapeutic impacts. Transl Lung Cancer Res. 2020;9(5):2233-2244. doi:10.21037/tlcr-20-269
- Lindsay CR, Shaw EC, Moore DA, et al. Large cell neuroendocrine lung carcinoma: consensus statement from The British Thoracic Oncology Group and the Association of Pulmonary Pathologists. Br J Cancer. 2021;125(9):1210-1216. doi:10.1038/s41416-021-01407-9
- Derks JL, Dingemans AC, van Suylen RJ, et al. Is the sum of positive neuroendocrine immunohistochemical stains useful for diagnosis of large cell neuroendocrine carcinoma (LCNEC) on biopsy specimens? Histopathology. 2019;74(4):555-566. doi:10.1111/his.13800
- George J, Walter V, Peifer M, et al. Integrative genomic profiling of large-cell neuroendocrine carcinomas reveals distinct subtypes of high-grade neuroendocrine lung tumors. Nat Commun. 2018;9(1):1048. doi:10.1038/s41467-018-03099-x
- Iyoda A, Hiroshima K, Moriya Y, et al. Prospective study of adjuvant chemotherapy for pulmonary large cell neuroendocrine carcinoma. Ann Thorac Surg. 2006;82(5):1802-1807. doi:10.1016/j.athoracsur.2006.05.109
- Cao L, Wu HF, Zhao L, et al. The role of radiotherapy in pulmonary large cell neuroendocrine carcinoma: propensity score matching analysis. J Radiat Res. 2020;61(4):594-601. doi:10.1093/jrr/rraa036
- Limonnik V, Abel S, Finley GG, Long GS, Wegner RE. Factors associated with treatment receipt and overall survival for patients with locally advanced large cell neuroendocrine carcinoma of the lung: a National Cancer Database analysis. Lung Cancer. 2020;150:107-113. doi:10.1016/j.lungcan.2020.10.001
- Shi Z, Wei J, Xu M, Song Z. Efficacy and safety of immune checkpoint inhibitors in lung large-cell neuroendocrine carcinoma. J Thorac Dis. 2023;15(8):4172-4181. doi:10.21037/jtd-23-348
- Chauhan A, Arnold SM, Kolesar J, Thomas HE, Evers M, Anthony L. Immune checkpoint inhibitors in large cell neuroendocrine carcinoma: current status. Oncotarget. 2018;9(18):14738-14740. doi:10.18632/oncotarget.24553
- Chen H, Ishihara M, Horita N, et al. Effect of adjuvant and palliative chemotherapy in large cell neuroendocrine carcinoma of the lung: a systematic review and metaanalysis. Cancers (Basel). 2021;13(23):5948. doi:10.3390/cancers13235948
- Ahn MJ, Cho BC, Felip E, et al. Tarlatamab for patients with previously treated small-cell lung cancer. N Engl J Med. 2023;389(22):2063-2075. doi:10.1056/NEJMoa2307980
- Patel SA, Whang Y, Medley C, et al. Tartalamab for large-cell neuroendocrine carcinoma in a young adult: a case report. JTO Clin Res Rep. 2024;5(10):100712. doi:10.1016/j.jtocrr.2024.100712
- Corbett V, Hallenbeck P, Rychahou P, Chauhan A. Evolving role of Seneca Valley virus and its biomarker TEM8/ANTXR1 in cancer therapeutics. Front Mol Biosci. 2022;9:930207. doi:10.3389/fmolb.2022.930207
- Kareff SA, Corbett V, Hallenbeck P, Chauhan A. TEM8 in oncogenesis: protein biology, pre-clinical agents, and clinical rationale. Cells. 2023;12(22):2623. doi:10.3390/cells12222623
- Sanders JJ, Temin S, Ghoshal A, et al. Palliative care for patients with cancer: ASCO guideline update. J Clin Oncol. 2024;42(19):2336-2357. doi:10.1200/JCO.24.00542
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
2024 Rare Diseases Report: Hematology and Oncology
2024 Rare Diseases Report: Hematology and Oncology
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
National Organization for Rare Disorders: Strengthening Rare Cancer Advocacy
By Alli Ward
NORD's Rare Cancer Coalition has transformed advocacy and awareness efforts, offering education and fostering research to address the challenges of rare cancers.
Treatment of Glioblastoma: A Potential Shift in Paradigm
By Jeffrey N. Bruce, MD
Immunotherapies and molecular profiling are paving the way for more targeted approaches in treating glioblastoma.
Emerging Insights and Therapeutic Strategies for Large Cell Neuroendocrine Carcinoma of the Lung
By Robert A. Ramirez, DO, FACP, and Aman Chauhan, MD
New diagnostic tools and precision medicine approaches are addressing the unique challenges of this aggressive neuroendocrine cancer.
Advancements in the Treatment of Malignant PEComas with mTOR Inhibitors
By Richard F. Riedel, MD
The use of mTOR inhibitors marks significant progress in managing advanced malignant PEComas, offering new hope for patients.
Cutaneous T-Cell Lymphomas Update: Benefits of a Multidisciplinary Care Approach
By Jina Chung, MD, and Eric Mou, MD
A multidisciplinary care model ensures optimal outcomes for patients with cutaneous T-cell lymphomas, addressing both medical and emotional needs.
Optimizing Myelofibrosis Care in the Age of JAK Inhibitors
By Douglas Tremblay, MD
JAK inhibitors are central to myelofibrosis management, with personalized strategies helping to navigate resistance and improve quality of life.
Current Management and Future Directions in the Treatment of Gallbladder Cancer
By Ghassan K. Abou-Alfa, MD, MBA, JD, FASCO
Molecular profiling and immunotherapy are reshaping the treatment paradigm for gallbladder cancer, improving survival outcomes.
Improving Prognosis in Hepatoblastoma: Evolving Risk Stratification and Treatment Strategies
By Greg M. Tiao, MD
Risk stratification and individualized therapies are driving progress in treating hepatoblastoma, with promising advancements on the horizon.
2024 Rare Diseases Report: Hematology and Oncology
2024 Rare Diseases Report: Hematology and Oncology
New Cancer Drugs: Do Patients Prefer Faster Access or Clinical Benefit?
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
When the Food and Drug Administration (FDA) grants cancer drugs accelerated approval, a key aim is to provide patients faster access to therapies that can benefit them.
The downside of a speedier approval timeline, however, is that it’s often not yet clear whether the new drugs will actually allow a patient to live longer or better. Information on overall survival and quality of life typically comes years later, after drugs undergo confirmatory trials, or sometimes not at all, if companies fail to conduct these trials.
During this waiting period, patients may be receiving a cancer drug that provides no real clinical benefit but comes with a host of toxicities.
In fact, the odds are about as good as a coin flip. For cancer drugs that have confirmatory trial data, more than half don’t ultimately provide an overall survival or quality of life benefit.
Inherent to the accelerated approval process is the assumption that patients are willing to accept this uncertainty in exchange for faster access.
But is that really the case?
The researchers asked about 870 adults with experience of cancer challenges — either their own cancer diagnosis or that of family or a close friend — whether they valued faster access or certainty that a drug really works.
In the study, participants imagined they had been diagnosed with cancer and could choose between two cancer drugs under investigation in clinical trials but with uncertain effectiveness, and a current standard treatment. Participants had to make a series of choices based on five scenarios.
The first two scenarios were based on the impact of the current standard treatment: A patient’s life expectancy on the standard treatment (6 months up to 3 years), and a patient’s physical health on the standard treatment (functional status restricted only during strenuous activities up to completely disabled).
The remaining three scenarios dealt with the two new drugs: The effect of the new drugs on a surrogate endpoint, progression-free survival (whether the drugs slowed tumor growth for an extra month or 5 additional months compared with the standard treatment), certainty that slowing tumor growth will improve survival (very low to high), and the wait time to access the drugs (immediately to as long as 2 years).
The researchers assessed the relative importance of survival benefit certainty vs wait time and how that balance shifted depending on the different scenarios.
Overall, the researchers found that, if there was no evidence linking the surrogate endpoint (progression-free survival) to overall survival, patients were willing to wait about 8 months for weak evidence of an overall survival benefit (ie, low certainty the drug will extend survival by 1-5 months), about 16 months for moderate certainty, and almost 22 months for high certainty.
Despite a willingness to wait for greater certainty, participants did value speed as well. Overall, respondents showed a strong preference against a 1-year delay in FDA approval time. People who were aged 55 years or more and were non-White individuals made less than $40,000 year as well as those with the lowest life expectancy on a current standard treatment were most sensitive to wait times while those with better functional status and longer life expectancies on a current treatment were less sensitive to longer wait times.
“Our results indicate that some patients (except those with the poorest prognoses) would find the additional time required to generate evidence on the survival benefit of new cancer drugs an acceptable tradeoff,” the study authors concluded.
Although people do place high value on timely access to new cancer drugs, especially if there are limited treatment options, many are willing to wait for greater certainty that a new drug provides an overall survival benefit, lead author Robin Forrest, MSc, with the Department of Health Policy, London School of Economics in England, said in an interview.
In the study, respondents also did not place significant value on whether the drug substantially slowed cancer growth. “In other words, substantial progression-free survival benefit of a drug did not compensate for lack of certainty about a drug’s benefit on survival in respondents’ drug choices,” the authors explained.
“In an effort to move quickly, we have accepted progression-free survival [as a surrogate endpoint],” Jyoti D. Patel, MD, oncologist with Northwestern Memorial Hospital, Chicago, Illinois, who wasn’t involved in the study. But a growing body of evidence indicates that progression-free survival is often a poor surrogate for overall survival. And what this study suggests is that “patients uniformly care about improvements in overall survival and the quality of that survival,” Patel said.
Bishal Gyawali, MD, PhD, was not surprised by the findings.
“I always thought this was the real-world scenario, but the problem is the voices of ordinary patients are not heard,” Gyawali, with Queen’s University, Kingston, Ontario, Canada, who also wasn’t involved in the study, said in an interview.
“What is heard is the loud noise of ‘we need access now, today, yesterday’ — ‘we don’t care if the drug doesn’t improve overall survival, we just need a drug, any drug’ — ‘we don’t care how much it costs, we need access today,’ ” Gyawali said. “Not saying this is wrong, but this is not the representation of all patients.”
However, the voices of patients who are more cautious and want evidence of benefit before accepting toxicities don’t make headlines, he added.
What this survey means from a policy perspective, said Gyawali, is that accelerated approvals that do not mandate survival endpoint in confirmatory trials are ignoring the need of many patients who prioritize certainty of benefit over speed of access.
The study was funded by the London School of Economics and Political Science Phelan United States Centre. Forrest had no relevant disclosures. Gyawali has received consulting fees from Vivio Health. Patel has various relationships with AbbVie, Anheart, AstraZeneca, Bristol-Myers Squibb, Guardant, Tempus, Sanofi, BluePrint, Takeda, and Gilead.
A version of this article first appeared on Medscape.com.
FROM THE LANCET ONCOLOGY
Dying in the Hospital: A Necessary Choice?
More than a third of all patients with cancer die in hospitals, a figure that has increased slightly in recent years, while deaths at home have decreased. These findings come from a recent study published in Cancer Epidemiology, which analyzed data on the different places in Italy where end of life occurs.
“Place of death is relevant both for individuals and for the society. Home is universally considered the optimal place of death, while dying in a hospital may be a signal of inappropriate end-of-life care,” wrote the authors, led by Gianmauro Numico, MD, head of the Oncology Department at the Santa Croce e Carle General Hospital in Cuneo, Italy.
“Despite the general trend toward strengthening community-based networks and the increasing number of hospice and long-term care facilities, we oncologists are facing an opposite trend, with many patients spending their last days in the hospital,” Numico explained to Univadis Italy. This observation led to the questions that prompted the study: Is this only a perception among doctors, or is it a real phenomenon? If the latter, why is it happening?
What’s Preferable
For their analysis, Numico and colleagues relied on death certificates published by the Italian National Institute of Statistics from 2015 to 2019, excluding data from the pandemic years to avoid potential biases.
The analysis of data pertaining to cancer deaths revealed that approximately 35% of Italian patients with cancer die in hospitals, with a slight increase over the study period. Of the patients who die elsewhere, 40% die at home and 20% die in hospice or other long-term care facilities. Home deaths have decreased by 3.09%, while those in hospices and long-term care facilities have increased by 2.71%, and hospital deaths have risen by 0.3%.
The study also highlighted notable geographical differences: Hospital deaths are more frequent in the north, while in the south, home deaths remain predominant, although hospital admissions are on the rise. “These differences reflect not only access to facilities but also cultural and social variables,” explained Numico. “Some end-of-life issues with cancer patients are more straightforward, while others are difficult to manage outside the hospital,” he said, recalling that many family members and caregivers are afraid they won’t be able to care for their loved ones properly without the support of an appropriate facility and skilled personnel.
Social factors also contribute to the increased use of hospitals for end-of-life care: Without a social and family network, it is often impossible to manage the final stages of life at home. “We cannot guarantee that dying at home is better for everyone; in some cases, the home cannot provide the necessary care and emotional support,” Numico added.
Attitudes Need Change
Looking beyond Italy, it is clear that this trend exists in other countries as well. For example, in the Netherlands — where community-based care is highly developed and includes practices such as euthanasia — hospital death rates are higher than those in Italy. In the United States, the trend is different, but this is largely due to the structure of the US healthcare system, where patients bear much of the financial burden of hospital admissions.
“The basic requests of patients and families are clear: They want a safe place that is adequately staffed and where the patient won’t suffer,” said Numico, questioning whether the home is truly the best place to die. “In reality, this is not always the case, and it’s important to focus on the quality of care in the final days rather than just the place of care,” he added.
Ruling out hospitals a priori as a place to die is not a winning strategy, according to the expert. Instead of trying to reverse the trend, he suggests integrating the hospital into a care network that prioritizes the patient’s well-being, regardless of the setting. “Our goal should not be to eliminate hospital deaths — a common request from hospital administrations — but rather to ensure that end-of-life care in hospitals is a dignified experience that respects the needs of the dying and their loved ones,” Numico said. “We must ensure that, wherever the end-of-life process occurs, it should happen in the best way possible, and the hospital must be a part of this overall framework,” he concluded.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
More than a third of all patients with cancer die in hospitals, a figure that has increased slightly in recent years, while deaths at home have decreased. These findings come from a recent study published in Cancer Epidemiology, which analyzed data on the different places in Italy where end of life occurs.
“Place of death is relevant both for individuals and for the society. Home is universally considered the optimal place of death, while dying in a hospital may be a signal of inappropriate end-of-life care,” wrote the authors, led by Gianmauro Numico, MD, head of the Oncology Department at the Santa Croce e Carle General Hospital in Cuneo, Italy.
“Despite the general trend toward strengthening community-based networks and the increasing number of hospice and long-term care facilities, we oncologists are facing an opposite trend, with many patients spending their last days in the hospital,” Numico explained to Univadis Italy. This observation led to the questions that prompted the study: Is this only a perception among doctors, or is it a real phenomenon? If the latter, why is it happening?
What’s Preferable
For their analysis, Numico and colleagues relied on death certificates published by the Italian National Institute of Statistics from 2015 to 2019, excluding data from the pandemic years to avoid potential biases.
The analysis of data pertaining to cancer deaths revealed that approximately 35% of Italian patients with cancer die in hospitals, with a slight increase over the study period. Of the patients who die elsewhere, 40% die at home and 20% die in hospice or other long-term care facilities. Home deaths have decreased by 3.09%, while those in hospices and long-term care facilities have increased by 2.71%, and hospital deaths have risen by 0.3%.
The study also highlighted notable geographical differences: Hospital deaths are more frequent in the north, while in the south, home deaths remain predominant, although hospital admissions are on the rise. “These differences reflect not only access to facilities but also cultural and social variables,” explained Numico. “Some end-of-life issues with cancer patients are more straightforward, while others are difficult to manage outside the hospital,” he said, recalling that many family members and caregivers are afraid they won’t be able to care for their loved ones properly without the support of an appropriate facility and skilled personnel.
Social factors also contribute to the increased use of hospitals for end-of-life care: Without a social and family network, it is often impossible to manage the final stages of life at home. “We cannot guarantee that dying at home is better for everyone; in some cases, the home cannot provide the necessary care and emotional support,” Numico added.
Attitudes Need Change
Looking beyond Italy, it is clear that this trend exists in other countries as well. For example, in the Netherlands — where community-based care is highly developed and includes practices such as euthanasia — hospital death rates are higher than those in Italy. In the United States, the trend is different, but this is largely due to the structure of the US healthcare system, where patients bear much of the financial burden of hospital admissions.
“The basic requests of patients and families are clear: They want a safe place that is adequately staffed and where the patient won’t suffer,” said Numico, questioning whether the home is truly the best place to die. “In reality, this is not always the case, and it’s important to focus on the quality of care in the final days rather than just the place of care,” he added.
Ruling out hospitals a priori as a place to die is not a winning strategy, according to the expert. Instead of trying to reverse the trend, he suggests integrating the hospital into a care network that prioritizes the patient’s well-being, regardless of the setting. “Our goal should not be to eliminate hospital deaths — a common request from hospital administrations — but rather to ensure that end-of-life care in hospitals is a dignified experience that respects the needs of the dying and their loved ones,” Numico said. “We must ensure that, wherever the end-of-life process occurs, it should happen in the best way possible, and the hospital must be a part of this overall framework,” he concluded.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
More than a third of all patients with cancer die in hospitals, a figure that has increased slightly in recent years, while deaths at home have decreased. These findings come from a recent study published in Cancer Epidemiology, which analyzed data on the different places in Italy where end of life occurs.
“Place of death is relevant both for individuals and for the society. Home is universally considered the optimal place of death, while dying in a hospital may be a signal of inappropriate end-of-life care,” wrote the authors, led by Gianmauro Numico, MD, head of the Oncology Department at the Santa Croce e Carle General Hospital in Cuneo, Italy.
“Despite the general trend toward strengthening community-based networks and the increasing number of hospice and long-term care facilities, we oncologists are facing an opposite trend, with many patients spending their last days in the hospital,” Numico explained to Univadis Italy. This observation led to the questions that prompted the study: Is this only a perception among doctors, or is it a real phenomenon? If the latter, why is it happening?
What’s Preferable
For their analysis, Numico and colleagues relied on death certificates published by the Italian National Institute of Statistics from 2015 to 2019, excluding data from the pandemic years to avoid potential biases.
The analysis of data pertaining to cancer deaths revealed that approximately 35% of Italian patients with cancer die in hospitals, with a slight increase over the study period. Of the patients who die elsewhere, 40% die at home and 20% die in hospice or other long-term care facilities. Home deaths have decreased by 3.09%, while those in hospices and long-term care facilities have increased by 2.71%, and hospital deaths have risen by 0.3%.
The study also highlighted notable geographical differences: Hospital deaths are more frequent in the north, while in the south, home deaths remain predominant, although hospital admissions are on the rise. “These differences reflect not only access to facilities but also cultural and social variables,” explained Numico. “Some end-of-life issues with cancer patients are more straightforward, while others are difficult to manage outside the hospital,” he said, recalling that many family members and caregivers are afraid they won’t be able to care for their loved ones properly without the support of an appropriate facility and skilled personnel.
Social factors also contribute to the increased use of hospitals for end-of-life care: Without a social and family network, it is often impossible to manage the final stages of life at home. “We cannot guarantee that dying at home is better for everyone; in some cases, the home cannot provide the necessary care and emotional support,” Numico added.
Attitudes Need Change
Looking beyond Italy, it is clear that this trend exists in other countries as well. For example, in the Netherlands — where community-based care is highly developed and includes practices such as euthanasia — hospital death rates are higher than those in Italy. In the United States, the trend is different, but this is largely due to the structure of the US healthcare system, where patients bear much of the financial burden of hospital admissions.
“The basic requests of patients and families are clear: They want a safe place that is adequately staffed and where the patient won’t suffer,” said Numico, questioning whether the home is truly the best place to die. “In reality, this is not always the case, and it’s important to focus on the quality of care in the final days rather than just the place of care,” he added.
Ruling out hospitals a priori as a place to die is not a winning strategy, according to the expert. Instead of trying to reverse the trend, he suggests integrating the hospital into a care network that prioritizes the patient’s well-being, regardless of the setting. “Our goal should not be to eliminate hospital deaths — a common request from hospital administrations — but rather to ensure that end-of-life care in hospitals is a dignified experience that respects the needs of the dying and their loved ones,” Numico said. “We must ensure that, wherever the end-of-life process occurs, it should happen in the best way possible, and the hospital must be a part of this overall framework,” he concluded.
This story was translated from Univadis Italy using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
FDA Approves Durvalumab for Limited-Stage SCLC
The Food and Drug Administration approval makes the monoclonal antibody — which is already approved for multiple tumor types — the first immunotherapy regimen approved in this setting, AstraZeneca noted in a press release.
“Durvalumab is the first and only systemic treatment following curative-intent, platinum-based chemoradiotherapy to show improved survival for patients with this aggressive form of lung cancer,” international coordinating investigator on the trial, Suresh Senan, PhD, stated in the press release. “This finding represents the first advance for this disease in 4 decades.”
Approval, which followed Priority Review and Breakthrough Therapy Designation, was based on findings from the phase 3 ADRIATIC trial showing a 27% reduction in the risk for death with durvalumab vs placebo.
Findings from the trial were reported during a plenary session at the 2024 American Society of Clinical Oncology conference, and subsequently published in The New England Journal of Medicine.
In 730 patients with LS-SCLC who were randomized 1:1:1 to receive single-agent durvalumab, durvalumab in combination with tremelimumab, or placebo, overall survival (OS) and progression-free survival (PFS) were significantly improved with durvalumab alone vs placebo (hazard ratio, 0.73 and 0.76, for OS and PFS, respectively). Median OS was 55.9 months vs 33.4 months with durvalumab vs placebo, and PFS was 16.6 vs 9.2 months, respectively.
Senan, a professor of clinical experimental radiotherapy at the Amsterdam University Medical Center in the Netherlands, noted in the press release that 57% of patients were still alive at 3 years after being treated with durvalumab, which underscores the practice-changing potential of this medicine in this setting.
“This new treatment option is a game changer for patients with limited-stage small cell lung cancer, a disease known for its high rate of recurrence,” Dusty Donaldson, founder and executive director of the nonprofit advocacy organization LiveLung, stated in the release. “Historically, more often than not, clinical trials to identify new treatment options for this type of cancer have failed to show benefit. We are therefore so excited that many more people will now have the opportunity to access this immunotherapy treatment that holds the potential to significantly improve outcomes.”
Adverse reactions occurring in at least 20% of patients in the ADRIATIC trial included pneumonitis or radiation pneumonitis and fatigue.
The recommended durvalumab dose, according to prescribing information, is 1500 mg every 4 weeks for patients weighing at least 30 kg and 20 mg/kg every 4 weeks for those weighing less than 30 kg, until disease progression or unacceptable toxicity or a maximum of 24 months.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration approval makes the monoclonal antibody — which is already approved for multiple tumor types — the first immunotherapy regimen approved in this setting, AstraZeneca noted in a press release.
“Durvalumab is the first and only systemic treatment following curative-intent, platinum-based chemoradiotherapy to show improved survival for patients with this aggressive form of lung cancer,” international coordinating investigator on the trial, Suresh Senan, PhD, stated in the press release. “This finding represents the first advance for this disease in 4 decades.”
Approval, which followed Priority Review and Breakthrough Therapy Designation, was based on findings from the phase 3 ADRIATIC trial showing a 27% reduction in the risk for death with durvalumab vs placebo.
Findings from the trial were reported during a plenary session at the 2024 American Society of Clinical Oncology conference, and subsequently published in The New England Journal of Medicine.
In 730 patients with LS-SCLC who were randomized 1:1:1 to receive single-agent durvalumab, durvalumab in combination with tremelimumab, or placebo, overall survival (OS) and progression-free survival (PFS) were significantly improved with durvalumab alone vs placebo (hazard ratio, 0.73 and 0.76, for OS and PFS, respectively). Median OS was 55.9 months vs 33.4 months with durvalumab vs placebo, and PFS was 16.6 vs 9.2 months, respectively.
Senan, a professor of clinical experimental radiotherapy at the Amsterdam University Medical Center in the Netherlands, noted in the press release that 57% of patients were still alive at 3 years after being treated with durvalumab, which underscores the practice-changing potential of this medicine in this setting.
“This new treatment option is a game changer for patients with limited-stage small cell lung cancer, a disease known for its high rate of recurrence,” Dusty Donaldson, founder and executive director of the nonprofit advocacy organization LiveLung, stated in the release. “Historically, more often than not, clinical trials to identify new treatment options for this type of cancer have failed to show benefit. We are therefore so excited that many more people will now have the opportunity to access this immunotherapy treatment that holds the potential to significantly improve outcomes.”
Adverse reactions occurring in at least 20% of patients in the ADRIATIC trial included pneumonitis or radiation pneumonitis and fatigue.
The recommended durvalumab dose, according to prescribing information, is 1500 mg every 4 weeks for patients weighing at least 30 kg and 20 mg/kg every 4 weeks for those weighing less than 30 kg, until disease progression or unacceptable toxicity or a maximum of 24 months.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration approval makes the monoclonal antibody — which is already approved for multiple tumor types — the first immunotherapy regimen approved in this setting, AstraZeneca noted in a press release.
“Durvalumab is the first and only systemic treatment following curative-intent, platinum-based chemoradiotherapy to show improved survival for patients with this aggressive form of lung cancer,” international coordinating investigator on the trial, Suresh Senan, PhD, stated in the press release. “This finding represents the first advance for this disease in 4 decades.”
Approval, which followed Priority Review and Breakthrough Therapy Designation, was based on findings from the phase 3 ADRIATIC trial showing a 27% reduction in the risk for death with durvalumab vs placebo.
Findings from the trial were reported during a plenary session at the 2024 American Society of Clinical Oncology conference, and subsequently published in The New England Journal of Medicine.
In 730 patients with LS-SCLC who were randomized 1:1:1 to receive single-agent durvalumab, durvalumab in combination with tremelimumab, or placebo, overall survival (OS) and progression-free survival (PFS) were significantly improved with durvalumab alone vs placebo (hazard ratio, 0.73 and 0.76, for OS and PFS, respectively). Median OS was 55.9 months vs 33.4 months with durvalumab vs placebo, and PFS was 16.6 vs 9.2 months, respectively.
Senan, a professor of clinical experimental radiotherapy at the Amsterdam University Medical Center in the Netherlands, noted in the press release that 57% of patients were still alive at 3 years after being treated with durvalumab, which underscores the practice-changing potential of this medicine in this setting.
“This new treatment option is a game changer for patients with limited-stage small cell lung cancer, a disease known for its high rate of recurrence,” Dusty Donaldson, founder and executive director of the nonprofit advocacy organization LiveLung, stated in the release. “Historically, more often than not, clinical trials to identify new treatment options for this type of cancer have failed to show benefit. We are therefore so excited that many more people will now have the opportunity to access this immunotherapy treatment that holds the potential to significantly improve outcomes.”
Adverse reactions occurring in at least 20% of patients in the ADRIATIC trial included pneumonitis or radiation pneumonitis and fatigue.
The recommended durvalumab dose, according to prescribing information, is 1500 mg every 4 weeks for patients weighing at least 30 kg and 20 mg/kg every 4 weeks for those weighing less than 30 kg, until disease progression or unacceptable toxicity or a maximum of 24 months.
A version of this article first appeared on Medscape.com.
New Cancer Vaccines on the Horizon: Renewed Hope or Hype?
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
Vaccines for treating and preventing cancer have long been considered a holy grail in oncology.
But aside from a few notable exceptions — including the human papillomavirus (HPV) vaccine, which has dramatically reduced the incidence of HPV-related cancers, and a Bacillus Calmette-Guerin vaccine, which helps prevent early-stage bladder cancer recurrence — most have failed to deliver.
Following a string of disappointments over the past decade, recent advances in the immunotherapy space are bringing renewed hope for progress.
In an American Association for Cancer Research (AACR) series earlier in 2024, Catherine J. Wu, MD, predicted big strides for cancer vaccines, especially for personalized vaccines that target patient-specific neoantigens — the proteins that form on cancer cells — as well as vaccines that can treat diverse tumor types.
said Wu, the Lavine Family Chair of Preventative Cancer Therapies at Dana-Farber Cancer Institute and a professor of medicine at Harvard Medical School, both in Boston, Massachusetts.
A prime example is a personalized, messenger RNA (mRNA)–based vaccine designed to prevent melanoma recurrence. The mRNA-4157 vaccine encodes up to 34 different patient-specific neoantigens.
“This is one of the most exciting developments in modern cancer therapy,” said Lawrence Young, a virologist and professor of molecular oncology at the University of Warwick, Coventry, England, who commented on the investigational vaccine via the UK-based Science Media Centre.
Other promising options are on the horizon as well. In August, BioNTech announced a phase 1 global trial to study BNT116 — a vaccine to treat non–small cell lung cancer (NSCLC). BNT116, like mRNA-4157, targets specific antigens in the lung cancer cells.
“This technology is the next big phase of cancer treatment,” Siow Ming Lee, MD, a consultant medical oncologist at University College London Hospitals in England, which is leading the UK trial for the lung cancer and melanoma vaccines, told The Guardian. “We are now entering this very exciting new era of mRNA-based immunotherapy clinical trials to investigate the treatment of lung cancer.”
Still, these predictions have a familiar ring. While the prospects are exciting, delivering on them is another story. There are simply no guarantees these strategies will work as hoped.
Then: Where We Were
Cancer vaccine research began to ramp up in the 2000s, and in 2006, the first-generation HPV vaccine, Gardasil, was approved. Gardasil prevents infection from four strains of HPV that cause about 80% of cervical cancer cases.
In 2010, the Food and Drug Administration approved sipuleucel-T, the first therapeutic cancer vaccine, which improved overall survival in patients with hormone-refractory prostate cancer.
Researchers predicted this approval would “pave the way for developing innovative, next generation of vaccines with enhanced antitumor potency.”
In a 2015 AACR research forecast report, Drew Pardoll, MD, PhD, co-director of the Cancer Immunology and Hematopoiesis Program at Johns Hopkins University, Baltimore, Maryland, said that “we can expect to see encouraging results from studies using cancer vaccines.”
Despite the excitement surrounding cancer vaccines alongside a few successes, the next decade brought a longer string of late-phase disappointments.
In 2016, the phase 3 ACT IV trial of a therapeutic vaccine to treat glioblastoma multiforme (CDX-110) was terminated after it failed to demonstrate improved survival.
In 2017, a phase 3 trial of the therapeutic pancreatic cancer vaccine, GVAX, was stopped early for lack of efficacy.
That year, an attenuated Listeria monocytogenes vaccine to treat pancreatic cancer and mesothelioma also failed to come to fruition. In late 2017, concerns over listeria infections prompted Aduro Biotech to cancel its listeria-based cancer treatment program.
In 2018, a phase 3 trial of belagenpumatucel-L, a therapeutic NSCLC vaccine, failed to demonstrate a significant improvement in survival and further study was discontinued.
And in 2019, a vaccine targeting MAGE-A3, a cancer-testis antigen present in multiple tumor types, failed to meet endpoints for improved survival in a phase 3 trial, leading to discontinuation of the vaccine program.
But these disappointments and failures are normal parts of medical research and drug development and have allowed for incremental advances that helped fuel renewed interest and hope for cancer vaccines, when the timing was right, explained vaccine pioneer Larry W. Kwak, MD, PhD, deputy director of the Comprehensive Cancer Center at City of Hope, Duarte, California.
When it comes to vaccine progress, timing makes a difference. In 2011, Kwak and colleagues published promising phase 3 trial results on a personalized vaccine. The vaccine was a patient-specific tumor-derived antigen for patients with follicular lymphoma in their first remission following chemotherapy. Patients who received the vaccine demonstrated significantly longer disease-free survival.
But, at the time, personalized vaccines faced strong headwinds due, largely, to high costs, and commercial interest failed to materialize. “That’s been the major hurdle for a long time,” said Kwak.
Now, however, interest has returned alongside advances in technology and research. The big shift has been the emergence of lower-cost rapid-production mRNA and DNA platforms and a better understanding of how vaccines and potent immune stimulants, like checkpoint inhibitors, can work together to improve outcomes, he explained.
“The timing wasn’t right” back then, Kwak noted. “Now, it’s a different environment and a different time.”
A Turning Point?
Indeed, a decade later, cancer vaccine development appears to be headed in a more promising direction.
Among key cancer vaccines to watch is the mRNA-4157 vaccine, developed by Merck and Moderna, designed to prevent melanoma recurrence. In a recent phase 2 study, patients receiving the mRNA-4157 vaccine alongside pembrolizumab had nearly half the risk for melanoma recurrence or death at 3 years compared with those receiving pembrolizumab alone. Investigators are now evaluating the vaccine in a global phase 3 study in patients with high-risk, stage IIB to IV melanoma following surgery.
Another one to watch is the BNT116 NSCLC vaccine from BioNTech. This vaccine presents the immune system with NSCLC tumor markers to encourage the body to fight cancer cells expressing those markers while ignoring healthy cells. BioNTech also launched a global clinical trial for its vaccine this year.
Other notables include a pancreatic cancer mRNA vaccine, which has shown promising early results in a small trial of 16 patients. Of 16 patients who received the vaccine alongside chemotherapy and after surgery and immunotherapy, 8 responded. Of these eight, six remained recurrence free at 3 years. Investigators noted that the vaccine appeared to stimulate a durable T-cell response in patients who responded.
Kwak has also continued his work on lymphoma vaccines. In August, his team published promising first-in-human data on the use of personalized neoantigen vaccines as an early intervention in untreated patients with lymphoplasmacytic lymphoma. Among nine asymptomatic patients who received the vaccine, all achieved stable disease or better, with no dose-limiting toxicities. One patient had a minor response, and the median time to progression was greater than 72 months.
“The current setting is more for advanced disease,” Kwak explained. “It’s a tougher task, but combined with checkpoint blockade, it may be potent enough to work.”
Still, caution is important. Despite early promise, it’s too soon to tell which, if any, of these investigational vaccines will pan out in the long run. Like investigational drugs, cancer vaccines may show big promising initially but then fail in larger trials.
One key to success, according to Kwak, is to design trials so that even negative results will inform next steps.
But, he noted, failures in large clinical trials will “put a chilling effect on cancer vaccine research again.”
“That’s what keeps me up at night,” he said. “We know the science is fundamentally sound and we have seen glimpses over decades of research that cancer vaccines can work, so it’s really just a matter of tweaking things to optimize trial design.”
Companies tend to design trials to test if a vaccine works or not, without trying to understand why, he said.
“What we need to do is design those so that we can learn from negative results,” he said. That’s what he and his colleagues attempted to do in their recent trial. “We didn’t just look at clinical results; we’re interrogating the actual tumor environment to understand what worked and didn’t and how to tweak that for the next trial.”
Kwak and his colleagues found, for instance, that the vaccine had a greater effect on B cell–derived tumor cells than on cells of plasma origin, so “the most rational design for the next iteration is to combine the vaccine with agents that work directly against plasma cells,” he explained.
As for what’s next, Kwak said: “We’re just focused on trying to do good science and understand. We’ve seen glimpses of success. That’s where we are.”
A version of this article first appeared on Medscape.com.
FDA Approves Bizengri for NSCLC and Pancreatic Cancers Harboring NRG1 Gene Fusion
Specifically, the systemic agent was approved for those with advanced, unresectable, or metastatic NSCLC or pancreatic adenocarcinoma harboring a neuregulin 1 (NRG1) gene fusion who progress on or after prior systemic therapy, according to the FDA.
The approval, based on findings from the multicenter, open-label eNRGy study, is the first from the FDA for a systemic therapy in this setting. In the multicohort study, treatment was associated with an overall response rate of 33% and 40% in 64 patients with NSCLC and 40 patients with pancreatic adenocarcinoma, respectively. Median duration of response was 7.4 months in the NSCLC patients and ranged from 3.7 to 16.6 months in those with pancreatic adenocarcinoma.
Adverse reactions occurring in at least 10% of patients included diarrhea, musculoskeletal pain, fatigue, nausea, infusion-related reactions, dyspnea, rash, constipation, vomiting, abdominal pain, and edema. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included increased gamma-glutamyl transferase and decreased hemoglobin, sodium, and platelets.
“The Personalized Medicine Coalition applauds the approval of BIZENGRI®,” Edward Abrahams, president of the Personalized Medicine Coalition, a Washington-based education and advocacy organization, stated in a press release from Merus. “In keeping with the growing number of personalized medicines on the market today, BIZENGRI® offers the only approved NRG1+ therapy for patients with these difficult-to-treat cancers.”
The agent is expected to be available for use in the “coming weeks,” according to Merus.
“The FDA approval of BIZENGRI® marks an important milestone for patients with pancreatic adenocarcinoma or NSCLC that is advanced unresectable or metastatic and harbors the NRG1 gene fusion,” noted Alison Schram, MD, an attending medical oncologist in the Early Drug Development Service at Memorial Sloan Kettering Cancer Center, New York City, and a principal investigator for the ongoing eNRGy trial. “I have seen firsthand how treatment with BIZENGRI® can deliver clinically meaningful outcomes for patients.”
Prescribing information for zenocutuzumab-zbco includes a Boxed Warning for embryo-fetal toxicity. The recommended treatment dose is 750 mg every 2 weeks until disease progression or unacceptable toxicity.
A version of this article first appeared on Medscape.com.
Specifically, the systemic agent was approved for those with advanced, unresectable, or metastatic NSCLC or pancreatic adenocarcinoma harboring a neuregulin 1 (NRG1) gene fusion who progress on or after prior systemic therapy, according to the FDA.
The approval, based on findings from the multicenter, open-label eNRGy study, is the first from the FDA for a systemic therapy in this setting. In the multicohort study, treatment was associated with an overall response rate of 33% and 40% in 64 patients with NSCLC and 40 patients with pancreatic adenocarcinoma, respectively. Median duration of response was 7.4 months in the NSCLC patients and ranged from 3.7 to 16.6 months in those with pancreatic adenocarcinoma.
Adverse reactions occurring in at least 10% of patients included diarrhea, musculoskeletal pain, fatigue, nausea, infusion-related reactions, dyspnea, rash, constipation, vomiting, abdominal pain, and edema. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included increased gamma-glutamyl transferase and decreased hemoglobin, sodium, and platelets.
“The Personalized Medicine Coalition applauds the approval of BIZENGRI®,” Edward Abrahams, president of the Personalized Medicine Coalition, a Washington-based education and advocacy organization, stated in a press release from Merus. “In keeping with the growing number of personalized medicines on the market today, BIZENGRI® offers the only approved NRG1+ therapy for patients with these difficult-to-treat cancers.”
The agent is expected to be available for use in the “coming weeks,” according to Merus.
“The FDA approval of BIZENGRI® marks an important milestone for patients with pancreatic adenocarcinoma or NSCLC that is advanced unresectable or metastatic and harbors the NRG1 gene fusion,” noted Alison Schram, MD, an attending medical oncologist in the Early Drug Development Service at Memorial Sloan Kettering Cancer Center, New York City, and a principal investigator for the ongoing eNRGy trial. “I have seen firsthand how treatment with BIZENGRI® can deliver clinically meaningful outcomes for patients.”
Prescribing information for zenocutuzumab-zbco includes a Boxed Warning for embryo-fetal toxicity. The recommended treatment dose is 750 mg every 2 weeks until disease progression or unacceptable toxicity.
A version of this article first appeared on Medscape.com.
Specifically, the systemic agent was approved for those with advanced, unresectable, or metastatic NSCLC or pancreatic adenocarcinoma harboring a neuregulin 1 (NRG1) gene fusion who progress on or after prior systemic therapy, according to the FDA.
The approval, based on findings from the multicenter, open-label eNRGy study, is the first from the FDA for a systemic therapy in this setting. In the multicohort study, treatment was associated with an overall response rate of 33% and 40% in 64 patients with NSCLC and 40 patients with pancreatic adenocarcinoma, respectively. Median duration of response was 7.4 months in the NSCLC patients and ranged from 3.7 to 16.6 months in those with pancreatic adenocarcinoma.
Adverse reactions occurring in at least 10% of patients included diarrhea, musculoskeletal pain, fatigue, nausea, infusion-related reactions, dyspnea, rash, constipation, vomiting, abdominal pain, and edema. Grade 3 or 4 laboratory abnormalities occurring in at least 10% of patients included increased gamma-glutamyl transferase and decreased hemoglobin, sodium, and platelets.
“The Personalized Medicine Coalition applauds the approval of BIZENGRI®,” Edward Abrahams, president of the Personalized Medicine Coalition, a Washington-based education and advocacy organization, stated in a press release from Merus. “In keeping with the growing number of personalized medicines on the market today, BIZENGRI® offers the only approved NRG1+ therapy for patients with these difficult-to-treat cancers.”
The agent is expected to be available for use in the “coming weeks,” according to Merus.
“The FDA approval of BIZENGRI® marks an important milestone for patients with pancreatic adenocarcinoma or NSCLC that is advanced unresectable or metastatic and harbors the NRG1 gene fusion,” noted Alison Schram, MD, an attending medical oncologist in the Early Drug Development Service at Memorial Sloan Kettering Cancer Center, New York City, and a principal investigator for the ongoing eNRGy trial. “I have seen firsthand how treatment with BIZENGRI® can deliver clinically meaningful outcomes for patients.”
Prescribing information for zenocutuzumab-zbco includes a Boxed Warning for embryo-fetal toxicity. The recommended treatment dose is 750 mg every 2 weeks until disease progression or unacceptable toxicity.
A version of this article first appeared on Medscape.com.
Inside the Patient-Oncologist Bond: Why It’s Often So Strong
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Rose Gerber was 39, mother to a third grader and a kindergartener, when the diagnosis came: Advanced HER2-positive breast cancer.
“On one of my first or second appointments, I took in a little picture of Alexander and Isabella,” Gerber said. Gerber showed her oncologist the picture and told her: “I’ll do anything. I just want to be there for them.”
That was 21 years ago. Today, her current cancer status is “no evidence of disease.”
Over the past 2 decades, Gerber has gotten to be there for her children. Her youngest is now a television producer and her oldest, a CPA.
In that time,
“I’ve seen multiple physicians over my 21 years, but my oncologist has always been the focal point, guiding me in the right direction,” Gerber said in an interview.
Over the years, Jaga guided Gerber through a range of treatment decisions, including a Herceptin clinical trial that the mom of two views as lifesaving. Jaga often took on the role of both doctor and therapist, even providing comfort in the smaller moments when Gerber would fret about her weight gain.
The oncologist-patient “bond is very, very, very special,” said Gerber, who now works as director of patient advocacy and education at the Community Oncology Alliance.
Gerber isn’t alone in calling out the depth of the oncologist-patient bond.
Over years, sometimes decades, patients and oncologists can experience a whole world together: The treatment successes, relapses, uncertainties, and tough calls. As a result, a deep therapeutic alliance often develops. And with each new hurdle or decision, that collaborative, human connection between doctor and patient continues to form new layers.
“It’s like a shared bonding experience over trauma, like strangers trapped on a subway and then we get out, and we’re now on the other side, celebrating together,” said Saad Khan, MD, an associate professor of medicine (oncology) at Stanford University in California.
Connecting Through Stress
Although studies exploring the oncologist-patient bond are limited, some research suggests that a strong therapeutic alliance between patients and oncologists not only provides a foundation for quality care but can also help improve patients’ quality of life, protect against suicidal ideation, and increase treatment adherence.
Because of how stressful and frightening a cancer diagnosis can be, creating “a trusting, uninterrupted, almost sacred environment for them” is paramount for Khan. “I have no doubt that the most important part of their treatment is that they find an oncologist in whom they have total confidence,” Khan wrote in a blog.
The stress that patients with cancer experience is well documented, but oncologists take on a lot themselves and can also experience intense stress (.
“I consider my patient’s battles to be my battles,” Khan wrote.
The stress can start with the daily schedule. Oncologists often have a high volume of patients and tend to spend more time with each individual than most.
According to a 2023 survey, oncologists see about 68 patients a week, on average, but some oncologists, like Khan, have many more. Khan typically sees 20-30 patients a day and continues to care for many over years.
The survey also found that oncologists tend to spend a lot of time with their patients. Compared with other physicians, oncologists are two times more likely to spend at least 25 minutes with each patient.
With this kind of patient volume and time, Khan said, “you’re going to be exhausted.”
What can compound the exhaustion are the occasions oncologists need to deliver bad news — this treatment isn’t working, your cancer has come roaring back and, perhaps the hardest, we have no therapeutic options left. The end-of-life conversations, in particular, can be heartbreaking, especially when a patient is young and not ready to stop trying.
“It can be hard for doctors to discuss the end of life,” Don Dizon, MD, director of the Pelvic Malignancies Program at Lifespan Cancer Institute and director of Medical Oncology at Rhode Island Hospital, Providence, wrote in a column in 2023. Instead, it can be tempting and is often easier to focus on the next treatment, “instilling hope that there’s more that can be done,” even if doing more will only do harm.
In the face of these challenging decisions, growing a personal connection with patients over time can help keep oncologists going.
“We’re not just chemotherapy salesmen,” Khan said in an interview. “We get to know their social support network, who’s going to be driving them [to and from appointments], where they go on vacation, their cat’s name, who their neighbors are.”
A ‘Special Relationship’
Ralph V. Boccia, MD, is often asked what he does.
The next question that often comes — “Why do I do what I do?” — is Boccia’s favorite.
“Someone needs to take these patients through their journey,” Boccia, the founder of The Center for Cancer and Blood Disorders, Bethesda, Maryland, typically responds. He also often notes that “it is a special relationship you develop with the patient and their families.”
Boccia thinks about one long-term patient who captures this bond.
Joan Pinson, 70, was diagnosed with multiple myeloma about 25 years ago, when patients’ average survival was about 4 years.
Over a quarter century, Pinson has pivoted to different treatments, amid multiple relapses and remissions. Throughout most of this cancer journey, Boccia has been her primary oncologist, performing a stem cell transplant in 2000 and steering her to six clinical trials.
Her last relapse was 2 years ago, and since then she has been doing well on oral chemotherapy.
“Every time I relapsed, by the next appointment, he’d say, ‘here is what we are going to do,’ ” Pinson recalled. “I never worried, I never panicked. I knew he would take care of me.”
Over the years, Pinson and Boccia have shared many personal moments, sometimes by accident. One special moment happened early on in Pinson’s cancer journey. During an appointment, Boccia had “one ear to the phone” as his wife was about to deliver their first baby, Pinson recalled.
Later, Pinson met that child as a young man working in Boccia’s lab. She has also met Boccia’s wife, a nurse, when she filled in one day in the chemotherapy room.
Boccia now also treats Pinson’s husband who has prostate cancer, and he ruled out cancer when Pinson’s son, now in his 40s, had some worrisome symptoms.
More than 2 decades ago, Pinson told Boccia her goal was to see her youngest child graduate from high school. Now, six grandsons later, she has lived far beyond that goal.
“He has kept me alive,” said Pinson.
The Dying Patient
Harsha Vyas, MD, FACP, remembers the first encounter his office had with a 29-year-old woman referred with a diagnosis of stage IV breast cancer.
After just 15 minutes in the waiting room, the woman announced she was leaving. Although office staff assured the woman that she was next, the patient walked out.
Several months later, Vyas was called for an inpatient consult. It was the same woman.
Her lungs were full of fluid, and she was struggling to breathe, said Vyas, president and CEO of the Cancer Center of Middle Georgia, Dublin, and assistant professor at Augusta University in Georgia.
The woman, a single mother, told Vyas about her three young kids at home and asked him, “Doc, do something, please help me,” he recalled.
“Absolutely,” Vyas told her. But he had to be brutally honest about her prognosis and firm that she needed to follow his instructions. “You have a breast cancer I cannot cure,” he said. “All I can do is control the disease.”
From that first day, until the day she died, she came to every appointment and followed the treatment plan Vyas laid out.
For about 2 years, she responded well to treatment. And as the time passed and the trust grew, she began to open up to him. She showed him pictures. She talked about her children and being a mother.
“I’ve got to get my kids in a better place. I’m going to be there for them,” he recalled her saying.
Vyas admired her resourcefulness. She held down a part-time job, working retail and at a local restaurant. She figured out childcare so she could get to her chemotherapy appointments every 3 weeks and manage the copays.
Several years later, when she knew she was approaching the end of her life, she asked Vyas a question that hit hard.
“Doc, I don’t want to die and my kids find me dead. What can we do about it?”
Vyas, who has three daughters, imagined how traumatic this would be for a child. She and Vyas made the shared decision to cease treatment and begin home hospice. When the end was approaching, a hospice worker took over, waiting for bodily functions to cease.
When news of a death comes, “I say a little prayer, it’s almost like a send-off for that soul. That helps me absorb the news ... and let it go.”
But when the bond grows strong over time, as with his patient with breast cancer, Vyas said, “a piece of her is still with me.”
Khan had no relevant disclosures. Boccia and Vyas had no disclosures.
A version of this article appeared on Medscape.com.
Thrombocytosis and Cancer Risk: Management in Primary Care
This transcript has been edited for clarity.
In this podcast, I’m going to talk about unexplained high platelet counts, or thrombocytosis, and the risk for cancer in primary care. Let’s start with a typical case we all might see in primary care.
Louisa is 47 years old and is the chief financial officer for a tech startup company. She presents to us in primary care feeling tired all the time — a very common presentation in primary care — with associated reduced appetite. Past medical history includes irritable bowel syndrome, and she’s an ex-smoker.
Systemic inquiry is unremarkable. Specifically, there is no history of weight loss. Louisa has not been prescribed any medication and uses over-the-counter remedies for her irritable bowel syndrome. Examination is also unremarkable. Blood tests were checked, which were all reassuring, except for a platelet count of 612 × 109 cells/L (usual normal range, about 150-450).
What do we do next? Do we refer for an urgent chest x-ray to exclude lung cancer? Do we check a quantitative immunohistochemical fecal occult blood test (qFIT) to identify any occult bleeding in her stool? Do we refer for a routine upper gastrointestinal endoscopy or pelvic ultrasound scan to exclude any upper gastrointestinal or endometrial malignancy?
Do we simply repeat the bloods? If so, do we repeat them routinely or urgently, and indeed, which ones should we recheck?
Louisa has an unexplained thrombocytosis. How do we manage this in primary care? Thrombocytosis is generally defined as a raised platelet count over 450. Importantly, thrombocytosis is a common incidental finding in around 2% of those over 40 years of age attending primary care. Reassuringly, 80%-90% of thrombocytosis is reactive, secondary to acute blood loss, infection, or inflammation, and the majority of cases resolve within 3 months.
Why the concern with Louisa then? Although most cases are reactive, clinical guidance (for example, NICE suspected cancer guidance in the UK and Scottish suspected cancer guidance in Scotland) reminds us that unexplained thrombocytosis is a risk marker for some solid-tumor malignancies.
Previous studies have demonstrated that unexplained thrombocytosis is associated with a 1-year cancer incidence of 11.6% in males and 6.2% in females, well exceeding the standard 3% threshold warranting investigation for underlying malignancy. However, thrombocytosis should not be used as a stand-alone diagnostic or screening test for cancer, or indeed to rule out cancer.
Instead, unexplained thrombocytosis should prompt us to think cancer. The Scottish suspected cancer referral guidelines include thrombocytosis in the investigation criteria for what they call the LEGO-C cancers — L for lung, E for endometrial, G for gastric, O for oesophageal, and C for colorectal, which is a useful reminder for us all.
What further history, examination, and investigations might we consider in primary care if we identify an unexplained high platelet count? As always, we should use our clinical judgment and trust our clinical acumen.
We should consider all the possible underlying causes, including infection, inflammation, and blood loss, including menstrual blood loss in women; myeloproliferative disorders such as polycythemia rubra vera, chronic myeloid leukemia, and essential thrombocythemia; and, of course, underlying malignancy. If a likely underlying reversible cause is present (for example, a recent lower respiratory tract infection), simply repeating the full blood count in 4-6 weeks is quite appropriate to see if the thrombocytosis has resolved.
Remember, 80%-90% of cases are reactive thrombocytosis, and most cases resolve within 3 months. If thrombocytosis is unexplained or not resolving, consider checking ferritin levels to exclude iron deficiency. Consider checking C-reactive protein (CRP) levels to exclude any inflammation, and also consider checking a blood film to exclude any hematologic disorders, in addition, of course, to more detailed history-taking and examination to elicit any red flags.
We can also consider a JAK2 gene mutation test, if it is available to you locally, or a hematology referral if we suspect a myeloproliferative disorder. JAK2 is a genetic mutation that may be present in people with essential thrombocythemia and can indicate a diagnosis of polycythemia rubra vera.
Subsequent to this, and again using our clinical judgment, we then need to exclude the LEGO-C cancers. Consider urgent chest x-ray to exclude lung cancer or pelvic ultrasound in women to exclude endometrial cancer. Also, we should consider an upper gastrointestinal endoscopy, particularly in those individuals who have associated upper gastrointestinal symptoms and/or weight loss.
Finally, consider a qFIT to identify any occult bleeding in the stool, again if it’s available to you, or certainly if not, urgent lower gastrointestinal investigations to exclude colorectal cancer.
Alongside these possible investigations, as always, we should safety-net appropriately within agreed timeframes and check for resolution of the thrombocytosis according to the condition being suspected. Remember, most cases resolve within 3 months.
Returning to Louisa, what did I do? After seeing a platelet count of 600, I subsequently telephoned her and reexplored her history, which yielded nil else of note. Specifically, there was no history of unexplained weight loss, no history of upper or lower gastrointestinal symptoms, and certainly nothing significantly different from her usual irritable bowel syndrome symptoms. There were also no respiratory or genitourinary symptoms of note.
I did arrange for Louisa to undergo a chest x-ray over the next few days, though, as she was an ex-smoker. This was subsequently reported as normal. I appreciate chest x-rays have poor sensitivity for detecting lung cancer, as highlighted in a number of recent papers, but it was mutually agreed with Louisa that we would simply repeat her blood test in around 6 weeks. As well as repeating the full blood count, I arranged to check her ferritin, CRP, and a blood film, and then I was planning to reassess her clinically in person.
These bloods and my subsequent clinical review were reassuring. In fact, her platelet count had normalized after that 6 weeks had elapsed. Her thrombocytosis had resolved.
I didn’t arrange any further follow-up for her, but I did give her the usual safety netting advice to re-present to me or one of my colleagues if she does develop any worrying symptoms or signs.
I appreciate these scenarios are not always this straightforward, but I wanted to outline what investigations and referrals we may need to consider in primary care if we encounter an unexplained high platelet count.
There are a couple of quality-improvement activities for us all to consider in primary care. Consider as a team how we would respond to an incidental finding of thrombocytosis on a full blood count. Also consider what are our safety-netting options for those found to have raised platelet counts but no other symptoms or risk factors for underlying malignancy.
Finally, I’ve produced a Medscape UK primary care hack or clinical aide-memoire on managing unexplained thrombocytosis and associated cancer risk in primary care for all healthcare professionals working in primary care. This can be found online. I hope you find this resource helpful.
Dr. Kevin Fernando, General practitioner partner with specialist interests in cardiovascular, renal, and metabolic medicine, North Berwick Group Practice in Scotland, has disclosed relevant financial relationships with Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Dexcom, Lilly, Menarini, Novartis, Novo Nordisk, Roche Diagnostics, Embecta, Roche Diabetes Care, Sanofi Menarini, and Daiichi Sankyo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
In this podcast, I’m going to talk about unexplained high platelet counts, or thrombocytosis, and the risk for cancer in primary care. Let’s start with a typical case we all might see in primary care.
Louisa is 47 years old and is the chief financial officer for a tech startup company. She presents to us in primary care feeling tired all the time — a very common presentation in primary care — with associated reduced appetite. Past medical history includes irritable bowel syndrome, and she’s an ex-smoker.
Systemic inquiry is unremarkable. Specifically, there is no history of weight loss. Louisa has not been prescribed any medication and uses over-the-counter remedies for her irritable bowel syndrome. Examination is also unremarkable. Blood tests were checked, which were all reassuring, except for a platelet count of 612 × 109 cells/L (usual normal range, about 150-450).
What do we do next? Do we refer for an urgent chest x-ray to exclude lung cancer? Do we check a quantitative immunohistochemical fecal occult blood test (qFIT) to identify any occult bleeding in her stool? Do we refer for a routine upper gastrointestinal endoscopy or pelvic ultrasound scan to exclude any upper gastrointestinal or endometrial malignancy?
Do we simply repeat the bloods? If so, do we repeat them routinely or urgently, and indeed, which ones should we recheck?
Louisa has an unexplained thrombocytosis. How do we manage this in primary care? Thrombocytosis is generally defined as a raised platelet count over 450. Importantly, thrombocytosis is a common incidental finding in around 2% of those over 40 years of age attending primary care. Reassuringly, 80%-90% of thrombocytosis is reactive, secondary to acute blood loss, infection, or inflammation, and the majority of cases resolve within 3 months.
Why the concern with Louisa then? Although most cases are reactive, clinical guidance (for example, NICE suspected cancer guidance in the UK and Scottish suspected cancer guidance in Scotland) reminds us that unexplained thrombocytosis is a risk marker for some solid-tumor malignancies.
Previous studies have demonstrated that unexplained thrombocytosis is associated with a 1-year cancer incidence of 11.6% in males and 6.2% in females, well exceeding the standard 3% threshold warranting investigation for underlying malignancy. However, thrombocytosis should not be used as a stand-alone diagnostic or screening test for cancer, or indeed to rule out cancer.
Instead, unexplained thrombocytosis should prompt us to think cancer. The Scottish suspected cancer referral guidelines include thrombocytosis in the investigation criteria for what they call the LEGO-C cancers — L for lung, E for endometrial, G for gastric, O for oesophageal, and C for colorectal, which is a useful reminder for us all.
What further history, examination, and investigations might we consider in primary care if we identify an unexplained high platelet count? As always, we should use our clinical judgment and trust our clinical acumen.
We should consider all the possible underlying causes, including infection, inflammation, and blood loss, including menstrual blood loss in women; myeloproliferative disorders such as polycythemia rubra vera, chronic myeloid leukemia, and essential thrombocythemia; and, of course, underlying malignancy. If a likely underlying reversible cause is present (for example, a recent lower respiratory tract infection), simply repeating the full blood count in 4-6 weeks is quite appropriate to see if the thrombocytosis has resolved.
Remember, 80%-90% of cases are reactive thrombocytosis, and most cases resolve within 3 months. If thrombocytosis is unexplained or not resolving, consider checking ferritin levels to exclude iron deficiency. Consider checking C-reactive protein (CRP) levels to exclude any inflammation, and also consider checking a blood film to exclude any hematologic disorders, in addition, of course, to more detailed history-taking and examination to elicit any red flags.
We can also consider a JAK2 gene mutation test, if it is available to you locally, or a hematology referral if we suspect a myeloproliferative disorder. JAK2 is a genetic mutation that may be present in people with essential thrombocythemia and can indicate a diagnosis of polycythemia rubra vera.
Subsequent to this, and again using our clinical judgment, we then need to exclude the LEGO-C cancers. Consider urgent chest x-ray to exclude lung cancer or pelvic ultrasound in women to exclude endometrial cancer. Also, we should consider an upper gastrointestinal endoscopy, particularly in those individuals who have associated upper gastrointestinal symptoms and/or weight loss.
Finally, consider a qFIT to identify any occult bleeding in the stool, again if it’s available to you, or certainly if not, urgent lower gastrointestinal investigations to exclude colorectal cancer.
Alongside these possible investigations, as always, we should safety-net appropriately within agreed timeframes and check for resolution of the thrombocytosis according to the condition being suspected. Remember, most cases resolve within 3 months.
Returning to Louisa, what did I do? After seeing a platelet count of 600, I subsequently telephoned her and reexplored her history, which yielded nil else of note. Specifically, there was no history of unexplained weight loss, no history of upper or lower gastrointestinal symptoms, and certainly nothing significantly different from her usual irritable bowel syndrome symptoms. There were also no respiratory or genitourinary symptoms of note.
I did arrange for Louisa to undergo a chest x-ray over the next few days, though, as she was an ex-smoker. This was subsequently reported as normal. I appreciate chest x-rays have poor sensitivity for detecting lung cancer, as highlighted in a number of recent papers, but it was mutually agreed with Louisa that we would simply repeat her blood test in around 6 weeks. As well as repeating the full blood count, I arranged to check her ferritin, CRP, and a blood film, and then I was planning to reassess her clinically in person.
These bloods and my subsequent clinical review were reassuring. In fact, her platelet count had normalized after that 6 weeks had elapsed. Her thrombocytosis had resolved.
I didn’t arrange any further follow-up for her, but I did give her the usual safety netting advice to re-present to me or one of my colleagues if she does develop any worrying symptoms or signs.
I appreciate these scenarios are not always this straightforward, but I wanted to outline what investigations and referrals we may need to consider in primary care if we encounter an unexplained high platelet count.
There are a couple of quality-improvement activities for us all to consider in primary care. Consider as a team how we would respond to an incidental finding of thrombocytosis on a full blood count. Also consider what are our safety-netting options for those found to have raised platelet counts but no other symptoms or risk factors for underlying malignancy.
Finally, I’ve produced a Medscape UK primary care hack or clinical aide-memoire on managing unexplained thrombocytosis and associated cancer risk in primary care for all healthcare professionals working in primary care. This can be found online. I hope you find this resource helpful.
Dr. Kevin Fernando, General practitioner partner with specialist interests in cardiovascular, renal, and metabolic medicine, North Berwick Group Practice in Scotland, has disclosed relevant financial relationships with Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Dexcom, Lilly, Menarini, Novartis, Novo Nordisk, Roche Diagnostics, Embecta, Roche Diabetes Care, Sanofi Menarini, and Daiichi Sankyo.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
In this podcast, I’m going to talk about unexplained high platelet counts, or thrombocytosis, and the risk for cancer in primary care. Let’s start with a typical case we all might see in primary care.
Louisa is 47 years old and is the chief financial officer for a tech startup company. She presents to us in primary care feeling tired all the time — a very common presentation in primary care — with associated reduced appetite. Past medical history includes irritable bowel syndrome, and she’s an ex-smoker.
Systemic inquiry is unremarkable. Specifically, there is no history of weight loss. Louisa has not been prescribed any medication and uses over-the-counter remedies for her irritable bowel syndrome. Examination is also unremarkable. Blood tests were checked, which were all reassuring, except for a platelet count of 612 × 109 cells/L (usual normal range, about 150-450).
What do we do next? Do we refer for an urgent chest x-ray to exclude lung cancer? Do we check a quantitative immunohistochemical fecal occult blood test (qFIT) to identify any occult bleeding in her stool? Do we refer for a routine upper gastrointestinal endoscopy or pelvic ultrasound scan to exclude any upper gastrointestinal or endometrial malignancy?
Do we simply repeat the bloods? If so, do we repeat them routinely or urgently, and indeed, which ones should we recheck?
Louisa has an unexplained thrombocytosis. How do we manage this in primary care? Thrombocytosis is generally defined as a raised platelet count over 450. Importantly, thrombocytosis is a common incidental finding in around 2% of those over 40 years of age attending primary care. Reassuringly, 80%-90% of thrombocytosis is reactive, secondary to acute blood loss, infection, or inflammation, and the majority of cases resolve within 3 months.
Why the concern with Louisa then? Although most cases are reactive, clinical guidance (for example, NICE suspected cancer guidance in the UK and Scottish suspected cancer guidance in Scotland) reminds us that unexplained thrombocytosis is a risk marker for some solid-tumor malignancies.
Previous studies have demonstrated that unexplained thrombocytosis is associated with a 1-year cancer incidence of 11.6% in males and 6.2% in females, well exceeding the standard 3% threshold warranting investigation for underlying malignancy. However, thrombocytosis should not be used as a stand-alone diagnostic or screening test for cancer, or indeed to rule out cancer.
Instead, unexplained thrombocytosis should prompt us to think cancer. The Scottish suspected cancer referral guidelines include thrombocytosis in the investigation criteria for what they call the LEGO-C cancers — L for lung, E for endometrial, G for gastric, O for oesophageal, and C for colorectal, which is a useful reminder for us all.
What further history, examination, and investigations might we consider in primary care if we identify an unexplained high platelet count? As always, we should use our clinical judgment and trust our clinical acumen.
We should consider all the possible underlying causes, including infection, inflammation, and blood loss, including menstrual blood loss in women; myeloproliferative disorders such as polycythemia rubra vera, chronic myeloid leukemia, and essential thrombocythemia; and, of course, underlying malignancy. If a likely underlying reversible cause is present (for example, a recent lower respiratory tract infection), simply repeating the full blood count in 4-6 weeks is quite appropriate to see if the thrombocytosis has resolved.
Remember, 80%-90% of cases are reactive thrombocytosis, and most cases resolve within 3 months. If thrombocytosis is unexplained or not resolving, consider checking ferritin levels to exclude iron deficiency. Consider checking C-reactive protein (CRP) levels to exclude any inflammation, and also consider checking a blood film to exclude any hematologic disorders, in addition, of course, to more detailed history-taking and examination to elicit any red flags.
We can also consider a JAK2 gene mutation test, if it is available to you locally, or a hematology referral if we suspect a myeloproliferative disorder. JAK2 is a genetic mutation that may be present in people with essential thrombocythemia and can indicate a diagnosis of polycythemia rubra vera.
Subsequent to this, and again using our clinical judgment, we then need to exclude the LEGO-C cancers. Consider urgent chest x-ray to exclude lung cancer or pelvic ultrasound in women to exclude endometrial cancer. Also, we should consider an upper gastrointestinal endoscopy, particularly in those individuals who have associated upper gastrointestinal symptoms and/or weight loss.
Finally, consider a qFIT to identify any occult bleeding in the stool, again if it’s available to you, or certainly if not, urgent lower gastrointestinal investigations to exclude colorectal cancer.
Alongside these possible investigations, as always, we should safety-net appropriately within agreed timeframes and check for resolution of the thrombocytosis according to the condition being suspected. Remember, most cases resolve within 3 months.
Returning to Louisa, what did I do? After seeing a platelet count of 600, I subsequently telephoned her and reexplored her history, which yielded nil else of note. Specifically, there was no history of unexplained weight loss, no history of upper or lower gastrointestinal symptoms, and certainly nothing significantly different from her usual irritable bowel syndrome symptoms. There were also no respiratory or genitourinary symptoms of note.
I did arrange for Louisa to undergo a chest x-ray over the next few days, though, as she was an ex-smoker. This was subsequently reported as normal. I appreciate chest x-rays have poor sensitivity for detecting lung cancer, as highlighted in a number of recent papers, but it was mutually agreed with Louisa that we would simply repeat her blood test in around 6 weeks. As well as repeating the full blood count, I arranged to check her ferritin, CRP, and a blood film, and then I was planning to reassess her clinically in person.
These bloods and my subsequent clinical review were reassuring. In fact, her platelet count had normalized after that 6 weeks had elapsed. Her thrombocytosis had resolved.
I didn’t arrange any further follow-up for her, but I did give her the usual safety netting advice to re-present to me or one of my colleagues if she does develop any worrying symptoms or signs.
I appreciate these scenarios are not always this straightforward, but I wanted to outline what investigations and referrals we may need to consider in primary care if we encounter an unexplained high platelet count.
There are a couple of quality-improvement activities for us all to consider in primary care. Consider as a team how we would respond to an incidental finding of thrombocytosis on a full blood count. Also consider what are our safety-netting options for those found to have raised platelet counts but no other symptoms or risk factors for underlying malignancy.
Finally, I’ve produced a Medscape UK primary care hack or clinical aide-memoire on managing unexplained thrombocytosis and associated cancer risk in primary care for all healthcare professionals working in primary care. This can be found online. I hope you find this resource helpful.
Dr. Kevin Fernando, General practitioner partner with specialist interests in cardiovascular, renal, and metabolic medicine, North Berwick Group Practice in Scotland, has disclosed relevant financial relationships with Amarin, Amgen, AstraZeneca, Bayer, Boehringer Ingelheim, Dexcom, Lilly, Menarini, Novartis, Novo Nordisk, Roche Diagnostics, Embecta, Roche Diabetes Care, Sanofi Menarini, and Daiichi Sankyo.
A version of this article first appeared on Medscape.com.


