User login
FDA approves infection-detecting system

Staphylococcus infection
Credit: Bill Branson
The US Food and Drug Administration (FDA) has approved the first mass spectrometer system for automated identification of bacteria and yeasts that are known to cause serious illnesses in humans.
The system, called VITEK MS, can identify 193 different microorganisms and perform up to 192 different tests, each of which takes about 1 minute.
The VITEK MS can identify yeasts and bacteria associated with skin infections, pneumonia, meningitis, and bloodstream infections.
Patients whose immune systems are compromised or weakened by HIV/AIDS, cancer treatment, or antirejection therapy following transplants are particularly vulnerable to these infections.
“The ability for laboratories to use 1 device to identify almost 200 different microorganisms is a significant advance in the timely identification of pathogenic microorganisms,” said Alberto Gutierrez, PhD, director of the Office of In Vitro Diagnostics and Radiological Health at FDA’s Center for Devices and Radiological Health.
The VITEK MS incorporates a technology called matrix-assisted laser desorption/ionization–time of flight mass spectrometry. The technology uses a laser to break yeast and bacteria specimens into small particles that form a pattern unique to the microorganism.
The VITEK MS automatically compares the microorganism pattern to 193 known yeasts and bacteria in the system’s database to identify the microorganism.
Compared to other identification methods that require abundant organism growth for testing, mass spectrometry requires only a small amount of yeast or bacterial growth.
So testing can start as soon as growth is visible, generally within 18 to 24 hours. Traditional methods can take up to 5 days to produce the same identification results.
The FDA reviewed the VITEK MS through its de novo classification process, a regulatory pathway for some novel, low-to-moderate-risk medical devices that are not substantially equivalent to an already legally marketed device.
The FDA based its decision on the results of a study of 7068 microorganisms. When compared to sequencing and biochemical testing, the VITEK MS correctly identified the scientific group or family 93.6% of the time (with 87.5% of microorganisms identified to species level).
The system provided a “no identification” result for 3.2% of the microorganisms in the study, 0.8% of the test results were incorrect, and 2.4% were low discrimination with no correct result.
The VITEK MS is manufactured by bioMerieux, Inc., located in Durham, North Carolina. ![]()

Staphylococcus infection
Credit: Bill Branson
The US Food and Drug Administration (FDA) has approved the first mass spectrometer system for automated identification of bacteria and yeasts that are known to cause serious illnesses in humans.
The system, called VITEK MS, can identify 193 different microorganisms and perform up to 192 different tests, each of which takes about 1 minute.
The VITEK MS can identify yeasts and bacteria associated with skin infections, pneumonia, meningitis, and bloodstream infections.
Patients whose immune systems are compromised or weakened by HIV/AIDS, cancer treatment, or antirejection therapy following transplants are particularly vulnerable to these infections.
“The ability for laboratories to use 1 device to identify almost 200 different microorganisms is a significant advance in the timely identification of pathogenic microorganisms,” said Alberto Gutierrez, PhD, director of the Office of In Vitro Diagnostics and Radiological Health at FDA’s Center for Devices and Radiological Health.
The VITEK MS incorporates a technology called matrix-assisted laser desorption/ionization–time of flight mass spectrometry. The technology uses a laser to break yeast and bacteria specimens into small particles that form a pattern unique to the microorganism.
The VITEK MS automatically compares the microorganism pattern to 193 known yeasts and bacteria in the system’s database to identify the microorganism.
Compared to other identification methods that require abundant organism growth for testing, mass spectrometry requires only a small amount of yeast or bacterial growth.
So testing can start as soon as growth is visible, generally within 18 to 24 hours. Traditional methods can take up to 5 days to produce the same identification results.
The FDA reviewed the VITEK MS through its de novo classification process, a regulatory pathway for some novel, low-to-moderate-risk medical devices that are not substantially equivalent to an already legally marketed device.
The FDA based its decision on the results of a study of 7068 microorganisms. When compared to sequencing and biochemical testing, the VITEK MS correctly identified the scientific group or family 93.6% of the time (with 87.5% of microorganisms identified to species level).
The system provided a “no identification” result for 3.2% of the microorganisms in the study, 0.8% of the test results were incorrect, and 2.4% were low discrimination with no correct result.
The VITEK MS is manufactured by bioMerieux, Inc., located in Durham, North Carolina. ![]()

Staphylococcus infection
Credit: Bill Branson
The US Food and Drug Administration (FDA) has approved the first mass spectrometer system for automated identification of bacteria and yeasts that are known to cause serious illnesses in humans.
The system, called VITEK MS, can identify 193 different microorganisms and perform up to 192 different tests, each of which takes about 1 minute.
The VITEK MS can identify yeasts and bacteria associated with skin infections, pneumonia, meningitis, and bloodstream infections.
Patients whose immune systems are compromised or weakened by HIV/AIDS, cancer treatment, or antirejection therapy following transplants are particularly vulnerable to these infections.
“The ability for laboratories to use 1 device to identify almost 200 different microorganisms is a significant advance in the timely identification of pathogenic microorganisms,” said Alberto Gutierrez, PhD, director of the Office of In Vitro Diagnostics and Radiological Health at FDA’s Center for Devices and Radiological Health.
The VITEK MS incorporates a technology called matrix-assisted laser desorption/ionization–time of flight mass spectrometry. The technology uses a laser to break yeast and bacteria specimens into small particles that form a pattern unique to the microorganism.
The VITEK MS automatically compares the microorganism pattern to 193 known yeasts and bacteria in the system’s database to identify the microorganism.
Compared to other identification methods that require abundant organism growth for testing, mass spectrometry requires only a small amount of yeast or bacterial growth.
So testing can start as soon as growth is visible, generally within 18 to 24 hours. Traditional methods can take up to 5 days to produce the same identification results.
The FDA reviewed the VITEK MS through its de novo classification process, a regulatory pathway for some novel, low-to-moderate-risk medical devices that are not substantially equivalent to an already legally marketed device.
The FDA based its decision on the results of a study of 7068 microorganisms. When compared to sequencing and biochemical testing, the VITEK MS correctly identified the scientific group or family 93.6% of the time (with 87.5% of microorganisms identified to species level).
The system provided a “no identification” result for 3.2% of the microorganisms in the study, 0.8% of the test results were incorrect, and 2.4% were low discrimination with no correct result.
The VITEK MS is manufactured by bioMerieux, Inc., located in Durham, North Carolina. ![]()
Investigational vaccine can prevent malaria
Changing the route of administration has significantly improved the efficacy of an investigational malaria vaccine, according to a study published in Science.
The vaccine, called PfSPZ, is composed of live but weakened sporozoites of Plasmodium falciparum.
In a previous clinical trial, the vaccine proved largely ineffective at preventing malaria. But in that study, PfSPZ was administered either intradermally or subcutaneously.
In a new phase 1 study, researchers administered the vaccine intravenously at varying doses. And they found that, at high doses, PfSPZ offered protection against malaria in most subjects.
“In this trial, we showed, in principle, that sporozoites can be developed into a malaria vaccine that confers high levels of protection . . . ,” said study author Robert A. Seder, MD, chief of the Cellular Immunology Section of the NIAID Vaccine Research Center in Bethesda, Maryland.
Dr Seder and his colleagues tested the vaccine in 57 healthy adult volunteers, aged 18 to 45 years, who never had malaria. Forty of the subjects received the vaccine, and 17 did not.
Adverse reactions
To evaluate safety, the researchers divided recipients into groups receiving 2 to 6 intravenous doses of PfSPZ vaccine at increasing dosages. After vaccination, the team monitored subjects closely for 7 days.
There were no malaria infections related to vaccination. However, there were a number of adverse events.
Nine subjects had mild pain/tenderness or bruising, and 1 had moderate bruising at the injection site. Sixteen participants had mild solicited systemic reactogenicity, 4 had moderate, and 1 had severe reactogenicity.
Sixteen subjects also had transient, asymptomatic increases in aspartate aminotransferase and/or alanine aminotransferase that was possibly related to vaccination.
Response and protection
Based on blood measurements, the researchers found that subjects who received a higher total dosage of PfSPZ vaccine generated more antibodies against malaria and more T cells specific to the vaccine.
To evaluate whether and how well the PfSPZ vaccine prevented malaria infection, each participant—both vaccinated individuals and the control group—was exposed to bites by 5 mosquitoes carrying the P falciparum strain.
This took place 3 weeks after participants received their final vaccination. The researchers monitored the subjects as outpatients for 7 days and then admitted them to the clinic, where they stayed until they were diagnosed with malaria, treated with antimalarial drugs and cured of infection, or shown to be free of infection.
The researchers found that higher dosages of PfSPZ vaccine were associated with protection against malaria infection. Three of the 15 participants who received higher dosages of the vaccine became infected, compared to 16 of 17 subjects in the lower-dosage group.
Among the 12 participants who received no vaccine, 11 became infected with malaria after the mosquito challenge.
“[T]hese trial results are a promising first step in generating high-level protection against malaria, and they allow for future studies to optimize the dose, schedule, and delivery route of the candidate vaccine,” Dr Seder said.
A number of follow-up studies are planned, including research to evaluate the vaccine’s different dose schedules, possible protection against other Plasmodium strains, and the durability of protection.
The researchers may also evaluate whether higher doses administered subcutaneously or intradermally provide the same level of protection as that found in this study.
The PfSPZ vaccine was developed by scientists at Sanaria Inc., of Rockville, Maryland. ![]()
Changing the route of administration has significantly improved the efficacy of an investigational malaria vaccine, according to a study published in Science.
The vaccine, called PfSPZ, is composed of live but weakened sporozoites of Plasmodium falciparum.
In a previous clinical trial, the vaccine proved largely ineffective at preventing malaria. But in that study, PfSPZ was administered either intradermally or subcutaneously.
In a new phase 1 study, researchers administered the vaccine intravenously at varying doses. And they found that, at high doses, PfSPZ offered protection against malaria in most subjects.
“In this trial, we showed, in principle, that sporozoites can be developed into a malaria vaccine that confers high levels of protection . . . ,” said study author Robert A. Seder, MD, chief of the Cellular Immunology Section of the NIAID Vaccine Research Center in Bethesda, Maryland.
Dr Seder and his colleagues tested the vaccine in 57 healthy adult volunteers, aged 18 to 45 years, who never had malaria. Forty of the subjects received the vaccine, and 17 did not.
Adverse reactions
To evaluate safety, the researchers divided recipients into groups receiving 2 to 6 intravenous doses of PfSPZ vaccine at increasing dosages. After vaccination, the team monitored subjects closely for 7 days.
There were no malaria infections related to vaccination. However, there were a number of adverse events.
Nine subjects had mild pain/tenderness or bruising, and 1 had moderate bruising at the injection site. Sixteen participants had mild solicited systemic reactogenicity, 4 had moderate, and 1 had severe reactogenicity.
Sixteen subjects also had transient, asymptomatic increases in aspartate aminotransferase and/or alanine aminotransferase that was possibly related to vaccination.
Response and protection
Based on blood measurements, the researchers found that subjects who received a higher total dosage of PfSPZ vaccine generated more antibodies against malaria and more T cells specific to the vaccine.
To evaluate whether and how well the PfSPZ vaccine prevented malaria infection, each participant—both vaccinated individuals and the control group—was exposed to bites by 5 mosquitoes carrying the P falciparum strain.
This took place 3 weeks after participants received their final vaccination. The researchers monitored the subjects as outpatients for 7 days and then admitted them to the clinic, where they stayed until they were diagnosed with malaria, treated with antimalarial drugs and cured of infection, or shown to be free of infection.
The researchers found that higher dosages of PfSPZ vaccine were associated with protection against malaria infection. Three of the 15 participants who received higher dosages of the vaccine became infected, compared to 16 of 17 subjects in the lower-dosage group.
Among the 12 participants who received no vaccine, 11 became infected with malaria after the mosquito challenge.
“[T]hese trial results are a promising first step in generating high-level protection against malaria, and they allow for future studies to optimize the dose, schedule, and delivery route of the candidate vaccine,” Dr Seder said.
A number of follow-up studies are planned, including research to evaluate the vaccine’s different dose schedules, possible protection against other Plasmodium strains, and the durability of protection.
The researchers may also evaluate whether higher doses administered subcutaneously or intradermally provide the same level of protection as that found in this study.
The PfSPZ vaccine was developed by scientists at Sanaria Inc., of Rockville, Maryland. ![]()
Changing the route of administration has significantly improved the efficacy of an investigational malaria vaccine, according to a study published in Science.
The vaccine, called PfSPZ, is composed of live but weakened sporozoites of Plasmodium falciparum.
In a previous clinical trial, the vaccine proved largely ineffective at preventing malaria. But in that study, PfSPZ was administered either intradermally or subcutaneously.
In a new phase 1 study, researchers administered the vaccine intravenously at varying doses. And they found that, at high doses, PfSPZ offered protection against malaria in most subjects.
“In this trial, we showed, in principle, that sporozoites can be developed into a malaria vaccine that confers high levels of protection . . . ,” said study author Robert A. Seder, MD, chief of the Cellular Immunology Section of the NIAID Vaccine Research Center in Bethesda, Maryland.
Dr Seder and his colleagues tested the vaccine in 57 healthy adult volunteers, aged 18 to 45 years, who never had malaria. Forty of the subjects received the vaccine, and 17 did not.
Adverse reactions
To evaluate safety, the researchers divided recipients into groups receiving 2 to 6 intravenous doses of PfSPZ vaccine at increasing dosages. After vaccination, the team monitored subjects closely for 7 days.
There were no malaria infections related to vaccination. However, there were a number of adverse events.
Nine subjects had mild pain/tenderness or bruising, and 1 had moderate bruising at the injection site. Sixteen participants had mild solicited systemic reactogenicity, 4 had moderate, and 1 had severe reactogenicity.
Sixteen subjects also had transient, asymptomatic increases in aspartate aminotransferase and/or alanine aminotransferase that was possibly related to vaccination.
Response and protection
Based on blood measurements, the researchers found that subjects who received a higher total dosage of PfSPZ vaccine generated more antibodies against malaria and more T cells specific to the vaccine.
To evaluate whether and how well the PfSPZ vaccine prevented malaria infection, each participant—both vaccinated individuals and the control group—was exposed to bites by 5 mosquitoes carrying the P falciparum strain.
This took place 3 weeks after participants received their final vaccination. The researchers monitored the subjects as outpatients for 7 days and then admitted them to the clinic, where they stayed until they were diagnosed with malaria, treated with antimalarial drugs and cured of infection, or shown to be free of infection.
The researchers found that higher dosages of PfSPZ vaccine were associated with protection against malaria infection. Three of the 15 participants who received higher dosages of the vaccine became infected, compared to 16 of 17 subjects in the lower-dosage group.
Among the 12 participants who received no vaccine, 11 became infected with malaria after the mosquito challenge.
“[T]hese trial results are a promising first step in generating high-level protection against malaria, and they allow for future studies to optimize the dose, schedule, and delivery route of the candidate vaccine,” Dr Seder said.
A number of follow-up studies are planned, including research to evaluate the vaccine’s different dose schedules, possible protection against other Plasmodium strains, and the durability of protection.
The researchers may also evaluate whether higher doses administered subcutaneously or intradermally provide the same level of protection as that found in this study.
The PfSPZ vaccine was developed by scientists at Sanaria Inc., of Rockville, Maryland. ![]()
Survey suggests docs could improve trial enrollment
Results of a nationwide poll indicate that many Americans believe participation in clinical trials is important.
And most of the individuals surveyed said they would participate in a trial if their doctor recommended it.
However, many respondents said they learned of clinical trials via the Internet, not from discussions with their physician. And most said a physician’s recommendation was an important factor in the decision to enroll in a clinical trial.
This poll was conducted online by Zogby Analytics. The results are available on the Research!America website.
The goal of the survey was to evaluate respondents’ perceptions about clinical trials and determine if perceptions differed according to racial groups. The survey included 684 non-Hispanic whites, 406 Hispanics, 403 African-Americans, and 300 Asians.
A willingness to participate
Most of the African-Americans (62%) and Hispanics (57%) polled said it’s very important to participate in a clinical trial to improve the health of others. Fifty percent of Asians agreed, as did 49% of non-Hispanic whites.
Furthermore, 75% of Hispanics, 72% of African-Americans, 71% of non-Hispanic whites, and 65% of Asians said they would likely participate in a clinical trial if it was recommended by a doctor.
And about three-quarters of all respondents said, assuming the correct privacy protections were in place, they would be willing to share personal health information to advance medical research.
“The poll reveals a willingness among minorities to participate in clinical trials to improve the quality of healthcare, but enrollment remains stubbornly low,” said Mary Woolley, president and CEO of Research!America.
Among those individuals surveyed, only 17% of Hispanics, 15% of African-Americans, 15% of non-Hispanic whites, and 11% of Asians said they or a family member had participated in a clinical trial.
When asked to give a reason for low enrollment in clinical trials, most respondents cited a lack of trust—61% of African-Americans, 54% of non-Hispanic whites, 52% of Hispanics, and 51% of Asians.
The importance of physicians, institutions
Many of the individuals surveyed said they had learned of clinical trials via the Internet—56% of Asians, 52% of non-Hispanic whites, 50% of African-Americans, and 45% of Hispanics.
Far fewer said they had heard of trials during discussions with their doctors—22% of Hispanics, 22% of non-Hispanic whites, 20% of Asians, and 17% of African-Americans.
And most respondents said a doctor’s recommendation is a “very” or “somewhat” important factor in the decision to participate in a clinical trial—81% of Hispanics, 81% of non-Hispanic whites, 80% of Asians, and 78% of African-Americans.
Institutional reputation was also deemed an important factor by 74% of African-Americans, 72% of non-Hispanic whites, 66% of Hispanics, and 66% of Asians.
And many respondents said healthcare providers should play a major role in raising awareness of clinical trials. In fact, 42% of non-Hispanic whites, 38% of Hispanics, 36% of Asians, and 33% of African-Americans said providers have the greatest responsibility in educating the public about clinical trials.
“It’s imperative that healthcare providers and others help patients gain a deeper knowledge of clinical trials so all Americans can benefit from life-saving treatments,” Woolley said.
This survey was conducted by Zogby Analytics for Research!America, the Association of Clinical Research Organizations, the Clinical Research Forum, the Friends of the National Library of Medicine, and the Clinical Trials Transformation Initiative. ![]()
Results of a nationwide poll indicate that many Americans believe participation in clinical trials is important.
And most of the individuals surveyed said they would participate in a trial if their doctor recommended it.
However, many respondents said they learned of clinical trials via the Internet, not from discussions with their physician. And most said a physician’s recommendation was an important factor in the decision to enroll in a clinical trial.
This poll was conducted online by Zogby Analytics. The results are available on the Research!America website.
The goal of the survey was to evaluate respondents’ perceptions about clinical trials and determine if perceptions differed according to racial groups. The survey included 684 non-Hispanic whites, 406 Hispanics, 403 African-Americans, and 300 Asians.
A willingness to participate
Most of the African-Americans (62%) and Hispanics (57%) polled said it’s very important to participate in a clinical trial to improve the health of others. Fifty percent of Asians agreed, as did 49% of non-Hispanic whites.
Furthermore, 75% of Hispanics, 72% of African-Americans, 71% of non-Hispanic whites, and 65% of Asians said they would likely participate in a clinical trial if it was recommended by a doctor.
And about three-quarters of all respondents said, assuming the correct privacy protections were in place, they would be willing to share personal health information to advance medical research.
“The poll reveals a willingness among minorities to participate in clinical trials to improve the quality of healthcare, but enrollment remains stubbornly low,” said Mary Woolley, president and CEO of Research!America.
Among those individuals surveyed, only 17% of Hispanics, 15% of African-Americans, 15% of non-Hispanic whites, and 11% of Asians said they or a family member had participated in a clinical trial.
When asked to give a reason for low enrollment in clinical trials, most respondents cited a lack of trust—61% of African-Americans, 54% of non-Hispanic whites, 52% of Hispanics, and 51% of Asians.
The importance of physicians, institutions
Many of the individuals surveyed said they had learned of clinical trials via the Internet—56% of Asians, 52% of non-Hispanic whites, 50% of African-Americans, and 45% of Hispanics.
Far fewer said they had heard of trials during discussions with their doctors—22% of Hispanics, 22% of non-Hispanic whites, 20% of Asians, and 17% of African-Americans.
And most respondents said a doctor’s recommendation is a “very” or “somewhat” important factor in the decision to participate in a clinical trial—81% of Hispanics, 81% of non-Hispanic whites, 80% of Asians, and 78% of African-Americans.
Institutional reputation was also deemed an important factor by 74% of African-Americans, 72% of non-Hispanic whites, 66% of Hispanics, and 66% of Asians.
And many respondents said healthcare providers should play a major role in raising awareness of clinical trials. In fact, 42% of non-Hispanic whites, 38% of Hispanics, 36% of Asians, and 33% of African-Americans said providers have the greatest responsibility in educating the public about clinical trials.
“It’s imperative that healthcare providers and others help patients gain a deeper knowledge of clinical trials so all Americans can benefit from life-saving treatments,” Woolley said.
This survey was conducted by Zogby Analytics for Research!America, the Association of Clinical Research Organizations, the Clinical Research Forum, the Friends of the National Library of Medicine, and the Clinical Trials Transformation Initiative. ![]()
Results of a nationwide poll indicate that many Americans believe participation in clinical trials is important.
And most of the individuals surveyed said they would participate in a trial if their doctor recommended it.
However, many respondents said they learned of clinical trials via the Internet, not from discussions with their physician. And most said a physician’s recommendation was an important factor in the decision to enroll in a clinical trial.
This poll was conducted online by Zogby Analytics. The results are available on the Research!America website.
The goal of the survey was to evaluate respondents’ perceptions about clinical trials and determine if perceptions differed according to racial groups. The survey included 684 non-Hispanic whites, 406 Hispanics, 403 African-Americans, and 300 Asians.
A willingness to participate
Most of the African-Americans (62%) and Hispanics (57%) polled said it’s very important to participate in a clinical trial to improve the health of others. Fifty percent of Asians agreed, as did 49% of non-Hispanic whites.
Furthermore, 75% of Hispanics, 72% of African-Americans, 71% of non-Hispanic whites, and 65% of Asians said they would likely participate in a clinical trial if it was recommended by a doctor.
And about three-quarters of all respondents said, assuming the correct privacy protections were in place, they would be willing to share personal health information to advance medical research.
“The poll reveals a willingness among minorities to participate in clinical trials to improve the quality of healthcare, but enrollment remains stubbornly low,” said Mary Woolley, president and CEO of Research!America.
Among those individuals surveyed, only 17% of Hispanics, 15% of African-Americans, 15% of non-Hispanic whites, and 11% of Asians said they or a family member had participated in a clinical trial.
When asked to give a reason for low enrollment in clinical trials, most respondents cited a lack of trust—61% of African-Americans, 54% of non-Hispanic whites, 52% of Hispanics, and 51% of Asians.
The importance of physicians, institutions
Many of the individuals surveyed said they had learned of clinical trials via the Internet—56% of Asians, 52% of non-Hispanic whites, 50% of African-Americans, and 45% of Hispanics.
Far fewer said they had heard of trials during discussions with their doctors—22% of Hispanics, 22% of non-Hispanic whites, 20% of Asians, and 17% of African-Americans.
And most respondents said a doctor’s recommendation is a “very” or “somewhat” important factor in the decision to participate in a clinical trial—81% of Hispanics, 81% of non-Hispanic whites, 80% of Asians, and 78% of African-Americans.
Institutional reputation was also deemed an important factor by 74% of African-Americans, 72% of non-Hispanic whites, 66% of Hispanics, and 66% of Asians.
And many respondents said healthcare providers should play a major role in raising awareness of clinical trials. In fact, 42% of non-Hispanic whites, 38% of Hispanics, 36% of Asians, and 33% of African-Americans said providers have the greatest responsibility in educating the public about clinical trials.
“It’s imperative that healthcare providers and others help patients gain a deeper knowledge of clinical trials so all Americans can benefit from life-saving treatments,” Woolley said.
This survey was conducted by Zogby Analytics for Research!America, the Association of Clinical Research Organizations, the Clinical Research Forum, the Friends of the National Library of Medicine, and the Clinical Trials Transformation Initiative. ![]()
Findings could revolutionize malaria drug testing

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have reported that injecting cryopreserved malaria sporozoites in human subjects can result in controlled infection.
This indicates that direct injection of sporozoites can be used in lieu of mosquito bites to test the efficacy of malaria drugs or vaccines in human volunteers.
The findings from this study were reported at the annual meeting of the American Society of Tropical Medicine and Hygiene and published online in the American Journal of Tropical Medicine and Hygiene.
“Our study shows it’s possible to manufacture and then administer controlled doses of malaria parasites using a needle and syringe to deliver a formulation that can meet regulatory standards for purity and dose consistency,” said study author Meta Roestenberg, MD, of the Radboud University Nijmegen Medical Center in The Netherlands.
She and her colleagues noted that the current method of testing malaria drugs and vaccines by exposing subjects to mosquito bites is technically complex and costly. And there are only a few places in the world today where such work is being done.
In addition, when using mosquitoes to deliver malaria parasites, it can be difficult to ensure that all subjects receive the same level of infection. And that can influence the outcome of the treatment.
So Dr Roestenberg and her colleagues set out to find a better way. They tested cryopreserved Plasmodium falciparum sporozoites that were harvested from the salivary glands of infected mosquitoes and had been frozen for more than 2 years. The sporozoites were manufactured by the Maryland-based biotechnology company Sanaria Inc.
The researchers enrolled 18 healthy volunteers and divided them into 3 groups. The first group received 2500 sporozoites, the second received 10,000 sporozoites, and the third received 25,000 sporozoites.
The team found that 84% of participants—5 of the 6 volunteers in each group—were safely and successfully infected with malaria. And there were no differences among the groups in the time it took for the infection to develop or the presentation of symptoms. The volunteers who developed infections subsequently received treatment and quickly recovered without incident.
“We have demonstrated the potential to develop what you might call ‘the human challenge trial in a bottle’ that could be available to scientists anywhere who need to know how a new drug or vaccine would fare against a real but carefully controlled and calibrated malaria infection,” said study author Stephen L. Hoffman, MD, of Sanaria Inc.
The researchers also said these results could aid the development of whole parasite vaccines.
“A major challenge to realizing the potential of whole parasite vaccines is the development of a stable, consistent formulation of sporozoites that can be manufactured, preserved, and used like any other vaccine,” said study author Robert W. Sauerwein, MD, PhD, also of Radboud University Nijmegen Medical Center.
Sanaria is currently pursuing clinical trials to test 2 different approaches to whole parasite vaccination: irradiated sporozoites and inducing controlled infections in tandem with the administration of antimalarial drugs.
Researchers are also planning additional trials to ensure the infection produced with the cryopreserved sporozoites mirrors what one would experience through bites from infected mosquitoes. ![]()

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have reported that injecting cryopreserved malaria sporozoites in human subjects can result in controlled infection.
This indicates that direct injection of sporozoites can be used in lieu of mosquito bites to test the efficacy of malaria drugs or vaccines in human volunteers.
The findings from this study were reported at the annual meeting of the American Society of Tropical Medicine and Hygiene and published online in the American Journal of Tropical Medicine and Hygiene.
“Our study shows it’s possible to manufacture and then administer controlled doses of malaria parasites using a needle and syringe to deliver a formulation that can meet regulatory standards for purity and dose consistency,” said study author Meta Roestenberg, MD, of the Radboud University Nijmegen Medical Center in The Netherlands.
She and her colleagues noted that the current method of testing malaria drugs and vaccines by exposing subjects to mosquito bites is technically complex and costly. And there are only a few places in the world today where such work is being done.
In addition, when using mosquitoes to deliver malaria parasites, it can be difficult to ensure that all subjects receive the same level of infection. And that can influence the outcome of the treatment.
So Dr Roestenberg and her colleagues set out to find a better way. They tested cryopreserved Plasmodium falciparum sporozoites that were harvested from the salivary glands of infected mosquitoes and had been frozen for more than 2 years. The sporozoites were manufactured by the Maryland-based biotechnology company Sanaria Inc.
The researchers enrolled 18 healthy volunteers and divided them into 3 groups. The first group received 2500 sporozoites, the second received 10,000 sporozoites, and the third received 25,000 sporozoites.
The team found that 84% of participants—5 of the 6 volunteers in each group—were safely and successfully infected with malaria. And there were no differences among the groups in the time it took for the infection to develop or the presentation of symptoms. The volunteers who developed infections subsequently received treatment and quickly recovered without incident.
“We have demonstrated the potential to develop what you might call ‘the human challenge trial in a bottle’ that could be available to scientists anywhere who need to know how a new drug or vaccine would fare against a real but carefully controlled and calibrated malaria infection,” said study author Stephen L. Hoffman, MD, of Sanaria Inc.
The researchers also said these results could aid the development of whole parasite vaccines.
“A major challenge to realizing the potential of whole parasite vaccines is the development of a stable, consistent formulation of sporozoites that can be manufactured, preserved, and used like any other vaccine,” said study author Robert W. Sauerwein, MD, PhD, also of Radboud University Nijmegen Medical Center.
Sanaria is currently pursuing clinical trials to test 2 different approaches to whole parasite vaccination: irradiated sporozoites and inducing controlled infections in tandem with the administration of antimalarial drugs.
Researchers are also planning additional trials to ensure the infection produced with the cryopreserved sporozoites mirrors what one would experience through bites from infected mosquitoes. ![]()

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have reported that injecting cryopreserved malaria sporozoites in human subjects can result in controlled infection.
This indicates that direct injection of sporozoites can be used in lieu of mosquito bites to test the efficacy of malaria drugs or vaccines in human volunteers.
The findings from this study were reported at the annual meeting of the American Society of Tropical Medicine and Hygiene and published online in the American Journal of Tropical Medicine and Hygiene.
“Our study shows it’s possible to manufacture and then administer controlled doses of malaria parasites using a needle and syringe to deliver a formulation that can meet regulatory standards for purity and dose consistency,” said study author Meta Roestenberg, MD, of the Radboud University Nijmegen Medical Center in The Netherlands.
She and her colleagues noted that the current method of testing malaria drugs and vaccines by exposing subjects to mosquito bites is technically complex and costly. And there are only a few places in the world today where such work is being done.
In addition, when using mosquitoes to deliver malaria parasites, it can be difficult to ensure that all subjects receive the same level of infection. And that can influence the outcome of the treatment.
So Dr Roestenberg and her colleagues set out to find a better way. They tested cryopreserved Plasmodium falciparum sporozoites that were harvested from the salivary glands of infected mosquitoes and had been frozen for more than 2 years. The sporozoites were manufactured by the Maryland-based biotechnology company Sanaria Inc.
The researchers enrolled 18 healthy volunteers and divided them into 3 groups. The first group received 2500 sporozoites, the second received 10,000 sporozoites, and the third received 25,000 sporozoites.
The team found that 84% of participants—5 of the 6 volunteers in each group—were safely and successfully infected with malaria. And there were no differences among the groups in the time it took for the infection to develop or the presentation of symptoms. The volunteers who developed infections subsequently received treatment and quickly recovered without incident.
“We have demonstrated the potential to develop what you might call ‘the human challenge trial in a bottle’ that could be available to scientists anywhere who need to know how a new drug or vaccine would fare against a real but carefully controlled and calibrated malaria infection,” said study author Stephen L. Hoffman, MD, of Sanaria Inc.
The researchers also said these results could aid the development of whole parasite vaccines.
“A major challenge to realizing the potential of whole parasite vaccines is the development of a stable, consistent formulation of sporozoites that can be manufactured, preserved, and used like any other vaccine,” said study author Robert W. Sauerwein, MD, PhD, also of Radboud University Nijmegen Medical Center.
Sanaria is currently pursuing clinical trials to test 2 different approaches to whole parasite vaccination: irradiated sporozoites and inducing controlled infections in tandem with the administration of antimalarial drugs.
Researchers are also planning additional trials to ensure the infection produced with the cryopreserved sporozoites mirrors what one would experience through bites from infected mosquitoes. ![]()
FDA approves drugs faster than EMA, Health Canada
The FDA generally approves drugs faster than its Canadian and European counterparts, according to a study published in this week’s edition of NEJM.
The researchers say these results refute criticisms that the drug approval process in the US is slow and that agencies in other countries tend to approve new therapies first.
“The perception that the FDA is too slow implies that sick patients are waiting unnecessarily for regulators to complete their review of new drug applications,” said lead study author Nicholas Downing, a medical student at Yale University.
He and his colleagues decided to conduct this study because there have been no recent comparisons of the FDA’s review speed with that of agencies in other countries.
So the researchers reviewed drug approval decisions made by the FDA, Health Canada, and the European Medicines Agency (EMA) between 2001 and 2010. The team said they chose Health Canada and the EMA as comparisons because these agencies face similar pressures to approve new drugs quickly while ensuring they don’t put patients at risk.
The investigators studied each regulator’s database of drug approvals to identify novel therapeutics, as well as the timing of key regulatory events. They then calculated each agency’s review speed.
The median total time to review a new drug application was 322 days at the FDA, 366 days at the EMA, and 393 days at Health Canada.
“Among the subsample of drugs approved for all 3 regulators, the FDA’s reviews were over 3 months faster than those of the EMA or Health Canada,” Downing said. “The total review time at the FDA was faster than EMA, despite the FDA’s far higher proportion of applications requiring multiple regulatory reviews.”
The researchers also found that, during the review period, the FDA approved 225 new drugs, the EMA approved 186, and Health Canada approved 99. Additionally, of the therapies that have been approved by all 3 agencies, most drugs were first approved in the US.
“[W]e found that 64% of medicines approved in both the US and in Europe were approved for US patients first,” Downing said. “And 86% of medicines approved in both the US and Canada were also approved first in the US.”
Downing and his colleagues noted that this study has 2 key limitations. First, the researchers didn’t account for drugs that were ultimately rejected, as the regulatory agencies don’t release review times for drugs that are never approved. However, the team also pointed out that the FDA approves more than 80% of its applications, so the exclusion may not have made much of an impact.
Secondly, the study included only new molecular entities and original biologic agents. In order to get a more accurate reading on the regulatory review process, research would need to evaluate the review of generic drugs, reformulated drugs, combination therapies, and medical devices. ![]()
The FDA generally approves drugs faster than its Canadian and European counterparts, according to a study published in this week’s edition of NEJM.
The researchers say these results refute criticisms that the drug approval process in the US is slow and that agencies in other countries tend to approve new therapies first.
“The perception that the FDA is too slow implies that sick patients are waiting unnecessarily for regulators to complete their review of new drug applications,” said lead study author Nicholas Downing, a medical student at Yale University.
He and his colleagues decided to conduct this study because there have been no recent comparisons of the FDA’s review speed with that of agencies in other countries.
So the researchers reviewed drug approval decisions made by the FDA, Health Canada, and the European Medicines Agency (EMA) between 2001 and 2010. The team said they chose Health Canada and the EMA as comparisons because these agencies face similar pressures to approve new drugs quickly while ensuring they don’t put patients at risk.
The investigators studied each regulator’s database of drug approvals to identify novel therapeutics, as well as the timing of key regulatory events. They then calculated each agency’s review speed.
The median total time to review a new drug application was 322 days at the FDA, 366 days at the EMA, and 393 days at Health Canada.
“Among the subsample of drugs approved for all 3 regulators, the FDA’s reviews were over 3 months faster than those of the EMA or Health Canada,” Downing said. “The total review time at the FDA was faster than EMA, despite the FDA’s far higher proportion of applications requiring multiple regulatory reviews.”
The researchers also found that, during the review period, the FDA approved 225 new drugs, the EMA approved 186, and Health Canada approved 99. Additionally, of the therapies that have been approved by all 3 agencies, most drugs were first approved in the US.
“[W]e found that 64% of medicines approved in both the US and in Europe were approved for US patients first,” Downing said. “And 86% of medicines approved in both the US and Canada were also approved first in the US.”
Downing and his colleagues noted that this study has 2 key limitations. First, the researchers didn’t account for drugs that were ultimately rejected, as the regulatory agencies don’t release review times for drugs that are never approved. However, the team also pointed out that the FDA approves more than 80% of its applications, so the exclusion may not have made much of an impact.
Secondly, the study included only new molecular entities and original biologic agents. In order to get a more accurate reading on the regulatory review process, research would need to evaluate the review of generic drugs, reformulated drugs, combination therapies, and medical devices. ![]()
The FDA generally approves drugs faster than its Canadian and European counterparts, according to a study published in this week’s edition of NEJM.
The researchers say these results refute criticisms that the drug approval process in the US is slow and that agencies in other countries tend to approve new therapies first.
“The perception that the FDA is too slow implies that sick patients are waiting unnecessarily for regulators to complete their review of new drug applications,” said lead study author Nicholas Downing, a medical student at Yale University.
He and his colleagues decided to conduct this study because there have been no recent comparisons of the FDA’s review speed with that of agencies in other countries.
So the researchers reviewed drug approval decisions made by the FDA, Health Canada, and the European Medicines Agency (EMA) between 2001 and 2010. The team said they chose Health Canada and the EMA as comparisons because these agencies face similar pressures to approve new drugs quickly while ensuring they don’t put patients at risk.
The investigators studied each regulator’s database of drug approvals to identify novel therapeutics, as well as the timing of key regulatory events. They then calculated each agency’s review speed.
The median total time to review a new drug application was 322 days at the FDA, 366 days at the EMA, and 393 days at Health Canada.
“Among the subsample of drugs approved for all 3 regulators, the FDA’s reviews were over 3 months faster than those of the EMA or Health Canada,” Downing said. “The total review time at the FDA was faster than EMA, despite the FDA’s far higher proportion of applications requiring multiple regulatory reviews.”
The researchers also found that, during the review period, the FDA approved 225 new drugs, the EMA approved 186, and Health Canada approved 99. Additionally, of the therapies that have been approved by all 3 agencies, most drugs were first approved in the US.
“[W]e found that 64% of medicines approved in both the US and in Europe were approved for US patients first,” Downing said. “And 86% of medicines approved in both the US and Canada were also approved first in the US.”
Downing and his colleagues noted that this study has 2 key limitations. First, the researchers didn’t account for drugs that were ultimately rejected, as the regulatory agencies don’t release review times for drugs that are never approved. However, the team also pointed out that the FDA approves more than 80% of its applications, so the exclusion may not have made much of an impact.
Secondly, the study included only new molecular entities and original biologic agents. In order to get a more accurate reading on the regulatory review process, research would need to evaluate the review of generic drugs, reformulated drugs, combination therapies, and medical devices. ![]()
Vaccine candidate reduces malaria risk

Credit: St Jude Children’s
Research Hospital
First results from a phase 3 trial of the vaccine candidate RTS,S/AS01 indicate it provides young African children with protection against clinical and severe malaria.
The researchers also said RTS,S/AS01 has an acceptable safety and tolerability profile.
These results were announced October 19 at the Malaria Forum, hosted by the Bill & Melinda Gates Foundation in Seattle, Washington.
The findings were also published online in The New England Journal of Medicine.
“The publication of the first results in children aged 5 to 17 months marks an important milestone in the development of RTS,S/AS01,” said Irving Hoffman, PA, MPH, co-principal investigator at a study site in Lilongwe, Malawi.
The trial is still ongoing, being conducted at 11 sites in 7 countries across sub-Saharan Africa. But the researchers have performed an initial analysis of results in the first 6000 children enrolled, who were aged 5 months to 17 months at the time of enrollment.
The children received 3 doses of RTS,S/AS01 and were followed for a 12-month period. RTS,S/AS01 reduced the risk of clinical malaria in these children by 56% and the risk of severe malaria by 47%.
“These results confirm findings from previous phase 2 studies and support ongoing efforts to advance the development of this malaria vaccine candidate,” Hoffman said.
Efficacy and safety results in 6- to 12-week-old infants are expected by the end of 2012, according to the investigators. However, they have performed an analysis of severe malaria episodes reported thus far in all 15,460 children enrolled in the trial, ranging from 6 weeks to 17 months of age.
The analysis showed that RTS,S/AS01 had 35% efficacy over a follow-up period ranging between 0 months and 22 months (average, 11.5 months). Further information about the longer-term effects of RTS,S/AS01—30 months after the third dose—should be available by the end of 2014, the researchers said.
The overall incidence of serious adverse events in this trial was comparable between RTS,S/AS01 recipients (18%) and those receiving a control vaccine (22%)
There were differences in the rates of certain serious adverse events between the vaccine groups. Seizures and meningitis were both more frequent in the RTS,S/AS01 group. Seizures were linked to fever, and meningitis was considered unlikely to be vaccine-related.
RTS,S/AS01 is being developed by GlaxoSmithKline and the PATH Malaria Vaccine Initiative, together with African research centers. The partners are all represented on the Clinical Trials Partnership Committee, which is responsible for the conduct of the trial.
Major funding for clinical development comes from a grant by the Bill & Melinda Gates Foundation. ![]()

Credit: St Jude Children’s
Research Hospital
First results from a phase 3 trial of the vaccine candidate RTS,S/AS01 indicate it provides young African children with protection against clinical and severe malaria.
The researchers also said RTS,S/AS01 has an acceptable safety and tolerability profile.
These results were announced October 19 at the Malaria Forum, hosted by the Bill & Melinda Gates Foundation in Seattle, Washington.
The findings were also published online in The New England Journal of Medicine.
“The publication of the first results in children aged 5 to 17 months marks an important milestone in the development of RTS,S/AS01,” said Irving Hoffman, PA, MPH, co-principal investigator at a study site in Lilongwe, Malawi.
The trial is still ongoing, being conducted at 11 sites in 7 countries across sub-Saharan Africa. But the researchers have performed an initial analysis of results in the first 6000 children enrolled, who were aged 5 months to 17 months at the time of enrollment.
The children received 3 doses of RTS,S/AS01 and were followed for a 12-month period. RTS,S/AS01 reduced the risk of clinical malaria in these children by 56% and the risk of severe malaria by 47%.
“These results confirm findings from previous phase 2 studies and support ongoing efforts to advance the development of this malaria vaccine candidate,” Hoffman said.
Efficacy and safety results in 6- to 12-week-old infants are expected by the end of 2012, according to the investigators. However, they have performed an analysis of severe malaria episodes reported thus far in all 15,460 children enrolled in the trial, ranging from 6 weeks to 17 months of age.
The analysis showed that RTS,S/AS01 had 35% efficacy over a follow-up period ranging between 0 months and 22 months (average, 11.5 months). Further information about the longer-term effects of RTS,S/AS01—30 months after the third dose—should be available by the end of 2014, the researchers said.
The overall incidence of serious adverse events in this trial was comparable between RTS,S/AS01 recipients (18%) and those receiving a control vaccine (22%)
There were differences in the rates of certain serious adverse events between the vaccine groups. Seizures and meningitis were both more frequent in the RTS,S/AS01 group. Seizures were linked to fever, and meningitis was considered unlikely to be vaccine-related.
RTS,S/AS01 is being developed by GlaxoSmithKline and the PATH Malaria Vaccine Initiative, together with African research centers. The partners are all represented on the Clinical Trials Partnership Committee, which is responsible for the conduct of the trial.
Major funding for clinical development comes from a grant by the Bill & Melinda Gates Foundation. ![]()

Credit: St Jude Children’s
Research Hospital
First results from a phase 3 trial of the vaccine candidate RTS,S/AS01 indicate it provides young African children with protection against clinical and severe malaria.
The researchers also said RTS,S/AS01 has an acceptable safety and tolerability profile.
These results were announced October 19 at the Malaria Forum, hosted by the Bill & Melinda Gates Foundation in Seattle, Washington.
The findings were also published online in The New England Journal of Medicine.
“The publication of the first results in children aged 5 to 17 months marks an important milestone in the development of RTS,S/AS01,” said Irving Hoffman, PA, MPH, co-principal investigator at a study site in Lilongwe, Malawi.
The trial is still ongoing, being conducted at 11 sites in 7 countries across sub-Saharan Africa. But the researchers have performed an initial analysis of results in the first 6000 children enrolled, who were aged 5 months to 17 months at the time of enrollment.
The children received 3 doses of RTS,S/AS01 and were followed for a 12-month period. RTS,S/AS01 reduced the risk of clinical malaria in these children by 56% and the risk of severe malaria by 47%.
“These results confirm findings from previous phase 2 studies and support ongoing efforts to advance the development of this malaria vaccine candidate,” Hoffman said.
Efficacy and safety results in 6- to 12-week-old infants are expected by the end of 2012, according to the investigators. However, they have performed an analysis of severe malaria episodes reported thus far in all 15,460 children enrolled in the trial, ranging from 6 weeks to 17 months of age.
The analysis showed that RTS,S/AS01 had 35% efficacy over a follow-up period ranging between 0 months and 22 months (average, 11.5 months). Further information about the longer-term effects of RTS,S/AS01—30 months after the third dose—should be available by the end of 2014, the researchers said.
The overall incidence of serious adverse events in this trial was comparable between RTS,S/AS01 recipients (18%) and those receiving a control vaccine (22%)
There were differences in the rates of certain serious adverse events between the vaccine groups. Seizures and meningitis were both more frequent in the RTS,S/AS01 group. Seizures were linked to fever, and meningitis was considered unlikely to be vaccine-related.
RTS,S/AS01 is being developed by GlaxoSmithKline and the PATH Malaria Vaccine Initiative, together with African research centers. The partners are all represented on the Clinical Trials Partnership Committee, which is responsible for the conduct of the trial.
Major funding for clinical development comes from a grant by the Bill & Melinda Gates Foundation. ![]()
Researchers reveal structure of CXCR4
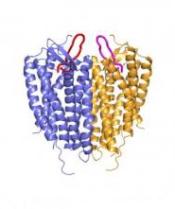
Credit: Raymond Stevens
Scripps Research Institute
A team of researchers has uncovered the structure of a cell surface receptor, CXCR4, which guides blood and immune cell movement throughout the body.
CXCR4 is also found on the surface of the human immunodeficiency virus (HIV), and helps the virus to enter blood cells.
The receptor is part of a group of about 700 proteins known as G protein-coupled receptors (GPCRs).
The team, led by Raymond C. Stevens, PhD, of Scripps Research Institute in La Jolla, California, and senior author of the study, already found the structures of two other GPCRs: the adrenergic receptor and A2A adenosine receptor.
CXCR4 belongs to a different group of GPCRs, one that binds to protein molecules called chemokines, responsible for steering blood and immune cells where they are needed.
The team used GPCR biochemistry, receptor stabilization, and X-ray crystallography to capture the first visual of a chemokine receptor bound to a ligand.
Unlike the adrenergic receptor and the A2A adenosine receptor, CXCR4 likes to form dimers.
“The dimerization observation was very intriguing,” said Dr Stevens. “We solved 5 different crystal structures in multiple crystal forms, and each one had the same dimer interface. It has long been debated how GPCRs might dimerize, if they did at all. This is the first solid observation about a consistent structural GPCR dimer.”
The team believes preventing dimerization might provide a new way to block CXCR4, which results in the release of hematopoietic stem cells from bone marrow into the bloodstream.
Currently, plerixafor injection is the only drug on the market that blocks CXCR4. Therapy that assists in the release of hematopoietic stem cells to the bloodstream is very useful following stem cell transplant.
Drugs that block CXCR4 are also useful in treating HIV infection.
Their findings were published in the October 7 issue of Science. ![]()

Credit: Raymond Stevens
Scripps Research Institute
A team of researchers has uncovered the structure of a cell surface receptor, CXCR4, which guides blood and immune cell movement throughout the body.
CXCR4 is also found on the surface of the human immunodeficiency virus (HIV), and helps the virus to enter blood cells.
The receptor is part of a group of about 700 proteins known as G protein-coupled receptors (GPCRs).
The team, led by Raymond C. Stevens, PhD, of Scripps Research Institute in La Jolla, California, and senior author of the study, already found the structures of two other GPCRs: the adrenergic receptor and A2A adenosine receptor.
CXCR4 belongs to a different group of GPCRs, one that binds to protein molecules called chemokines, responsible for steering blood and immune cells where they are needed.
The team used GPCR biochemistry, receptor stabilization, and X-ray crystallography to capture the first visual of a chemokine receptor bound to a ligand.
Unlike the adrenergic receptor and the A2A adenosine receptor, CXCR4 likes to form dimers.
“The dimerization observation was very intriguing,” said Dr Stevens. “We solved 5 different crystal structures in multiple crystal forms, and each one had the same dimer interface. It has long been debated how GPCRs might dimerize, if they did at all. This is the first solid observation about a consistent structural GPCR dimer.”
The team believes preventing dimerization might provide a new way to block CXCR4, which results in the release of hematopoietic stem cells from bone marrow into the bloodstream.
Currently, plerixafor injection is the only drug on the market that blocks CXCR4. Therapy that assists in the release of hematopoietic stem cells to the bloodstream is very useful following stem cell transplant.
Drugs that block CXCR4 are also useful in treating HIV infection.
Their findings were published in the October 7 issue of Science. ![]()

Credit: Raymond Stevens
Scripps Research Institute
A team of researchers has uncovered the structure of a cell surface receptor, CXCR4, which guides blood and immune cell movement throughout the body.
CXCR4 is also found on the surface of the human immunodeficiency virus (HIV), and helps the virus to enter blood cells.
The receptor is part of a group of about 700 proteins known as G protein-coupled receptors (GPCRs).
The team, led by Raymond C. Stevens, PhD, of Scripps Research Institute in La Jolla, California, and senior author of the study, already found the structures of two other GPCRs: the adrenergic receptor and A2A adenosine receptor.
CXCR4 belongs to a different group of GPCRs, one that binds to protein molecules called chemokines, responsible for steering blood and immune cells where they are needed.
The team used GPCR biochemistry, receptor stabilization, and X-ray crystallography to capture the first visual of a chemokine receptor bound to a ligand.
Unlike the adrenergic receptor and the A2A adenosine receptor, CXCR4 likes to form dimers.
“The dimerization observation was very intriguing,” said Dr Stevens. “We solved 5 different crystal structures in multiple crystal forms, and each one had the same dimer interface. It has long been debated how GPCRs might dimerize, if they did at all. This is the first solid observation about a consistent structural GPCR dimer.”
The team believes preventing dimerization might provide a new way to block CXCR4, which results in the release of hematopoietic stem cells from bone marrow into the bloodstream.
Currently, plerixafor injection is the only drug on the market that blocks CXCR4. Therapy that assists in the release of hematopoietic stem cells to the bloodstream is very useful following stem cell transplant.
Drugs that block CXCR4 are also useful in treating HIV infection.
Their findings were published in the October 7 issue of Science. ![]()

