User login
No need to switch antibiotics, study shows

Staphylococcus
infectionCredit: Bill Branson
New research suggests the antibiotic vancomycin is still effective in treating Staphylococcus aureus bloodstream (SAB) infections, despite increases in minimum inhibitory concentration (MIC) values.
Researchers found no difference in mortality between patients with low-vancomycin MIC and those with high-vancomycin MIC.
So it seems physicians can continue using vancomycin when MIC values are elevated but within the susceptible range, rather than
switching to newer antibiotics.
Andre Kalil, MD, of the University of Nebraska Medical Center in Omaha, and his colleagues described this research in JAMA.
In recent years, physicians treating Staphylococcus infections with vancomycin have seen an increase in the MIC, the lowest concentration of an antimicrobial agent that inhibits the growth of a microorganism.
MIC values lower than 4 mg/L suggest Staphylococcus is susceptible to vancomycin. However, when the MIC value exceeds 1.5 mg/L, some physicians have taken it as an indication that vancomycin may not be working at maximum effectiveness.
Some reports have suggested that elevations in vancomycin MIC values may be associated with increased treatment failure and death.
To determine the effectiveness of vancomycin, Dr Kalil and his colleagues analyzed data from 38 studies covering 8291 episodes of SAB infection.
The team evaluated the association between vancomycin MIC elevation and mortality. Among all SAB infections studied, the overall mortality was 26.1%.
The adjusted absolute risk of mortality did not differ significantly between patients with high-vancomycin MIC and those with low-vancomycin MIC—26.8% and 25.8%, respectively.
In studies that included only methicillin-resistant Staphylococcus aureus infections, the mortality among SAB episodes in patients with high-vancomycin MIC was 27.6%, compared with a mortality of 27.4% among patients with low-vancomycin MIC.
“The study provides strong evidence that vancomycin remains highly useful,” Dr Kalil said. “Even though vancomycin is an older drug, it is still killing staph very efficiently. There are newer antibiotics available to treat Staphylococcus aureus infections, but this study demonstrates that physicians don’t necessarily need to switch to these new drugs when the MIC is increased but still within the susceptible range.”
“The prevention of a rapid switch to newer drugs has another great benefit to our patients—less unnecessary exposure to these drugs, which will translate into less development of antibiotic resistance.”
Dr Kalil said the study may have implications for clinical practice and public health.
The results suggest standards for vancomycin MIC likely do not need to be lowered, routine differentiation of MIC values between 1 mg/L and 2 mg/L appears unnecessary, and the use of alternative drugs may not be required for Staphylococcus aureus isolates with elevated but susceptible vancomycin MIC values. ![]()

Staphylococcus
infectionCredit: Bill Branson
New research suggests the antibiotic vancomycin is still effective in treating Staphylococcus aureus bloodstream (SAB) infections, despite increases in minimum inhibitory concentration (MIC) values.
Researchers found no difference in mortality between patients with low-vancomycin MIC and those with high-vancomycin MIC.
So it seems physicians can continue using vancomycin when MIC values are elevated but within the susceptible range, rather than
switching to newer antibiotics.
Andre Kalil, MD, of the University of Nebraska Medical Center in Omaha, and his colleagues described this research in JAMA.
In recent years, physicians treating Staphylococcus infections with vancomycin have seen an increase in the MIC, the lowest concentration of an antimicrobial agent that inhibits the growth of a microorganism.
MIC values lower than 4 mg/L suggest Staphylococcus is susceptible to vancomycin. However, when the MIC value exceeds 1.5 mg/L, some physicians have taken it as an indication that vancomycin may not be working at maximum effectiveness.
Some reports have suggested that elevations in vancomycin MIC values may be associated with increased treatment failure and death.
To determine the effectiveness of vancomycin, Dr Kalil and his colleagues analyzed data from 38 studies covering 8291 episodes of SAB infection.
The team evaluated the association between vancomycin MIC elevation and mortality. Among all SAB infections studied, the overall mortality was 26.1%.
The adjusted absolute risk of mortality did not differ significantly between patients with high-vancomycin MIC and those with low-vancomycin MIC—26.8% and 25.8%, respectively.
In studies that included only methicillin-resistant Staphylococcus aureus infections, the mortality among SAB episodes in patients with high-vancomycin MIC was 27.6%, compared with a mortality of 27.4% among patients with low-vancomycin MIC.
“The study provides strong evidence that vancomycin remains highly useful,” Dr Kalil said. “Even though vancomycin is an older drug, it is still killing staph very efficiently. There are newer antibiotics available to treat Staphylococcus aureus infections, but this study demonstrates that physicians don’t necessarily need to switch to these new drugs when the MIC is increased but still within the susceptible range.”
“The prevention of a rapid switch to newer drugs has another great benefit to our patients—less unnecessary exposure to these drugs, which will translate into less development of antibiotic resistance.”
Dr Kalil said the study may have implications for clinical practice and public health.
The results suggest standards for vancomycin MIC likely do not need to be lowered, routine differentiation of MIC values between 1 mg/L and 2 mg/L appears unnecessary, and the use of alternative drugs may not be required for Staphylococcus aureus isolates with elevated but susceptible vancomycin MIC values. ![]()

Staphylococcus
infectionCredit: Bill Branson
New research suggests the antibiotic vancomycin is still effective in treating Staphylococcus aureus bloodstream (SAB) infections, despite increases in minimum inhibitory concentration (MIC) values.
Researchers found no difference in mortality between patients with low-vancomycin MIC and those with high-vancomycin MIC.
So it seems physicians can continue using vancomycin when MIC values are elevated but within the susceptible range, rather than
switching to newer antibiotics.
Andre Kalil, MD, of the University of Nebraska Medical Center in Omaha, and his colleagues described this research in JAMA.
In recent years, physicians treating Staphylococcus infections with vancomycin have seen an increase in the MIC, the lowest concentration of an antimicrobial agent that inhibits the growth of a microorganism.
MIC values lower than 4 mg/L suggest Staphylococcus is susceptible to vancomycin. However, when the MIC value exceeds 1.5 mg/L, some physicians have taken it as an indication that vancomycin may not be working at maximum effectiveness.
Some reports have suggested that elevations in vancomycin MIC values may be associated with increased treatment failure and death.
To determine the effectiveness of vancomycin, Dr Kalil and his colleagues analyzed data from 38 studies covering 8291 episodes of SAB infection.
The team evaluated the association between vancomycin MIC elevation and mortality. Among all SAB infections studied, the overall mortality was 26.1%.
The adjusted absolute risk of mortality did not differ significantly between patients with high-vancomycin MIC and those with low-vancomycin MIC—26.8% and 25.8%, respectively.
In studies that included only methicillin-resistant Staphylococcus aureus infections, the mortality among SAB episodes in patients with high-vancomycin MIC was 27.6%, compared with a mortality of 27.4% among patients with low-vancomycin MIC.
“The study provides strong evidence that vancomycin remains highly useful,” Dr Kalil said. “Even though vancomycin is an older drug, it is still killing staph very efficiently. There are newer antibiotics available to treat Staphylococcus aureus infections, but this study demonstrates that physicians don’t necessarily need to switch to these new drugs when the MIC is increased but still within the susceptible range.”
“The prevention of a rapid switch to newer drugs has another great benefit to our patients—less unnecessary exposure to these drugs, which will translate into less development of antibiotic resistance.”
Dr Kalil said the study may have implications for clinical practice and public health.
The results suggest standards for vancomycin MIC likely do not need to be lowered, routine differentiation of MIC values between 1 mg/L and 2 mg/L appears unnecessary, and the use of alternative drugs may not be required for Staphylococcus aureus isolates with elevated but susceptible vancomycin MIC values. ![]()
New insight into T-cell development

Credit: NIAID
A fundamental theory about how the thymus “educates” T cells appears to be wrong, according to research published in Nature Communications.
It’s known that stem cells leave the bone marrow and travel to the thymus to become one of two CD4 T-cell types: effector cells or regulatory T cells (Tregs).
One widely held concept of why the cells become one type or the other is that they are exposed to different ligands in the thymus, said Leszek Ignatowicz, PhD, of Georgia Regents University in Augusta.
But when he and his colleagues limited the cells’ exposure to only one ligand, the same mix of T cells still emerged.
“We asked a simple question, ‘Is it going to affect their development,’ and the answer was ‘no,’” Dr Ignatowicz said. “The cells still mature in the thymus, so something else must be determining it.”
The finding provides more insight into immunity that could one day enable a new approach to vaccines that steer the thymus to produce more of whatever T-cell type a patient needs: more effector cells if they have an infection or cancer, more Tregs if they have autoimmune diseases such as arthritis and multiple sclerosis.
“We could help steer the education process in the desired direction,” Dr Ignatowicz said.
To uncover their findings, he and his colleagues studied two types of mice, each expressing a single ligand in the thymus. The researchers thought one would prompt strong ligand binding and favor Treg development, and the other would favor a weaker ligand bond and effector cell development.
The mix of resulting T cells was the same as if both were exposed to the usual thousands of ligands, although the experiment did reveal one difference.
Ligands—and, eventually, bacteria and other invaders—get the attention of T cells by activating their receptors. Both CD4 T-cell types generally have the same receptors, but they are organized differently.
The researchers found that as long as the binding was weak, as it was in the first mouse, there was a lot of overlap in the receptors the ligand bound to in both T-cell types. However, in the second mouse, where the ligand prompted strong binding, there was far less overlap.
“We are now trying to find what causes that difference,” Dr Ignatowicz said. ![]()

Credit: NIAID
A fundamental theory about how the thymus “educates” T cells appears to be wrong, according to research published in Nature Communications.
It’s known that stem cells leave the bone marrow and travel to the thymus to become one of two CD4 T-cell types: effector cells or regulatory T cells (Tregs).
One widely held concept of why the cells become one type or the other is that they are exposed to different ligands in the thymus, said Leszek Ignatowicz, PhD, of Georgia Regents University in Augusta.
But when he and his colleagues limited the cells’ exposure to only one ligand, the same mix of T cells still emerged.
“We asked a simple question, ‘Is it going to affect their development,’ and the answer was ‘no,’” Dr Ignatowicz said. “The cells still mature in the thymus, so something else must be determining it.”
The finding provides more insight into immunity that could one day enable a new approach to vaccines that steer the thymus to produce more of whatever T-cell type a patient needs: more effector cells if they have an infection or cancer, more Tregs if they have autoimmune diseases such as arthritis and multiple sclerosis.
“We could help steer the education process in the desired direction,” Dr Ignatowicz said.
To uncover their findings, he and his colleagues studied two types of mice, each expressing a single ligand in the thymus. The researchers thought one would prompt strong ligand binding and favor Treg development, and the other would favor a weaker ligand bond and effector cell development.
The mix of resulting T cells was the same as if both were exposed to the usual thousands of ligands, although the experiment did reveal one difference.
Ligands—and, eventually, bacteria and other invaders—get the attention of T cells by activating their receptors. Both CD4 T-cell types generally have the same receptors, but they are organized differently.
The researchers found that as long as the binding was weak, as it was in the first mouse, there was a lot of overlap in the receptors the ligand bound to in both T-cell types. However, in the second mouse, where the ligand prompted strong binding, there was far less overlap.
“We are now trying to find what causes that difference,” Dr Ignatowicz said. ![]()

Credit: NIAID
A fundamental theory about how the thymus “educates” T cells appears to be wrong, according to research published in Nature Communications.
It’s known that stem cells leave the bone marrow and travel to the thymus to become one of two CD4 T-cell types: effector cells or regulatory T cells (Tregs).
One widely held concept of why the cells become one type or the other is that they are exposed to different ligands in the thymus, said Leszek Ignatowicz, PhD, of Georgia Regents University in Augusta.
But when he and his colleagues limited the cells’ exposure to only one ligand, the same mix of T cells still emerged.
“We asked a simple question, ‘Is it going to affect their development,’ and the answer was ‘no,’” Dr Ignatowicz said. “The cells still mature in the thymus, so something else must be determining it.”
The finding provides more insight into immunity that could one day enable a new approach to vaccines that steer the thymus to produce more of whatever T-cell type a patient needs: more effector cells if they have an infection or cancer, more Tregs if they have autoimmune diseases such as arthritis and multiple sclerosis.
“We could help steer the education process in the desired direction,” Dr Ignatowicz said.
To uncover their findings, he and his colleagues studied two types of mice, each expressing a single ligand in the thymus. The researchers thought one would prompt strong ligand binding and favor Treg development, and the other would favor a weaker ligand bond and effector cell development.
The mix of resulting T cells was the same as if both were exposed to the usual thousands of ligands, although the experiment did reveal one difference.
Ligands—and, eventually, bacteria and other invaders—get the attention of T cells by activating their receptors. Both CD4 T-cell types generally have the same receptors, but they are organized differently.
The researchers found that as long as the binding was weak, as it was in the first mouse, there was a lot of overlap in the receptors the ligand bound to in both T-cell types. However, in the second mouse, where the ligand prompted strong binding, there was far less overlap.
“We are now trying to find what causes that difference,” Dr Ignatowicz said. ![]()
Microscopy advances net Nobel Prize

Credit: Max Planck Institute
for Biophysical Chemistry
Three scientists have received the 2014 Nobel Prize in Chemistry for aiding the development of super-resolved fluorescence microscopy.
For a long time, optical microscopy was held back by a presumed limitation: that it would never obtain a better resolution than half the wavelength
of light.
Working separately, this year’s Nobel Laureates in Chemistry circumvented this limitation and brought optical microscopy into the nanodimension.
Now, scientists can monitor the interplay between individual molecules inside cells, watch disease-related proteins aggregate, and track cell division at the nanolevel.
For enabling these advances, Eric Betzig, PhD, of the Howard Hughes Medical Institute in Ashburn, Virginia; Stefan W. Hell, PhD, of the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany; and William E. Moerner, PhD, of Stanford University in California, received the prize. The prize amount was SEK 8 million, to be shared equally among the Laureates.
The work in brief
In 1873, the microscopist Ernst Abbe stipulated a physical limit for the maximum resolution of traditional optical microscopy—0.2 micrometers. Drs Moerner, Hell, and Betzig were able to bypass this limit.
Dr Hell developed stimulated emission depletion (STED) microscopy. This method employs 2 laser beams. One stimulates fluorescent molecules to glow, and another cancels out all fluorescence except for that in a nanometer-sized volume.
Scanning over the sample, nanometer for nanometer, yields an image with a resolution better than Abbe’s stipulated limit.
Drs Betzig and Moerner laid the foundation for another method, single-molecule microscopy. This method relies upon the possibility to turn the fluorescence of individual molecules on and off.
Scientists image the same area multiple times, letting just a few interspersed molecules glow each time. Superimposing these images yields a dense super-image resolved at the nanolevel. In 2006, Dr Betzig used this method for the first time.
For more details on the Nobel Laureates and their work, visit Nobelprize.org. ![]()

Credit: Max Planck Institute
for Biophysical Chemistry
Three scientists have received the 2014 Nobel Prize in Chemistry for aiding the development of super-resolved fluorescence microscopy.
For a long time, optical microscopy was held back by a presumed limitation: that it would never obtain a better resolution than half the wavelength
of light.
Working separately, this year’s Nobel Laureates in Chemistry circumvented this limitation and brought optical microscopy into the nanodimension.
Now, scientists can monitor the interplay between individual molecules inside cells, watch disease-related proteins aggregate, and track cell division at the nanolevel.
For enabling these advances, Eric Betzig, PhD, of the Howard Hughes Medical Institute in Ashburn, Virginia; Stefan W. Hell, PhD, of the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany; and William E. Moerner, PhD, of Stanford University in California, received the prize. The prize amount was SEK 8 million, to be shared equally among the Laureates.
The work in brief
In 1873, the microscopist Ernst Abbe stipulated a physical limit for the maximum resolution of traditional optical microscopy—0.2 micrometers. Drs Moerner, Hell, and Betzig were able to bypass this limit.
Dr Hell developed stimulated emission depletion (STED) microscopy. This method employs 2 laser beams. One stimulates fluorescent molecules to glow, and another cancels out all fluorescence except for that in a nanometer-sized volume.
Scanning over the sample, nanometer for nanometer, yields an image with a resolution better than Abbe’s stipulated limit.
Drs Betzig and Moerner laid the foundation for another method, single-molecule microscopy. This method relies upon the possibility to turn the fluorescence of individual molecules on and off.
Scientists image the same area multiple times, letting just a few interspersed molecules glow each time. Superimposing these images yields a dense super-image resolved at the nanolevel. In 2006, Dr Betzig used this method for the first time.
For more details on the Nobel Laureates and their work, visit Nobelprize.org. ![]()

Credit: Max Planck Institute
for Biophysical Chemistry
Three scientists have received the 2014 Nobel Prize in Chemistry for aiding the development of super-resolved fluorescence microscopy.
For a long time, optical microscopy was held back by a presumed limitation: that it would never obtain a better resolution than half the wavelength
of light.
Working separately, this year’s Nobel Laureates in Chemistry circumvented this limitation and brought optical microscopy into the nanodimension.
Now, scientists can monitor the interplay between individual molecules inside cells, watch disease-related proteins aggregate, and track cell division at the nanolevel.
For enabling these advances, Eric Betzig, PhD, of the Howard Hughes Medical Institute in Ashburn, Virginia; Stefan W. Hell, PhD, of the Max Planck Institute for Biophysical Chemistry in Göttingen, Germany; and William E. Moerner, PhD, of Stanford University in California, received the prize. The prize amount was SEK 8 million, to be shared equally among the Laureates.
The work in brief
In 1873, the microscopist Ernst Abbe stipulated a physical limit for the maximum resolution of traditional optical microscopy—0.2 micrometers. Drs Moerner, Hell, and Betzig were able to bypass this limit.
Dr Hell developed stimulated emission depletion (STED) microscopy. This method employs 2 laser beams. One stimulates fluorescent molecules to glow, and another cancels out all fluorescence except for that in a nanometer-sized volume.
Scanning over the sample, nanometer for nanometer, yields an image with a resolution better than Abbe’s stipulated limit.
Drs Betzig and Moerner laid the foundation for another method, single-molecule microscopy. This method relies upon the possibility to turn the fluorescence of individual molecules on and off.
Scientists image the same area multiple times, letting just a few interspersed molecules glow each time. Superimposing these images yields a dense super-image resolved at the nanolevel. In 2006, Dr Betzig used this method for the first time.
For more details on the Nobel Laureates and their work, visit Nobelprize.org. ![]()
Price increases drive spending on cancer drugs

Credit: Steven Harbour
The recent surge in spending on oral anticancer drugs in the US exceeds the increase in use of these drugs, new research shows.
Average quarterly national spending on oral oncologics increased 37% during the period studied, from $940 million in the first quarter of 2006 to $1.4 billion in the third quarter of 2011.
But the average quarterly use of these drugs in the same time period increased by only 10%.
This suggests price increases are a significant driver of spending trends.
Rena M. Conti, PhD, of the University of Chicago in Illinois, and her colleagues examined recent trends in spending and use of oral oncologics and disclosed their findings in Health Affairs.
Of the 47 drugs analyzed, most were targeted agents (30%), hormonal agents (26%), and alkylating agents (19%).
The researchers observed a significant increase in national spending on oral oncologics from 2006 to 2011—an estimated average quarterly increase of $20 million.
This was driven by brand-name, patent-protected drugs, but the use of these drugs climbed a comparatively small amount. Average quarterly spending of patent-protected drugs increased 61%, and average quarterly use increased 30% between 2006 and the period from September 2010 to September 2011.
“This is an exciting time, an era of breakthrough cancer drugs,” Dr Conti said. “Some of these medications have extended the lives of many people with certain types of cancer. However, spending on these brand-name oral oncologics is outstripping national spending on all pharmaceuticals and all medical care spending generally.”
The researchers also discovered that when oncologics lose patent protection, spending takes a nosedive. The use of newly off-patent drugs increased by 16%, but average quarterly spending on those drugs fell by 65%.
Another finding was that US spending on targeted anticancer agents increased from 35% of all oral cancer drugs in 2006 to nearly 60% in 2011.
Meanwhile, spending on hormonal agents decreased from 42% of total spending to 19%, spending on antimetabolites increased from 11% to 12%, and spending on alkylating agents decreased from 10% to 8%. ![]()

Credit: Steven Harbour
The recent surge in spending on oral anticancer drugs in the US exceeds the increase in use of these drugs, new research shows.
Average quarterly national spending on oral oncologics increased 37% during the period studied, from $940 million in the first quarter of 2006 to $1.4 billion in the third quarter of 2011.
But the average quarterly use of these drugs in the same time period increased by only 10%.
This suggests price increases are a significant driver of spending trends.
Rena M. Conti, PhD, of the University of Chicago in Illinois, and her colleagues examined recent trends in spending and use of oral oncologics and disclosed their findings in Health Affairs.
Of the 47 drugs analyzed, most were targeted agents (30%), hormonal agents (26%), and alkylating agents (19%).
The researchers observed a significant increase in national spending on oral oncologics from 2006 to 2011—an estimated average quarterly increase of $20 million.
This was driven by brand-name, patent-protected drugs, but the use of these drugs climbed a comparatively small amount. Average quarterly spending of patent-protected drugs increased 61%, and average quarterly use increased 30% between 2006 and the period from September 2010 to September 2011.
“This is an exciting time, an era of breakthrough cancer drugs,” Dr Conti said. “Some of these medications have extended the lives of many people with certain types of cancer. However, spending on these brand-name oral oncologics is outstripping national spending on all pharmaceuticals and all medical care spending generally.”
The researchers also discovered that when oncologics lose patent protection, spending takes a nosedive. The use of newly off-patent drugs increased by 16%, but average quarterly spending on those drugs fell by 65%.
Another finding was that US spending on targeted anticancer agents increased from 35% of all oral cancer drugs in 2006 to nearly 60% in 2011.
Meanwhile, spending on hormonal agents decreased from 42% of total spending to 19%, spending on antimetabolites increased from 11% to 12%, and spending on alkylating agents decreased from 10% to 8%. ![]()

Credit: Steven Harbour
The recent surge in spending on oral anticancer drugs in the US exceeds the increase in use of these drugs, new research shows.
Average quarterly national spending on oral oncologics increased 37% during the period studied, from $940 million in the first quarter of 2006 to $1.4 billion in the third quarter of 2011.
But the average quarterly use of these drugs in the same time period increased by only 10%.
This suggests price increases are a significant driver of spending trends.
Rena M. Conti, PhD, of the University of Chicago in Illinois, and her colleagues examined recent trends in spending and use of oral oncologics and disclosed their findings in Health Affairs.
Of the 47 drugs analyzed, most were targeted agents (30%), hormonal agents (26%), and alkylating agents (19%).
The researchers observed a significant increase in national spending on oral oncologics from 2006 to 2011—an estimated average quarterly increase of $20 million.
This was driven by brand-name, patent-protected drugs, but the use of these drugs climbed a comparatively small amount. Average quarterly spending of patent-protected drugs increased 61%, and average quarterly use increased 30% between 2006 and the period from September 2010 to September 2011.
“This is an exciting time, an era of breakthrough cancer drugs,” Dr Conti said. “Some of these medications have extended the lives of many people with certain types of cancer. However, spending on these brand-name oral oncologics is outstripping national spending on all pharmaceuticals and all medical care spending generally.”
The researchers also discovered that when oncologics lose patent protection, spending takes a nosedive. The use of newly off-patent drugs increased by 16%, but average quarterly spending on those drugs fell by 65%.
Another finding was that US spending on targeted anticancer agents increased from 35% of all oral cancer drugs in 2006 to nearly 60% in 2011.
Meanwhile, spending on hormonal agents decreased from 42% of total spending to 19%, spending on antimetabolites increased from 11% to 12%, and spending on alkylating agents decreased from 10% to 8%. ![]()
Program for poor can boost hospital profits

Credit: Petr Kratochvil
A federal program designed to help the poor may actually be used to help US hospitals increase their profits, according to research published in Health Affairs.
Researchers examined enrollment in the 340B program, which provides deep discounts on outpatient drug purchases.
They found that hospitals and clinics that joined the program since 2004 currently serve more affluent and well-insured communities than those that qualified for the program in previous years.
This supports the idea that the program is changing from one that serves patients in need to one that enriches hospitals and their affiliated clinics, according to the researchers.
“This study provides the first nationally representative empirical evidence suggesting that the program’s original intent is being eroded by the actions of certain hospitals,” said study author Rena M. Conti, PhD, of the University of Chicago in Illinois.
This study follows work by Dr Conti and Peter B. Bach, MD, of Memorial Sloan-Kettering Cancer Center in New York, that was published in JAMA last year.
The JAMA study explained how 340B-qualified hospital-affiliated clinics can boost profits thanks to discounts on expensive anticancer drugs. The facilities receive the discounts under the expectation that the savings will be passed on to poor patients.
“Hospitals qualify for the program based on the poverty of their inpatient census only,” Dr Conti said. “The affiliated clinics are the only 340B institutions not required to pass the discounts off to patients or their insurers, nor do they have to report to the government exactly how these profits are used to serve the poor. Insurers’ and their patients’ payments for outpatient drug treatment don’t reflect the discounts the hospital receives.”
The 340B program, which began in 1992, was designed to help selected hospitals and their outpatient clinics serve low-income and uninsured patients by providing discounts of 30% to 50% on outpatient drugs.
About a decade ago, enrollment in 340B began to increase rapidly. Now, more than a third of the 4375 US non-federal hospitals are 340B-qualified. Recent Congressional and news reports suggest that, for selected hospitals, profits off the 340B program can be significant.
For their new study, Drs Conti and Bach examined the populations served by hospitals and clinics qualifying for 340B before and after the decade-long growth spurt. They matched data for all hospitals and clinics registered with the 340B program to socioeconomic data from the US Census Bureau.
The results showed that communities served by hospital-affiliated clinics joining the program in 2004 or later tended to have higher household incomes, much less unemployment, and higher rates of health insurance.
The researchers said their findings are consistent with recent complaints that, rather than serving vulnerable communities, the 340B program is being used to increase profits for hospitals and their affiliated clinics. ![]()

Credit: Petr Kratochvil
A federal program designed to help the poor may actually be used to help US hospitals increase their profits, according to research published in Health Affairs.
Researchers examined enrollment in the 340B program, which provides deep discounts on outpatient drug purchases.
They found that hospitals and clinics that joined the program since 2004 currently serve more affluent and well-insured communities than those that qualified for the program in previous years.
This supports the idea that the program is changing from one that serves patients in need to one that enriches hospitals and their affiliated clinics, according to the researchers.
“This study provides the first nationally representative empirical evidence suggesting that the program’s original intent is being eroded by the actions of certain hospitals,” said study author Rena M. Conti, PhD, of the University of Chicago in Illinois.
This study follows work by Dr Conti and Peter B. Bach, MD, of Memorial Sloan-Kettering Cancer Center in New York, that was published in JAMA last year.
The JAMA study explained how 340B-qualified hospital-affiliated clinics can boost profits thanks to discounts on expensive anticancer drugs. The facilities receive the discounts under the expectation that the savings will be passed on to poor patients.
“Hospitals qualify for the program based on the poverty of their inpatient census only,” Dr Conti said. “The affiliated clinics are the only 340B institutions not required to pass the discounts off to patients or their insurers, nor do they have to report to the government exactly how these profits are used to serve the poor. Insurers’ and their patients’ payments for outpatient drug treatment don’t reflect the discounts the hospital receives.”
The 340B program, which began in 1992, was designed to help selected hospitals and their outpatient clinics serve low-income and uninsured patients by providing discounts of 30% to 50% on outpatient drugs.
About a decade ago, enrollment in 340B began to increase rapidly. Now, more than a third of the 4375 US non-federal hospitals are 340B-qualified. Recent Congressional and news reports suggest that, for selected hospitals, profits off the 340B program can be significant.
For their new study, Drs Conti and Bach examined the populations served by hospitals and clinics qualifying for 340B before and after the decade-long growth spurt. They matched data for all hospitals and clinics registered with the 340B program to socioeconomic data from the US Census Bureau.
The results showed that communities served by hospital-affiliated clinics joining the program in 2004 or later tended to have higher household incomes, much less unemployment, and higher rates of health insurance.
The researchers said their findings are consistent with recent complaints that, rather than serving vulnerable communities, the 340B program is being used to increase profits for hospitals and their affiliated clinics. ![]()

Credit: Petr Kratochvil
A federal program designed to help the poor may actually be used to help US hospitals increase their profits, according to research published in Health Affairs.
Researchers examined enrollment in the 340B program, which provides deep discounts on outpatient drug purchases.
They found that hospitals and clinics that joined the program since 2004 currently serve more affluent and well-insured communities than those that qualified for the program in previous years.
This supports the idea that the program is changing from one that serves patients in need to one that enriches hospitals and their affiliated clinics, according to the researchers.
“This study provides the first nationally representative empirical evidence suggesting that the program’s original intent is being eroded by the actions of certain hospitals,” said study author Rena M. Conti, PhD, of the University of Chicago in Illinois.
This study follows work by Dr Conti and Peter B. Bach, MD, of Memorial Sloan-Kettering Cancer Center in New York, that was published in JAMA last year.
The JAMA study explained how 340B-qualified hospital-affiliated clinics can boost profits thanks to discounts on expensive anticancer drugs. The facilities receive the discounts under the expectation that the savings will be passed on to poor patients.
“Hospitals qualify for the program based on the poverty of their inpatient census only,” Dr Conti said. “The affiliated clinics are the only 340B institutions not required to pass the discounts off to patients or their insurers, nor do they have to report to the government exactly how these profits are used to serve the poor. Insurers’ and their patients’ payments for outpatient drug treatment don’t reflect the discounts the hospital receives.”
The 340B program, which began in 1992, was designed to help selected hospitals and their outpatient clinics serve low-income and uninsured patients by providing discounts of 30% to 50% on outpatient drugs.
About a decade ago, enrollment in 340B began to increase rapidly. Now, more than a third of the 4375 US non-federal hospitals are 340B-qualified. Recent Congressional and news reports suggest that, for selected hospitals, profits off the 340B program can be significant.
For their new study, Drs Conti and Bach examined the populations served by hospitals and clinics qualifying for 340B before and after the decade-long growth spurt. They matched data for all hospitals and clinics registered with the 340B program to socioeconomic data from the US Census Bureau.
The results showed that communities served by hospital-affiliated clinics joining the program in 2004 or later tended to have higher household incomes, much less unemployment, and higher rates of health insurance.
The researchers said their findings are consistent with recent complaints that, rather than serving vulnerable communities, the 340B program is being used to increase profits for hospitals and their affiliated clinics. ![]()
Mental health challenges fairly common in cancer patients

chemotherapy
Credit: Rhoda Baer
In a large German study, investigators found that nearly a third of cancer patients experienced some form of clinically relevant mental health
challenge.
Of the more than 2100 cancer patients interviewed, 32% had experienced a clinically meaningful level of mental or emotional distress in the previous 4 weeks.
This prevalence is higher than that observed in the general population, and the difference is primarily due to a higher rate of anxiety and adjustment disorders.
The incidence of mental health issues varied by cancer type. The highest was among patients with breast cancer (42%) and head and neck cancer (41%), followed by malignant melanoma (39%).
The lowest prevalence was seen among patients with prostate cancer (22%), stomach cancers (21%), and pancreatic cancer (20%).
These results appear in the Journal of Clinical Oncology.
“These findings reinforce that, as doctors, we need to be very aware of signs and symptoms of mental and emotional distress,” said lead study author Anja Mehnert, PhD, of the University of Leipzig in Germany.
“We must encourage patients to seek evaluation, support, and treatment if necessary, as there are long-term risks often associated with more severe, untreated mental health disorders. This research also sheds light on which patients we should watch more closely.”
Dr Mehnert and her colleagues conducted this study in 2141 cancer patients who were 18 to 75 years of age. The team conducted face-to-face interviews in hospitals, outpatient cancer care centers, and rehabilitation centers in Germany.
Interview answers were immediately entered into a computer based-diagnostic program. The test assessed various psychological symptoms over the previous 4-week period. Patients’ diagnoses were classified according to the Diagnostic and Statistical Manual of Mental Disorders, the standard classification used by mental health professionals.
The patients had a range of cancer types, with the most common being breast cancer (44%), prostate cancer (15%), and colorectal cancer (14%). The average time since cancer diagnosis was 13.5 months, and 51% of the participants were women.
The researchers found that 32% of patients experienced at least one clinically meaningful mental health issue (defined in the study as a mental health disorder). This is a higher prevalence than in the general population, in which 18% to 20% of people are estimated to have a clinically meaningful mental disorder.
In the 4-week period prior to the interview, 11.5% of patients experienced an anxiety disorder. Eleven percent met the criteria for an adjustment disorder, a predominantly mixed anxiety-depressive syndrome that persisted for at least 4 weeks in response to a significant life change. And 6.5% of patients had signs of a mood disorder such as major depression.
The 11.5% rate of anxiety disorders—such as phobia, panic, or generalized anxiety disorder—was slightly higher than in the general population (9%), while the prevalence of other mental health diagnoses was similar to rates in the general population.
It is likely that the prevalence of adjustment disorders (11%), which is rarely assessed in general population surveys, significantly contributed to the overall higher prevalence rate of mental disorders in this population of patients with cancer.
Dr Mehnert said it was surprising that patients with a more treatable malignancy, such as breast cancer, experienced more distress than people with cancers that are more challenging to treat, such as stomach and pancreatic cancers. So more research is needed to interpret these findings.
The investigators believe the study’s results may be useful for planning future support programs for cancer patients, and they can provide additional information to guide programs for people with specific cancer types.
The team also believes the findings can likely be generalized to patients in the US because the prevalence of mental health diagnoses is similar between the 2 countries. ![]()

chemotherapy
Credit: Rhoda Baer
In a large German study, investigators found that nearly a third of cancer patients experienced some form of clinically relevant mental health
challenge.
Of the more than 2100 cancer patients interviewed, 32% had experienced a clinically meaningful level of mental or emotional distress in the previous 4 weeks.
This prevalence is higher than that observed in the general population, and the difference is primarily due to a higher rate of anxiety and adjustment disorders.
The incidence of mental health issues varied by cancer type. The highest was among patients with breast cancer (42%) and head and neck cancer (41%), followed by malignant melanoma (39%).
The lowest prevalence was seen among patients with prostate cancer (22%), stomach cancers (21%), and pancreatic cancer (20%).
These results appear in the Journal of Clinical Oncology.
“These findings reinforce that, as doctors, we need to be very aware of signs and symptoms of mental and emotional distress,” said lead study author Anja Mehnert, PhD, of the University of Leipzig in Germany.
“We must encourage patients to seek evaluation, support, and treatment if necessary, as there are long-term risks often associated with more severe, untreated mental health disorders. This research also sheds light on which patients we should watch more closely.”
Dr Mehnert and her colleagues conducted this study in 2141 cancer patients who were 18 to 75 years of age. The team conducted face-to-face interviews in hospitals, outpatient cancer care centers, and rehabilitation centers in Germany.
Interview answers were immediately entered into a computer based-diagnostic program. The test assessed various psychological symptoms over the previous 4-week period. Patients’ diagnoses were classified according to the Diagnostic and Statistical Manual of Mental Disorders, the standard classification used by mental health professionals.
The patients had a range of cancer types, with the most common being breast cancer (44%), prostate cancer (15%), and colorectal cancer (14%). The average time since cancer diagnosis was 13.5 months, and 51% of the participants were women.
The researchers found that 32% of patients experienced at least one clinically meaningful mental health issue (defined in the study as a mental health disorder). This is a higher prevalence than in the general population, in which 18% to 20% of people are estimated to have a clinically meaningful mental disorder.
In the 4-week period prior to the interview, 11.5% of patients experienced an anxiety disorder. Eleven percent met the criteria for an adjustment disorder, a predominantly mixed anxiety-depressive syndrome that persisted for at least 4 weeks in response to a significant life change. And 6.5% of patients had signs of a mood disorder such as major depression.
The 11.5% rate of anxiety disorders—such as phobia, panic, or generalized anxiety disorder—was slightly higher than in the general population (9%), while the prevalence of other mental health diagnoses was similar to rates in the general population.
It is likely that the prevalence of adjustment disorders (11%), which is rarely assessed in general population surveys, significantly contributed to the overall higher prevalence rate of mental disorders in this population of patients with cancer.
Dr Mehnert said it was surprising that patients with a more treatable malignancy, such as breast cancer, experienced more distress than people with cancers that are more challenging to treat, such as stomach and pancreatic cancers. So more research is needed to interpret these findings.
The investigators believe the study’s results may be useful for planning future support programs for cancer patients, and they can provide additional information to guide programs for people with specific cancer types.
The team also believes the findings can likely be generalized to patients in the US because the prevalence of mental health diagnoses is similar between the 2 countries. ![]()

chemotherapy
Credit: Rhoda Baer
In a large German study, investigators found that nearly a third of cancer patients experienced some form of clinically relevant mental health
challenge.
Of the more than 2100 cancer patients interviewed, 32% had experienced a clinically meaningful level of mental or emotional distress in the previous 4 weeks.
This prevalence is higher than that observed in the general population, and the difference is primarily due to a higher rate of anxiety and adjustment disorders.
The incidence of mental health issues varied by cancer type. The highest was among patients with breast cancer (42%) and head and neck cancer (41%), followed by malignant melanoma (39%).
The lowest prevalence was seen among patients with prostate cancer (22%), stomach cancers (21%), and pancreatic cancer (20%).
These results appear in the Journal of Clinical Oncology.
“These findings reinforce that, as doctors, we need to be very aware of signs and symptoms of mental and emotional distress,” said lead study author Anja Mehnert, PhD, of the University of Leipzig in Germany.
“We must encourage patients to seek evaluation, support, and treatment if necessary, as there are long-term risks often associated with more severe, untreated mental health disorders. This research also sheds light on which patients we should watch more closely.”
Dr Mehnert and her colleagues conducted this study in 2141 cancer patients who were 18 to 75 years of age. The team conducted face-to-face interviews in hospitals, outpatient cancer care centers, and rehabilitation centers in Germany.
Interview answers were immediately entered into a computer based-diagnostic program. The test assessed various psychological symptoms over the previous 4-week period. Patients’ diagnoses were classified according to the Diagnostic and Statistical Manual of Mental Disorders, the standard classification used by mental health professionals.
The patients had a range of cancer types, with the most common being breast cancer (44%), prostate cancer (15%), and colorectal cancer (14%). The average time since cancer diagnosis was 13.5 months, and 51% of the participants were women.
The researchers found that 32% of patients experienced at least one clinically meaningful mental health issue (defined in the study as a mental health disorder). This is a higher prevalence than in the general population, in which 18% to 20% of people are estimated to have a clinically meaningful mental disorder.
In the 4-week period prior to the interview, 11.5% of patients experienced an anxiety disorder. Eleven percent met the criteria for an adjustment disorder, a predominantly mixed anxiety-depressive syndrome that persisted for at least 4 weeks in response to a significant life change. And 6.5% of patients had signs of a mood disorder such as major depression.
The 11.5% rate of anxiety disorders—such as phobia, panic, or generalized anxiety disorder—was slightly higher than in the general population (9%), while the prevalence of other mental health diagnoses was similar to rates in the general population.
It is likely that the prevalence of adjustment disorders (11%), which is rarely assessed in general population surveys, significantly contributed to the overall higher prevalence rate of mental disorders in this population of patients with cancer.
Dr Mehnert said it was surprising that patients with a more treatable malignancy, such as breast cancer, experienced more distress than people with cancers that are more challenging to treat, such as stomach and pancreatic cancers. So more research is needed to interpret these findings.
The investigators believe the study’s results may be useful for planning future support programs for cancer patients, and they can provide additional information to guide programs for people with specific cancer types.
The team also believes the findings can likely be generalized to patients in the US because the prevalence of mental health diagnoses is similar between the 2 countries. ![]()
Survey shows lack of adherence to safety guidelines

Credit: Bill Branson
Healthcare professionals do not consistently follow the recommended safe handling practices for antineoplastic drugs, according to a survey published in the Journal of Occupational and Environmental Hygiene.
Researchers surveyed more than 2000 healthcare workers and found that a majority do not always use the recommended personal protective equipment when they are administering antineoplastic drugs.
Furthermore, some respondents reported spills or leaks of a drug during administration, and a small percentage said they had experienced skin
contact with an antineoplastic drug.
“Chemotherapy drugs save lives of cancer patients but also can result in adverse health outcomes in workers who are exposed to these drugs, including cancer, reproductive problems, and organ damage when recommended safe handling guidelines are not followed,” said John Howard, MD, director of the National Institute for Occupational Safety and Health (NIOSH).
NIOSH researchers conducted this study, which included 2069 healthcare personnel who completed the 2011 Health and Safety Practices Survey of Healthcare Workers.
Results showed that, despite the longstanding availability of authoritative safe handling guidelines (ASHP, NIOSH, ONS, OSHA), recommended exposure controls were not always used.
For example, 80% of respondents said they do not always wear 2 pairs of chemotherapy gloves, and 15% said they don’t always wear a single pair.
Forty-two percent of respondents said they don’t always wear a nonabsorbent gown with a closed front and tight-fitting cuffs, and 12% had taken home potentially contaminated clothing.
Twelve percent of respondents reported a spill or leak of an antineoplastic drug during administration, and 4% reported skin contact with an antineoplastic drug.
Six percent of respondents said they primed intravenous tubing with an antineoplastic drug instead of a non-drug containing liquid, and 12% said the pharmacy department followed this practice.
Taking these and other findings into account, the researchers concluded that better risk communication is needed to ensure that employers and employees are fully aware of the hazards and the availability of precautionary measures to minimize exposure to antineoplastic drugs. ![]()

Credit: Bill Branson
Healthcare professionals do not consistently follow the recommended safe handling practices for antineoplastic drugs, according to a survey published in the Journal of Occupational and Environmental Hygiene.
Researchers surveyed more than 2000 healthcare workers and found that a majority do not always use the recommended personal protective equipment when they are administering antineoplastic drugs.
Furthermore, some respondents reported spills or leaks of a drug during administration, and a small percentage said they had experienced skin
contact with an antineoplastic drug.
“Chemotherapy drugs save lives of cancer patients but also can result in adverse health outcomes in workers who are exposed to these drugs, including cancer, reproductive problems, and organ damage when recommended safe handling guidelines are not followed,” said John Howard, MD, director of the National Institute for Occupational Safety and Health (NIOSH).
NIOSH researchers conducted this study, which included 2069 healthcare personnel who completed the 2011 Health and Safety Practices Survey of Healthcare Workers.
Results showed that, despite the longstanding availability of authoritative safe handling guidelines (ASHP, NIOSH, ONS, OSHA), recommended exposure controls were not always used.
For example, 80% of respondents said they do not always wear 2 pairs of chemotherapy gloves, and 15% said they don’t always wear a single pair.
Forty-two percent of respondents said they don’t always wear a nonabsorbent gown with a closed front and tight-fitting cuffs, and 12% had taken home potentially contaminated clothing.
Twelve percent of respondents reported a spill or leak of an antineoplastic drug during administration, and 4% reported skin contact with an antineoplastic drug.
Six percent of respondents said they primed intravenous tubing with an antineoplastic drug instead of a non-drug containing liquid, and 12% said the pharmacy department followed this practice.
Taking these and other findings into account, the researchers concluded that better risk communication is needed to ensure that employers and employees are fully aware of the hazards and the availability of precautionary measures to minimize exposure to antineoplastic drugs. ![]()

Credit: Bill Branson
Healthcare professionals do not consistently follow the recommended safe handling practices for antineoplastic drugs, according to a survey published in the Journal of Occupational and Environmental Hygiene.
Researchers surveyed more than 2000 healthcare workers and found that a majority do not always use the recommended personal protective equipment when they are administering antineoplastic drugs.
Furthermore, some respondents reported spills or leaks of a drug during administration, and a small percentage said they had experienced skin
contact with an antineoplastic drug.
“Chemotherapy drugs save lives of cancer patients but also can result in adverse health outcomes in workers who are exposed to these drugs, including cancer, reproductive problems, and organ damage when recommended safe handling guidelines are not followed,” said John Howard, MD, director of the National Institute for Occupational Safety and Health (NIOSH).
NIOSH researchers conducted this study, which included 2069 healthcare personnel who completed the 2011 Health and Safety Practices Survey of Healthcare Workers.
Results showed that, despite the longstanding availability of authoritative safe handling guidelines (ASHP, NIOSH, ONS, OSHA), recommended exposure controls were not always used.
For example, 80% of respondents said they do not always wear 2 pairs of chemotherapy gloves, and 15% said they don’t always wear a single pair.
Forty-two percent of respondents said they don’t always wear a nonabsorbent gown with a closed front and tight-fitting cuffs, and 12% had taken home potentially contaminated clothing.
Twelve percent of respondents reported a spill or leak of an antineoplastic drug during administration, and 4% reported skin contact with an antineoplastic drug.
Six percent of respondents said they primed intravenous tubing with an antineoplastic drug instead of a non-drug containing liquid, and 12% said the pharmacy department followed this practice.
Taking these and other findings into account, the researchers concluded that better risk communication is needed to ensure that employers and employees are fully aware of the hazards and the availability of precautionary measures to minimize exposure to antineoplastic drugs.
Animal studies help explain chemo brain

californica
releasing inkafter being disturbed
Results of preclinical research appear to explain how the anticancer agent doxorubicin can cause chemo brain.
Neuroscientists conducted experiments in cells from rats and Aplysia californica, a marine mollusk that has many of the same memory mechanisms as humans.
This revealed memory mechanisms that are inhibited by doxorubicin, as well as a method of unblocking these mechanisms—administering a drug known as SB203580.
“Our research has implications in the care of people given to cognitive deficits following drug treatment for cancer,” said John H. Byrne, PhD, of the University of Texas Health Medical School.
He added that understanding how drugs like doxorubicin impact the brain is an important first step in alleviating chemo brain, which is characterized by forgetfulness, trouble concentrating, and difficulty multitasking.
Dr Byrne and his colleagues explained this first step in The Journal of Neuroscience.
The researchers knew that, in non-neuronal cells, doxorubicin inhibits the expression of MAPK phosphatases, thereby inhibiting the dephosphorylation of ERK and p38 MAPK, 2 MAPK isoforms that are important for long-term memory.
To evaluate doxorubicin’s effects on levels of phosphorylated ERK and p38 MAPK, the team used cultures of cortical neurons from rats and sensory neurons from Aplysia californica.
Experiments showed that doxorubicin elevated levels of phosphorylated ERK and phosphorylated p38 MAPK in sensory neurons and cortical neurons. In addition, the drug increased phosphorylation of the downstream transcriptional repressor CREB2 in sensory neurons.
The researchers also assessed doxorubicin’s effects on long-term enhanced excitability, long-term synaptic facilitation, and long-term
synaptic depression.
They found that doxorubicin enhanced long-term synaptic depression induced by the neuropeptide Phe-Met-Arg-Phe-NH2. And the drug inhibited long-term synaptic facilitation induced by serotonin.
However, the researchers were able to restore long-term synaptic facilitation with SB203580, an inhibitor of p38 MAPK.
Unfortunately, SB203580 would not be appropriate for human use, Dr Byrne noted, adding that his team would like to identify other drugs that might have the same effect as SB203580.
The researchers also hope to determine if doxorubicin works the same way in humans as it did in these experiments.

californica
releasing inkafter being disturbed
Results of preclinical research appear to explain how the anticancer agent doxorubicin can cause chemo brain.
Neuroscientists conducted experiments in cells from rats and Aplysia californica, a marine mollusk that has many of the same memory mechanisms as humans.
This revealed memory mechanisms that are inhibited by doxorubicin, as well as a method of unblocking these mechanisms—administering a drug known as SB203580.
“Our research has implications in the care of people given to cognitive deficits following drug treatment for cancer,” said John H. Byrne, PhD, of the University of Texas Health Medical School.
He added that understanding how drugs like doxorubicin impact the brain is an important first step in alleviating chemo brain, which is characterized by forgetfulness, trouble concentrating, and difficulty multitasking.
Dr Byrne and his colleagues explained this first step in The Journal of Neuroscience.
The researchers knew that, in non-neuronal cells, doxorubicin inhibits the expression of MAPK phosphatases, thereby inhibiting the dephosphorylation of ERK and p38 MAPK, 2 MAPK isoforms that are important for long-term memory.
To evaluate doxorubicin’s effects on levels of phosphorylated ERK and p38 MAPK, the team used cultures of cortical neurons from rats and sensory neurons from Aplysia californica.
Experiments showed that doxorubicin elevated levels of phosphorylated ERK and phosphorylated p38 MAPK in sensory neurons and cortical neurons. In addition, the drug increased phosphorylation of the downstream transcriptional repressor CREB2 in sensory neurons.
The researchers also assessed doxorubicin’s effects on long-term enhanced excitability, long-term synaptic facilitation, and long-term
synaptic depression.
They found that doxorubicin enhanced long-term synaptic depression induced by the neuropeptide Phe-Met-Arg-Phe-NH2. And the drug inhibited long-term synaptic facilitation induced by serotonin.
However, the researchers were able to restore long-term synaptic facilitation with SB203580, an inhibitor of p38 MAPK.
Unfortunately, SB203580 would not be appropriate for human use, Dr Byrne noted, adding that his team would like to identify other drugs that might have the same effect as SB203580.
The researchers also hope to determine if doxorubicin works the same way in humans as it did in these experiments.

californica
releasing inkafter being disturbed
Results of preclinical research appear to explain how the anticancer agent doxorubicin can cause chemo brain.
Neuroscientists conducted experiments in cells from rats and Aplysia californica, a marine mollusk that has many of the same memory mechanisms as humans.
This revealed memory mechanisms that are inhibited by doxorubicin, as well as a method of unblocking these mechanisms—administering a drug known as SB203580.
“Our research has implications in the care of people given to cognitive deficits following drug treatment for cancer,” said John H. Byrne, PhD, of the University of Texas Health Medical School.
He added that understanding how drugs like doxorubicin impact the brain is an important first step in alleviating chemo brain, which is characterized by forgetfulness, trouble concentrating, and difficulty multitasking.
Dr Byrne and his colleagues explained this first step in The Journal of Neuroscience.
The researchers knew that, in non-neuronal cells, doxorubicin inhibits the expression of MAPK phosphatases, thereby inhibiting the dephosphorylation of ERK and p38 MAPK, 2 MAPK isoforms that are important for long-term memory.
To evaluate doxorubicin’s effects on levels of phosphorylated ERK and p38 MAPK, the team used cultures of cortical neurons from rats and sensory neurons from Aplysia californica.
Experiments showed that doxorubicin elevated levels of phosphorylated ERK and phosphorylated p38 MAPK in sensory neurons and cortical neurons. In addition, the drug increased phosphorylation of the downstream transcriptional repressor CREB2 in sensory neurons.
The researchers also assessed doxorubicin’s effects on long-term enhanced excitability, long-term synaptic facilitation, and long-term
synaptic depression.
They found that doxorubicin enhanced long-term synaptic depression induced by the neuropeptide Phe-Met-Arg-Phe-NH2. And the drug inhibited long-term synaptic facilitation induced by serotonin.
However, the researchers were able to restore long-term synaptic facilitation with SB203580, an inhibitor of p38 MAPK.
Unfortunately, SB203580 would not be appropriate for human use, Dr Byrne noted, adding that his team would like to identify other drugs that might have the same effect as SB203580.
The researchers also hope to determine if doxorubicin works the same way in humans as it did in these experiments.
New insight into HSCs’ role in hematopoiesis
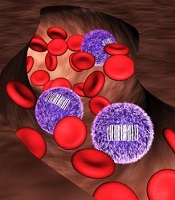
allows scientists to identify
differences in blood cell origin
Credit: Camargo Lab
By developing a tracking system for stem cells, researchers may have discovered previously unrecognized features of hematopoiesis.
Their work suggests the main drivers of steady-state hematopoiesis are not hematopoietic stem cells (HSCs) but their less pluripotent descendants, progenitor cells.
The team speculates that stable populations of long-lived progenitor cells are responsible for giving rise to specific blood cell types, while HSCs likely act as essential reserves.
The research, published in Nature, indicates that progenitor cells could be just as valuable as HSCs for blood regeneration therapies.
The work challenges what textbooks have long read: that HSCs maintain the day-to-day renewal of blood, a conclusion drawn from their importance in re-establishing blood cell populations after bone marrow transplants.
Due to a lack of tools to study how blood forms in a normal context, no one was able to track the origin of blood cells without doing a transplant.
Fernando Camargo, PhD, of Boston Children’s Hospital in Massachusetts, and his colleagues addressed this problem with a tool that generates a unique barcode in the DNA of all HSCs and their progenitor cells in a mouse.
When a tagged cell divides, all of its descendant cells possess the same barcode. This biological inventory system makes it possible to determine the number of HSCs/progenitors being used to make blood and how long they live, as well as answer fundamental questions about where individual blood cells come from.
“There’s never been such a robust experimental method that could allow people to look at lineage relationships between mature cell types in the body without doing transplantation,” said study author Jianlong Sun, PhD, also of Boston Children’s Hospital.
“One of the major directions we can now go is to revisit the entire blood cell hierarchy and see how the current knowledge holds true when we use this internal labeling system.”
“People have tried using viruses to tag blood cells in the past, but the cells needed to be taken out of the body, infected, and re-transplanted, which raised a number of issues,” Dr Camargo noted. “I wanted to figure out a way to label blood cells inside of the body, and the best idea I had was to use mobile genetic elements called transposons.”
A transposon is a piece of genetic code that can jump to a random point in DNA when exposed to the enzyme transposase. Dr Camargo’s approach works using transgenic mice that possess a single fish-derived transposon in all of their blood cells.
When one of these mice is exposed to transposase, each of its blood cells’ transposons changes location. The location in the DNA where a transposon moves acts as an individual cell’s barcode, so that if the mouse’s blood is taken a few months later, any cell with the same transposon location can be linked back to its parent cell.
Now, the researchers are planning to explore more applications for their barcode tool.
“We are also tremendously excited to use this tool to barcode and track descendants of different stem cells or progenitor cells for a range of conditions, from aging to the normal immune response,” Dr Sun said.
“We first used this technology for blood analysis. However, this system can also help address basic questions about cell populations in solid tissue. You can imagine being able to look at tumor progression or identify the precise origins of cancer cells that have broken off from a tumor and are now circulating in the blood.”

allows scientists to identify
differences in blood cell origin
Credit: Camargo Lab
By developing a tracking system for stem cells, researchers may have discovered previously unrecognized features of hematopoiesis.
Their work suggests the main drivers of steady-state hematopoiesis are not hematopoietic stem cells (HSCs) but their less pluripotent descendants, progenitor cells.
The team speculates that stable populations of long-lived progenitor cells are responsible for giving rise to specific blood cell types, while HSCs likely act as essential reserves.
The research, published in Nature, indicates that progenitor cells could be just as valuable as HSCs for blood regeneration therapies.
The work challenges what textbooks have long read: that HSCs maintain the day-to-day renewal of blood, a conclusion drawn from their importance in re-establishing blood cell populations after bone marrow transplants.
Due to a lack of tools to study how blood forms in a normal context, no one was able to track the origin of blood cells without doing a transplant.
Fernando Camargo, PhD, of Boston Children’s Hospital in Massachusetts, and his colleagues addressed this problem with a tool that generates a unique barcode in the DNA of all HSCs and their progenitor cells in a mouse.
When a tagged cell divides, all of its descendant cells possess the same barcode. This biological inventory system makes it possible to determine the number of HSCs/progenitors being used to make blood and how long they live, as well as answer fundamental questions about where individual blood cells come from.
“There’s never been such a robust experimental method that could allow people to look at lineage relationships between mature cell types in the body without doing transplantation,” said study author Jianlong Sun, PhD, also of Boston Children’s Hospital.
“One of the major directions we can now go is to revisit the entire blood cell hierarchy and see how the current knowledge holds true when we use this internal labeling system.”
“People have tried using viruses to tag blood cells in the past, but the cells needed to be taken out of the body, infected, and re-transplanted, which raised a number of issues,” Dr Camargo noted. “I wanted to figure out a way to label blood cells inside of the body, and the best idea I had was to use mobile genetic elements called transposons.”
A transposon is a piece of genetic code that can jump to a random point in DNA when exposed to the enzyme transposase. Dr Camargo’s approach works using transgenic mice that possess a single fish-derived transposon in all of their blood cells.
When one of these mice is exposed to transposase, each of its blood cells’ transposons changes location. The location in the DNA where a transposon moves acts as an individual cell’s barcode, so that if the mouse’s blood is taken a few months later, any cell with the same transposon location can be linked back to its parent cell.
Now, the researchers are planning to explore more applications for their barcode tool.
“We are also tremendously excited to use this tool to barcode and track descendants of different stem cells or progenitor cells for a range of conditions, from aging to the normal immune response,” Dr Sun said.
“We first used this technology for blood analysis. However, this system can also help address basic questions about cell populations in solid tissue. You can imagine being able to look at tumor progression or identify the precise origins of cancer cells that have broken off from a tumor and are now circulating in the blood.”

allows scientists to identify
differences in blood cell origin
Credit: Camargo Lab
By developing a tracking system for stem cells, researchers may have discovered previously unrecognized features of hematopoiesis.
Their work suggests the main drivers of steady-state hematopoiesis are not hematopoietic stem cells (HSCs) but their less pluripotent descendants, progenitor cells.
The team speculates that stable populations of long-lived progenitor cells are responsible for giving rise to specific blood cell types, while HSCs likely act as essential reserves.
The research, published in Nature, indicates that progenitor cells could be just as valuable as HSCs for blood regeneration therapies.
The work challenges what textbooks have long read: that HSCs maintain the day-to-day renewal of blood, a conclusion drawn from their importance in re-establishing blood cell populations after bone marrow transplants.
Due to a lack of tools to study how blood forms in a normal context, no one was able to track the origin of blood cells without doing a transplant.
Fernando Camargo, PhD, of Boston Children’s Hospital in Massachusetts, and his colleagues addressed this problem with a tool that generates a unique barcode in the DNA of all HSCs and their progenitor cells in a mouse.
When a tagged cell divides, all of its descendant cells possess the same barcode. This biological inventory system makes it possible to determine the number of HSCs/progenitors being used to make blood and how long they live, as well as answer fundamental questions about where individual blood cells come from.
“There’s never been such a robust experimental method that could allow people to look at lineage relationships between mature cell types in the body without doing transplantation,” said study author Jianlong Sun, PhD, also of Boston Children’s Hospital.
“One of the major directions we can now go is to revisit the entire blood cell hierarchy and see how the current knowledge holds true when we use this internal labeling system.”
“People have tried using viruses to tag blood cells in the past, but the cells needed to be taken out of the body, infected, and re-transplanted, which raised a number of issues,” Dr Camargo noted. “I wanted to figure out a way to label blood cells inside of the body, and the best idea I had was to use mobile genetic elements called transposons.”
A transposon is a piece of genetic code that can jump to a random point in DNA when exposed to the enzyme transposase. Dr Camargo’s approach works using transgenic mice that possess a single fish-derived transposon in all of their blood cells.
When one of these mice is exposed to transposase, each of its blood cells’ transposons changes location. The location in the DNA where a transposon moves acts as an individual cell’s barcode, so that if the mouse’s blood is taken a few months later, any cell with the same transposon location can be linked back to its parent cell.
Now, the researchers are planning to explore more applications for their barcode tool.
“We are also tremendously excited to use this tool to barcode and track descendants of different stem cells or progenitor cells for a range of conditions, from aging to the normal immune response,” Dr Sun said.
“We first used this technology for blood analysis. However, this system can also help address basic questions about cell populations in solid tissue. You can imagine being able to look at tumor progression or identify the precise origins of cancer cells that have broken off from a tumor and are now circulating in the blood.”
Novel capsule could replace injections

Credit: USDA
Scientists have created a novel drug capsule coated with tiny needles that can inject drugs directly into the lining of the stomach after the capsule is swallowed.
In experiments with pigs, the capsule delivered insulin more efficiently than an injection under the skin, and there were no harmful side effects as the capsule passed through the digestive system.
The researchers anticipate the capsule would be most useful for delivering biopharmaceuticals such as antibodies to treat cancers and other disorders.
“This could be a way that the patient can circumvent the need to have an infusion or subcutaneous administration of a drug,” said Giovanni Traverso, MB BChir, PhD, of Massachusetts General Hospital in Boston.
He and his colleagues described their capsule in the Journal of Pharmaceutical Sciences.
The team had set out to design a capsule that would serve as a platform for the delivery of a wide range of therapeutics, prevent degradation of the drugs, and inject the payload directly into the lining of the gastrointestinal tract.
Their prototype acrylic capsule, 2 cm long and 1 cm in diameter, includes a reservoir for the drug and is coated with hollow, stainless steel needles about 5 mm long.
Previous studies of accidental ingestion of sharp objects in human patients have suggested that it could be safe to swallow a capsule coated with short needles. Because there are no pain receptors in the gastrointestinal tract, patients would not feel any pain from the drug injection.
To test whether this type of capsule could allow safe and effective drug delivery, the researchers tested it in pigs, with insulin as the drug payload.
It took more than a week for the capsules to move through the entire digestive tract, and the researchers found no traces of tissue damage, supporting the potential safety of this novel approach.
They also found the microneedles successfully injected insulin into the lining of the stomach, small intestine, and colon, causing the animals’ blood glucose levels to drop. This reduction in blood glucose was faster and larger than the drop seen when the same amount of glucose was given by subcutaneous injection.
“The kinetics are much better, and much faster-onset, than those seen with traditional under-the-skin administration,” Dr Traverso said. “For molecules that are particularly difficult to absorb, this would be a way of actually administering them at much higher efficiency.”
This approach could also be used to administer vaccines that normally have to be injected, the researchers said.
The team now plans to modify the capsule so that peristalsis would slowly squeeze the drug out of the capsule as it travels through the digestive tract.
They are also working on capsules with needles made of degradable polymers and sugar that would break off and become embedded in the gut lining, where they would slowly disintegrate and release the drug. This would further minimize any safety concern.

Credit: USDA
Scientists have created a novel drug capsule coated with tiny needles that can inject drugs directly into the lining of the stomach after the capsule is swallowed.
In experiments with pigs, the capsule delivered insulin more efficiently than an injection under the skin, and there were no harmful side effects as the capsule passed through the digestive system.
The researchers anticipate the capsule would be most useful for delivering biopharmaceuticals such as antibodies to treat cancers and other disorders.
“This could be a way that the patient can circumvent the need to have an infusion or subcutaneous administration of a drug,” said Giovanni Traverso, MB BChir, PhD, of Massachusetts General Hospital in Boston.
He and his colleagues described their capsule in the Journal of Pharmaceutical Sciences.
The team had set out to design a capsule that would serve as a platform for the delivery of a wide range of therapeutics, prevent degradation of the drugs, and inject the payload directly into the lining of the gastrointestinal tract.
Their prototype acrylic capsule, 2 cm long and 1 cm in diameter, includes a reservoir for the drug and is coated with hollow, stainless steel needles about 5 mm long.
Previous studies of accidental ingestion of sharp objects in human patients have suggested that it could be safe to swallow a capsule coated with short needles. Because there are no pain receptors in the gastrointestinal tract, patients would not feel any pain from the drug injection.
To test whether this type of capsule could allow safe and effective drug delivery, the researchers tested it in pigs, with insulin as the drug payload.
It took more than a week for the capsules to move through the entire digestive tract, and the researchers found no traces of tissue damage, supporting the potential safety of this novel approach.
They also found the microneedles successfully injected insulin into the lining of the stomach, small intestine, and colon, causing the animals’ blood glucose levels to drop. This reduction in blood glucose was faster and larger than the drop seen when the same amount of glucose was given by subcutaneous injection.
“The kinetics are much better, and much faster-onset, than those seen with traditional under-the-skin administration,” Dr Traverso said. “For molecules that are particularly difficult to absorb, this would be a way of actually administering them at much higher efficiency.”
This approach could also be used to administer vaccines that normally have to be injected, the researchers said.
The team now plans to modify the capsule so that peristalsis would slowly squeeze the drug out of the capsule as it travels through the digestive tract.
They are also working on capsules with needles made of degradable polymers and sugar that would break off and become embedded in the gut lining, where they would slowly disintegrate and release the drug. This would further minimize any safety concern.

Credit: USDA
Scientists have created a novel drug capsule coated with tiny needles that can inject drugs directly into the lining of the stomach after the capsule is swallowed.
In experiments with pigs, the capsule delivered insulin more efficiently than an injection under the skin, and there were no harmful side effects as the capsule passed through the digestive system.
The researchers anticipate the capsule would be most useful for delivering biopharmaceuticals such as antibodies to treat cancers and other disorders.
“This could be a way that the patient can circumvent the need to have an infusion or subcutaneous administration of a drug,” said Giovanni Traverso, MB BChir, PhD, of Massachusetts General Hospital in Boston.
He and his colleagues described their capsule in the Journal of Pharmaceutical Sciences.
The team had set out to design a capsule that would serve as a platform for the delivery of a wide range of therapeutics, prevent degradation of the drugs, and inject the payload directly into the lining of the gastrointestinal tract.
Their prototype acrylic capsule, 2 cm long and 1 cm in diameter, includes a reservoir for the drug and is coated with hollow, stainless steel needles about 5 mm long.
Previous studies of accidental ingestion of sharp objects in human patients have suggested that it could be safe to swallow a capsule coated with short needles. Because there are no pain receptors in the gastrointestinal tract, patients would not feel any pain from the drug injection.
To test whether this type of capsule could allow safe and effective drug delivery, the researchers tested it in pigs, with insulin as the drug payload.
It took more than a week for the capsules to move through the entire digestive tract, and the researchers found no traces of tissue damage, supporting the potential safety of this novel approach.
They also found the microneedles successfully injected insulin into the lining of the stomach, small intestine, and colon, causing the animals’ blood glucose levels to drop. This reduction in blood glucose was faster and larger than the drop seen when the same amount of glucose was given by subcutaneous injection.
“The kinetics are much better, and much faster-onset, than those seen with traditional under-the-skin administration,” Dr Traverso said. “For molecules that are particularly difficult to absorb, this would be a way of actually administering them at much higher efficiency.”
This approach could also be used to administer vaccines that normally have to be injected, the researchers said.
The team now plans to modify the capsule so that peristalsis would slowly squeeze the drug out of the capsule as it travels through the digestive tract.
They are also working on capsules with needles made of degradable polymers and sugar that would break off and become embedded in the gut lining, where they would slowly disintegrate and release the drug. This would further minimize any safety concern.