User login
How NK cells kill abnormal blood cells

Credit: Bjorn Onfelt/Dan Davis
New research provides additional insight into how natural killer (NK) cells eliminate abnormal hematopoietic cells.
The investigators evaluated 2 molecules that are known to play important roles in this process.
Ewing’s sarcoma-associated transcript 2 (EAT-2) and signaling lymphocytic activation molecule (SLAM)–associated protein (SAP) are expressed in NK cells, and their combined expression is essential for NK cells to kill abnormal hematopoietic cells.
“We knew that EAT-2 cooperates with SAP, and, with this research project, we wanted to better understand why they are both required for the proper functioning of NK cells,” said study author André Veillette, PhD, of the Institut de Recherches Cliniques de Montréal (IRCM) in Canada.
Dr Veillette and his colleagues described this research in the Journal of Experimental Medicine.
“We identified the molecular chain of events that occur and showed that EAT-2 and SAP perform different functions using distinct mechanisms,” Dr Veillette said. “These findings explain the cooperative and essential function of these 2 molecules in activating NK cells, thereby allowing them to kill abnormal blood cells.”
The investigators noted that SAP couples SLAM family receptors to the protein tyrosine kinase Fyn and the exchange factor Vav, thereby promoting conjugate formation between NK cells and target hematopoietic cells.
EAT-2, on the other hand, works by accelerating the polarization and exocytosis of cytotoxic granules toward hematopoietic cells.
EAT-2 mediates its effects in NK cells by linking SLAM family receptors to phospholipase Cγ, calcium fluxes, and Erk kinase. These signals are triggered by 1 or 2 tyrosines that are located in the carboxyl-terminal tail of EAT-2.
Dr Veillete pointed out that, although EAT-2 and SAP behave differently, both are linked to receptors of the SLAM family on the cell surface.
“Because they can make better drug targets, our future work will focus on these receptors,” he said, “which could eventually lead to identifying new potential treatment avenues for blood cancers such as leukemia and lymphoma.” ![]()

Credit: Bjorn Onfelt/Dan Davis
New research provides additional insight into how natural killer (NK) cells eliminate abnormal hematopoietic cells.
The investigators evaluated 2 molecules that are known to play important roles in this process.
Ewing’s sarcoma-associated transcript 2 (EAT-2) and signaling lymphocytic activation molecule (SLAM)–associated protein (SAP) are expressed in NK cells, and their combined expression is essential for NK cells to kill abnormal hematopoietic cells.
“We knew that EAT-2 cooperates with SAP, and, with this research project, we wanted to better understand why they are both required for the proper functioning of NK cells,” said study author André Veillette, PhD, of the Institut de Recherches Cliniques de Montréal (IRCM) in Canada.
Dr Veillette and his colleagues described this research in the Journal of Experimental Medicine.
“We identified the molecular chain of events that occur and showed that EAT-2 and SAP perform different functions using distinct mechanisms,” Dr Veillette said. “These findings explain the cooperative and essential function of these 2 molecules in activating NK cells, thereby allowing them to kill abnormal blood cells.”
The investigators noted that SAP couples SLAM family receptors to the protein tyrosine kinase Fyn and the exchange factor Vav, thereby promoting conjugate formation between NK cells and target hematopoietic cells.
EAT-2, on the other hand, works by accelerating the polarization and exocytosis of cytotoxic granules toward hematopoietic cells.
EAT-2 mediates its effects in NK cells by linking SLAM family receptors to phospholipase Cγ, calcium fluxes, and Erk kinase. These signals are triggered by 1 or 2 tyrosines that are located in the carboxyl-terminal tail of EAT-2.
Dr Veillete pointed out that, although EAT-2 and SAP behave differently, both are linked to receptors of the SLAM family on the cell surface.
“Because they can make better drug targets, our future work will focus on these receptors,” he said, “which could eventually lead to identifying new potential treatment avenues for blood cancers such as leukemia and lymphoma.” ![]()

Credit: Bjorn Onfelt/Dan Davis
New research provides additional insight into how natural killer (NK) cells eliminate abnormal hematopoietic cells.
The investigators evaluated 2 molecules that are known to play important roles in this process.
Ewing’s sarcoma-associated transcript 2 (EAT-2) and signaling lymphocytic activation molecule (SLAM)–associated protein (SAP) are expressed in NK cells, and their combined expression is essential for NK cells to kill abnormal hematopoietic cells.
“We knew that EAT-2 cooperates with SAP, and, with this research project, we wanted to better understand why they are both required for the proper functioning of NK cells,” said study author André Veillette, PhD, of the Institut de Recherches Cliniques de Montréal (IRCM) in Canada.
Dr Veillette and his colleagues described this research in the Journal of Experimental Medicine.
“We identified the molecular chain of events that occur and showed that EAT-2 and SAP perform different functions using distinct mechanisms,” Dr Veillette said. “These findings explain the cooperative and essential function of these 2 molecules in activating NK cells, thereby allowing them to kill abnormal blood cells.”
The investigators noted that SAP couples SLAM family receptors to the protein tyrosine kinase Fyn and the exchange factor Vav, thereby promoting conjugate formation between NK cells and target hematopoietic cells.
EAT-2, on the other hand, works by accelerating the polarization and exocytosis of cytotoxic granules toward hematopoietic cells.
EAT-2 mediates its effects in NK cells by linking SLAM family receptors to phospholipase Cγ, calcium fluxes, and Erk kinase. These signals are triggered by 1 or 2 tyrosines that are located in the carboxyl-terminal tail of EAT-2.
Dr Veillete pointed out that, although EAT-2 and SAP behave differently, both are linked to receptors of the SLAM family on the cell surface.
“Because they can make better drug targets, our future work will focus on these receptors,” he said, “which could eventually lead to identifying new potential treatment avenues for blood cancers such as leukemia and lymphoma.” ![]()
Targeting sperm protection to combat malaria

Credit: CDC
New research has revealed a strategy for impairing fertility in malaria-carrying mosquitoes, potentially providing a tactic for combatting the disease.
Anopheles gambiae mosquitoes are the main transmitters of malaria, and the females mate only once during their lives.
They store the sperm from this single mating in an organ called the spermatheca, from which they repeatedly take sperm over the course of their lifetime to fertilize the eggs they lay.
The new research, published in PNAS, reveals that the sperm are partly protected by the actions of an enzyme called HPX15.
When researchers interfered with HPX15 in female A gambiae mosquitoes in the lab, the mosquitoes fertilized fewer eggs and, therefore, produced fewer offspring.
The team injected the female mosquitoes with an inhibitor to reduce the levels of HPX15. Normally, around 3% of the eggs a female lays do not develop into offspring. When the researchers reduced the levels of HPX15 in female mosquitoes, 20% of the mosquitoes’ eggs were infertile.
“[W]e reduced the number of offspring by a fifth, and that’s not a huge reduction,” said study author Flaminia Catteruccia, PhD, of the Harvard School of Public Health in Boston and the University of Perugia in Italy.
“But mosquitoes in the laboratory are subjected to much less stress than those in the wild, so we suspect that this kind of intervention would have a bigger impact on the fertility of wild mosquitoes. That’s something we would ultimately hope to investigate.”
The researchers also discovered how HPX15 is activated, suggesting another possible target for immobilizing the enzyme. The male mosquito transfers the hormone 20E to the female during mating, and it is this hormone that induces the expression of HPX15 in the female.
“The next step for this research is to think about how we could prevent activation of either the enzyme that protects the sperm, HPX15, or of the male trigger, 20E, that kicks that enzyme into action,” Dr Catteruccia said. “There may also be other pathways that we could target, and this is something that we’re keen to investigate.” ![]()

Credit: CDC
New research has revealed a strategy for impairing fertility in malaria-carrying mosquitoes, potentially providing a tactic for combatting the disease.
Anopheles gambiae mosquitoes are the main transmitters of malaria, and the females mate only once during their lives.
They store the sperm from this single mating in an organ called the spermatheca, from which they repeatedly take sperm over the course of their lifetime to fertilize the eggs they lay.
The new research, published in PNAS, reveals that the sperm are partly protected by the actions of an enzyme called HPX15.
When researchers interfered with HPX15 in female A gambiae mosquitoes in the lab, the mosquitoes fertilized fewer eggs and, therefore, produced fewer offspring.
The team injected the female mosquitoes with an inhibitor to reduce the levels of HPX15. Normally, around 3% of the eggs a female lays do not develop into offspring. When the researchers reduced the levels of HPX15 in female mosquitoes, 20% of the mosquitoes’ eggs were infertile.
“[W]e reduced the number of offspring by a fifth, and that’s not a huge reduction,” said study author Flaminia Catteruccia, PhD, of the Harvard School of Public Health in Boston and the University of Perugia in Italy.
“But mosquitoes in the laboratory are subjected to much less stress than those in the wild, so we suspect that this kind of intervention would have a bigger impact on the fertility of wild mosquitoes. That’s something we would ultimately hope to investigate.”
The researchers also discovered how HPX15 is activated, suggesting another possible target for immobilizing the enzyme. The male mosquito transfers the hormone 20E to the female during mating, and it is this hormone that induces the expression of HPX15 in the female.
“The next step for this research is to think about how we could prevent activation of either the enzyme that protects the sperm, HPX15, or of the male trigger, 20E, that kicks that enzyme into action,” Dr Catteruccia said. “There may also be other pathways that we could target, and this is something that we’re keen to investigate.” ![]()

Credit: CDC
New research has revealed a strategy for impairing fertility in malaria-carrying mosquitoes, potentially providing a tactic for combatting the disease.
Anopheles gambiae mosquitoes are the main transmitters of malaria, and the females mate only once during their lives.
They store the sperm from this single mating in an organ called the spermatheca, from which they repeatedly take sperm over the course of their lifetime to fertilize the eggs they lay.
The new research, published in PNAS, reveals that the sperm are partly protected by the actions of an enzyme called HPX15.
When researchers interfered with HPX15 in female A gambiae mosquitoes in the lab, the mosquitoes fertilized fewer eggs and, therefore, produced fewer offspring.
The team injected the female mosquitoes with an inhibitor to reduce the levels of HPX15. Normally, around 3% of the eggs a female lays do not develop into offspring. When the researchers reduced the levels of HPX15 in female mosquitoes, 20% of the mosquitoes’ eggs were infertile.
“[W]e reduced the number of offspring by a fifth, and that’s not a huge reduction,” said study author Flaminia Catteruccia, PhD, of the Harvard School of Public Health in Boston and the University of Perugia in Italy.
“But mosquitoes in the laboratory are subjected to much less stress than those in the wild, so we suspect that this kind of intervention would have a bigger impact on the fertility of wild mosquitoes. That’s something we would ultimately hope to investigate.”
The researchers also discovered how HPX15 is activated, suggesting another possible target for immobilizing the enzyme. The male mosquito transfers the hormone 20E to the female during mating, and it is this hormone that induces the expression of HPX15 in the female.
“The next step for this research is to think about how we could prevent activation of either the enzyme that protects the sperm, HPX15, or of the male trigger, 20E, that kicks that enzyme into action,” Dr Catteruccia said. “There may also be other pathways that we could target, and this is something that we’re keen to investigate.” ![]()
Team creates 3D model of malaria parasite genome

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have generated a 3D model of the human malaria parasite genome at 3 different stages in the parasite’s life cycle, according to a report in Genome Research.
The team said this model of Plasmodium falciparum is the first to be generated during the progression of a parasite’s life cycle.
“We successfully mapped all physical interactions between genetic elements in the parasite nucleus,” said study author Karine Le Roch, PhD, of the University of California, Riverside.
“To do so, we used a chromosome conformation capture method, followed by high-throughput sequencing technology—a recently developed methodology to analyze the organization of chromosomes in the natural state of the cell. We then used the maps of all physical interactions to generate a 3D model of the genome for each stage of the parasite life cycle analyzed.”
The model revealed that genes that need to be highly expressed in the malaria parasite—for example, genes involved in translation—tend to cluster in the same area of the cell nucleus, while genes that need to be tightly repressed—for example, genes involved in virulence—are found elsewhere in the 3D structure in a “repression center.”
The 3D structure had one major repression center. And the researchers found that virulence genes, which are all organized into that one repression center in a distinct area in the nucleus, seem to drive the full genome organization of the parasite.
“If we understand how the malaria parasite genome is organized in the nucleus and which components control this organization, we may be able to disrupt this architecture and disrupt, too, the parasite development,” Dr Le Roch said.
“We know that the genome architecture is critical in regulating gene expression and, more important, in regulating genes that are critical for parasite virulence. Now, we can more carefully search for components or drugs that can disrupt this organization, helping in the identification of new antimalaria strategies.”
Dr Le Roch’s lab is now looking at other stages of the malaria life cycle in order to identify components responsible for the 3D genome architecture.
“The importance of the genome architecture was initially thought to be critical for only higher eukaryotes,” she explained. “But we found, to our surprise, that the genome architecture is closely linked to virulence, even in the case of the malaria parasite.” ![]()

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have generated a 3D model of the human malaria parasite genome at 3 different stages in the parasite’s life cycle, according to a report in Genome Research.
The team said this model of Plasmodium falciparum is the first to be generated during the progression of a parasite’s life cycle.
“We successfully mapped all physical interactions between genetic elements in the parasite nucleus,” said study author Karine Le Roch, PhD, of the University of California, Riverside.
“To do so, we used a chromosome conformation capture method, followed by high-throughput sequencing technology—a recently developed methodology to analyze the organization of chromosomes in the natural state of the cell. We then used the maps of all physical interactions to generate a 3D model of the genome for each stage of the parasite life cycle analyzed.”
The model revealed that genes that need to be highly expressed in the malaria parasite—for example, genes involved in translation—tend to cluster in the same area of the cell nucleus, while genes that need to be tightly repressed—for example, genes involved in virulence—are found elsewhere in the 3D structure in a “repression center.”
The 3D structure had one major repression center. And the researchers found that virulence genes, which are all organized into that one repression center in a distinct area in the nucleus, seem to drive the full genome organization of the parasite.
“If we understand how the malaria parasite genome is organized in the nucleus and which components control this organization, we may be able to disrupt this architecture and disrupt, too, the parasite development,” Dr Le Roch said.
“We know that the genome architecture is critical in regulating gene expression and, more important, in regulating genes that are critical for parasite virulence. Now, we can more carefully search for components or drugs that can disrupt this organization, helping in the identification of new antimalaria strategies.”
Dr Le Roch’s lab is now looking at other stages of the malaria life cycle in order to identify components responsible for the 3D genome architecture.
“The importance of the genome architecture was initially thought to be critical for only higher eukaryotes,” she explained. “But we found, to our surprise, that the genome architecture is closely linked to virulence, even in the case of the malaria parasite.” ![]()

a red blood cell; Credit: St Jude
Children’s Research Hospital
Scientists have generated a 3D model of the human malaria parasite genome at 3 different stages in the parasite’s life cycle, according to a report in Genome Research.
The team said this model of Plasmodium falciparum is the first to be generated during the progression of a parasite’s life cycle.
“We successfully mapped all physical interactions between genetic elements in the parasite nucleus,” said study author Karine Le Roch, PhD, of the University of California, Riverside.
“To do so, we used a chromosome conformation capture method, followed by high-throughput sequencing technology—a recently developed methodology to analyze the organization of chromosomes in the natural state of the cell. We then used the maps of all physical interactions to generate a 3D model of the genome for each stage of the parasite life cycle analyzed.”
The model revealed that genes that need to be highly expressed in the malaria parasite—for example, genes involved in translation—tend to cluster in the same area of the cell nucleus, while genes that need to be tightly repressed—for example, genes involved in virulence—are found elsewhere in the 3D structure in a “repression center.”
The 3D structure had one major repression center. And the researchers found that virulence genes, which are all organized into that one repression center in a distinct area in the nucleus, seem to drive the full genome organization of the parasite.
“If we understand how the malaria parasite genome is organized in the nucleus and which components control this organization, we may be able to disrupt this architecture and disrupt, too, the parasite development,” Dr Le Roch said.
“We know that the genome architecture is critical in regulating gene expression and, more important, in regulating genes that are critical for parasite virulence. Now, we can more carefully search for components or drugs that can disrupt this organization, helping in the identification of new antimalaria strategies.”
Dr Le Roch’s lab is now looking at other stages of the malaria life cycle in order to identify components responsible for the 3D genome architecture.
“The importance of the genome architecture was initially thought to be critical for only higher eukaryotes,” she explained. “But we found, to our surprise, that the genome architecture is closely linked to virulence, even in the case of the malaria parasite.” ![]()
Good or bad, immune responses to cancer are similar

Credit: Aaron Logan
SAN DIEGO—Researchers have found evidence to suggest there may be little difference between an immune response that kills cancer cells and one that stimulates tumor growth.
The team set out to determine whether the immune responses that mediate cancer immunosurveillance and those responsible for inflammatory facilitation are qualitatively or quantitatively distinct.
They tested antibodies in mouse models of a few different cancers, including rituximab in Burkitt lymphoma.
And they found that lower antibody concentrations stimulated tumor growth, while higher concentrations inhibited growth, and the dose range was “surprisingly narrow.”
The researchers reported these findings in a paper published in PNAS and a poster presentation at the AACR Annual Meeting 2014 (abstract 1063).
“We have found that the intensity difference between an immune response that stimulates cancer and one that kills it may not be very much,” said principal investigator Ajit Varki, MD, of the University of California, San Diego School of Medicine.
“This may come as a surprise to researchers exploring two areas typically considered distinct: the role of the immune system in preventing and killing cancers and the role of chronic inflammation in stimulating cancers. As always, it turns out that the immune system is a double-edged sword.”
The concept of naturally occurring immunosurveillance against malignancies is not new, and there is compelling evidence for it. But understanding this process is confounded by the fact that some types of immune reaction promote tumor development.
Dr Varki and his colleagues looked specifically at a non-human sialic acid sugar molecule called Neu5Gc. Previous research showed that Neu5Gc accumulates in human tumors from dietary sources, despite an ongoing antibody response against it.
The researchers deployed antibodies against Neu5Gc in a mouse tumor model to determine whether and to what degree the antibodies altered tumor progression. The team found that low antibody doses stimulated growth, but high doses inhibited it.
The effect occurred over a “linear and remarkably narrow range,” according to Dr Varki, generating an immune response curve or “inverse hormesis.” Moreover, this curve could be shifted to the left or right simply by modifying the quality of the immune response.
The researchers uncovered similar findings in experiments with mouse models of colon and lung cancer, as well as when they used rituximab in a model of Burkitt lymphoma.
Dr Varki said these results could have implications for all aspects of cancer research. The immune response can have multiple roles in the genesis of cancers, in altering the progress of established tumors and in anticancer therapies that use antibodies as drugs.
Dr Varki is a co-founder of the company Sialix, Inc., which has licensed UC San Diego technologies related to anti-Neu5Gc antibodies in cancer. ![]()

Credit: Aaron Logan
SAN DIEGO—Researchers have found evidence to suggest there may be little difference between an immune response that kills cancer cells and one that stimulates tumor growth.
The team set out to determine whether the immune responses that mediate cancer immunosurveillance and those responsible for inflammatory facilitation are qualitatively or quantitatively distinct.
They tested antibodies in mouse models of a few different cancers, including rituximab in Burkitt lymphoma.
And they found that lower antibody concentrations stimulated tumor growth, while higher concentrations inhibited growth, and the dose range was “surprisingly narrow.”
The researchers reported these findings in a paper published in PNAS and a poster presentation at the AACR Annual Meeting 2014 (abstract 1063).
“We have found that the intensity difference between an immune response that stimulates cancer and one that kills it may not be very much,” said principal investigator Ajit Varki, MD, of the University of California, San Diego School of Medicine.
“This may come as a surprise to researchers exploring two areas typically considered distinct: the role of the immune system in preventing and killing cancers and the role of chronic inflammation in stimulating cancers. As always, it turns out that the immune system is a double-edged sword.”
The concept of naturally occurring immunosurveillance against malignancies is not new, and there is compelling evidence for it. But understanding this process is confounded by the fact that some types of immune reaction promote tumor development.
Dr Varki and his colleagues looked specifically at a non-human sialic acid sugar molecule called Neu5Gc. Previous research showed that Neu5Gc accumulates in human tumors from dietary sources, despite an ongoing antibody response against it.
The researchers deployed antibodies against Neu5Gc in a mouse tumor model to determine whether and to what degree the antibodies altered tumor progression. The team found that low antibody doses stimulated growth, but high doses inhibited it.
The effect occurred over a “linear and remarkably narrow range,” according to Dr Varki, generating an immune response curve or “inverse hormesis.” Moreover, this curve could be shifted to the left or right simply by modifying the quality of the immune response.
The researchers uncovered similar findings in experiments with mouse models of colon and lung cancer, as well as when they used rituximab in a model of Burkitt lymphoma.
Dr Varki said these results could have implications for all aspects of cancer research. The immune response can have multiple roles in the genesis of cancers, in altering the progress of established tumors and in anticancer therapies that use antibodies as drugs.
Dr Varki is a co-founder of the company Sialix, Inc., which has licensed UC San Diego technologies related to anti-Neu5Gc antibodies in cancer. ![]()

Credit: Aaron Logan
SAN DIEGO—Researchers have found evidence to suggest there may be little difference between an immune response that kills cancer cells and one that stimulates tumor growth.
The team set out to determine whether the immune responses that mediate cancer immunosurveillance and those responsible for inflammatory facilitation are qualitatively or quantitatively distinct.
They tested antibodies in mouse models of a few different cancers, including rituximab in Burkitt lymphoma.
And they found that lower antibody concentrations stimulated tumor growth, while higher concentrations inhibited growth, and the dose range was “surprisingly narrow.”
The researchers reported these findings in a paper published in PNAS and a poster presentation at the AACR Annual Meeting 2014 (abstract 1063).
“We have found that the intensity difference between an immune response that stimulates cancer and one that kills it may not be very much,” said principal investigator Ajit Varki, MD, of the University of California, San Diego School of Medicine.
“This may come as a surprise to researchers exploring two areas typically considered distinct: the role of the immune system in preventing and killing cancers and the role of chronic inflammation in stimulating cancers. As always, it turns out that the immune system is a double-edged sword.”
The concept of naturally occurring immunosurveillance against malignancies is not new, and there is compelling evidence for it. But understanding this process is confounded by the fact that some types of immune reaction promote tumor development.
Dr Varki and his colleagues looked specifically at a non-human sialic acid sugar molecule called Neu5Gc. Previous research showed that Neu5Gc accumulates in human tumors from dietary sources, despite an ongoing antibody response against it.
The researchers deployed antibodies against Neu5Gc in a mouse tumor model to determine whether and to what degree the antibodies altered tumor progression. The team found that low antibody doses stimulated growth, but high doses inhibited it.
The effect occurred over a “linear and remarkably narrow range,” according to Dr Varki, generating an immune response curve or “inverse hormesis.” Moreover, this curve could be shifted to the left or right simply by modifying the quality of the immune response.
The researchers uncovered similar findings in experiments with mouse models of colon and lung cancer, as well as when they used rituximab in a model of Burkitt lymphoma.
Dr Varki said these results could have implications for all aspects of cancer research. The immune response can have multiple roles in the genesis of cancers, in altering the progress of established tumors and in anticancer therapies that use antibodies as drugs.
Dr Varki is a co-founder of the company Sialix, Inc., which has licensed UC San Diego technologies related to anti-Neu5Gc antibodies in cancer. ![]()
How autophagy helps cancer cells evade death

Credit: Sarah Pfau
SAN DIEGO—New research suggests that autophagy may allow cancer cells to recover and divide, rather than die, when faced with chemotherapy.
“What we showed is that if this mechanism doesn’t work right—for example, if autophagy is too high or if the target regulated by autophagy isn’t around—cancer cells may be able to rescue themselves from death caused by chemotherapies,” said study author Andrew Thorburn, PhD, of the University of Colorado Denver.
He and his colleagues believe this finding has important implications. It demonstrates a mechanism whereby autophagy controls cell death, and it further reinforces the clinical potential of inhibiting autophagy to sensitize cancer cells to chemotherapy.
Dr Thorburn and his colleagues recounted their research in Cell Reports and presented it in an education session at the AACR Annual Meeting 2014.
The researchers had set out to examine how autophagy affects canonical death receptor-induced mitochondrial outer membrane permeabilization (MOMP) and apoptosis. They found that MOMP occurs at variable times in cells, and it’s delayed by autophagy.
Furthermore, autophagy leads to inefficient MOMP. This causes some cells to die via a slower process than typical apoptosis, which allows them to eventually recover and divide.
Specifically, the researchers found that, as a cancer cell begins to die, mitochondrial cell walls break down. And the cell’s mitochondria release proteins via MOMP.
But then, high autophagy allows the cell to encapsulate and “digest” these released proteins before MOMP can keep the cell well and truly dead. The cell recovers and goes on to divide.
“The implication here is that if you inhibit autophagy, you’d make this less likely to happen; ie, when you kill cancer cells, they would stay dead,” Dr Thorburn said.
He and his colleagues also found that autophagy depends on the target PUMA to regulate cell death. When PUMA is absent, it doesn’t matter if autophagy is inhibited. Without the communicating action of PUMA, cancer cells evade apoptosis and continue to survive.
The researchers said this suggests autophagy can control apoptosis via a regulator that makes MOMP faster and more efficient, thus ensuring the rapid completion of apoptosis.
“Autophagy is complex and, as yet, not fully understood,” Dr Thorburn said. “But now that we see a molecular mechanism whereby cell fate can be determined by autophagy, we hope to discover patient populations that could benefit from drugs that inhibit this action.” ![]()

Credit: Sarah Pfau
SAN DIEGO—New research suggests that autophagy may allow cancer cells to recover and divide, rather than die, when faced with chemotherapy.
“What we showed is that if this mechanism doesn’t work right—for example, if autophagy is too high or if the target regulated by autophagy isn’t around—cancer cells may be able to rescue themselves from death caused by chemotherapies,” said study author Andrew Thorburn, PhD, of the University of Colorado Denver.
He and his colleagues believe this finding has important implications. It demonstrates a mechanism whereby autophagy controls cell death, and it further reinforces the clinical potential of inhibiting autophagy to sensitize cancer cells to chemotherapy.
Dr Thorburn and his colleagues recounted their research in Cell Reports and presented it in an education session at the AACR Annual Meeting 2014.
The researchers had set out to examine how autophagy affects canonical death receptor-induced mitochondrial outer membrane permeabilization (MOMP) and apoptosis. They found that MOMP occurs at variable times in cells, and it’s delayed by autophagy.
Furthermore, autophagy leads to inefficient MOMP. This causes some cells to die via a slower process than typical apoptosis, which allows them to eventually recover and divide.
Specifically, the researchers found that, as a cancer cell begins to die, mitochondrial cell walls break down. And the cell’s mitochondria release proteins via MOMP.
But then, high autophagy allows the cell to encapsulate and “digest” these released proteins before MOMP can keep the cell well and truly dead. The cell recovers and goes on to divide.
“The implication here is that if you inhibit autophagy, you’d make this less likely to happen; ie, when you kill cancer cells, they would stay dead,” Dr Thorburn said.
He and his colleagues also found that autophagy depends on the target PUMA to regulate cell death. When PUMA is absent, it doesn’t matter if autophagy is inhibited. Without the communicating action of PUMA, cancer cells evade apoptosis and continue to survive.
The researchers said this suggests autophagy can control apoptosis via a regulator that makes MOMP faster and more efficient, thus ensuring the rapid completion of apoptosis.
“Autophagy is complex and, as yet, not fully understood,” Dr Thorburn said. “But now that we see a molecular mechanism whereby cell fate can be determined by autophagy, we hope to discover patient populations that could benefit from drugs that inhibit this action.” ![]()

Credit: Sarah Pfau
SAN DIEGO—New research suggests that autophagy may allow cancer cells to recover and divide, rather than die, when faced with chemotherapy.
“What we showed is that if this mechanism doesn’t work right—for example, if autophagy is too high or if the target regulated by autophagy isn’t around—cancer cells may be able to rescue themselves from death caused by chemotherapies,” said study author Andrew Thorburn, PhD, of the University of Colorado Denver.
He and his colleagues believe this finding has important implications. It demonstrates a mechanism whereby autophagy controls cell death, and it further reinforces the clinical potential of inhibiting autophagy to sensitize cancer cells to chemotherapy.
Dr Thorburn and his colleagues recounted their research in Cell Reports and presented it in an education session at the AACR Annual Meeting 2014.
The researchers had set out to examine how autophagy affects canonical death receptor-induced mitochondrial outer membrane permeabilization (MOMP) and apoptosis. They found that MOMP occurs at variable times in cells, and it’s delayed by autophagy.
Furthermore, autophagy leads to inefficient MOMP. This causes some cells to die via a slower process than typical apoptosis, which allows them to eventually recover and divide.
Specifically, the researchers found that, as a cancer cell begins to die, mitochondrial cell walls break down. And the cell’s mitochondria release proteins via MOMP.
But then, high autophagy allows the cell to encapsulate and “digest” these released proteins before MOMP can keep the cell well and truly dead. The cell recovers and goes on to divide.
“The implication here is that if you inhibit autophagy, you’d make this less likely to happen; ie, when you kill cancer cells, they would stay dead,” Dr Thorburn said.
He and his colleagues also found that autophagy depends on the target PUMA to regulate cell death. When PUMA is absent, it doesn’t matter if autophagy is inhibited. Without the communicating action of PUMA, cancer cells evade apoptosis and continue to survive.
The researchers said this suggests autophagy can control apoptosis via a regulator that makes MOMP faster and more efficient, thus ensuring the rapid completion of apoptosis.
“Autophagy is complex and, as yet, not fully understood,” Dr Thorburn said. “But now that we see a molecular mechanism whereby cell fate can be determined by autophagy, we hope to discover patient populations that could benefit from drugs that inhibit this action.” ![]()
Pharmacogenomic studies follow 90/10 rule

Credit: NIGMS
Few pharmacogenomic studies focus on orphan or tropical diseases prevalent in developing countries, according to research published in Global Public Health.
Researchers found that, from 1997 to 2010, pharmacogenomics research most commonly focused on cancers, depression or psychological disorders, and cardiovascular disease.
Less than 4% of publications dealt with orphan or infectious diseases.
According to the researchers, this suggests pharmacogenomic research follows the 90/10 rule.
“It is recognized that the distribution of technology and research follows the so-called 90/10 ratio rule; that is, 90% of global funding for health research, including the development drugs, is invested to treat 10% of the world’s population,” said study author Catherine Olivier, a PhD candidate at the University of Montreal’s School of Public Health.
This inequality between rich and poor countries led the United Nations (UN) to make the fight against HIV-AIDS, malaria, and neglected tropical diseases one of its 8 Millennium Development Goals, adopted in September 2000 by the 189 UN member states.
To verify the extent to which pharmacogenomic research has addressed orphan and tropical diseases, Olivier searched for pharmacogenomic studies published from 1997 to 2010. She identified 626 studies in 171 journals.
Each study was analyzed according to the type of disease it concerned, the origin of its authors, and their affiliation with pharmaceutical companies, if any.
“The information collected allowed us to draw a map showing current and historical trends in the development of pharmacogenomic research,” Olivier said.
She found that, from 1997 to 2003, there were 401 publications on pharmacogenomics in the PubMed database. And the majority of them (67%) were published in a single journal, Pharmacogenetics. Then, from 2003 to 2010, the number of studies doubled.
However, the apparent enthusiasm for this type of research seems to have been artificially inflated. Olivier noted that the percentage of nonoriginal publications, including reviews, meta-analyses, and debates, increased from 15% in 1997 to 51% in 2010.
“The number of original articles—that is, studies focusing on a new aspect of pharmacogenomics—began to decline after 2002,” Olivier said.
Moreover, during the period analyzed, nearly 23% of published studies in pharmacogenomics dealt with the area of oncology, followed by depression and psychological disorders (14.7%), and cardiovascular disorders (13.6%).
“Rare diseases, tropical infections, and maternal health, which should have benefited from pharmacogenomic research under the Millennium Development Goals, represented only 3.8% of published studies,” Olivier explained.
She noted that investigators from countries most likely to be interested in these areas of research conducted few studies on rare diseases and tropical infections.
“Of the 65 publications from BRICS countries—Brazil, Russia, India, China, and South Africa—only 2 concerned rare diseases and tropical infections,” Olivier said.
Yet these diseases represented nearly half (45.5%) of the main causes of mortality in underdeveloped countries, and 15% in developing countries, according to 2008 data issued by the UN.
“Unfortunately, our study indicates that we are far from fulfilling the promise to reduce health inequalities in the world,” Olivier said, “a promise which was made before the adoption of the Millennium Declaration.” ![]()

Credit: NIGMS
Few pharmacogenomic studies focus on orphan or tropical diseases prevalent in developing countries, according to research published in Global Public Health.
Researchers found that, from 1997 to 2010, pharmacogenomics research most commonly focused on cancers, depression or psychological disorders, and cardiovascular disease.
Less than 4% of publications dealt with orphan or infectious diseases.
According to the researchers, this suggests pharmacogenomic research follows the 90/10 rule.
“It is recognized that the distribution of technology and research follows the so-called 90/10 ratio rule; that is, 90% of global funding for health research, including the development drugs, is invested to treat 10% of the world’s population,” said study author Catherine Olivier, a PhD candidate at the University of Montreal’s School of Public Health.
This inequality between rich and poor countries led the United Nations (UN) to make the fight against HIV-AIDS, malaria, and neglected tropical diseases one of its 8 Millennium Development Goals, adopted in September 2000 by the 189 UN member states.
To verify the extent to which pharmacogenomic research has addressed orphan and tropical diseases, Olivier searched for pharmacogenomic studies published from 1997 to 2010. She identified 626 studies in 171 journals.
Each study was analyzed according to the type of disease it concerned, the origin of its authors, and their affiliation with pharmaceutical companies, if any.
“The information collected allowed us to draw a map showing current and historical trends in the development of pharmacogenomic research,” Olivier said.
She found that, from 1997 to 2003, there were 401 publications on pharmacogenomics in the PubMed database. And the majority of them (67%) were published in a single journal, Pharmacogenetics. Then, from 2003 to 2010, the number of studies doubled.
However, the apparent enthusiasm for this type of research seems to have been artificially inflated. Olivier noted that the percentage of nonoriginal publications, including reviews, meta-analyses, and debates, increased from 15% in 1997 to 51% in 2010.
“The number of original articles—that is, studies focusing on a new aspect of pharmacogenomics—began to decline after 2002,” Olivier said.
Moreover, during the period analyzed, nearly 23% of published studies in pharmacogenomics dealt with the area of oncology, followed by depression and psychological disorders (14.7%), and cardiovascular disorders (13.6%).
“Rare diseases, tropical infections, and maternal health, which should have benefited from pharmacogenomic research under the Millennium Development Goals, represented only 3.8% of published studies,” Olivier explained.
She noted that investigators from countries most likely to be interested in these areas of research conducted few studies on rare diseases and tropical infections.
“Of the 65 publications from BRICS countries—Brazil, Russia, India, China, and South Africa—only 2 concerned rare diseases and tropical infections,” Olivier said.
Yet these diseases represented nearly half (45.5%) of the main causes of mortality in underdeveloped countries, and 15% in developing countries, according to 2008 data issued by the UN.
“Unfortunately, our study indicates that we are far from fulfilling the promise to reduce health inequalities in the world,” Olivier said, “a promise which was made before the adoption of the Millennium Declaration.” ![]()

Credit: NIGMS
Few pharmacogenomic studies focus on orphan or tropical diseases prevalent in developing countries, according to research published in Global Public Health.
Researchers found that, from 1997 to 2010, pharmacogenomics research most commonly focused on cancers, depression or psychological disorders, and cardiovascular disease.
Less than 4% of publications dealt with orphan or infectious diseases.
According to the researchers, this suggests pharmacogenomic research follows the 90/10 rule.
“It is recognized that the distribution of technology and research follows the so-called 90/10 ratio rule; that is, 90% of global funding for health research, including the development drugs, is invested to treat 10% of the world’s population,” said study author Catherine Olivier, a PhD candidate at the University of Montreal’s School of Public Health.
This inequality between rich and poor countries led the United Nations (UN) to make the fight against HIV-AIDS, malaria, and neglected tropical diseases one of its 8 Millennium Development Goals, adopted in September 2000 by the 189 UN member states.
To verify the extent to which pharmacogenomic research has addressed orphan and tropical diseases, Olivier searched for pharmacogenomic studies published from 1997 to 2010. She identified 626 studies in 171 journals.
Each study was analyzed according to the type of disease it concerned, the origin of its authors, and their affiliation with pharmaceutical companies, if any.
“The information collected allowed us to draw a map showing current and historical trends in the development of pharmacogenomic research,” Olivier said.
She found that, from 1997 to 2003, there were 401 publications on pharmacogenomics in the PubMed database. And the majority of them (67%) were published in a single journal, Pharmacogenetics. Then, from 2003 to 2010, the number of studies doubled.
However, the apparent enthusiasm for this type of research seems to have been artificially inflated. Olivier noted that the percentage of nonoriginal publications, including reviews, meta-analyses, and debates, increased from 15% in 1997 to 51% in 2010.
“The number of original articles—that is, studies focusing on a new aspect of pharmacogenomics—began to decline after 2002,” Olivier said.
Moreover, during the period analyzed, nearly 23% of published studies in pharmacogenomics dealt with the area of oncology, followed by depression and psychological disorders (14.7%), and cardiovascular disorders (13.6%).
“Rare diseases, tropical infections, and maternal health, which should have benefited from pharmacogenomic research under the Millennium Development Goals, represented only 3.8% of published studies,” Olivier explained.
She noted that investigators from countries most likely to be interested in these areas of research conducted few studies on rare diseases and tropical infections.
“Of the 65 publications from BRICS countries—Brazil, Russia, India, China, and South Africa—only 2 concerned rare diseases and tropical infections,” Olivier said.
Yet these diseases represented nearly half (45.5%) of the main causes of mortality in underdeveloped countries, and 15% in developing countries, according to 2008 data issued by the UN.
“Unfortunately, our study indicates that we are far from fulfilling the promise to reduce health inequalities in the world,” Olivier said, “a promise which was made before the adoption of the Millennium Declaration.” ![]()
Nanocapsules prevent release of nontargeted radiation

Credit: Rhoda Baer
A novel type of nanocapsule can safely and effectively store isotopes that emit ionizing radiation, according to a paper published in Biochimica et Biophysica Acta.
In experiments, the nanocapsules were taken up by cells, accumulated near the perinuclear region, and persisted without degrading.
They also prevented nontargeted, radioactive daughter ions from escaping. These ions could cause significant damage if released, such as prompting the development of leukemia.
This research suggests the nanocapsules have the potential to advance radiation therapy, according to study author John M. Tomich, PhD, of Kansas State University’s Johnson Cancer Research Center.
Dr Tomich and his colleagues created the nanocapsules, called branched amphiphilic peptide capsules (BAPCs), by combining 2 related sequences of amino acids—bis(FLIVI)-K-KKKK and bis(FLIVIGSII)-K-KKKK.
“We found that the 2 sequences come together to form a thin membrane that assembled into little spheres, which we call capsules,” Dr Tomich said. “While other vesicles have been created from lipids, most are much less stable and break down. Ours are like stones, though. They’re incredibly stable and are not destroyed by cells in the body.”
The capsules’ ability to stay intact with the isotope inside and remain undetected by the body’s clearance systems prompted Dr Tomich to investigate using BAPCs for radiation therapies.
“The problem with current alpha-particle radiation therapies used to treat cancer is that they lead to the release of nontargeted, radioactive daughter ions into the body,” Dr Tomich said. “Radioactive atoms break down to form new atoms, called daughter ions, with the release of some form of energy or energetic particles. Alpha emitters give off an energetic particle that comes off at nearly the speed of light.”
The alpha particle destroys DNA and whatever vital cellular components are in its path. Similarly, the daughter ions recoil with high energy on ejection of the alpha particle. The daughter ions have enough energy to escape the targeting and containment molecules that are currently in use.
“Once freed, the daughter isotopes can end up in places you don’t want them, like bone marrow, which can then lead to leukemia and new challenges,” Dr Tomich said.
To see if the BAPCs could prevent the release of daughter isotopes, the researchers loaded the nanoparticles with 225Actinium. Upon decay, this compound releases 4 alpha particles and numerous daughter ions.
The team found that BAPCs loaded with the compound readily entered cells and migrated to a position alongside the nucleus.
As the alpha-particle-emitting isotopes decayed, the recoiled daughter ions collided with the capsule walls, essentially bouncing off them, and remained trapped inside the BAPCs. This completely blocked the release of the daughter ions, which prevented uptake in nontarget tissues.
Dr Tomich said more studies are needed to add target molecules to the surface of the BAPCs. But he believes the particles could provide a safer option for treating tumors with radiation therapy by reducing the amount of radioisotope required for killing cancer cells and reducing the side effects caused by off-target accumulation of radioisotopes.
“These capsules are easy to make and easy to work with,” Dr Tomich said. “I think we’re just scratching the surface of what we can do with them to improve human health and nanomaterials.” ![]()

Credit: Rhoda Baer
A novel type of nanocapsule can safely and effectively store isotopes that emit ionizing radiation, according to a paper published in Biochimica et Biophysica Acta.
In experiments, the nanocapsules were taken up by cells, accumulated near the perinuclear region, and persisted without degrading.
They also prevented nontargeted, radioactive daughter ions from escaping. These ions could cause significant damage if released, such as prompting the development of leukemia.
This research suggests the nanocapsules have the potential to advance radiation therapy, according to study author John M. Tomich, PhD, of Kansas State University’s Johnson Cancer Research Center.
Dr Tomich and his colleagues created the nanocapsules, called branched amphiphilic peptide capsules (BAPCs), by combining 2 related sequences of amino acids—bis(FLIVI)-K-KKKK and bis(FLIVIGSII)-K-KKKK.
“We found that the 2 sequences come together to form a thin membrane that assembled into little spheres, which we call capsules,” Dr Tomich said. “While other vesicles have been created from lipids, most are much less stable and break down. Ours are like stones, though. They’re incredibly stable and are not destroyed by cells in the body.”
The capsules’ ability to stay intact with the isotope inside and remain undetected by the body’s clearance systems prompted Dr Tomich to investigate using BAPCs for radiation therapies.
“The problem with current alpha-particle radiation therapies used to treat cancer is that they lead to the release of nontargeted, radioactive daughter ions into the body,” Dr Tomich said. “Radioactive atoms break down to form new atoms, called daughter ions, with the release of some form of energy or energetic particles. Alpha emitters give off an energetic particle that comes off at nearly the speed of light.”
The alpha particle destroys DNA and whatever vital cellular components are in its path. Similarly, the daughter ions recoil with high energy on ejection of the alpha particle. The daughter ions have enough energy to escape the targeting and containment molecules that are currently in use.
“Once freed, the daughter isotopes can end up in places you don’t want them, like bone marrow, which can then lead to leukemia and new challenges,” Dr Tomich said.
To see if the BAPCs could prevent the release of daughter isotopes, the researchers loaded the nanoparticles with 225Actinium. Upon decay, this compound releases 4 alpha particles and numerous daughter ions.
The team found that BAPCs loaded with the compound readily entered cells and migrated to a position alongside the nucleus.
As the alpha-particle-emitting isotopes decayed, the recoiled daughter ions collided with the capsule walls, essentially bouncing off them, and remained trapped inside the BAPCs. This completely blocked the release of the daughter ions, which prevented uptake in nontarget tissues.
Dr Tomich said more studies are needed to add target molecules to the surface of the BAPCs. But he believes the particles could provide a safer option for treating tumors with radiation therapy by reducing the amount of radioisotope required for killing cancer cells and reducing the side effects caused by off-target accumulation of radioisotopes.
“These capsules are easy to make and easy to work with,” Dr Tomich said. “I think we’re just scratching the surface of what we can do with them to improve human health and nanomaterials.” ![]()

Credit: Rhoda Baer
A novel type of nanocapsule can safely and effectively store isotopes that emit ionizing radiation, according to a paper published in Biochimica et Biophysica Acta.
In experiments, the nanocapsules were taken up by cells, accumulated near the perinuclear region, and persisted without degrading.
They also prevented nontargeted, radioactive daughter ions from escaping. These ions could cause significant damage if released, such as prompting the development of leukemia.
This research suggests the nanocapsules have the potential to advance radiation therapy, according to study author John M. Tomich, PhD, of Kansas State University’s Johnson Cancer Research Center.
Dr Tomich and his colleagues created the nanocapsules, called branched amphiphilic peptide capsules (BAPCs), by combining 2 related sequences of amino acids—bis(FLIVI)-K-KKKK and bis(FLIVIGSII)-K-KKKK.
“We found that the 2 sequences come together to form a thin membrane that assembled into little spheres, which we call capsules,” Dr Tomich said. “While other vesicles have been created from lipids, most are much less stable and break down. Ours are like stones, though. They’re incredibly stable and are not destroyed by cells in the body.”
The capsules’ ability to stay intact with the isotope inside and remain undetected by the body’s clearance systems prompted Dr Tomich to investigate using BAPCs for radiation therapies.
“The problem with current alpha-particle radiation therapies used to treat cancer is that they lead to the release of nontargeted, radioactive daughter ions into the body,” Dr Tomich said. “Radioactive atoms break down to form new atoms, called daughter ions, with the release of some form of energy or energetic particles. Alpha emitters give off an energetic particle that comes off at nearly the speed of light.”
The alpha particle destroys DNA and whatever vital cellular components are in its path. Similarly, the daughter ions recoil with high energy on ejection of the alpha particle. The daughter ions have enough energy to escape the targeting and containment molecules that are currently in use.
“Once freed, the daughter isotopes can end up in places you don’t want them, like bone marrow, which can then lead to leukemia and new challenges,” Dr Tomich said.
To see if the BAPCs could prevent the release of daughter isotopes, the researchers loaded the nanoparticles with 225Actinium. Upon decay, this compound releases 4 alpha particles and numerous daughter ions.
The team found that BAPCs loaded with the compound readily entered cells and migrated to a position alongside the nucleus.
As the alpha-particle-emitting isotopes decayed, the recoiled daughter ions collided with the capsule walls, essentially bouncing off them, and remained trapped inside the BAPCs. This completely blocked the release of the daughter ions, which prevented uptake in nontarget tissues.
Dr Tomich said more studies are needed to add target molecules to the surface of the BAPCs. But he believes the particles could provide a safer option for treating tumors with radiation therapy by reducing the amount of radioisotope required for killing cancer cells and reducing the side effects caused by off-target accumulation of radioisotopes.
“These capsules are easy to make and easy to work with,” Dr Tomich said. “I think we’re just scratching the surface of what we can do with them to improve human health and nanomaterials.”
Group uncovers inconsistent reporting of AEs, deaths

OHSU School of Medicine
A new analysis indicates that researchers sometimes exclude unfavorable trial data, whether reporting results in medical journals or on ClinicalTrials.gov.
A group of investigators analyzed trials in which data was reported both on the government website and in journals. And they found that discrepancies between the 2 sources were common.
Adverse events (AEs) were more likely to be reported on ClinicalTrials.gov and excluded from reports in journals.
But deaths seemed to be underreported or inconsistently reported on ClinicalTrials.gov when compared to journals.
“This is the most comprehensive study of ClinicalTrials.gov to date,” said study author Mark Helfand, MD, of Oregon Health & Science University.
“It shows that patients and clinicians could use [the site] to find information that is not available in the published literature, particularly to get more complete information about the harms of various treatment options. It also shows that, to best serve the public, death rates and some other items in ClinicalTrials.gov should be audited to keep them up to date.”
Dr Helfand and his colleagues reported these findings in Annals of Internal Medicine.
The researchers evaluated 110 trials that were completed by January 1, 2009, and reported on ClinicalTrials.gov. The team looked only at trials completed by 2009 to allow for the results to be later published in medical journals. Most of the trials were industry-sponsored.
Analyses revealed a number of discrepancies between data on ClinicalTrials.gov and in medical journals. For instance, 80% (n=88) of the trials had inconsistencies in secondary outcome measures.
In 15% (n=16) of trials, there were inconsistencies in the description of the primary outcome. And in 20% (n=22) of trials, there were inconsistencies in the primary outcome value. Still, in most cases, these discrepancies were small and did not affect the statistical significance of the results.
There were inconsistencies in AE reporting as well. Of the 84 trials in which a serious AE was reported on ClinicalTrials.gov, 11 published papers did not mention serious AEs, 5 reported that there were no serious AEs, and 21 reported a different
number of serious AEs.
So of the trials that had inconsistent AE reporting, 87% had more serious AEs listed on ClinicalTrials.gov than in the journal.
On the other hand, deaths seemed to be underreported on ClinicalTrials.gov compared to journals. For instance, in 17% of trials that did not report deaths on ClinicalTrials.gov, deaths were reported in the journal article.
Prior studies have indicated ClinicalTrials.gov does not have a uniform way of reporting deaths, and that may lead to inconsistencies.

OHSU School of Medicine
A new analysis indicates that researchers sometimes exclude unfavorable trial data, whether reporting results in medical journals or on ClinicalTrials.gov.
A group of investigators analyzed trials in which data was reported both on the government website and in journals. And they found that discrepancies between the 2 sources were common.
Adverse events (AEs) were more likely to be reported on ClinicalTrials.gov and excluded from reports in journals.
But deaths seemed to be underreported or inconsistently reported on ClinicalTrials.gov when compared to journals.
“This is the most comprehensive study of ClinicalTrials.gov to date,” said study author Mark Helfand, MD, of Oregon Health & Science University.
“It shows that patients and clinicians could use [the site] to find information that is not available in the published literature, particularly to get more complete information about the harms of various treatment options. It also shows that, to best serve the public, death rates and some other items in ClinicalTrials.gov should be audited to keep them up to date.”
Dr Helfand and his colleagues reported these findings in Annals of Internal Medicine.
The researchers evaluated 110 trials that were completed by January 1, 2009, and reported on ClinicalTrials.gov. The team looked only at trials completed by 2009 to allow for the results to be later published in medical journals. Most of the trials were industry-sponsored.
Analyses revealed a number of discrepancies between data on ClinicalTrials.gov and in medical journals. For instance, 80% (n=88) of the trials had inconsistencies in secondary outcome measures.
In 15% (n=16) of trials, there were inconsistencies in the description of the primary outcome. And in 20% (n=22) of trials, there were inconsistencies in the primary outcome value. Still, in most cases, these discrepancies were small and did not affect the statistical significance of the results.
There were inconsistencies in AE reporting as well. Of the 84 trials in which a serious AE was reported on ClinicalTrials.gov, 11 published papers did not mention serious AEs, 5 reported that there were no serious AEs, and 21 reported a different
number of serious AEs.
So of the trials that had inconsistent AE reporting, 87% had more serious AEs listed on ClinicalTrials.gov than in the journal.
On the other hand, deaths seemed to be underreported on ClinicalTrials.gov compared to journals. For instance, in 17% of trials that did not report deaths on ClinicalTrials.gov, deaths were reported in the journal article.
Prior studies have indicated ClinicalTrials.gov does not have a uniform way of reporting deaths, and that may lead to inconsistencies.

OHSU School of Medicine
A new analysis indicates that researchers sometimes exclude unfavorable trial data, whether reporting results in medical journals or on ClinicalTrials.gov.
A group of investigators analyzed trials in which data was reported both on the government website and in journals. And they found that discrepancies between the 2 sources were common.
Adverse events (AEs) were more likely to be reported on ClinicalTrials.gov and excluded from reports in journals.
But deaths seemed to be underreported or inconsistently reported on ClinicalTrials.gov when compared to journals.
“This is the most comprehensive study of ClinicalTrials.gov to date,” said study author Mark Helfand, MD, of Oregon Health & Science University.
“It shows that patients and clinicians could use [the site] to find information that is not available in the published literature, particularly to get more complete information about the harms of various treatment options. It also shows that, to best serve the public, death rates and some other items in ClinicalTrials.gov should be audited to keep them up to date.”
Dr Helfand and his colleagues reported these findings in Annals of Internal Medicine.
The researchers evaluated 110 trials that were completed by January 1, 2009, and reported on ClinicalTrials.gov. The team looked only at trials completed by 2009 to allow for the results to be later published in medical journals. Most of the trials were industry-sponsored.
Analyses revealed a number of discrepancies between data on ClinicalTrials.gov and in medical journals. For instance, 80% (n=88) of the trials had inconsistencies in secondary outcome measures.
In 15% (n=16) of trials, there were inconsistencies in the description of the primary outcome. And in 20% (n=22) of trials, there were inconsistencies in the primary outcome value. Still, in most cases, these discrepancies were small and did not affect the statistical significance of the results.
There were inconsistencies in AE reporting as well. Of the 84 trials in which a serious AE was reported on ClinicalTrials.gov, 11 published papers did not mention serious AEs, 5 reported that there were no serious AEs, and 21 reported a different
number of serious AEs.
So of the trials that had inconsistent AE reporting, 87% had more serious AEs listed on ClinicalTrials.gov than in the journal.
On the other hand, deaths seemed to be underreported on ClinicalTrials.gov compared to journals. For instance, in 17% of trials that did not report deaths on ClinicalTrials.gov, deaths were reported in the journal article.
Prior studies have indicated ClinicalTrials.gov does not have a uniform way of reporting deaths, and that may lead to inconsistencies.
Investigation suggests misconduct in STAP study
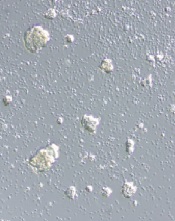
alleged STAP cells
Credit: Haruko Obokata
The Japanese research institute RIKEN has completed its investigation of the research on STAP cells (stimulus-triggered acquisition of pluripotency cells) and claims that study authors are guilty of misconduct and negligence.
According to RIKEN, lead study author Haruko Obokata, PhD, is guilty of misconduct because she manipulated study data.
And 2 other authors—Yoshiki Sasai, MD, PhD, and Teruhiko Wakayama, PhD—should have recognized the data manipulation.
Dr Obokata denied the allegations and said she plans to appeal this judgment. Drs Wakayama and Sasai wrote letters of apology, but Dr Sasai said he still believes the research is valid.
A group of RIKEN researchers has joined other groups in the attempt to replicate Dr Obokata’s experiments and determine if STAP cells can be generated.
About the investigation
RIKEN’s investigation began shortly after Dr Obokata and her colleagues reported the creation of STAP cells. The researchers said they could induce pluripotency by introducing somatic cells to a low-pH environment, and they described this discovery in an article and a letter to Nature.
Not long after the papers were published, members of the scientific community began questioning the validity of the research, citing issues with images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation into 6 issues with the papers. Early results of the investigation revealed 2 cases of “data mishandling” but no misconduct. And the final results of the investigation suggest 2 of the other issues were simply errors.
However, the 2 remaining issues constituted acts of misconduct, according to RIKEN. In the first case, Dr Obokata switched 1 lane of a diagram for another. She said it was for the purpose of image clarity, but the committee said this change was “intentionally misleading.”
Dr Obokata also used an image of a teratoma from her doctoral thesis (which involved different research). She said she mistakenly used the wrong image, but RIKEN said it was a fraudulent act because the caption on the image had been changed.
Next steps
It is still unclear whether the issues identified affect the results of this research. Dr Obokata and other study authors said the errors do not alter the study’s findings, and creating STAP cells is possible.
Other research groups have attempted to create STAP cells, and most results have suggested it is not possible when using the methods described in the Nature paper (or variations of those methods).
RIKEN has established an internal group that is attempting to verify the results of the STAP experiments. The group expects this process to take about a year.
As for dealing with the alleged misconduct, RIKEN said it will allow for an appeal. If the appeal is unsuccessful, the institution will call for the Nature papers to be retracted and take disciplinary action against Dr Obokata.
Drs Sasai and Wakayama may be subject to disciplinary measures as well. RIKEN said that, although the authors are not guilty of research misconduct, they “still bear heavy responsibility for their administrative negligence.”

alleged STAP cells
Credit: Haruko Obokata
The Japanese research institute RIKEN has completed its investigation of the research on STAP cells (stimulus-triggered acquisition of pluripotency cells) and claims that study authors are guilty of misconduct and negligence.
According to RIKEN, lead study author Haruko Obokata, PhD, is guilty of misconduct because she manipulated study data.
And 2 other authors—Yoshiki Sasai, MD, PhD, and Teruhiko Wakayama, PhD—should have recognized the data manipulation.
Dr Obokata denied the allegations and said she plans to appeal this judgment. Drs Wakayama and Sasai wrote letters of apology, but Dr Sasai said he still believes the research is valid.
A group of RIKEN researchers has joined other groups in the attempt to replicate Dr Obokata’s experiments and determine if STAP cells can be generated.
About the investigation
RIKEN’s investigation began shortly after Dr Obokata and her colleagues reported the creation of STAP cells. The researchers said they could induce pluripotency by introducing somatic cells to a low-pH environment, and they described this discovery in an article and a letter to Nature.
Not long after the papers were published, members of the scientific community began questioning the validity of the research, citing issues with images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation into 6 issues with the papers. Early results of the investigation revealed 2 cases of “data mishandling” but no misconduct. And the final results of the investigation suggest 2 of the other issues were simply errors.
However, the 2 remaining issues constituted acts of misconduct, according to RIKEN. In the first case, Dr Obokata switched 1 lane of a diagram for another. She said it was for the purpose of image clarity, but the committee said this change was “intentionally misleading.”
Dr Obokata also used an image of a teratoma from her doctoral thesis (which involved different research). She said she mistakenly used the wrong image, but RIKEN said it was a fraudulent act because the caption on the image had been changed.
Next steps
It is still unclear whether the issues identified affect the results of this research. Dr Obokata and other study authors said the errors do not alter the study’s findings, and creating STAP cells is possible.
Other research groups have attempted to create STAP cells, and most results have suggested it is not possible when using the methods described in the Nature paper (or variations of those methods).
RIKEN has established an internal group that is attempting to verify the results of the STAP experiments. The group expects this process to take about a year.
As for dealing with the alleged misconduct, RIKEN said it will allow for an appeal. If the appeal is unsuccessful, the institution will call for the Nature papers to be retracted and take disciplinary action against Dr Obokata.
Drs Sasai and Wakayama may be subject to disciplinary measures as well. RIKEN said that, although the authors are not guilty of research misconduct, they “still bear heavy responsibility for their administrative negligence.”

alleged STAP cells
Credit: Haruko Obokata
The Japanese research institute RIKEN has completed its investigation of the research on STAP cells (stimulus-triggered acquisition of pluripotency cells) and claims that study authors are guilty of misconduct and negligence.
According to RIKEN, lead study author Haruko Obokata, PhD, is guilty of misconduct because she manipulated study data.
And 2 other authors—Yoshiki Sasai, MD, PhD, and Teruhiko Wakayama, PhD—should have recognized the data manipulation.
Dr Obokata denied the allegations and said she plans to appeal this judgment. Drs Wakayama and Sasai wrote letters of apology, but Dr Sasai said he still believes the research is valid.
A group of RIKEN researchers has joined other groups in the attempt to replicate Dr Obokata’s experiments and determine if STAP cells can be generated.
About the investigation
RIKEN’s investigation began shortly after Dr Obokata and her colleagues reported the creation of STAP cells. The researchers said they could induce pluripotency by introducing somatic cells to a low-pH environment, and they described this discovery in an article and a letter to Nature.
Not long after the papers were published, members of the scientific community began questioning the validity of the research, citing issues with images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation into 6 issues with the papers. Early results of the investigation revealed 2 cases of “data mishandling” but no misconduct. And the final results of the investigation suggest 2 of the other issues were simply errors.
However, the 2 remaining issues constituted acts of misconduct, according to RIKEN. In the first case, Dr Obokata switched 1 lane of a diagram for another. She said it was for the purpose of image clarity, but the committee said this change was “intentionally misleading.”
Dr Obokata also used an image of a teratoma from her doctoral thesis (which involved different research). She said she mistakenly used the wrong image, but RIKEN said it was a fraudulent act because the caption on the image had been changed.
Next steps
It is still unclear whether the issues identified affect the results of this research. Dr Obokata and other study authors said the errors do not alter the study’s findings, and creating STAP cells is possible.
Other research groups have attempted to create STAP cells, and most results have suggested it is not possible when using the methods described in the Nature paper (or variations of those methods).
RIKEN has established an internal group that is attempting to verify the results of the STAP experiments. The group expects this process to take about a year.
As for dealing with the alleged misconduct, RIKEN said it will allow for an appeal. If the appeal is unsuccessful, the institution will call for the Nature papers to be retracted and take disciplinary action against Dr Obokata.
Drs Sasai and Wakayama may be subject to disciplinary measures as well. RIKEN said that, although the authors are not guilty of research misconduct, they “still bear heavy responsibility for their administrative negligence.”
Congress passes another SGR patch

Credit: NIH
The US House and Senate have passed a bill that will delay for another year the 24% cut in Medicare reimbursements that was set to take effect April 1 as part of the sustainable growth rate (SGR) formula.
Leaders from the House and Senate recently agreed on a plan to permanently replace the SGR, but they could not agree on a way to pay for it.
The current bill delays the cuts to Medicare reimbursements and extends other expiring healthcare provisions, such as higher payment rates for rural hospitals and ambulance rides in rural areas.
In total, this is expected to cost $21 billion. It will be paid for by cuts to healthcare providers, but half of the cuts won’t take effect for 10 years.
The SGR calls for annual, automatic cuts in Medicare payments to physicians, but these cuts have accumulated over the years. This marks the seventeenth temporary “patch” to the SGR.
For more details on this year’s fix, see the bill: Protecting Access to Medicare Act of 2014 (H.R. 4302).

Credit: NIH
The US House and Senate have passed a bill that will delay for another year the 24% cut in Medicare reimbursements that was set to take effect April 1 as part of the sustainable growth rate (SGR) formula.
Leaders from the House and Senate recently agreed on a plan to permanently replace the SGR, but they could not agree on a way to pay for it.
The current bill delays the cuts to Medicare reimbursements and extends other expiring healthcare provisions, such as higher payment rates for rural hospitals and ambulance rides in rural areas.
In total, this is expected to cost $21 billion. It will be paid for by cuts to healthcare providers, but half of the cuts won’t take effect for 10 years.
The SGR calls for annual, automatic cuts in Medicare payments to physicians, but these cuts have accumulated over the years. This marks the seventeenth temporary “patch” to the SGR.
For more details on this year’s fix, see the bill: Protecting Access to Medicare Act of 2014 (H.R. 4302).

Credit: NIH
The US House and Senate have passed a bill that will delay for another year the 24% cut in Medicare reimbursements that was set to take effect April 1 as part of the sustainable growth rate (SGR) formula.
Leaders from the House and Senate recently agreed on a plan to permanently replace the SGR, but they could not agree on a way to pay for it.
The current bill delays the cuts to Medicare reimbursements and extends other expiring healthcare provisions, such as higher payment rates for rural hospitals and ambulance rides in rural areas.
In total, this is expected to cost $21 billion. It will be paid for by cuts to healthcare providers, but half of the cuts won’t take effect for 10 years.
The SGR calls for annual, automatic cuts in Medicare payments to physicians, but these cuts have accumulated over the years. This marks the seventeenth temporary “patch” to the SGR.
For more details on this year’s fix, see the bill: Protecting Access to Medicare Act of 2014 (H.R. 4302).