User login
Video glasses can curb patient anxiety

Credit: CDC
SAN DIEGO—Watching videos through special glasses can calm patients undergoing a biopsy or other minimally invasive treatment, according to research presented at the Society of Interventional Radiology’s 39th Annual Scientific Meeting.
Researchers have explored strategies other than medication to reduce anxiety in these patients, including having patients listen to music or undergo hypnosis. But these methods have had modest benefits.
“Our study—the first of its kind for interventional radiology treatments—puts a spin on using modern technology to provide a safe, potentially cost-effective strategy of reducing anxiety, which can help and improve patient care,” said David L. Waldman, MD, PhD, of the University of Rochester Medical Center in New York.
“Whether they were watching a children’s movie or a nature show, patients wearing video glasses were successful at tuning out their surroundings. It’s an effective distraction technique that helps focus the individual’s attention away from the treatment.”
The study involved 49 patients (33 men and 16 women, ages 18-87) who were undergoing an outpatient interventional radiology treatment, such as a biopsy or placement of a catheter in the arm or chest to receive medication for treating cancer or infection.
Twenty-five of the patients donned video glasses prior to undergoing the treatment, and 24 did not. The video viewers chose from 20 videos, none of which were violent.
All of the patients filled out a standard 20-question test called the State-Trait Anxiety Inventory Form Y before and after the procedure to assess their level of anxiety.
Patients who wore the video glasses were 18.1% less anxious after the treatment than they were before, while those who didn’t wear video glasses were 7.5% less anxious afterward.
And the presence of the video glasses did not bother either the patient or the doctor, Dr Waldman said.
The glasses had no significant effect on blood pressure, heart rate, respiratory rate, pain, procedure time, or the amount of sedation or pain medication used.
“Patients told us the video glasses really helped calm them down and took their mind off the treatment, and we now offer video glasses to help distract patients from medical treatment going on mere inches away,” Dr Waldman said. “It is really comforting for patients, especially the ones who tend to be more nervous.
Dr Waldman and his colleagues presented these results at the meeting as abstract 126. ![]()

Credit: CDC
SAN DIEGO—Watching videos through special glasses can calm patients undergoing a biopsy or other minimally invasive treatment, according to research presented at the Society of Interventional Radiology’s 39th Annual Scientific Meeting.
Researchers have explored strategies other than medication to reduce anxiety in these patients, including having patients listen to music or undergo hypnosis. But these methods have had modest benefits.
“Our study—the first of its kind for interventional radiology treatments—puts a spin on using modern technology to provide a safe, potentially cost-effective strategy of reducing anxiety, which can help and improve patient care,” said David L. Waldman, MD, PhD, of the University of Rochester Medical Center in New York.
“Whether they were watching a children’s movie or a nature show, patients wearing video glasses were successful at tuning out their surroundings. It’s an effective distraction technique that helps focus the individual’s attention away from the treatment.”
The study involved 49 patients (33 men and 16 women, ages 18-87) who were undergoing an outpatient interventional radiology treatment, such as a biopsy or placement of a catheter in the arm or chest to receive medication for treating cancer or infection.
Twenty-five of the patients donned video glasses prior to undergoing the treatment, and 24 did not. The video viewers chose from 20 videos, none of which were violent.
All of the patients filled out a standard 20-question test called the State-Trait Anxiety Inventory Form Y before and after the procedure to assess their level of anxiety.
Patients who wore the video glasses were 18.1% less anxious after the treatment than they were before, while those who didn’t wear video glasses were 7.5% less anxious afterward.
And the presence of the video glasses did not bother either the patient or the doctor, Dr Waldman said.
The glasses had no significant effect on blood pressure, heart rate, respiratory rate, pain, procedure time, or the amount of sedation or pain medication used.
“Patients told us the video glasses really helped calm them down and took their mind off the treatment, and we now offer video glasses to help distract patients from medical treatment going on mere inches away,” Dr Waldman said. “It is really comforting for patients, especially the ones who tend to be more nervous.
Dr Waldman and his colleagues presented these results at the meeting as abstract 126. ![]()

Credit: CDC
SAN DIEGO—Watching videos through special glasses can calm patients undergoing a biopsy or other minimally invasive treatment, according to research presented at the Society of Interventional Radiology’s 39th Annual Scientific Meeting.
Researchers have explored strategies other than medication to reduce anxiety in these patients, including having patients listen to music or undergo hypnosis. But these methods have had modest benefits.
“Our study—the first of its kind for interventional radiology treatments—puts a spin on using modern technology to provide a safe, potentially cost-effective strategy of reducing anxiety, which can help and improve patient care,” said David L. Waldman, MD, PhD, of the University of Rochester Medical Center in New York.
“Whether they were watching a children’s movie or a nature show, patients wearing video glasses were successful at tuning out their surroundings. It’s an effective distraction technique that helps focus the individual’s attention away from the treatment.”
The study involved 49 patients (33 men and 16 women, ages 18-87) who were undergoing an outpatient interventional radiology treatment, such as a biopsy or placement of a catheter in the arm or chest to receive medication for treating cancer or infection.
Twenty-five of the patients donned video glasses prior to undergoing the treatment, and 24 did not. The video viewers chose from 20 videos, none of which were violent.
All of the patients filled out a standard 20-question test called the State-Trait Anxiety Inventory Form Y before and after the procedure to assess their level of anxiety.
Patients who wore the video glasses were 18.1% less anxious after the treatment than they were before, while those who didn’t wear video glasses were 7.5% less anxious afterward.
And the presence of the video glasses did not bother either the patient or the doctor, Dr Waldman said.
The glasses had no significant effect on blood pressure, heart rate, respiratory rate, pain, procedure time, or the amount of sedation or pain medication used.
“Patients told us the video glasses really helped calm them down and took their mind off the treatment, and we now offer video glasses to help distract patients from medical treatment going on mere inches away,” Dr Waldman said. “It is really comforting for patients, especially the ones who tend to be more nervous.
Dr Waldman and his colleagues presented these results at the meeting as abstract 126. ![]()
Some docs are wary of multiplex genomic testing

General Medical Sciences
Results of a survey suggest that some physicians aren’t ready to embrace multiplex genomic testing in cancer patients, while others plan to offer the testing to most of their patients.
The variation in attitudes is, in part, associated with physician confidence.
Physicians who reported having a lot of confidence in their ability to use and explain genomic findings were more likely to want to prescribe tests and consider using test results when making treatment recommendations.
But physicians with lower levels of confidence were more reluctant to offer such testing.
Researchers were surprised by these findings because the survey was carried out at the Dana-Farber/Brigham and Women’s Cancer Center, which has a research program that allows all consenting patients to undergo tests to reveal mutations and other DNA changes that drive their cancer.
The team was perplexed by another finding as well. Forty-two percent of responding oncologists approved of telling patients about test results even when their significance for the patient’s outlook and treatment is uncertain.
This issue comes with the growing use of multiplex testing, which can hunt for tens or hundreds of mutant genes simultaneously and often detects rare DNA variants that may or may not be relevant to an individual’s cancer.
“Some oncologists said we shouldn’t return these results to the patient, and others say, ‘Of course, we should give them to the patient,’” said study author Stacy W. Gray, MD, of the Dana-Farber Cancer Institute in Boston.
“The fact that we found so much variation in physicians’ confidence about their ability to use genetic data at a tertiary care National Cancer Institute-designated Comprehensive Cancer Center makes us pause and wonder about how confident physicians in the community are about dealing with this. It begs the question at a national level: how are we going to make sure that this technology for cancer care is adequately delivered?”
The survey was conducted in 2011 and early 2012 as a baseline assessment of physicians’ attitudes prior to the rollout of a tumor profiling project called Profile. The Profile technology platform includes complete DNA sequencing of more than 300 genomic regions to detect known and unknown cancer-related mutations.
The technology can also examine those regions for gains and losses of DNA sequences and rearrangements of DNA on chromosomes. The results are entered into a database for research purposes, but, if patients agree, the clinically important findings can also be returned to their doctor for use in the clinic.
A total of 160 cancer physicians—including medical oncologists, surgeons, and radiation oncologists—participated in the survey.
They were asked about their current use of somatic testing, their attitudes about multiplex testing, and their confidence in their ability to understand and use genomic data. The survey did not include a direct test of the physicians’ knowledge.
Respondents said they ordered tumor genomic testing on an average of 24% of patients. Twenty-two percent of the doctors reported low confidence in their knowledge about genomics.
Fourteen percent lacked knowledge in explaining these concepts to patients, and 26% doubted their ability to make treatment recommendations based on genomic data. Perhaps for these reasons, 18% of the physicians said they planned to use multiplex tumor testing infrequently.
Dr Gray and her colleagues concluded that there is “little consensus” on how physicians plan to use this technology for personalized cancer care and suggested the need for evidence-based guidelines to help doctors determine when testing is indicated.
The researchers reported their findings in the Journal of Clinical Oncology. A related editorial is available in the journal as well. ![]()

General Medical Sciences
Results of a survey suggest that some physicians aren’t ready to embrace multiplex genomic testing in cancer patients, while others plan to offer the testing to most of their patients.
The variation in attitudes is, in part, associated with physician confidence.
Physicians who reported having a lot of confidence in their ability to use and explain genomic findings were more likely to want to prescribe tests and consider using test results when making treatment recommendations.
But physicians with lower levels of confidence were more reluctant to offer such testing.
Researchers were surprised by these findings because the survey was carried out at the Dana-Farber/Brigham and Women’s Cancer Center, which has a research program that allows all consenting patients to undergo tests to reveal mutations and other DNA changes that drive their cancer.
The team was perplexed by another finding as well. Forty-two percent of responding oncologists approved of telling patients about test results even when their significance for the patient’s outlook and treatment is uncertain.
This issue comes with the growing use of multiplex testing, which can hunt for tens or hundreds of mutant genes simultaneously and often detects rare DNA variants that may or may not be relevant to an individual’s cancer.
“Some oncologists said we shouldn’t return these results to the patient, and others say, ‘Of course, we should give them to the patient,’” said study author Stacy W. Gray, MD, of the Dana-Farber Cancer Institute in Boston.
“The fact that we found so much variation in physicians’ confidence about their ability to use genetic data at a tertiary care National Cancer Institute-designated Comprehensive Cancer Center makes us pause and wonder about how confident physicians in the community are about dealing with this. It begs the question at a national level: how are we going to make sure that this technology for cancer care is adequately delivered?”
The survey was conducted in 2011 and early 2012 as a baseline assessment of physicians’ attitudes prior to the rollout of a tumor profiling project called Profile. The Profile technology platform includes complete DNA sequencing of more than 300 genomic regions to detect known and unknown cancer-related mutations.
The technology can also examine those regions for gains and losses of DNA sequences and rearrangements of DNA on chromosomes. The results are entered into a database for research purposes, but, if patients agree, the clinically important findings can also be returned to their doctor for use in the clinic.
A total of 160 cancer physicians—including medical oncologists, surgeons, and radiation oncologists—participated in the survey.
They were asked about their current use of somatic testing, their attitudes about multiplex testing, and their confidence in their ability to understand and use genomic data. The survey did not include a direct test of the physicians’ knowledge.
Respondents said they ordered tumor genomic testing on an average of 24% of patients. Twenty-two percent of the doctors reported low confidence in their knowledge about genomics.
Fourteen percent lacked knowledge in explaining these concepts to patients, and 26% doubted their ability to make treatment recommendations based on genomic data. Perhaps for these reasons, 18% of the physicians said they planned to use multiplex tumor testing infrequently.
Dr Gray and her colleagues concluded that there is “little consensus” on how physicians plan to use this technology for personalized cancer care and suggested the need for evidence-based guidelines to help doctors determine when testing is indicated.
The researchers reported their findings in the Journal of Clinical Oncology. A related editorial is available in the journal as well. ![]()

General Medical Sciences
Results of a survey suggest that some physicians aren’t ready to embrace multiplex genomic testing in cancer patients, while others plan to offer the testing to most of their patients.
The variation in attitudes is, in part, associated with physician confidence.
Physicians who reported having a lot of confidence in their ability to use and explain genomic findings were more likely to want to prescribe tests and consider using test results when making treatment recommendations.
But physicians with lower levels of confidence were more reluctant to offer such testing.
Researchers were surprised by these findings because the survey was carried out at the Dana-Farber/Brigham and Women’s Cancer Center, which has a research program that allows all consenting patients to undergo tests to reveal mutations and other DNA changes that drive their cancer.
The team was perplexed by another finding as well. Forty-two percent of responding oncologists approved of telling patients about test results even when their significance for the patient’s outlook and treatment is uncertain.
This issue comes with the growing use of multiplex testing, which can hunt for tens or hundreds of mutant genes simultaneously and often detects rare DNA variants that may or may not be relevant to an individual’s cancer.
“Some oncologists said we shouldn’t return these results to the patient, and others say, ‘Of course, we should give them to the patient,’” said study author Stacy W. Gray, MD, of the Dana-Farber Cancer Institute in Boston.
“The fact that we found so much variation in physicians’ confidence about their ability to use genetic data at a tertiary care National Cancer Institute-designated Comprehensive Cancer Center makes us pause and wonder about how confident physicians in the community are about dealing with this. It begs the question at a national level: how are we going to make sure that this technology for cancer care is adequately delivered?”
The survey was conducted in 2011 and early 2012 as a baseline assessment of physicians’ attitudes prior to the rollout of a tumor profiling project called Profile. The Profile technology platform includes complete DNA sequencing of more than 300 genomic regions to detect known and unknown cancer-related mutations.
The technology can also examine those regions for gains and losses of DNA sequences and rearrangements of DNA on chromosomes. The results are entered into a database for research purposes, but, if patients agree, the clinically important findings can also be returned to their doctor for use in the clinic.
A total of 160 cancer physicians—including medical oncologists, surgeons, and radiation oncologists—participated in the survey.
They were asked about their current use of somatic testing, their attitudes about multiplex testing, and their confidence in their ability to understand and use genomic data. The survey did not include a direct test of the physicians’ knowledge.
Respondents said they ordered tumor genomic testing on an average of 24% of patients. Twenty-two percent of the doctors reported low confidence in their knowledge about genomics.
Fourteen percent lacked knowledge in explaining these concepts to patients, and 26% doubted their ability to make treatment recommendations based on genomic data. Perhaps for these reasons, 18% of the physicians said they planned to use multiplex tumor testing infrequently.
Dr Gray and her colleagues concluded that there is “little consensus” on how physicians plan to use this technology for personalized cancer care and suggested the need for evidence-based guidelines to help doctors determine when testing is indicated.
The researchers reported their findings in the Journal of Clinical Oncology. A related editorial is available in the journal as well. ![]()
Proteins appear necessary for stem cell formation
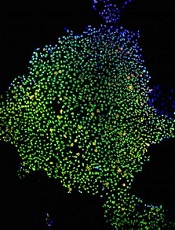
Credit: James Thomson
Proteins that regulate energy metabolism are essential for stem cell formation, according to a study published in Cell Stem Cell.
The researchers showed that hypoxia-induced factor 1α and 2α (HIF1α and HIF2α)—2 proteins that control how cells metabolize glucose—play a key role in the formation of stem cells.
The findings may advance our understanding of stem cell development, but they also suggest the proteins might be targets for new cancer therapies.
Julie Mathieu, PhD, of the University of Washington in Seattle, and her colleagues conducted this research, creating induced pluripotent stem cells (iPSCs) by reprogramming mature human tissue fibroblasts.
During reprogramming, the cells must go through a stage in which they shut down the metabolic pathway they use to generate energy from glucose that requires the presence of oxygen in mitochondria. The cells shift over to the glycolytic pathway, which generates less energy but does not require the presence of oxygen.
This shift may take place because in nature, embryonic and tissue stem cells often must survive in hypoxic conditions. This transition to a glycolytic state is of particular interest to cancer researchers because, as normal cells are transformed into cancer cells, they too go through a glycolytic phase.
For their study, Dr Mathieu and her colleagues focused on the function of HIF1α and HIF2α in this process. The researchers showed that each protein is required for iPSC generation.
To tease out the impact of HIF1α and 2α on cellular processes in more detail, the team stabilized the proteins in an active form and tested what each protein could do alone.
They found that when HIF1α was stabilized, the cells went into the glycolytic state and produced more iPSCs than normal. However, when the researchers activated HIF2α, the cells failed to develop into stem cells.
“This was a big surprise,” Dr Mathieu said. “These proteins are very similar, but HIF1α gives you lots of stem cells [and] HIF2α, none.”
If stabilized together, HIF2α won the battle, repressing all stem cell formation.
Further investigation revealed that HIF2α does indeed promote the shift to glycolysis in an early stage of the cells’ reprogramming. But if it persists too long, it has the opposite effect, blocking the progression to the stem cell state.
“HIF2α is like Darth Vader, originally a Jedi who falls to the dark side,” said study author Hannele Ruohola-Baker, PhD, also of the University of Washington.
“While HIF1α, the good guy, is beneficial for reprogramming throughout the process, HIF2α, if not eliminated, turns bad in the middle and represses pluripotency.”
HIF2α does this, in part, by upregulating production of the protein TRAIL, which is known to, among other things, induce apoptosis.
These findings suggest there may be proteins of other families that are playing alternating “good guy/bad guy” roles during stem cell development, according to study author Wenyu Zhou, PhD, of Stanford University in California.
“It is very intriguing that HIF2α has the capacity to both promote and repress pluripotency, doing so at different stages in a cellular reprogramming process,” she said.
The findings have implications for stem cell research, Dr Mathieu said. First, they indicate that it may be possible to use HIF1α to greatly increase the number of stem cells in a culture.
And second, they suggest it may be possible to induce stem cell formation with HIF proteins alone or in combination with other stimulating factors without inserting genes at the start of the reprogramming process.
But the findings may also have implications for cancer research. Both HIF1α and 2α are known to play a role in normal cells’ transformation to cancer stem cells. And the presence of activated HIF1α is known to be a marker for aggressive disease.
So the researchers believe it might be possible to interfere with cancer development by either blocking the effect of HIF1α in malignant cells early in the process or stimulating the effect of HIF2 at a later stage. ![]()

Credit: James Thomson
Proteins that regulate energy metabolism are essential for stem cell formation, according to a study published in Cell Stem Cell.
The researchers showed that hypoxia-induced factor 1α and 2α (HIF1α and HIF2α)—2 proteins that control how cells metabolize glucose—play a key role in the formation of stem cells.
The findings may advance our understanding of stem cell development, but they also suggest the proteins might be targets for new cancer therapies.
Julie Mathieu, PhD, of the University of Washington in Seattle, and her colleagues conducted this research, creating induced pluripotent stem cells (iPSCs) by reprogramming mature human tissue fibroblasts.
During reprogramming, the cells must go through a stage in which they shut down the metabolic pathway they use to generate energy from glucose that requires the presence of oxygen in mitochondria. The cells shift over to the glycolytic pathway, which generates less energy but does not require the presence of oxygen.
This shift may take place because in nature, embryonic and tissue stem cells often must survive in hypoxic conditions. This transition to a glycolytic state is of particular interest to cancer researchers because, as normal cells are transformed into cancer cells, they too go through a glycolytic phase.
For their study, Dr Mathieu and her colleagues focused on the function of HIF1α and HIF2α in this process. The researchers showed that each protein is required for iPSC generation.
To tease out the impact of HIF1α and 2α on cellular processes in more detail, the team stabilized the proteins in an active form and tested what each protein could do alone.
They found that when HIF1α was stabilized, the cells went into the glycolytic state and produced more iPSCs than normal. However, when the researchers activated HIF2α, the cells failed to develop into stem cells.
“This was a big surprise,” Dr Mathieu said. “These proteins are very similar, but HIF1α gives you lots of stem cells [and] HIF2α, none.”
If stabilized together, HIF2α won the battle, repressing all stem cell formation.
Further investigation revealed that HIF2α does indeed promote the shift to glycolysis in an early stage of the cells’ reprogramming. But if it persists too long, it has the opposite effect, blocking the progression to the stem cell state.
“HIF2α is like Darth Vader, originally a Jedi who falls to the dark side,” said study author Hannele Ruohola-Baker, PhD, also of the University of Washington.
“While HIF1α, the good guy, is beneficial for reprogramming throughout the process, HIF2α, if not eliminated, turns bad in the middle and represses pluripotency.”
HIF2α does this, in part, by upregulating production of the protein TRAIL, which is known to, among other things, induce apoptosis.
These findings suggest there may be proteins of other families that are playing alternating “good guy/bad guy” roles during stem cell development, according to study author Wenyu Zhou, PhD, of Stanford University in California.
“It is very intriguing that HIF2α has the capacity to both promote and repress pluripotency, doing so at different stages in a cellular reprogramming process,” she said.
The findings have implications for stem cell research, Dr Mathieu said. First, they indicate that it may be possible to use HIF1α to greatly increase the number of stem cells in a culture.
And second, they suggest it may be possible to induce stem cell formation with HIF proteins alone or in combination with other stimulating factors without inserting genes at the start of the reprogramming process.
But the findings may also have implications for cancer research. Both HIF1α and 2α are known to play a role in normal cells’ transformation to cancer stem cells. And the presence of activated HIF1α is known to be a marker for aggressive disease.
So the researchers believe it might be possible to interfere with cancer development by either blocking the effect of HIF1α in malignant cells early in the process or stimulating the effect of HIF2 at a later stage. ![]()

Credit: James Thomson
Proteins that regulate energy metabolism are essential for stem cell formation, according to a study published in Cell Stem Cell.
The researchers showed that hypoxia-induced factor 1α and 2α (HIF1α and HIF2α)—2 proteins that control how cells metabolize glucose—play a key role in the formation of stem cells.
The findings may advance our understanding of stem cell development, but they also suggest the proteins might be targets for new cancer therapies.
Julie Mathieu, PhD, of the University of Washington in Seattle, and her colleagues conducted this research, creating induced pluripotent stem cells (iPSCs) by reprogramming mature human tissue fibroblasts.
During reprogramming, the cells must go through a stage in which they shut down the metabolic pathway they use to generate energy from glucose that requires the presence of oxygen in mitochondria. The cells shift over to the glycolytic pathway, which generates less energy but does not require the presence of oxygen.
This shift may take place because in nature, embryonic and tissue stem cells often must survive in hypoxic conditions. This transition to a glycolytic state is of particular interest to cancer researchers because, as normal cells are transformed into cancer cells, they too go through a glycolytic phase.
For their study, Dr Mathieu and her colleagues focused on the function of HIF1α and HIF2α in this process. The researchers showed that each protein is required for iPSC generation.
To tease out the impact of HIF1α and 2α on cellular processes in more detail, the team stabilized the proteins in an active form and tested what each protein could do alone.
They found that when HIF1α was stabilized, the cells went into the glycolytic state and produced more iPSCs than normal. However, when the researchers activated HIF2α, the cells failed to develop into stem cells.
“This was a big surprise,” Dr Mathieu said. “These proteins are very similar, but HIF1α gives you lots of stem cells [and] HIF2α, none.”
If stabilized together, HIF2α won the battle, repressing all stem cell formation.
Further investigation revealed that HIF2α does indeed promote the shift to glycolysis in an early stage of the cells’ reprogramming. But if it persists too long, it has the opposite effect, blocking the progression to the stem cell state.
“HIF2α is like Darth Vader, originally a Jedi who falls to the dark side,” said study author Hannele Ruohola-Baker, PhD, also of the University of Washington.
“While HIF1α, the good guy, is beneficial for reprogramming throughout the process, HIF2α, if not eliminated, turns bad in the middle and represses pluripotency.”
HIF2α does this, in part, by upregulating production of the protein TRAIL, which is known to, among other things, induce apoptosis.
These findings suggest there may be proteins of other families that are playing alternating “good guy/bad guy” roles during stem cell development, according to study author Wenyu Zhou, PhD, of Stanford University in California.
“It is very intriguing that HIF2α has the capacity to both promote and repress pluripotency, doing so at different stages in a cellular reprogramming process,” she said.
The findings have implications for stem cell research, Dr Mathieu said. First, they indicate that it may be possible to use HIF1α to greatly increase the number of stem cells in a culture.
And second, they suggest it may be possible to induce stem cell formation with HIF proteins alone or in combination with other stimulating factors without inserting genes at the start of the reprogramming process.
But the findings may also have implications for cancer research. Both HIF1α and 2α are known to play a role in normal cells’ transformation to cancer stem cells. And the presence of activated HIF1α is known to be a marker for aggressive disease.
So the researchers believe it might be possible to interfere with cancer development by either blocking the effect of HIF1α in malignant cells early in the process or stimulating the effect of HIF2 at a later stage. ![]()
Finger prick yields ample iPSCs for banking

Credit: Salk Institute
Researchers say they’ve discovered an easy way to collect large quantities of viable, bankable stem cells.
Donors prick their own fingers to provide a single drop of blood, and the team generates induced pluripotent stem cells (iPSCs) from that sample.
“We show that a single drop of blood from a finger-prick sample is sufficient for performing cellular reprogramming, DNA sequencing, and blood typing in parallel,” said Jonathan Yuin-Han Loh, PhD, of the Agency for Science, Technology and Research (A*STAR) in Singapore.
“Our strategy has the potential of facilitating the development of large-scale human iPSC banking worldwide.”
The researchers described this strategy in STEM CELLS Translational Medicine.
“We gradually reduced the starting volume of blood (collected using a needle) and confirmed that reprogramming can be achieved with as little as 0.25 milliliters,” said Hong Kee Tan, a research officer in the Loh lab.
And this made the team wonder whether a do-it-yourself approach to blood collection might work too.
“To test this idea, we asked donors to prick their own fingers in a normal room environment and collect a single drop of blood sample into a tube,” Tan said. “The tube was placed on ice and delivered to the lab for reprogramming.”
The cells were treated with a buffer at 12-, 24- or 48-hour increments and observed under the microscope for viability and signs of contamination. After 12 days of expansion in medium, the cells appeared healthy and were actively dividing.
The researchers then succeeded in forcing the cells to become mesodermal, endodermal, and neural cells. They were also able to produce cells that gave rise to rhythmically beating cardiomyocytes.
The team said there was no noticeable reduction in reprogramming efficiency between the freshly collected finger-prick samples and the do-it-yourself samples.
“[W]e derived healthy iPSCs from tiny volumes of venipuncture and a single drop from finger-prick blood samples,” Dr Loh said. “We also report a high reprogramming yield of 100 to 600 colonies per milliliter of blood.” ![]()

Credit: Salk Institute
Researchers say they’ve discovered an easy way to collect large quantities of viable, bankable stem cells.
Donors prick their own fingers to provide a single drop of blood, and the team generates induced pluripotent stem cells (iPSCs) from that sample.
“We show that a single drop of blood from a finger-prick sample is sufficient for performing cellular reprogramming, DNA sequencing, and blood typing in parallel,” said Jonathan Yuin-Han Loh, PhD, of the Agency for Science, Technology and Research (A*STAR) in Singapore.
“Our strategy has the potential of facilitating the development of large-scale human iPSC banking worldwide.”
The researchers described this strategy in STEM CELLS Translational Medicine.
“We gradually reduced the starting volume of blood (collected using a needle) and confirmed that reprogramming can be achieved with as little as 0.25 milliliters,” said Hong Kee Tan, a research officer in the Loh lab.
And this made the team wonder whether a do-it-yourself approach to blood collection might work too.
“To test this idea, we asked donors to prick their own fingers in a normal room environment and collect a single drop of blood sample into a tube,” Tan said. “The tube was placed on ice and delivered to the lab for reprogramming.”
The cells were treated with a buffer at 12-, 24- or 48-hour increments and observed under the microscope for viability and signs of contamination. After 12 days of expansion in medium, the cells appeared healthy and were actively dividing.
The researchers then succeeded in forcing the cells to become mesodermal, endodermal, and neural cells. They were also able to produce cells that gave rise to rhythmically beating cardiomyocytes.
The team said there was no noticeable reduction in reprogramming efficiency between the freshly collected finger-prick samples and the do-it-yourself samples.
“[W]e derived healthy iPSCs from tiny volumes of venipuncture and a single drop from finger-prick blood samples,” Dr Loh said. “We also report a high reprogramming yield of 100 to 600 colonies per milliliter of blood.” ![]()

Credit: Salk Institute
Researchers say they’ve discovered an easy way to collect large quantities of viable, bankable stem cells.
Donors prick their own fingers to provide a single drop of blood, and the team generates induced pluripotent stem cells (iPSCs) from that sample.
“We show that a single drop of blood from a finger-prick sample is sufficient for performing cellular reprogramming, DNA sequencing, and blood typing in parallel,” said Jonathan Yuin-Han Loh, PhD, of the Agency for Science, Technology and Research (A*STAR) in Singapore.
“Our strategy has the potential of facilitating the development of large-scale human iPSC banking worldwide.”
The researchers described this strategy in STEM CELLS Translational Medicine.
“We gradually reduced the starting volume of blood (collected using a needle) and confirmed that reprogramming can be achieved with as little as 0.25 milliliters,” said Hong Kee Tan, a research officer in the Loh lab.
And this made the team wonder whether a do-it-yourself approach to blood collection might work too.
“To test this idea, we asked donors to prick their own fingers in a normal room environment and collect a single drop of blood sample into a tube,” Tan said. “The tube was placed on ice and delivered to the lab for reprogramming.”
The cells were treated with a buffer at 12-, 24- or 48-hour increments and observed under the microscope for viability and signs of contamination. After 12 days of expansion in medium, the cells appeared healthy and were actively dividing.
The researchers then succeeded in forcing the cells to become mesodermal, endodermal, and neural cells. They were also able to produce cells that gave rise to rhythmically beating cardiomyocytes.
The team said there was no noticeable reduction in reprogramming efficiency between the freshly collected finger-prick samples and the do-it-yourself samples.
“[W]e derived healthy iPSCs from tiny volumes of venipuncture and a single drop from finger-prick blood samples,” Dr Loh said. “We also report a high reprogramming yield of 100 to 600 colonies per milliliter of blood.” ![]()
Bloodstream infections treated ‘inappropriately’
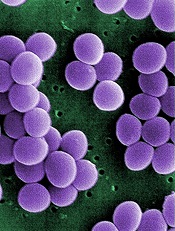
Credit: Janice Haney Carr
An analysis of 9 community hospitals showed that 1 in 3 patients with bloodstream infections received inappropriate therapy.
The study also revealed growing resistance to treatment and a high prevalence of Staphylococcus aureus bacteria in these hospitals.
Investigators said the findings, published in PLOS ONE, provide the most comprehensive look at bloodstream infections in community hospitals to date.
Much of the existing research on bloodstream infections focuses on tertiary care centers.
“Our study provides a much-needed update on what we’re seeing in community hospitals, and ultimately, we’re finding similar types of infections in these hospitals as in tertiary care centers,” said study author Deverick Anderson, MD, of Duke University in Durham North Carolina.
“It’s a challenge to identify bloodstream infections and treat them quickly and appropriately, but this study shows that there is room for improvement in both kinds of hospital settings.”
Types of infection
To better understand the types of bloodstream infections found in community hospitals, Dr Anderson and his colleagues collected information on patients treated at these hospitals in Virginia and North Carolina from 2003 to 2006.
The investigators focused on 1470 patients diagnosed with bloodstream infections. The infections were classified depending on where and when they were contracted.
Infections resulting from prior hospitalization, surgery, invasive devices (such as catheters), or living in long-term care facilities were designated healthcare-associated infections.
Community-acquired infections were contracted outside of medical settings or shortly after being admitted to a hospital. And hospital-onset infections occurred after being in a hospital for several days.
The investigators found that 56% of bloodstream infections were healthcare-associated, but symptoms began prior to hospital admission. Community-acquired infections unrelated to medical care were seen in 29% of patients. And 15% had hospital-onset healthcare-associated infections.
S aureus was the most common pathogen, causing 28% of bloodstream infections. This was closely followed by Escherichia coli, which was found in 24% of patients.
Bloodstream infections due to multidrug-resistant pathogens occurred in 23% of patients—an increase over earlier studies. And methicillin-resistant S aureus (MRSA) was the most common multidrug-resistant pathogen.
“Similar patterns of pathogens and drug resistance have been observed in tertiary care centers, suggesting that bloodstream infections in community hospitals aren’t that different from tertiary care centers,” Dr Anderson said.
“There’s a misconception that community hospitals don’t have to deal with S aureus and MRSA, but our findings dispel that myth, since community hospitals also see these serious infections.”
Inappropriate therapy
The investigators also found that approximately 38% of patients with bloodstream infections received inappropriate empiric antimicrobial therapy or were not initially prescribed an effective antibiotic while the cause of the infection was still unknown.
A multivariate analysis revealed several factors associated with receiving inappropriate therapy, including the hospital where the patient received care (P<0.001), the need for assistance with 3 or more “daily living” activities (P=0.005), and a high Charlson score (P=0.05).
Community-onset healthcare-associated infections (P=0.01) and hospital-onset healthcare-associated infections (P=0.02) were associated with the failure to receive appropriate therapy, when community-acquired infections were used as the reference.
The investigators also incorporated drug resistance into their analysis. And they found that infection due to a multidrug-resistant organism was strongly associated with the failure to receive appropriate therapy (P<0.0001).
But most of the predictors the team initially identified retained their significance. The patient’s hospital (P<0.001), need for assistance with activities (P=0.02), and type of infection remained significant (P=0.04), but the Charlson score did not (P=0.07).
Dr Anderson recommended that clinicians in community hospitals focus on these risk factors when choosing antibiotic therapy for patients with bloodstream infections. He noted that most risk factors for receiving inappropriate therapy are already recorded in electronic health records.
“Developing an intervention where electronic records automatically alert clinicians to these risk factors when they’re choosing antibiotics could help reduce the problem,” he said. “This is just a place to start, but it’s an example of an area where we could improve how we treat patients with bloodstream infections.” ![]()

Credit: Janice Haney Carr
An analysis of 9 community hospitals showed that 1 in 3 patients with bloodstream infections received inappropriate therapy.
The study also revealed growing resistance to treatment and a high prevalence of Staphylococcus aureus bacteria in these hospitals.
Investigators said the findings, published in PLOS ONE, provide the most comprehensive look at bloodstream infections in community hospitals to date.
Much of the existing research on bloodstream infections focuses on tertiary care centers.
“Our study provides a much-needed update on what we’re seeing in community hospitals, and ultimately, we’re finding similar types of infections in these hospitals as in tertiary care centers,” said study author Deverick Anderson, MD, of Duke University in Durham North Carolina.
“It’s a challenge to identify bloodstream infections and treat them quickly and appropriately, but this study shows that there is room for improvement in both kinds of hospital settings.”
Types of infection
To better understand the types of bloodstream infections found in community hospitals, Dr Anderson and his colleagues collected information on patients treated at these hospitals in Virginia and North Carolina from 2003 to 2006.
The investigators focused on 1470 patients diagnosed with bloodstream infections. The infections were classified depending on where and when they were contracted.
Infections resulting from prior hospitalization, surgery, invasive devices (such as catheters), or living in long-term care facilities were designated healthcare-associated infections.
Community-acquired infections were contracted outside of medical settings or shortly after being admitted to a hospital. And hospital-onset infections occurred after being in a hospital for several days.
The investigators found that 56% of bloodstream infections were healthcare-associated, but symptoms began prior to hospital admission. Community-acquired infections unrelated to medical care were seen in 29% of patients. And 15% had hospital-onset healthcare-associated infections.
S aureus was the most common pathogen, causing 28% of bloodstream infections. This was closely followed by Escherichia coli, which was found in 24% of patients.
Bloodstream infections due to multidrug-resistant pathogens occurred in 23% of patients—an increase over earlier studies. And methicillin-resistant S aureus (MRSA) was the most common multidrug-resistant pathogen.
“Similar patterns of pathogens and drug resistance have been observed in tertiary care centers, suggesting that bloodstream infections in community hospitals aren’t that different from tertiary care centers,” Dr Anderson said.
“There’s a misconception that community hospitals don’t have to deal with S aureus and MRSA, but our findings dispel that myth, since community hospitals also see these serious infections.”
Inappropriate therapy
The investigators also found that approximately 38% of patients with bloodstream infections received inappropriate empiric antimicrobial therapy or were not initially prescribed an effective antibiotic while the cause of the infection was still unknown.
A multivariate analysis revealed several factors associated with receiving inappropriate therapy, including the hospital where the patient received care (P<0.001), the need for assistance with 3 or more “daily living” activities (P=0.005), and a high Charlson score (P=0.05).
Community-onset healthcare-associated infections (P=0.01) and hospital-onset healthcare-associated infections (P=0.02) were associated with the failure to receive appropriate therapy, when community-acquired infections were used as the reference.
The investigators also incorporated drug resistance into their analysis. And they found that infection due to a multidrug-resistant organism was strongly associated with the failure to receive appropriate therapy (P<0.0001).
But most of the predictors the team initially identified retained their significance. The patient’s hospital (P<0.001), need for assistance with activities (P=0.02), and type of infection remained significant (P=0.04), but the Charlson score did not (P=0.07).
Dr Anderson recommended that clinicians in community hospitals focus on these risk factors when choosing antibiotic therapy for patients with bloodstream infections. He noted that most risk factors for receiving inappropriate therapy are already recorded in electronic health records.
“Developing an intervention where electronic records automatically alert clinicians to these risk factors when they’re choosing antibiotics could help reduce the problem,” he said. “This is just a place to start, but it’s an example of an area where we could improve how we treat patients with bloodstream infections.” ![]()

Credit: Janice Haney Carr
An analysis of 9 community hospitals showed that 1 in 3 patients with bloodstream infections received inappropriate therapy.
The study also revealed growing resistance to treatment and a high prevalence of Staphylococcus aureus bacteria in these hospitals.
Investigators said the findings, published in PLOS ONE, provide the most comprehensive look at bloodstream infections in community hospitals to date.
Much of the existing research on bloodstream infections focuses on tertiary care centers.
“Our study provides a much-needed update on what we’re seeing in community hospitals, and ultimately, we’re finding similar types of infections in these hospitals as in tertiary care centers,” said study author Deverick Anderson, MD, of Duke University in Durham North Carolina.
“It’s a challenge to identify bloodstream infections and treat them quickly and appropriately, but this study shows that there is room for improvement in both kinds of hospital settings.”
Types of infection
To better understand the types of bloodstream infections found in community hospitals, Dr Anderson and his colleagues collected information on patients treated at these hospitals in Virginia and North Carolina from 2003 to 2006.
The investigators focused on 1470 patients diagnosed with bloodstream infections. The infections were classified depending on where and when they were contracted.
Infections resulting from prior hospitalization, surgery, invasive devices (such as catheters), or living in long-term care facilities were designated healthcare-associated infections.
Community-acquired infections were contracted outside of medical settings or shortly after being admitted to a hospital. And hospital-onset infections occurred after being in a hospital for several days.
The investigators found that 56% of bloodstream infections were healthcare-associated, but symptoms began prior to hospital admission. Community-acquired infections unrelated to medical care were seen in 29% of patients. And 15% had hospital-onset healthcare-associated infections.
S aureus was the most common pathogen, causing 28% of bloodstream infections. This was closely followed by Escherichia coli, which was found in 24% of patients.
Bloodstream infections due to multidrug-resistant pathogens occurred in 23% of patients—an increase over earlier studies. And methicillin-resistant S aureus (MRSA) was the most common multidrug-resistant pathogen.
“Similar patterns of pathogens and drug resistance have been observed in tertiary care centers, suggesting that bloodstream infections in community hospitals aren’t that different from tertiary care centers,” Dr Anderson said.
“There’s a misconception that community hospitals don’t have to deal with S aureus and MRSA, but our findings dispel that myth, since community hospitals also see these serious infections.”
Inappropriate therapy
The investigators also found that approximately 38% of patients with bloodstream infections received inappropriate empiric antimicrobial therapy or were not initially prescribed an effective antibiotic while the cause of the infection was still unknown.
A multivariate analysis revealed several factors associated with receiving inappropriate therapy, including the hospital where the patient received care (P<0.001), the need for assistance with 3 or more “daily living” activities (P=0.005), and a high Charlson score (P=0.05).
Community-onset healthcare-associated infections (P=0.01) and hospital-onset healthcare-associated infections (P=0.02) were associated with the failure to receive appropriate therapy, when community-acquired infections were used as the reference.
The investigators also incorporated drug resistance into their analysis. And they found that infection due to a multidrug-resistant organism was strongly associated with the failure to receive appropriate therapy (P<0.0001).
But most of the predictors the team initially identified retained their significance. The patient’s hospital (P<0.001), need for assistance with activities (P=0.02), and type of infection remained significant (P=0.04), but the Charlson score did not (P=0.07).
Dr Anderson recommended that clinicians in community hospitals focus on these risk factors when choosing antibiotic therapy for patients with bloodstream infections. He noted that most risk factors for receiving inappropriate therapy are already recorded in electronic health records.
“Developing an intervention where electronic records automatically alert clinicians to these risk factors when they’re choosing antibiotics could help reduce the problem,” he said. “This is just a place to start, but it’s an example of an area where we could improve how we treat patients with bloodstream infections.” ![]()
Group calls for standardized data collection practices across cancer centers

Credit: CDC
Researchers have identified significant variations in how cancer centers gather data, particularly that pertaining to racial and ethnic minorities.
Although racial and ethnic categories were similar across the centers, those categories were defined differently.
And the centers’ definitions of “catchment area,” the geographic region they expect to influence with their programs, differed widely.
This research, published in Cancer, was part of a national effort to recruit more racial/ethnic minorities into clinical trials and, ultimately, reduce the disproportional incidence of many cancers among those populations.
Five National Cancer Institute-designated comprehensive cancer centers participated in the endeavor, known as EMPaCT—Enhancing Minority Participation in Clinical Trials. They were:
- University of Minnesota, Minneapolis, which represents the Midwest and targets the accrual of Native Americans and African Americans
- University of Alabama, Birmingham, representing the Southeast, targeting African Americans
- Johns Hopkins University, representing the East, targeting African Americans
- University of Texas MD Anderson, Houston, representing the Southwest, targeting Latinos
- University of California, Davis, representing the West, targeting Asian Americans.
Ernest T. Hawk, MD, of the MD Anderson Cancer Center, and his colleagues reviewed the collection and reporting of patient data and other practices by these 5 centers.
This revealed significant variation in the centers’ methods of data collection. For example, patients’ insurance status was routinely documented at 2 centers, collected for non-research patients only at a third center, collected for billing of researcher enrollees at a fourth center, and not documented at all at a fifth center.
There were differences in data collection according to race/ethnicity as well. Racial/ethnic categories were generally similar across the centers—white, black/African American, Asian, Native American, Hispanic/Latino, and “other/unknown.”
However, the means of race/ethnicity data collection differed. Each center collected self-reported data on race/ethnicity, but 2 centers included data from staff observations.
Two centers compared the proportions of racial/ethnic groups enrolled in trials with those of their catchment area(s). But the others did not.
The centers also differed in how they defined their patient catchment area, in terms of their cancer patient-vs-general-population specificity, levels of specificity, and geographic coverage.
That merits notice, according to the researchers, because National Cancer Institute cancer centers are required to accrue women and minorities to clinical trials in rough proportion to the cancer patient population of the center’s primary catchment area.
Given these findings, the researchers recommended better standardization of data definition, collection, and reporting as an essential first step toward expanding minority participation in clinical trials.
The team also advised that cancer centers collect socioeconomic data, including a patient’s income and education levels, given past evidence of the strong link between socioeconomic status and cancer outcomes.
Finally, the group recommended collecting patient zip codes and insurance status to allow researchers to assess differences in access to clinical trials that may be related to geography and the availability of health insurance coverage. ![]()

Credit: CDC
Researchers have identified significant variations in how cancer centers gather data, particularly that pertaining to racial and ethnic minorities.
Although racial and ethnic categories were similar across the centers, those categories were defined differently.
And the centers’ definitions of “catchment area,” the geographic region they expect to influence with their programs, differed widely.
This research, published in Cancer, was part of a national effort to recruit more racial/ethnic minorities into clinical trials and, ultimately, reduce the disproportional incidence of many cancers among those populations.
Five National Cancer Institute-designated comprehensive cancer centers participated in the endeavor, known as EMPaCT—Enhancing Minority Participation in Clinical Trials. They were:
- University of Minnesota, Minneapolis, which represents the Midwest and targets the accrual of Native Americans and African Americans
- University of Alabama, Birmingham, representing the Southeast, targeting African Americans
- Johns Hopkins University, representing the East, targeting African Americans
- University of Texas MD Anderson, Houston, representing the Southwest, targeting Latinos
- University of California, Davis, representing the West, targeting Asian Americans.
Ernest T. Hawk, MD, of the MD Anderson Cancer Center, and his colleagues reviewed the collection and reporting of patient data and other practices by these 5 centers.
This revealed significant variation in the centers’ methods of data collection. For example, patients’ insurance status was routinely documented at 2 centers, collected for non-research patients only at a third center, collected for billing of researcher enrollees at a fourth center, and not documented at all at a fifth center.
There were differences in data collection according to race/ethnicity as well. Racial/ethnic categories were generally similar across the centers—white, black/African American, Asian, Native American, Hispanic/Latino, and “other/unknown.”
However, the means of race/ethnicity data collection differed. Each center collected self-reported data on race/ethnicity, but 2 centers included data from staff observations.
Two centers compared the proportions of racial/ethnic groups enrolled in trials with those of their catchment area(s). But the others did not.
The centers also differed in how they defined their patient catchment area, in terms of their cancer patient-vs-general-population specificity, levels of specificity, and geographic coverage.
That merits notice, according to the researchers, because National Cancer Institute cancer centers are required to accrue women and minorities to clinical trials in rough proportion to the cancer patient population of the center’s primary catchment area.
Given these findings, the researchers recommended better standardization of data definition, collection, and reporting as an essential first step toward expanding minority participation in clinical trials.
The team also advised that cancer centers collect socioeconomic data, including a patient’s income and education levels, given past evidence of the strong link between socioeconomic status and cancer outcomes.
Finally, the group recommended collecting patient zip codes and insurance status to allow researchers to assess differences in access to clinical trials that may be related to geography and the availability of health insurance coverage. ![]()

Credit: CDC
Researchers have identified significant variations in how cancer centers gather data, particularly that pertaining to racial and ethnic minorities.
Although racial and ethnic categories were similar across the centers, those categories were defined differently.
And the centers’ definitions of “catchment area,” the geographic region they expect to influence with their programs, differed widely.
This research, published in Cancer, was part of a national effort to recruit more racial/ethnic minorities into clinical trials and, ultimately, reduce the disproportional incidence of many cancers among those populations.
Five National Cancer Institute-designated comprehensive cancer centers participated in the endeavor, known as EMPaCT—Enhancing Minority Participation in Clinical Trials. They were:
- University of Minnesota, Minneapolis, which represents the Midwest and targets the accrual of Native Americans and African Americans
- University of Alabama, Birmingham, representing the Southeast, targeting African Americans
- Johns Hopkins University, representing the East, targeting African Americans
- University of Texas MD Anderson, Houston, representing the Southwest, targeting Latinos
- University of California, Davis, representing the West, targeting Asian Americans.
Ernest T. Hawk, MD, of the MD Anderson Cancer Center, and his colleagues reviewed the collection and reporting of patient data and other practices by these 5 centers.
This revealed significant variation in the centers’ methods of data collection. For example, patients’ insurance status was routinely documented at 2 centers, collected for non-research patients only at a third center, collected for billing of researcher enrollees at a fourth center, and not documented at all at a fifth center.
There were differences in data collection according to race/ethnicity as well. Racial/ethnic categories were generally similar across the centers—white, black/African American, Asian, Native American, Hispanic/Latino, and “other/unknown.”
However, the means of race/ethnicity data collection differed. Each center collected self-reported data on race/ethnicity, but 2 centers included data from staff observations.
Two centers compared the proportions of racial/ethnic groups enrolled in trials with those of their catchment area(s). But the others did not.
The centers also differed in how they defined their patient catchment area, in terms of their cancer patient-vs-general-population specificity, levels of specificity, and geographic coverage.
That merits notice, according to the researchers, because National Cancer Institute cancer centers are required to accrue women and minorities to clinical trials in rough proportion to the cancer patient population of the center’s primary catchment area.
Given these findings, the researchers recommended better standardization of data definition, collection, and reporting as an essential first step toward expanding minority participation in clinical trials.
The team also advised that cancer centers collect socioeconomic data, including a patient’s income and education levels, given past evidence of the strong link between socioeconomic status and cancer outcomes.
Finally, the group recommended collecting patient zip codes and insurance status to allow researchers to assess differences in access to clinical trials that may be related to geography and the availability of health insurance coverage. ![]()
Adult minorities underrepresented in cancer trials

Credit: Rhoda Baer
New research indicates that less than 2% of trials funded by the National Cancer Institute focus on racial and ethnic minorities, and minority participation in adult cancer trials is not representative of the US population.
The researchers said these findings suggest we must do more to promote minority-focused research and clinical trial recruitment, beyond the National Institutes of Health (NIH) Revitalization Act of 1993, which mandated the appropriate inclusion of minorities in all NIH-funded research.
“What is needed is deliberate effort,” said study author Moon Chen, Jr, PhD, of the University of California, Davis. “Minorities are not hard to reach. They are hardly reached.”
To assess minority inclusion in clinical trials, Dr Chen and his colleagues searched ClinicalTrials.gov, looking for trials sponsored by the National Cancer Institute that were available in January 2013.
They searched using terms for different minority groups, then counted the number of clinical trials with a primary focus on a particular ethnic or minority population. Roughly 150 trials out of 10,000—or less than 2%—met the criteria.
The researchers also reviewed abstracts and articles accessed from January through March 2013 on PubMed to find those that specifically examined minority accrual in clinical trials.
Of the 42 citations found, 5 included reports explicitly discussing participation levels by race and ethnicity. Those reports revealed an “encouraging but less than optimal” increase in specification of race or ethnicity in published results of clinical trials.
Dr Chen and his colleagues also reported that participation of adult minorities is not proportional to their representation in the US population.
For example, African Americans experience the highest cancer incidence of any racial group (593.7 cases per 100,000), but they have the lowest rates of cancer trial participation (tied with Hispanics), at 1.3%. It’s important to note, however, that clinical trial participation is low for all adult cancer patients, at 3% to 5%.
In contrast, the researchers pointed out that 60% of all patients under age 15 are enrolled in clinical trials. And minority representation among children is excellent, either equal to or greater than their proportion of the population.
To put the adult population on par with the pediatric population, researchers should design trials to include and focus on specific populations, Dr Chen said. Furthermore, scientific journals should insist on appropriate representation and analyses of NIH research by race and ethnicity.
“Whatever happens in the laboratory or in the clinic needs to be applied to solving real-world problems,” Dr Chen said. “And those relate to the disproportionate effects of cancer and other diseases on racial and ethnic minorities.”
Dr Chen and his colleagues reported this research in Cancer. ![]()

Credit: Rhoda Baer
New research indicates that less than 2% of trials funded by the National Cancer Institute focus on racial and ethnic minorities, and minority participation in adult cancer trials is not representative of the US population.
The researchers said these findings suggest we must do more to promote minority-focused research and clinical trial recruitment, beyond the National Institutes of Health (NIH) Revitalization Act of 1993, which mandated the appropriate inclusion of minorities in all NIH-funded research.
“What is needed is deliberate effort,” said study author Moon Chen, Jr, PhD, of the University of California, Davis. “Minorities are not hard to reach. They are hardly reached.”
To assess minority inclusion in clinical trials, Dr Chen and his colleagues searched ClinicalTrials.gov, looking for trials sponsored by the National Cancer Institute that were available in January 2013.
They searched using terms for different minority groups, then counted the number of clinical trials with a primary focus on a particular ethnic or minority population. Roughly 150 trials out of 10,000—or less than 2%—met the criteria.
The researchers also reviewed abstracts and articles accessed from January through March 2013 on PubMed to find those that specifically examined minority accrual in clinical trials.
Of the 42 citations found, 5 included reports explicitly discussing participation levels by race and ethnicity. Those reports revealed an “encouraging but less than optimal” increase in specification of race or ethnicity in published results of clinical trials.
Dr Chen and his colleagues also reported that participation of adult minorities is not proportional to their representation in the US population.
For example, African Americans experience the highest cancer incidence of any racial group (593.7 cases per 100,000), but they have the lowest rates of cancer trial participation (tied with Hispanics), at 1.3%. It’s important to note, however, that clinical trial participation is low for all adult cancer patients, at 3% to 5%.
In contrast, the researchers pointed out that 60% of all patients under age 15 are enrolled in clinical trials. And minority representation among children is excellent, either equal to or greater than their proportion of the population.
To put the adult population on par with the pediatric population, researchers should design trials to include and focus on specific populations, Dr Chen said. Furthermore, scientific journals should insist on appropriate representation and analyses of NIH research by race and ethnicity.
“Whatever happens in the laboratory or in the clinic needs to be applied to solving real-world problems,” Dr Chen said. “And those relate to the disproportionate effects of cancer and other diseases on racial and ethnic minorities.”
Dr Chen and his colleagues reported this research in Cancer. ![]()

Credit: Rhoda Baer
New research indicates that less than 2% of trials funded by the National Cancer Institute focus on racial and ethnic minorities, and minority participation in adult cancer trials is not representative of the US population.
The researchers said these findings suggest we must do more to promote minority-focused research and clinical trial recruitment, beyond the National Institutes of Health (NIH) Revitalization Act of 1993, which mandated the appropriate inclusion of minorities in all NIH-funded research.
“What is needed is deliberate effort,” said study author Moon Chen, Jr, PhD, of the University of California, Davis. “Minorities are not hard to reach. They are hardly reached.”
To assess minority inclusion in clinical trials, Dr Chen and his colleagues searched ClinicalTrials.gov, looking for trials sponsored by the National Cancer Institute that were available in January 2013.
They searched using terms for different minority groups, then counted the number of clinical trials with a primary focus on a particular ethnic or minority population. Roughly 150 trials out of 10,000—or less than 2%—met the criteria.
The researchers also reviewed abstracts and articles accessed from January through March 2013 on PubMed to find those that specifically examined minority accrual in clinical trials.
Of the 42 citations found, 5 included reports explicitly discussing participation levels by race and ethnicity. Those reports revealed an “encouraging but less than optimal” increase in specification of race or ethnicity in published results of clinical trials.
Dr Chen and his colleagues also reported that participation of adult minorities is not proportional to their representation in the US population.
For example, African Americans experience the highest cancer incidence of any racial group (593.7 cases per 100,000), but they have the lowest rates of cancer trial participation (tied with Hispanics), at 1.3%. It’s important to note, however, that clinical trial participation is low for all adult cancer patients, at 3% to 5%.
In contrast, the researchers pointed out that 60% of all patients under age 15 are enrolled in clinical trials. And minority representation among children is excellent, either equal to or greater than their proportion of the population.
To put the adult population on par with the pediatric population, researchers should design trials to include and focus on specific populations, Dr Chen said. Furthermore, scientific journals should insist on appropriate representation and analyses of NIH research by race and ethnicity.
“Whatever happens in the laboratory or in the clinic needs to be applied to solving real-world problems,” Dr Chen said. “And those relate to the disproportionate effects of cancer and other diseases on racial and ethnic minorities.”
Dr Chen and his colleagues reported this research in Cancer.
FDA approves drug for infantile hemangioma
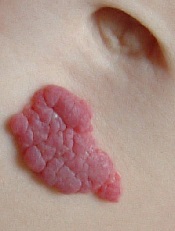
infant’s stomach
The US Food and Drug Administration has approved oral propranolol hydrochloride (Hemangeol) to treat proliferating infantile hemangiomas that require systemic therapy.
The drug, which is also under review in the European Union, will be available in the US in June.
Infantile hemangioma is the most common vascular benign tumor of infancy, affecting 3% to 10% of newborns. The lesions are rarely detectable at birth and start growing noticeably in the first 4 to 6 weeks of life.
While most infantile hemangiomas do not require treatment, approximately 12% do. Depending upon their location, infantile hemangiomas might impair breathing, eating, or vision, or become life-threatening.
Propranolol has long been used in cardiology, but its use in infantile hemangiomas is relatively new. In 2007, Christine Léauté-Labreze, MD, a dermatologist at the Bordeaux University Hospital in France, discovered that propranolol could treat infantile hemangiomas.
Since then, the drug has been used for this indication off-label. And in 2009, Pierre Fabre Dermatologie began developing propranolol hydrochloride for use in infantile hemangiomas.
In a study of 32 children, propranolol slowed the growth of infantile hemangiomas in 100% of patients. Patients had received propranolol at 2 to 3 mg/kg per day for a median of 6.1 months. Side effects were “limited and mild,” according to researchers (V Sans et al. Pediatrics 2009).
Researchers also conducted a randomized, controlled trial of the drug in infants 5 weeks to 5 months old at therapy initiation. The team compared 4 propranolol treatment protocols (1 or 3 mg/kg/day for 3 or 6 months) to placebo.
Propranolol at a daily dose of 3 mg/kg for 6 months had a 60.4% success rate, compared to 3.6% in the placebo group (P<0.0001). Success was defined as complete or nearly complete resolution of the target hemangioma. However, 11.4% of patients needed to be re-treated after stopping propranolol.
The drug is contraindicated in premature infants who have a corrected age of less than 5 weeks, weight less than 2 kg, known hypersensitivity to propranolol or any of its excipients, asthma or a history of bronchospasm, pheochromocytoma, blood pressure less than 50/30 mmHg, a heart rate less than 80 beats per minute, greater than first degree heart block, or decompensated heart failure.
Propranolol hydrochloride can cause serious side effects, including hypoglycemia, bradycardia, hypotension, and bronchospasm. The drug can worsen congestive heart failure and may increase the risk of stroke in children with PHACE syndrome.
The most frequently reported adverse reactions, occurring in more than 10% of infants receiving propranolol hydrochloride, were sleep disorders, diarrhea, vomiting, and aggravated respiratory tract infections, such as bronchitis and bronchiolitis associated with cough and fever. Adverse reactions led to treatment discontinuation in fewer than 2% of treated patients.

infant’s stomach
The US Food and Drug Administration has approved oral propranolol hydrochloride (Hemangeol) to treat proliferating infantile hemangiomas that require systemic therapy.
The drug, which is also under review in the European Union, will be available in the US in June.
Infantile hemangioma is the most common vascular benign tumor of infancy, affecting 3% to 10% of newborns. The lesions are rarely detectable at birth and start growing noticeably in the first 4 to 6 weeks of life.
While most infantile hemangiomas do not require treatment, approximately 12% do. Depending upon their location, infantile hemangiomas might impair breathing, eating, or vision, or become life-threatening.
Propranolol has long been used in cardiology, but its use in infantile hemangiomas is relatively new. In 2007, Christine Léauté-Labreze, MD, a dermatologist at the Bordeaux University Hospital in France, discovered that propranolol could treat infantile hemangiomas.
Since then, the drug has been used for this indication off-label. And in 2009, Pierre Fabre Dermatologie began developing propranolol hydrochloride for use in infantile hemangiomas.
In a study of 32 children, propranolol slowed the growth of infantile hemangiomas in 100% of patients. Patients had received propranolol at 2 to 3 mg/kg per day for a median of 6.1 months. Side effects were “limited and mild,” according to researchers (V Sans et al. Pediatrics 2009).
Researchers also conducted a randomized, controlled trial of the drug in infants 5 weeks to 5 months old at therapy initiation. The team compared 4 propranolol treatment protocols (1 or 3 mg/kg/day for 3 or 6 months) to placebo.
Propranolol at a daily dose of 3 mg/kg for 6 months had a 60.4% success rate, compared to 3.6% in the placebo group (P<0.0001). Success was defined as complete or nearly complete resolution of the target hemangioma. However, 11.4% of patients needed to be re-treated after stopping propranolol.
The drug is contraindicated in premature infants who have a corrected age of less than 5 weeks, weight less than 2 kg, known hypersensitivity to propranolol or any of its excipients, asthma or a history of bronchospasm, pheochromocytoma, blood pressure less than 50/30 mmHg, a heart rate less than 80 beats per minute, greater than first degree heart block, or decompensated heart failure.
Propranolol hydrochloride can cause serious side effects, including hypoglycemia, bradycardia, hypotension, and bronchospasm. The drug can worsen congestive heart failure and may increase the risk of stroke in children with PHACE syndrome.
The most frequently reported adverse reactions, occurring in more than 10% of infants receiving propranolol hydrochloride, were sleep disorders, diarrhea, vomiting, and aggravated respiratory tract infections, such as bronchitis and bronchiolitis associated with cough and fever. Adverse reactions led to treatment discontinuation in fewer than 2% of treated patients.

infant’s stomach
The US Food and Drug Administration has approved oral propranolol hydrochloride (Hemangeol) to treat proliferating infantile hemangiomas that require systemic therapy.
The drug, which is also under review in the European Union, will be available in the US in June.
Infantile hemangioma is the most common vascular benign tumor of infancy, affecting 3% to 10% of newborns. The lesions are rarely detectable at birth and start growing noticeably in the first 4 to 6 weeks of life.
While most infantile hemangiomas do not require treatment, approximately 12% do. Depending upon their location, infantile hemangiomas might impair breathing, eating, or vision, or become life-threatening.
Propranolol has long been used in cardiology, but its use in infantile hemangiomas is relatively new. In 2007, Christine Léauté-Labreze, MD, a dermatologist at the Bordeaux University Hospital in France, discovered that propranolol could treat infantile hemangiomas.
Since then, the drug has been used for this indication off-label. And in 2009, Pierre Fabre Dermatologie began developing propranolol hydrochloride for use in infantile hemangiomas.
In a study of 32 children, propranolol slowed the growth of infantile hemangiomas in 100% of patients. Patients had received propranolol at 2 to 3 mg/kg per day for a median of 6.1 months. Side effects were “limited and mild,” according to researchers (V Sans et al. Pediatrics 2009).
Researchers also conducted a randomized, controlled trial of the drug in infants 5 weeks to 5 months old at therapy initiation. The team compared 4 propranolol treatment protocols (1 or 3 mg/kg/day for 3 or 6 months) to placebo.
Propranolol at a daily dose of 3 mg/kg for 6 months had a 60.4% success rate, compared to 3.6% in the placebo group (P<0.0001). Success was defined as complete or nearly complete resolution of the target hemangioma. However, 11.4% of patients needed to be re-treated after stopping propranolol.
The drug is contraindicated in premature infants who have a corrected age of less than 5 weeks, weight less than 2 kg, known hypersensitivity to propranolol or any of its excipients, asthma or a history of bronchospasm, pheochromocytoma, blood pressure less than 50/30 mmHg, a heart rate less than 80 beats per minute, greater than first degree heart block, or decompensated heart failure.
Propranolol hydrochloride can cause serious side effects, including hypoglycemia, bradycardia, hypotension, and bronchospasm. The drug can worsen congestive heart failure and may increase the risk of stroke in children with PHACE syndrome.
The most frequently reported adverse reactions, occurring in more than 10% of infants receiving propranolol hydrochloride, were sleep disorders, diarrhea, vomiting, and aggravated respiratory tract infections, such as bronchitis and bronchiolitis associated with cough and fever. Adverse reactions led to treatment discontinuation in fewer than 2% of treated patients.
FDA approves IV formulation of antifungal agent

The US Food and Drug Administration has approved an intravenous formulation of posaconazole (Noxafil), which is expected to be available at wholesalers in mid-April.
The antifungal agent is already available as delayed-release tablets and in an oral suspension formulation.
In any formulation, posaconazole is indicated for prophylaxis of invasive Aspergillus and Candida infections in immunocompromised patients who are at high risk of developing these infections.
This includes patients who have developed graft-vs-host disease after hematopoietic stem cell transplant and patients with hematologic malignancies who have prolonged neutropenia resulting from chemotherapy.
Posaconazole injection is indicated for use in patients 18 years of age and older. The delayed-release tablets and oral suspension are indicated for patients 13 years of age and older.
Posaconazole injection is administered with a loading dose of 300 mg (one 300 mg vial) twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by slow intravenous infusion over approximately 90 minutes.
Once combined with a mixture of intravenous solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole injection should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2-8 degrees C (36-46 degrees F).
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
In clinical trials, the adverse reactions reported for posaconazole injection were generally similar to those reported in trials of posaconazole oral suspension. The most frequently reported adverse reactions with an onset during the posaconazole intravenous phase of dosing 300 mg once-daily therapy were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
Patients who are allergic to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given along with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
For more details, see the complete prescribing information. Posaconazole is marketed as Noxafil by Merck.

The US Food and Drug Administration has approved an intravenous formulation of posaconazole (Noxafil), which is expected to be available at wholesalers in mid-April.
The antifungal agent is already available as delayed-release tablets and in an oral suspension formulation.
In any formulation, posaconazole is indicated for prophylaxis of invasive Aspergillus and Candida infections in immunocompromised patients who are at high risk of developing these infections.
This includes patients who have developed graft-vs-host disease after hematopoietic stem cell transplant and patients with hematologic malignancies who have prolonged neutropenia resulting from chemotherapy.
Posaconazole injection is indicated for use in patients 18 years of age and older. The delayed-release tablets and oral suspension are indicated for patients 13 years of age and older.
Posaconazole injection is administered with a loading dose of 300 mg (one 300 mg vial) twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by slow intravenous infusion over approximately 90 minutes.
Once combined with a mixture of intravenous solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole injection should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2-8 degrees C (36-46 degrees F).
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
In clinical trials, the adverse reactions reported for posaconazole injection were generally similar to those reported in trials of posaconazole oral suspension. The most frequently reported adverse reactions with an onset during the posaconazole intravenous phase of dosing 300 mg once-daily therapy were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
Patients who are allergic to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given along with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
For more details, see the complete prescribing information. Posaconazole is marketed as Noxafil by Merck.

The US Food and Drug Administration has approved an intravenous formulation of posaconazole (Noxafil), which is expected to be available at wholesalers in mid-April.
The antifungal agent is already available as delayed-release tablets and in an oral suspension formulation.
In any formulation, posaconazole is indicated for prophylaxis of invasive Aspergillus and Candida infections in immunocompromised patients who are at high risk of developing these infections.
This includes patients who have developed graft-vs-host disease after hematopoietic stem cell transplant and patients with hematologic malignancies who have prolonged neutropenia resulting from chemotherapy.
Posaconazole injection is indicated for use in patients 18 years of age and older. The delayed-release tablets and oral suspension are indicated for patients 13 years of age and older.
Posaconazole injection is administered with a loading dose of 300 mg (one 300 mg vial) twice a day on the first day of therapy, then 300 mg once a day thereafter. It is given through a central venous line by slow intravenous infusion over approximately 90 minutes.
Once combined with a mixture of intravenous solution (150 mL of 5% dextrose in water or sodium chloride 0.9%), posaconazole injection should be administered immediately. If not used immediately, the solution can be stored up to 24 hours if refrigerated at 2-8 degrees C (36-46 degrees F).
Co-administration of drugs that can decrease the plasma concentration of posaconazole should be avoided unless the benefit outweighs the risk. If such drugs are necessary, patients should be monitored closely for breakthrough fungal infections.
In clinical trials, the adverse reactions reported for posaconazole injection were generally similar to those reported in trials of posaconazole oral suspension. The most frequently reported adverse reactions with an onset during the posaconazole intravenous phase of dosing 300 mg once-daily therapy were diarrhea (32%), hypokalemia (22%), fever (21%), and nausea (19%).
Patients who are allergic to posaconazole or other azole antifungal medicines should not receive posaconazole. The drug should not be given along with sirolimus, pimozide, quinidine, atorvastatin, lovastatin, simvastatin, or ergot alkaloids.
Drugs such as cyclosporine and tacrolimus require dose adjustments and frequent blood monitoring when administered with posaconazole. Serious side effects, including nephrotoxicity, leukoencephalopathy, and death, have been reported in patients with increased cyclosporine or tacrolimus blood levels.
Healthcare professionals should use caution when administering posaconazole to patients at risk of developing an irregular heart rhythm, as the drug has been shown to prolong the QT interval, and cases of potentially fatal irregular heart rhythm (torsades de pointes) have been reported in patients taking posaconazole.
For more details, see the complete prescribing information. Posaconazole is marketed as Noxafil by Merck.
Study may explain why targeted treatment falls short in angiosarcoma
Heiser & Robert Ackland
Multiple mutations drive the development of angiosarcoma, according to a study published in Nature Genetics.
Researchers identified driver mutations in several genes associated with angiogenesis, including PTPRB and PLCG1.
They also found that PLCG1 mutations only occurred alongside mutations in PTPRB.
The investigators believe these findings may explain why angiosarcoma therapies directed at a single target fail to eradicate the disease.
Angiosarcoma is a rare cancer of the blood vessels that can occur spontaneously or develop after radiotherapy or chronic lymphedema.
Previous research indicated that aberrant angiogenesis, including somatic mutations in angiogenesis-signaling genes, drives angiosarcoma. So researchers developed drugs targeting pathways involved in angiogenesis, but these drugs have had little or no success.
“Because this cancer doesn’t respond well to traditional chemotherapy and radiotherapy, it makes sense to develop drugs that target pathways that control blood vessel formation,” said study author Peter Campbell, MD, PhD, of the Wellcome Trust Sanger Institute in the UK.
“We found 2 novel cancer genes that control blood vessel formation which are mutated in this cancer and which could be targeted for treatment of this highly aggressive cancer.”
To identify these genes, Dr Campbell and his colleagues performed whole-genome, whole-exome, and targeted sequencing in samples from patients with angiosarcoma.
Thirty-eight percent of the samples (15/39) carried mutations in genes that control angiogenesis, including PLCG1 and PTPRB.
The researchers identified 14 PTPRB mutations in 10 samples. This included 8 nonsense variants, 3 missense variants, 2 essential splice-site variants, and 1 frameshift insertion.
The investigators also discovered a recurrent mutation in PLCG1, a missense variant encoding p.Arg707Gln, which was present in 3 patient samples. All 3 PLCG1 mutations co-occurred with PTPRB mutations.
The researchers said this discovery may explain why drugs developed for a single target are ineffective in some angiosarcoma patients.
“Not only does our study change the way people view the biology of this tumor, it acts as a guide for future drug trials in angiosarcoma patients,” said study author Adrian Harris, MD, DPhil, of the University of Oxford in the UK.
He noted that researchers can use information from this study to determine if existing drugs could be effective against angiosarcoma.

Heiser & Robert Ackland
Multiple mutations drive the development of angiosarcoma, according to a study published in Nature Genetics.
Researchers identified driver mutations in several genes associated with angiogenesis, including PTPRB and PLCG1.
They also found that PLCG1 mutations only occurred alongside mutations in PTPRB.
The investigators believe these findings may explain why angiosarcoma therapies directed at a single target fail to eradicate the disease.
Angiosarcoma is a rare cancer of the blood vessels that can occur spontaneously or develop after radiotherapy or chronic lymphedema.
Previous research indicated that aberrant angiogenesis, including somatic mutations in angiogenesis-signaling genes, drives angiosarcoma. So researchers developed drugs targeting pathways involved in angiogenesis, but these drugs have had little or no success.
“Because this cancer doesn’t respond well to traditional chemotherapy and radiotherapy, it makes sense to develop drugs that target pathways that control blood vessel formation,” said study author Peter Campbell, MD, PhD, of the Wellcome Trust Sanger Institute in the UK.
“We found 2 novel cancer genes that control blood vessel formation which are mutated in this cancer and which could be targeted for treatment of this highly aggressive cancer.”
To identify these genes, Dr Campbell and his colleagues performed whole-genome, whole-exome, and targeted sequencing in samples from patients with angiosarcoma.
Thirty-eight percent of the samples (15/39) carried mutations in genes that control angiogenesis, including PLCG1 and PTPRB.
The researchers identified 14 PTPRB mutations in 10 samples. This included 8 nonsense variants, 3 missense variants, 2 essential splice-site variants, and 1 frameshift insertion.
The investigators also discovered a recurrent mutation in PLCG1, a missense variant encoding p.Arg707Gln, which was present in 3 patient samples. All 3 PLCG1 mutations co-occurred with PTPRB mutations.
The researchers said this discovery may explain why drugs developed for a single target are ineffective in some angiosarcoma patients.
“Not only does our study change the way people view the biology of this tumor, it acts as a guide for future drug trials in angiosarcoma patients,” said study author Adrian Harris, MD, DPhil, of the University of Oxford in the UK.
He noted that researchers can use information from this study to determine if existing drugs could be effective against angiosarcoma.

Heiser & Robert Ackland
Multiple mutations drive the development of angiosarcoma, according to a study published in Nature Genetics.
Researchers identified driver mutations in several genes associated with angiogenesis, including PTPRB and PLCG1.
They also found that PLCG1 mutations only occurred alongside mutations in PTPRB.
The investigators believe these findings may explain why angiosarcoma therapies directed at a single target fail to eradicate the disease.
Angiosarcoma is a rare cancer of the blood vessels that can occur spontaneously or develop after radiotherapy or chronic lymphedema.
Previous research indicated that aberrant angiogenesis, including somatic mutations in angiogenesis-signaling genes, drives angiosarcoma. So researchers developed drugs targeting pathways involved in angiogenesis, but these drugs have had little or no success.
“Because this cancer doesn’t respond well to traditional chemotherapy and radiotherapy, it makes sense to develop drugs that target pathways that control blood vessel formation,” said study author Peter Campbell, MD, PhD, of the Wellcome Trust Sanger Institute in the UK.
“We found 2 novel cancer genes that control blood vessel formation which are mutated in this cancer and which could be targeted for treatment of this highly aggressive cancer.”
To identify these genes, Dr Campbell and his colleagues performed whole-genome, whole-exome, and targeted sequencing in samples from patients with angiosarcoma.
Thirty-eight percent of the samples (15/39) carried mutations in genes that control angiogenesis, including PLCG1 and PTPRB.
The researchers identified 14 PTPRB mutations in 10 samples. This included 8 nonsense variants, 3 missense variants, 2 essential splice-site variants, and 1 frameshift insertion.
The investigators also discovered a recurrent mutation in PLCG1, a missense variant encoding p.Arg707Gln, which was present in 3 patient samples. All 3 PLCG1 mutations co-occurred with PTPRB mutations.
The researchers said this discovery may explain why drugs developed for a single target are ineffective in some angiosarcoma patients.
“Not only does our study change the way people view the biology of this tumor, it acts as a guide for future drug trials in angiosarcoma patients,” said study author Adrian Harris, MD, DPhil, of the University of Oxford in the UK.
He noted that researchers can use information from this study to determine if existing drugs could be effective against angiosarcoma.