User login
Malaria maps show progress, room for improvement

Credit: CDC
Malaria prevalence maps indicate that, in 2010, nearly 184 million Africans were still living in areas where there is a high risk of contracting malaria, despite a decade of efforts to control the spread of the disease.
The maps showed that 40 African countries experienced reductions in childhood malaria transmission between 2000 and 2010.
Despite this progress, 57% of the population in malaria-endemic countries continued to live in areas of moderate to intense malaria transmission, with infection rates higher than 10%.
These findings are published in The Lancet.
Researchers compiled data from a collection of 26,746 community-based surveys of Plasmodium falciparum prevalence. The surveys covered 3,575,418 person observations from 44 malaria-endemic countries and territories in Africa since 1980.
“Health information systems in many African countries are weak, and it has been difficult to reliably estimate how many people get sick or die of malaria,” said study author Abdisalan Mohamed Noor, PhD, of the Kenya Medical Research Institute-Wellcome Trust Research Programme in Nairobi and the University of Oxford in the UK.
“The population surveys we used in this study are a more reliable indicator for tracking, and we hope our study will help countries assess their progress and adapt their strategies for more effective malaria control.”
Using model-based geostatistics, Dr Noor and his colleagues estimated the proportion of the population, aged 2 to 10 years old, infected with different levels of P falciparum across Africa in 2000 and 2010.
The researchers wanted to evaluate the effects of the Roll Back Malaria Partnership, which was launched in 2000 and resulted in a large increase in investments targeting malaria control.
The team found that the number of people living in high-risk areas, where more than 50% of the population is likely to carry infections, fell from 218.6 million in 2000 to 183.5 million in 2010—a 16% decrease.
But the population living in areas where the risk of infection is considered moderate to high grew from 178.6 million to 280.1 million—a 57% increase.
And the population living in areas where risk is regarded as very low grew from 78.2 million to 128.2 million—a 64% increase.
The researchers also discovered that 10 countries harbor 87% of the population remaining at high risk of malaria transmission. These countries are Guinea, Togo, Mali, Mozambique, Burkina Faso, Ghana, Côte d’Ivoire, Uganda, Nigeria, and the Democratic Republic of Congo.
On the other hand, the team noted that 7 countries have levels of malaria transmission so low that eliminating the disease is a realistic goal. These countries are Cape Verde, Eritrea, South Africa, Ethiopia, Swaziland, Djibouti, and Mayotte.
“The results of our analysis are pause for thought,” said study author Robert Snow, PhD, also of the Kenya Medical Research Institute-Wellcome Trust Research Programme and the University of Oxford.
“On the one hand, it’s a glass half full, with several countries showing significant reductions in malaria transmission. And on the other, it’s a glass half empty, where, despite a decade of massive investment in malaria control, the populations living in several African countries are as likely to be infected with malaria in 2000 as they were 10 years later.” ![]()

Credit: CDC
Malaria prevalence maps indicate that, in 2010, nearly 184 million Africans were still living in areas where there is a high risk of contracting malaria, despite a decade of efforts to control the spread of the disease.
The maps showed that 40 African countries experienced reductions in childhood malaria transmission between 2000 and 2010.
Despite this progress, 57% of the population in malaria-endemic countries continued to live in areas of moderate to intense malaria transmission, with infection rates higher than 10%.
These findings are published in The Lancet.
Researchers compiled data from a collection of 26,746 community-based surveys of Plasmodium falciparum prevalence. The surveys covered 3,575,418 person observations from 44 malaria-endemic countries and territories in Africa since 1980.
“Health information systems in many African countries are weak, and it has been difficult to reliably estimate how many people get sick or die of malaria,” said study author Abdisalan Mohamed Noor, PhD, of the Kenya Medical Research Institute-Wellcome Trust Research Programme in Nairobi and the University of Oxford in the UK.
“The population surveys we used in this study are a more reliable indicator for tracking, and we hope our study will help countries assess their progress and adapt their strategies for more effective malaria control.”
Using model-based geostatistics, Dr Noor and his colleagues estimated the proportion of the population, aged 2 to 10 years old, infected with different levels of P falciparum across Africa in 2000 and 2010.
The researchers wanted to evaluate the effects of the Roll Back Malaria Partnership, which was launched in 2000 and resulted in a large increase in investments targeting malaria control.
The team found that the number of people living in high-risk areas, where more than 50% of the population is likely to carry infections, fell from 218.6 million in 2000 to 183.5 million in 2010—a 16% decrease.
But the population living in areas where the risk of infection is considered moderate to high grew from 178.6 million to 280.1 million—a 57% increase.
And the population living in areas where risk is regarded as very low grew from 78.2 million to 128.2 million—a 64% increase.
The researchers also discovered that 10 countries harbor 87% of the population remaining at high risk of malaria transmission. These countries are Guinea, Togo, Mali, Mozambique, Burkina Faso, Ghana, Côte d’Ivoire, Uganda, Nigeria, and the Democratic Republic of Congo.
On the other hand, the team noted that 7 countries have levels of malaria transmission so low that eliminating the disease is a realistic goal. These countries are Cape Verde, Eritrea, South Africa, Ethiopia, Swaziland, Djibouti, and Mayotte.
“The results of our analysis are pause for thought,” said study author Robert Snow, PhD, also of the Kenya Medical Research Institute-Wellcome Trust Research Programme and the University of Oxford.
“On the one hand, it’s a glass half full, with several countries showing significant reductions in malaria transmission. And on the other, it’s a glass half empty, where, despite a decade of massive investment in malaria control, the populations living in several African countries are as likely to be infected with malaria in 2000 as they were 10 years later.” ![]()

Credit: CDC
Malaria prevalence maps indicate that, in 2010, nearly 184 million Africans were still living in areas where there is a high risk of contracting malaria, despite a decade of efforts to control the spread of the disease.
The maps showed that 40 African countries experienced reductions in childhood malaria transmission between 2000 and 2010.
Despite this progress, 57% of the population in malaria-endemic countries continued to live in areas of moderate to intense malaria transmission, with infection rates higher than 10%.
These findings are published in The Lancet.
Researchers compiled data from a collection of 26,746 community-based surveys of Plasmodium falciparum prevalence. The surveys covered 3,575,418 person observations from 44 malaria-endemic countries and territories in Africa since 1980.
“Health information systems in many African countries are weak, and it has been difficult to reliably estimate how many people get sick or die of malaria,” said study author Abdisalan Mohamed Noor, PhD, of the Kenya Medical Research Institute-Wellcome Trust Research Programme in Nairobi and the University of Oxford in the UK.
“The population surveys we used in this study are a more reliable indicator for tracking, and we hope our study will help countries assess their progress and adapt their strategies for more effective malaria control.”
Using model-based geostatistics, Dr Noor and his colleagues estimated the proportion of the population, aged 2 to 10 years old, infected with different levels of P falciparum across Africa in 2000 and 2010.
The researchers wanted to evaluate the effects of the Roll Back Malaria Partnership, which was launched in 2000 and resulted in a large increase in investments targeting malaria control.
The team found that the number of people living in high-risk areas, where more than 50% of the population is likely to carry infections, fell from 218.6 million in 2000 to 183.5 million in 2010—a 16% decrease.
But the population living in areas where the risk of infection is considered moderate to high grew from 178.6 million to 280.1 million—a 57% increase.
And the population living in areas where risk is regarded as very low grew from 78.2 million to 128.2 million—a 64% increase.
The researchers also discovered that 10 countries harbor 87% of the population remaining at high risk of malaria transmission. These countries are Guinea, Togo, Mali, Mozambique, Burkina Faso, Ghana, Côte d’Ivoire, Uganda, Nigeria, and the Democratic Republic of Congo.
On the other hand, the team noted that 7 countries have levels of malaria transmission so low that eliminating the disease is a realistic goal. These countries are Cape Verde, Eritrea, South Africa, Ethiopia, Swaziland, Djibouti, and Mayotte.
“The results of our analysis are pause for thought,” said study author Robert Snow, PhD, also of the Kenya Medical Research Institute-Wellcome Trust Research Programme and the University of Oxford.
“On the one hand, it’s a glass half full, with several countries showing significant reductions in malaria transmission. And on the other, it’s a glass half empty, where, despite a decade of massive investment in malaria control, the populations living in several African countries are as likely to be infected with malaria in 2000 as they were 10 years later.” ![]()
Environment may play role in malaria transmission
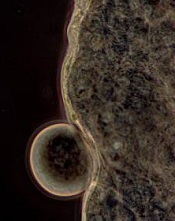
the wall of a mosquito midgut
Credit: Krijn Paaijmans
Researchers have found the environment can significantly influence whether or not Wolbachia bacteria will prevent mosquitoes from transmitting malaria.
“Bacteria in the genus Wolbachia represent a promising new tool for controlling malaria due to their demonstrated ability to block the development of the pathogen within Anopheles mosquitoes,” said study investigator Courtney Murdock, PhD, of Pennsylvania State University.
“However, much of the work on the Wolbachia-malaria interaction has been conducted under highly simplified laboratory conditions. In this study, we investigated the ability of Wolbachia to block transmission of malaria—Plasmodium—parasites across variable environmental conditions, which are more reflective of conditions in the field.”
Dr Murdock and her colleagues described this research in Nature Scientific Reports.
The researchers used the malaria parasite Plasmodium yoelii, which affects rodents, and the mosquito Anopheles stephensi as a model system to investigate whether Wolbachia would block the ability of the malaria parasite to infect the mosquitoes.
The team divided the mosquitoes into an uninfected control group and a group infected with Wolbachia. Next, they raised all groups of mosquitoes in incubators set to different experimental temperatures—68, 72, 75, 79, and 82 degrees Fahrenheit.
At 82 degrees, Wolbachia reduced the number of mosquitoes infected by malaria parasites, the number of malaria parasites within each mosquito, and the intensity of oocysts.
At 75 degrees, Wolbachia had no effect on the prevalence of malaria parasites but increased oocyst intensity. At 68 degrees, Wolbachia had no effect on the prevalence of parasites or the intensity of oocysts.
The researchers also identified a previously undiscovered effect of Wolbachia. Infection with the bacterium reduced the development of sporozoites across all temperatures. This suggests that Wolbachia and malaria parasites may compete for similar hosts.
“Typically, the more oocysts a mosquito has on its midgut, the more sporozoites it produces,” Dr Murdock said. “So, depending on the environmental temperature, Wolbachia either reduced, enhanced, or had no effect on the number of oocysts.”
“At 75 degrees Fahrenheit, Wolbachia-infected mosquitos had 3 times the numbers of oocysts relative to uninfected mosquitoes. Thus, we would predict these mosquitoes to produce more sporozoites. But instead, we see that this is not the case, and that is because Wolbachia infection significantly reduces the number of sporozoites produced per oocyst, regardless of the environmental temperature.”
“This effect counteracts the enhancement we see at 75 degrees Fahrenheit. How the influence of Wolbachia on parasite establishment and the production of sporozoites under different temperatures plays out to ultimately affect transmission remains to be determined.”
Dr Murdock and her colleagues plan to duplicate their experiment using a species of malaria parasite that affects humans to determine whether or not the temperature effects they observed occur in humans as well.
The team also intends to explore the effects of additional environmental variation—such as daily temperature fluctuation and differential access to food resources in the mosquito larval and adult environments—on the transmission-blocking ability of Wolbachia. ![]()

the wall of a mosquito midgut
Credit: Krijn Paaijmans
Researchers have found the environment can significantly influence whether or not Wolbachia bacteria will prevent mosquitoes from transmitting malaria.
“Bacteria in the genus Wolbachia represent a promising new tool for controlling malaria due to their demonstrated ability to block the development of the pathogen within Anopheles mosquitoes,” said study investigator Courtney Murdock, PhD, of Pennsylvania State University.
“However, much of the work on the Wolbachia-malaria interaction has been conducted under highly simplified laboratory conditions. In this study, we investigated the ability of Wolbachia to block transmission of malaria—Plasmodium—parasites across variable environmental conditions, which are more reflective of conditions in the field.”
Dr Murdock and her colleagues described this research in Nature Scientific Reports.
The researchers used the malaria parasite Plasmodium yoelii, which affects rodents, and the mosquito Anopheles stephensi as a model system to investigate whether Wolbachia would block the ability of the malaria parasite to infect the mosquitoes.
The team divided the mosquitoes into an uninfected control group and a group infected with Wolbachia. Next, they raised all groups of mosquitoes in incubators set to different experimental temperatures—68, 72, 75, 79, and 82 degrees Fahrenheit.
At 82 degrees, Wolbachia reduced the number of mosquitoes infected by malaria parasites, the number of malaria parasites within each mosquito, and the intensity of oocysts.
At 75 degrees, Wolbachia had no effect on the prevalence of malaria parasites but increased oocyst intensity. At 68 degrees, Wolbachia had no effect on the prevalence of parasites or the intensity of oocysts.
The researchers also identified a previously undiscovered effect of Wolbachia. Infection with the bacterium reduced the development of sporozoites across all temperatures. This suggests that Wolbachia and malaria parasites may compete for similar hosts.
“Typically, the more oocysts a mosquito has on its midgut, the more sporozoites it produces,” Dr Murdock said. “So, depending on the environmental temperature, Wolbachia either reduced, enhanced, or had no effect on the number of oocysts.”
“At 75 degrees Fahrenheit, Wolbachia-infected mosquitos had 3 times the numbers of oocysts relative to uninfected mosquitoes. Thus, we would predict these mosquitoes to produce more sporozoites. But instead, we see that this is not the case, and that is because Wolbachia infection significantly reduces the number of sporozoites produced per oocyst, regardless of the environmental temperature.”
“This effect counteracts the enhancement we see at 75 degrees Fahrenheit. How the influence of Wolbachia on parasite establishment and the production of sporozoites under different temperatures plays out to ultimately affect transmission remains to be determined.”
Dr Murdock and her colleagues plan to duplicate their experiment using a species of malaria parasite that affects humans to determine whether or not the temperature effects they observed occur in humans as well.
The team also intends to explore the effects of additional environmental variation—such as daily temperature fluctuation and differential access to food resources in the mosquito larval and adult environments—on the transmission-blocking ability of Wolbachia. ![]()

the wall of a mosquito midgut
Credit: Krijn Paaijmans
Researchers have found the environment can significantly influence whether or not Wolbachia bacteria will prevent mosquitoes from transmitting malaria.
“Bacteria in the genus Wolbachia represent a promising new tool for controlling malaria due to their demonstrated ability to block the development of the pathogen within Anopheles mosquitoes,” said study investigator Courtney Murdock, PhD, of Pennsylvania State University.
“However, much of the work on the Wolbachia-malaria interaction has been conducted under highly simplified laboratory conditions. In this study, we investigated the ability of Wolbachia to block transmission of malaria—Plasmodium—parasites across variable environmental conditions, which are more reflective of conditions in the field.”
Dr Murdock and her colleagues described this research in Nature Scientific Reports.
The researchers used the malaria parasite Plasmodium yoelii, which affects rodents, and the mosquito Anopheles stephensi as a model system to investigate whether Wolbachia would block the ability of the malaria parasite to infect the mosquitoes.
The team divided the mosquitoes into an uninfected control group and a group infected with Wolbachia. Next, they raised all groups of mosquitoes in incubators set to different experimental temperatures—68, 72, 75, 79, and 82 degrees Fahrenheit.
At 82 degrees, Wolbachia reduced the number of mosquitoes infected by malaria parasites, the number of malaria parasites within each mosquito, and the intensity of oocysts.
At 75 degrees, Wolbachia had no effect on the prevalence of malaria parasites but increased oocyst intensity. At 68 degrees, Wolbachia had no effect on the prevalence of parasites or the intensity of oocysts.
The researchers also identified a previously undiscovered effect of Wolbachia. Infection with the bacterium reduced the development of sporozoites across all temperatures. This suggests that Wolbachia and malaria parasites may compete for similar hosts.
“Typically, the more oocysts a mosquito has on its midgut, the more sporozoites it produces,” Dr Murdock said. “So, depending on the environmental temperature, Wolbachia either reduced, enhanced, or had no effect on the number of oocysts.”
“At 75 degrees Fahrenheit, Wolbachia-infected mosquitos had 3 times the numbers of oocysts relative to uninfected mosquitoes. Thus, we would predict these mosquitoes to produce more sporozoites. But instead, we see that this is not the case, and that is because Wolbachia infection significantly reduces the number of sporozoites produced per oocyst, regardless of the environmental temperature.”
“This effect counteracts the enhancement we see at 75 degrees Fahrenheit. How the influence of Wolbachia on parasite establishment and the production of sporozoites under different temperatures plays out to ultimately affect transmission remains to be determined.”
Dr Murdock and her colleagues plan to duplicate their experiment using a species of malaria parasite that affects humans to determine whether or not the temperature effects they observed occur in humans as well.
The team also intends to explore the effects of additional environmental variation—such as daily temperature fluctuation and differential access to food resources in the mosquito larval and adult environments—on the transmission-blocking ability of Wolbachia. ![]()
Group finds progenitors of ILCs

with ILCs (green), epithelial
cells (red), and nuclei (blue)
University of Pennsylvania
Scientists say they’ve discovered the progenitors of innate lymphoid cells (ILCs) in the liver of fetal mice and the bone marrow of adult mice.
ILCs are among the first components of the immune system to confront certain pathogens, yet the cells went undetected by researchers for a century.
“Scientists tend to look for immune cells in the blood, lymph nodes, or spleen,” said Albert Bendelac, PhD, of the University of Chicago in Illinois.
“That is precisely where you would not find these cells. Once they mature, they directly go to tissues, such as the gut or the skin. You seldom see them in blood.”
To understand how ILCs fit into the ecosystem of cells that fight off infections and cancers, Dr Bendelac’s team focused on finding ILCs’ source.
And they reported their findings in a letter to Nature.
The team noted that ILCs, which were first recognized 5 years ago, are rare. A mouse might have 200 million lymphocytes and only a few thousand ILCs.
But previous work on natural killer (NK) cells showed that ILCs express the transcription factor PLZF during their development.
So Dr Bendelac and his colleagues created mice with the gene for green fluorescent protein inserted into mouse DNA, just downstream from the PLZF gene. As a result, cells from mice that expressed PLZF appeared bright green under the microscope.
Nevertheless, finding the precursors to ILCs was not easy. The precursors are not in the blood, and, by the time they migrate to the lungs or gut, they have already matured into ILCs.
The researchers eventually found the precursor cells—known as ILCPs—in the liver of fetal mice and in the bone marrow of adult mice.
When the team purified the ILCPs, which still contained the GFP gene, and transferred them into mice that lacked ILCs, the precursors were able to reconstitute the 3 known types of ILCs—ILCs 1, 2, and 3.
“There were no B cells or T cells or myeloid cells—no other immune cells, just these,” Dr Bendelac said. “So we think the ILCP really is a committed precursor to innate lymphoid cells.”
To confirm their finding, the researchers designed mice in which PLZF gene expression was tied to the gene for diphtheria toxin. When the cells expressed PLZF, they also produced the toxin, which was lethal for those cells. The result was a mouse that had a normal immune system except that it completely lacked ILCs.
“ILCs are found in the most exposed tissues,” Dr Bendelac noted. “They are one of your first lines of defense. We now suspect they may also influence the ensuing adaptive immune response, priming the pump, influencing how T-helper cells respond.”
Each of the 3 types of ILCs has different properties and serves different functions. ILC1 cells help prevent viral infections and can detect and remove some cancerous cells. They are similar to NK cells, except that NK cells circulate in the blood, and ILC1s live in the gut and the liver.
ILC2s are found in the lungs, where they can detect and respond to parasites. But they can also initiate an allergic reaction and mucus hyper-secretion.
ILC3 cells cluster in the gut, where they help mediate interactions between the bowel and bacteria. When that balance is disturbed, they can accelerate inflammation and may play a role in inflammatory bowel disease.
Dr Bendelac said his group’s research provides “one more tool for understanding this complex system,” and it could help generate a “powerful new way to assess the function of innate lymphocytes.” ![]()

with ILCs (green), epithelial
cells (red), and nuclei (blue)
University of Pennsylvania
Scientists say they’ve discovered the progenitors of innate lymphoid cells (ILCs) in the liver of fetal mice and the bone marrow of adult mice.
ILCs are among the first components of the immune system to confront certain pathogens, yet the cells went undetected by researchers for a century.
“Scientists tend to look for immune cells in the blood, lymph nodes, or spleen,” said Albert Bendelac, PhD, of the University of Chicago in Illinois.
“That is precisely where you would not find these cells. Once they mature, they directly go to tissues, such as the gut or the skin. You seldom see them in blood.”
To understand how ILCs fit into the ecosystem of cells that fight off infections and cancers, Dr Bendelac’s team focused on finding ILCs’ source.
And they reported their findings in a letter to Nature.
The team noted that ILCs, which were first recognized 5 years ago, are rare. A mouse might have 200 million lymphocytes and only a few thousand ILCs.
But previous work on natural killer (NK) cells showed that ILCs express the transcription factor PLZF during their development.
So Dr Bendelac and his colleagues created mice with the gene for green fluorescent protein inserted into mouse DNA, just downstream from the PLZF gene. As a result, cells from mice that expressed PLZF appeared bright green under the microscope.
Nevertheless, finding the precursors to ILCs was not easy. The precursors are not in the blood, and, by the time they migrate to the lungs or gut, they have already matured into ILCs.
The researchers eventually found the precursor cells—known as ILCPs—in the liver of fetal mice and in the bone marrow of adult mice.
When the team purified the ILCPs, which still contained the GFP gene, and transferred them into mice that lacked ILCs, the precursors were able to reconstitute the 3 known types of ILCs—ILCs 1, 2, and 3.
“There were no B cells or T cells or myeloid cells—no other immune cells, just these,” Dr Bendelac said. “So we think the ILCP really is a committed precursor to innate lymphoid cells.”
To confirm their finding, the researchers designed mice in which PLZF gene expression was tied to the gene for diphtheria toxin. When the cells expressed PLZF, they also produced the toxin, which was lethal for those cells. The result was a mouse that had a normal immune system except that it completely lacked ILCs.
“ILCs are found in the most exposed tissues,” Dr Bendelac noted. “They are one of your first lines of defense. We now suspect they may also influence the ensuing adaptive immune response, priming the pump, influencing how T-helper cells respond.”
Each of the 3 types of ILCs has different properties and serves different functions. ILC1 cells help prevent viral infections and can detect and remove some cancerous cells. They are similar to NK cells, except that NK cells circulate in the blood, and ILC1s live in the gut and the liver.
ILC2s are found in the lungs, where they can detect and respond to parasites. But they can also initiate an allergic reaction and mucus hyper-secretion.
ILC3 cells cluster in the gut, where they help mediate interactions between the bowel and bacteria. When that balance is disturbed, they can accelerate inflammation and may play a role in inflammatory bowel disease.
Dr Bendelac said his group’s research provides “one more tool for understanding this complex system,” and it could help generate a “powerful new way to assess the function of innate lymphocytes.” ![]()

with ILCs (green), epithelial
cells (red), and nuclei (blue)
University of Pennsylvania
Scientists say they’ve discovered the progenitors of innate lymphoid cells (ILCs) in the liver of fetal mice and the bone marrow of adult mice.
ILCs are among the first components of the immune system to confront certain pathogens, yet the cells went undetected by researchers for a century.
“Scientists tend to look for immune cells in the blood, lymph nodes, or spleen,” said Albert Bendelac, PhD, of the University of Chicago in Illinois.
“That is precisely where you would not find these cells. Once they mature, they directly go to tissues, such as the gut or the skin. You seldom see them in blood.”
To understand how ILCs fit into the ecosystem of cells that fight off infections and cancers, Dr Bendelac’s team focused on finding ILCs’ source.
And they reported their findings in a letter to Nature.
The team noted that ILCs, which were first recognized 5 years ago, are rare. A mouse might have 200 million lymphocytes and only a few thousand ILCs.
But previous work on natural killer (NK) cells showed that ILCs express the transcription factor PLZF during their development.
So Dr Bendelac and his colleagues created mice with the gene for green fluorescent protein inserted into mouse DNA, just downstream from the PLZF gene. As a result, cells from mice that expressed PLZF appeared bright green under the microscope.
Nevertheless, finding the precursors to ILCs was not easy. The precursors are not in the blood, and, by the time they migrate to the lungs or gut, they have already matured into ILCs.
The researchers eventually found the precursor cells—known as ILCPs—in the liver of fetal mice and in the bone marrow of adult mice.
When the team purified the ILCPs, which still contained the GFP gene, and transferred them into mice that lacked ILCs, the precursors were able to reconstitute the 3 known types of ILCs—ILCs 1, 2, and 3.
“There were no B cells or T cells or myeloid cells—no other immune cells, just these,” Dr Bendelac said. “So we think the ILCP really is a committed precursor to innate lymphoid cells.”
To confirm their finding, the researchers designed mice in which PLZF gene expression was tied to the gene for diphtheria toxin. When the cells expressed PLZF, they also produced the toxin, which was lethal for those cells. The result was a mouse that had a normal immune system except that it completely lacked ILCs.
“ILCs are found in the most exposed tissues,” Dr Bendelac noted. “They are one of your first lines of defense. We now suspect they may also influence the ensuing adaptive immune response, priming the pump, influencing how T-helper cells respond.”
Each of the 3 types of ILCs has different properties and serves different functions. ILC1 cells help prevent viral infections and can detect and remove some cancerous cells. They are similar to NK cells, except that NK cells circulate in the blood, and ILC1s live in the gut and the liver.
ILC2s are found in the lungs, where they can detect and respond to parasites. But they can also initiate an allergic reaction and mucus hyper-secretion.
ILC3 cells cluster in the gut, where they help mediate interactions between the bowel and bacteria. When that balance is disturbed, they can accelerate inflammation and may play a role in inflammatory bowel disease.
Dr Bendelac said his group’s research provides “one more tool for understanding this complex system,” and it could help generate a “powerful new way to assess the function of innate lymphocytes.” ![]()
Rare outbreak in cancer clinic tied to saline flush

Credit: Rhoda Baer
The first reported outbreak of Tsukamurella species bloodstream infections was due to improper handling of intravenous saline, according to a report published in Infection Control and Hospital Epidemiology.
From September 2011 to May 2012, 15 immunocompromised patients treated at an outpatient oncology clinic in West Virginia developed infections with Tsukamurella, which are gram-positive bacteria that rarely cause disease in humans.
All patients had received a cancer diagnosis and had an indwelling central line, although central line types varied.
A case-control study revealed that the only risk factor for developing Tsukamurella infection was the receipt of a saline flush in September or October 2011, when clinic staff were using a common-source bag of saline.
Investigations by the West Virginia Bureau of Public Health (WVBPH) and the Centers for Disease Control and Prevention (CDC) uncovered several lapses in infection control procedures relating to the care of long-term intravenous catheters and preparation of chemotherapy for patients at the clinic.
However, these investigations also suggested that saline flush syringes were the likely source of infection.
So the WVBPH and the CDC recommended the clinic institute several changes to its infection prevention and control practices, including using pre-packaged, manufactured saline flushes.
After the clinic changed this practice, Tsukamurella bloodstream infections stopped occurring, further supporting the saline flush as the source of infection.
“This outbreak illustrates the need for outpatient clinics to follow proper infection control guidelines and medication preparation practices to minimize the risk of infection for patients with weakened immune systems,” said lead study author Isaac See, MD, of the CDC.
To that end, the CDC has developed a basic infection control plan tailored to outpatient oncology facilities.
The plan outlines policies and procedures needed to meet minimal requirements for patient safety, including the proper use and handling of injectable medications and correct procedures for assessing central lines. ![]()

Credit: Rhoda Baer
The first reported outbreak of Tsukamurella species bloodstream infections was due to improper handling of intravenous saline, according to a report published in Infection Control and Hospital Epidemiology.
From September 2011 to May 2012, 15 immunocompromised patients treated at an outpatient oncology clinic in West Virginia developed infections with Tsukamurella, which are gram-positive bacteria that rarely cause disease in humans.
All patients had received a cancer diagnosis and had an indwelling central line, although central line types varied.
A case-control study revealed that the only risk factor for developing Tsukamurella infection was the receipt of a saline flush in September or October 2011, when clinic staff were using a common-source bag of saline.
Investigations by the West Virginia Bureau of Public Health (WVBPH) and the Centers for Disease Control and Prevention (CDC) uncovered several lapses in infection control procedures relating to the care of long-term intravenous catheters and preparation of chemotherapy for patients at the clinic.
However, these investigations also suggested that saline flush syringes were the likely source of infection.
So the WVBPH and the CDC recommended the clinic institute several changes to its infection prevention and control practices, including using pre-packaged, manufactured saline flushes.
After the clinic changed this practice, Tsukamurella bloodstream infections stopped occurring, further supporting the saline flush as the source of infection.
“This outbreak illustrates the need for outpatient clinics to follow proper infection control guidelines and medication preparation practices to minimize the risk of infection for patients with weakened immune systems,” said lead study author Isaac See, MD, of the CDC.
To that end, the CDC has developed a basic infection control plan tailored to outpatient oncology facilities.
The plan outlines policies and procedures needed to meet minimal requirements for patient safety, including the proper use and handling of injectable medications and correct procedures for assessing central lines. ![]()

Credit: Rhoda Baer
The first reported outbreak of Tsukamurella species bloodstream infections was due to improper handling of intravenous saline, according to a report published in Infection Control and Hospital Epidemiology.
From September 2011 to May 2012, 15 immunocompromised patients treated at an outpatient oncology clinic in West Virginia developed infections with Tsukamurella, which are gram-positive bacteria that rarely cause disease in humans.
All patients had received a cancer diagnosis and had an indwelling central line, although central line types varied.
A case-control study revealed that the only risk factor for developing Tsukamurella infection was the receipt of a saline flush in September or October 2011, when clinic staff were using a common-source bag of saline.
Investigations by the West Virginia Bureau of Public Health (WVBPH) and the Centers for Disease Control and Prevention (CDC) uncovered several lapses in infection control procedures relating to the care of long-term intravenous catheters and preparation of chemotherapy for patients at the clinic.
However, these investigations also suggested that saline flush syringes were the likely source of infection.
So the WVBPH and the CDC recommended the clinic institute several changes to its infection prevention and control practices, including using pre-packaged, manufactured saline flushes.
After the clinic changed this practice, Tsukamurella bloodstream infections stopped occurring, further supporting the saline flush as the source of infection.
“This outbreak illustrates the need for outpatient clinics to follow proper infection control guidelines and medication preparation practices to minimize the risk of infection for patients with weakened immune systems,” said lead study author Isaac See, MD, of the CDC.
To that end, the CDC has developed a basic infection control plan tailored to outpatient oncology facilities.
The plan outlines policies and procedures needed to meet minimal requirements for patient safety, including the proper use and handling of injectable medications and correct procedures for assessing central lines. ![]()
Team develops new live-cell printing technology

Credit: Lidong Qin lab
A new technique allows scientists to print living cells onto any surface in virtually any shape, according to a paper published in Proceedings of the National Academy of Sciences.
The approach, called Block-Cell-Printing (BloC-Printing), produces 2-D cell arrays in as little as half an hour, prints the cells as close together as 5 μm, and allows for the use of different cell types.
And unlike similar work using inkjet printing approaches, almost all cells survive BloC-Printing.
“Cell printing is used in so many different ways now—for drug development and in studies of tissue regeneration, cell function, and cell-cell communication,” said study author Lidong Qin, PhD, of Houston Methodist Research Institute in Texas.
“Such things can only be done when cells are alive and active. A survival rate of 50% to 80% is typical as cells exit the inkjet nozzles. By comparison, we are seeing close to 100% of cells in BloC-Printing survive the printing process.”
On the other hand, Dr Qin noted that inkjet printing remains faster than BloC-Printing. And BloC-Printing cannot yet print multi-layer structures as inkjetting can.
BloC-Printing manipulates microfluidic physics to guide living cells into hook-like traps in a silicone mold. Cells flow down a column in the mold, past trapped cells to the next available slot, eventually creating a line of cells (in a grid of such lines).
The position and spacing of the traps and the shape of the channel navigated by the cells is fully configurable during the mold’s creation. When the mold is lifted away, the living cells remain behind, adhering to the growth medium or other substrate in prescribed formation.
Dr Qin’s group tested BloC-Printing for its utility in studying breast cancer cells and primary neurons.
By arranging the cancer cells in a grid and examining their growth in comparison with a non-metastatic control, the researchers found they could easily characterize the metastatic potential of the cancer cells.
“We looked at cancer cells for their protrusion generation capability, which correlates to their malignancy level,” Dr Qin said. “Longer protrusion means more aggressive cancer cells. The measurement may help to diagnose a cancer’s stage.”
The researchers also printed a grid of brain cells and gave the cells time to form synaptic and autaptic junctions.
“The cell junctions we created may be useful for future neuron signal transduction and axon regeneration studies,” Dr Qin said. “Such work could be helpful in understanding Alzheimer’s disease and other neurodegenerative diseases.”
While it is too early to predict the market cost of BloC-Printing, Dr Qin said the materials of a single BloC mold cost about $1. After the mold has been fabricated and delivered, a researcher only needs a syringe, a carefully prepared suspension of living cells, a Petri dish, and a steady hand.
“BloC-Printing can be combined with molecular printing for many types of drug screening, RNA interference, and molecule-cell interaction studies,” Dr Qin said. “We believe the technology has big potential.” ![]()

Credit: Lidong Qin lab
A new technique allows scientists to print living cells onto any surface in virtually any shape, according to a paper published in Proceedings of the National Academy of Sciences.
The approach, called Block-Cell-Printing (BloC-Printing), produces 2-D cell arrays in as little as half an hour, prints the cells as close together as 5 μm, and allows for the use of different cell types.
And unlike similar work using inkjet printing approaches, almost all cells survive BloC-Printing.
“Cell printing is used in so many different ways now—for drug development and in studies of tissue regeneration, cell function, and cell-cell communication,” said study author Lidong Qin, PhD, of Houston Methodist Research Institute in Texas.
“Such things can only be done when cells are alive and active. A survival rate of 50% to 80% is typical as cells exit the inkjet nozzles. By comparison, we are seeing close to 100% of cells in BloC-Printing survive the printing process.”
On the other hand, Dr Qin noted that inkjet printing remains faster than BloC-Printing. And BloC-Printing cannot yet print multi-layer structures as inkjetting can.
BloC-Printing manipulates microfluidic physics to guide living cells into hook-like traps in a silicone mold. Cells flow down a column in the mold, past trapped cells to the next available slot, eventually creating a line of cells (in a grid of such lines).
The position and spacing of the traps and the shape of the channel navigated by the cells is fully configurable during the mold’s creation. When the mold is lifted away, the living cells remain behind, adhering to the growth medium or other substrate in prescribed formation.
Dr Qin’s group tested BloC-Printing for its utility in studying breast cancer cells and primary neurons.
By arranging the cancer cells in a grid and examining their growth in comparison with a non-metastatic control, the researchers found they could easily characterize the metastatic potential of the cancer cells.
“We looked at cancer cells for their protrusion generation capability, which correlates to their malignancy level,” Dr Qin said. “Longer protrusion means more aggressive cancer cells. The measurement may help to diagnose a cancer’s stage.”
The researchers also printed a grid of brain cells and gave the cells time to form synaptic and autaptic junctions.
“The cell junctions we created may be useful for future neuron signal transduction and axon regeneration studies,” Dr Qin said. “Such work could be helpful in understanding Alzheimer’s disease and other neurodegenerative diseases.”
While it is too early to predict the market cost of BloC-Printing, Dr Qin said the materials of a single BloC mold cost about $1. After the mold has been fabricated and delivered, a researcher only needs a syringe, a carefully prepared suspension of living cells, a Petri dish, and a steady hand.
“BloC-Printing can be combined with molecular printing for many types of drug screening, RNA interference, and molecule-cell interaction studies,” Dr Qin said. “We believe the technology has big potential.” ![]()

Credit: Lidong Qin lab
A new technique allows scientists to print living cells onto any surface in virtually any shape, according to a paper published in Proceedings of the National Academy of Sciences.
The approach, called Block-Cell-Printing (BloC-Printing), produces 2-D cell arrays in as little as half an hour, prints the cells as close together as 5 μm, and allows for the use of different cell types.
And unlike similar work using inkjet printing approaches, almost all cells survive BloC-Printing.
“Cell printing is used in so many different ways now—for drug development and in studies of tissue regeneration, cell function, and cell-cell communication,” said study author Lidong Qin, PhD, of Houston Methodist Research Institute in Texas.
“Such things can only be done when cells are alive and active. A survival rate of 50% to 80% is typical as cells exit the inkjet nozzles. By comparison, we are seeing close to 100% of cells in BloC-Printing survive the printing process.”
On the other hand, Dr Qin noted that inkjet printing remains faster than BloC-Printing. And BloC-Printing cannot yet print multi-layer structures as inkjetting can.
BloC-Printing manipulates microfluidic physics to guide living cells into hook-like traps in a silicone mold. Cells flow down a column in the mold, past trapped cells to the next available slot, eventually creating a line of cells (in a grid of such lines).
The position and spacing of the traps and the shape of the channel navigated by the cells is fully configurable during the mold’s creation. When the mold is lifted away, the living cells remain behind, adhering to the growth medium or other substrate in prescribed formation.
Dr Qin’s group tested BloC-Printing for its utility in studying breast cancer cells and primary neurons.
By arranging the cancer cells in a grid and examining their growth in comparison with a non-metastatic control, the researchers found they could easily characterize the metastatic potential of the cancer cells.
“We looked at cancer cells for their protrusion generation capability, which correlates to their malignancy level,” Dr Qin said. “Longer protrusion means more aggressive cancer cells. The measurement may help to diagnose a cancer’s stage.”
The researchers also printed a grid of brain cells and gave the cells time to form synaptic and autaptic junctions.
“The cell junctions we created may be useful for future neuron signal transduction and axon regeneration studies,” Dr Qin said. “Such work could be helpful in understanding Alzheimer’s disease and other neurodegenerative diseases.”
While it is too early to predict the market cost of BloC-Printing, Dr Qin said the materials of a single BloC mold cost about $1. After the mold has been fabricated and delivered, a researcher only needs a syringe, a carefully prepared suspension of living cells, a Petri dish, and a steady hand.
“BloC-Printing can be combined with molecular printing for many types of drug screening, RNA interference, and molecule-cell interaction studies,” Dr Qin said. “We believe the technology has big potential.” ![]()
Improving the efficacy of etoposide

after treatment with etoposide
Credit: CNIO
A compound that interferes with the cell cycle can increase the antineoplastic effects of etoposide, according to research published in Cell Reports.
Etoposide works by inhibiting topoisomerase II (TOP2), a protein needed for DNA repair during cell division.
Researchers discovered a relationship between TOP2 and Cdh1, a protein that (along with Cdc20) controls cell division by activating the anaphase-promoting complex/cyclosome (APC/C).
So the team hypothesized that combining etoposide with a compound that inhibits Cdh1 might improve etoposide’s antineoplastic effects. Experiments in cancer cell lines confirmed this theory.
Marcos Malumbres, PhD, of the Spanish National Cancer Research Centre (CNIO) in Madrid, and his colleagues began this research by investigating Cdh1 in vitro and in mouse models.
The team found that a decrease in Cdh1 activity increases cells’ TOP2 levels. So they decided to combine etoposide with a Cdh1 inhibitor and evaluate the effect on cancer cells, which divide more than normal cells and therefore have a greater dependency on TOP2 to maintain DNA integrity.
The researchers tested proTAME, a small molecule that targets APC/C-Cdh1 and APC/C-Cdc20, in combination with etoposide. And they found the drugs had a synergistic effect against cancer cells.
In experiments with a lung cancer cell line (A549) and 2 breast cancer cell lines (HeLa and MCF7), administering proTAME and etoposide together proved more effective than administering either compound alone.
The researchers believe these findings could apply to other malignancies as well. Etoposide has demonstrated activity against a number of cancers, including leukemias, lymphomas, and multiple myeloma.
The team said their next step is to study the etoposide-proTAME combination in patients and investigate the malignancies in which this therapeutic strategy would be most effective.
The researchers also noted that previous studies have shown Cdh1 is inactive in some patients due to various oncogenic mutations. So stratifying patients according to their tumor’s Cdh1 status could optimize treatment with etoposide. ![]()

after treatment with etoposide
Credit: CNIO
A compound that interferes with the cell cycle can increase the antineoplastic effects of etoposide, according to research published in Cell Reports.
Etoposide works by inhibiting topoisomerase II (TOP2), a protein needed for DNA repair during cell division.
Researchers discovered a relationship between TOP2 and Cdh1, a protein that (along with Cdc20) controls cell division by activating the anaphase-promoting complex/cyclosome (APC/C).
So the team hypothesized that combining etoposide with a compound that inhibits Cdh1 might improve etoposide’s antineoplastic effects. Experiments in cancer cell lines confirmed this theory.
Marcos Malumbres, PhD, of the Spanish National Cancer Research Centre (CNIO) in Madrid, and his colleagues began this research by investigating Cdh1 in vitro and in mouse models.
The team found that a decrease in Cdh1 activity increases cells’ TOP2 levels. So they decided to combine etoposide with a Cdh1 inhibitor and evaluate the effect on cancer cells, which divide more than normal cells and therefore have a greater dependency on TOP2 to maintain DNA integrity.
The researchers tested proTAME, a small molecule that targets APC/C-Cdh1 and APC/C-Cdc20, in combination with etoposide. And they found the drugs had a synergistic effect against cancer cells.
In experiments with a lung cancer cell line (A549) and 2 breast cancer cell lines (HeLa and MCF7), administering proTAME and etoposide together proved more effective than administering either compound alone.
The researchers believe these findings could apply to other malignancies as well. Etoposide has demonstrated activity against a number of cancers, including leukemias, lymphomas, and multiple myeloma.
The team said their next step is to study the etoposide-proTAME combination in patients and investigate the malignancies in which this therapeutic strategy would be most effective.
The researchers also noted that previous studies have shown Cdh1 is inactive in some patients due to various oncogenic mutations. So stratifying patients according to their tumor’s Cdh1 status could optimize treatment with etoposide. ![]()

after treatment with etoposide
Credit: CNIO
A compound that interferes with the cell cycle can increase the antineoplastic effects of etoposide, according to research published in Cell Reports.
Etoposide works by inhibiting topoisomerase II (TOP2), a protein needed for DNA repair during cell division.
Researchers discovered a relationship between TOP2 and Cdh1, a protein that (along with Cdc20) controls cell division by activating the anaphase-promoting complex/cyclosome (APC/C).
So the team hypothesized that combining etoposide with a compound that inhibits Cdh1 might improve etoposide’s antineoplastic effects. Experiments in cancer cell lines confirmed this theory.
Marcos Malumbres, PhD, of the Spanish National Cancer Research Centre (CNIO) in Madrid, and his colleagues began this research by investigating Cdh1 in vitro and in mouse models.
The team found that a decrease in Cdh1 activity increases cells’ TOP2 levels. So they decided to combine etoposide with a Cdh1 inhibitor and evaluate the effect on cancer cells, which divide more than normal cells and therefore have a greater dependency on TOP2 to maintain DNA integrity.
The researchers tested proTAME, a small molecule that targets APC/C-Cdh1 and APC/C-Cdc20, in combination with etoposide. And they found the drugs had a synergistic effect against cancer cells.
In experiments with a lung cancer cell line (A549) and 2 breast cancer cell lines (HeLa and MCF7), administering proTAME and etoposide together proved more effective than administering either compound alone.
The researchers believe these findings could apply to other malignancies as well. Etoposide has demonstrated activity against a number of cancers, including leukemias, lymphomas, and multiple myeloma.
The team said their next step is to study the etoposide-proTAME combination in patients and investigate the malignancies in which this therapeutic strategy would be most effective.
The researchers also noted that previous studies have shown Cdh1 is inactive in some patients due to various oncogenic mutations. So stratifying patients according to their tumor’s Cdh1 status could optimize treatment with etoposide. ![]()
National plan to lower HAIs shows signs of success, investigators find

Credit: CDC
New research suggests a federally sponsored plan to decrease the incidence of healthcare-acquired infections (HAIs) in the US was successful in addressing the challenges of prioritizing and coordinating strategies.
The plan has also been associated with reductions in the rates of HAIs, with progress made toward most targets where data are available.
Descriptions of the plan and its initial results appear in a series of articles published in a supplement to the February issue of Medical Care.
“Much progress has been made in raising awareness of and developing strategies for curbing the life-threatening infections that strike patients too often when they are receiving medical care,” said Katherine Kahn, MD, a leader of the project and professor at the Geffen School of Medicine at the University of California, Los Angeles.
“In order to make even more progress, we need to build our systems of care to be safer within and across hospitals, nursing homes, clinics, and community settings.”
In 2009, the US Department of Health and Humans Services released a national plan aimed at preventing HAIs, called “National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.”
Researchers performed an evaluation of the first few years of the plan, reviewing the structure of the effort, as well as the results thus far.
The plan focuses on evidence-based strategies, such as considering the benefits and risks when deciding about the use and duration of treatments like antibiotics.
Most of the prevention initiatives have focused on hospital settings, but the action plan has focused attention on efforts in other care settings, such as outpatient surgery centers, kidney dialysis centers, and long-term care facilities.
The investigators said these efforts have likely contributed to stakeholders’ reported perceptions of greater momentum in adopting strategies to prevent HAIs.
The national plan has generated clinical, political, and financial support for the complex efforts required to eliminate HAIs across federal, regional, state, and local settings.
Despite an influx of federal funding to support elimination of HAIs, the researchers said ongoing dedicated resources will be required to maintain momentum and sustain efforts made to date.
On the other hand, because future funding for efforts to further reduce HAIs is unclear, the investigators said it may be best to incorporate the efforts into the overall movement to improve patient safety. ![]()

Credit: CDC
New research suggests a federally sponsored plan to decrease the incidence of healthcare-acquired infections (HAIs) in the US was successful in addressing the challenges of prioritizing and coordinating strategies.
The plan has also been associated with reductions in the rates of HAIs, with progress made toward most targets where data are available.
Descriptions of the plan and its initial results appear in a series of articles published in a supplement to the February issue of Medical Care.
“Much progress has been made in raising awareness of and developing strategies for curbing the life-threatening infections that strike patients too often when they are receiving medical care,” said Katherine Kahn, MD, a leader of the project and professor at the Geffen School of Medicine at the University of California, Los Angeles.
“In order to make even more progress, we need to build our systems of care to be safer within and across hospitals, nursing homes, clinics, and community settings.”
In 2009, the US Department of Health and Humans Services released a national plan aimed at preventing HAIs, called “National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.”
Researchers performed an evaluation of the first few years of the plan, reviewing the structure of the effort, as well as the results thus far.
The plan focuses on evidence-based strategies, such as considering the benefits and risks when deciding about the use and duration of treatments like antibiotics.
Most of the prevention initiatives have focused on hospital settings, but the action plan has focused attention on efforts in other care settings, such as outpatient surgery centers, kidney dialysis centers, and long-term care facilities.
The investigators said these efforts have likely contributed to stakeholders’ reported perceptions of greater momentum in adopting strategies to prevent HAIs.
The national plan has generated clinical, political, and financial support for the complex efforts required to eliminate HAIs across federal, regional, state, and local settings.
Despite an influx of federal funding to support elimination of HAIs, the researchers said ongoing dedicated resources will be required to maintain momentum and sustain efforts made to date.
On the other hand, because future funding for efforts to further reduce HAIs is unclear, the investigators said it may be best to incorporate the efforts into the overall movement to improve patient safety. ![]()

Credit: CDC
New research suggests a federally sponsored plan to decrease the incidence of healthcare-acquired infections (HAIs) in the US was successful in addressing the challenges of prioritizing and coordinating strategies.
The plan has also been associated with reductions in the rates of HAIs, with progress made toward most targets where data are available.
Descriptions of the plan and its initial results appear in a series of articles published in a supplement to the February issue of Medical Care.
“Much progress has been made in raising awareness of and developing strategies for curbing the life-threatening infections that strike patients too often when they are receiving medical care,” said Katherine Kahn, MD, a leader of the project and professor at the Geffen School of Medicine at the University of California, Los Angeles.
“In order to make even more progress, we need to build our systems of care to be safer within and across hospitals, nursing homes, clinics, and community settings.”
In 2009, the US Department of Health and Humans Services released a national plan aimed at preventing HAIs, called “National Action Plan to Prevent Health Care-Associated Infections: Road Map to Elimination.”
Researchers performed an evaluation of the first few years of the plan, reviewing the structure of the effort, as well as the results thus far.
The plan focuses on evidence-based strategies, such as considering the benefits and risks when deciding about the use and duration of treatments like antibiotics.
Most of the prevention initiatives have focused on hospital settings, but the action plan has focused attention on efforts in other care settings, such as outpatient surgery centers, kidney dialysis centers, and long-term care facilities.
The investigators said these efforts have likely contributed to stakeholders’ reported perceptions of greater momentum in adopting strategies to prevent HAIs.
The national plan has generated clinical, political, and financial support for the complex efforts required to eliminate HAIs across federal, regional, state, and local settings.
Despite an influx of federal funding to support elimination of HAIs, the researchers said ongoing dedicated resources will be required to maintain momentum and sustain efforts made to date.
On the other hand, because future funding for efforts to further reduce HAIs is unclear, the investigators said it may be best to incorporate the efforts into the overall movement to improve patient safety.
Team discovers global regulator of RNA editing
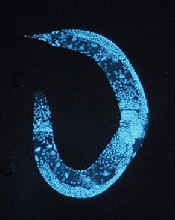
Credit: UC San Diego
School of Medicine
Scientists say they have identified a protein that broadly regulates how genetic information transcribed from DNA to messenger RNA (mRNA) is processed and ultimately translated into the myriad proteins necessary for life.
The group’s work shows that the protein ADR-1 binds to mRNA and then enhances RNA editing.
This process allows a gene to be present as multiple mRNAs that can each affect gene expression differently.
The research appears in Cell Reports.
“Problems with RNA editing show up in many human diseases, including those of neurodegeneration, cancer, and blood disorders,” said study author Gene Yeo, PhD, of the University of California, San Diego.
“This is the first time that a single protein has been identified that broadly regulates RNA editing. There are probably hundreds more. Our approach provides
a method to screen for them and opens up new ways to study human biology and disease.”
Using the model organism Caenorhabditis elegans, Dr Yeo and his colleagues identified more than 400 new mRNA editing sites. As the majority of them were regulated by ADR-1, the team declared the protein the first global regulator of RNA editing.
“What we’ve determined is that this protein’s ability to alter editing of mRNAs is not specific to just a few genes, but, instead, its ability to bind to mRNAs is required for proper RNA editing of most mRNAs,” said study author Michael C. Washburn, of Indiana University in Bloomington.
The group found that the region of ADR-1 protein that binds to target mRNAs in C elegans is required for regulating editing. This region is present in many human proteins, and a protein similar to ADR-1 is specifically expressed in human neurons.
“So it is likely that a similar mechanism exists to regulate editing in humans,” said study author Heather A. Hundley, PhD, also of Indiana University.
“Further work in our lab will be aimed at understanding the detailed mechanism of how these proteins regulate editing, in turn providing an inroad to developing therapeutics that modulate editing for the treatment of human diseases.”
C elegans, like humans, highly expresses a family of proteins in the nervous system called adenosine deaminases acting on RNA (ADARs), a family that includes ADR-1.
ADARs change specific nucleotides in RNA in a process called adenosine-to-inosine editing (A-to-I editing) that diversifies genetic information to specify different amino acids, splice sites, and structures.
Scientists currently estimate there are between 400,000 and 1 million A-to-I editing events in noncoding regions of the human transcriptome.
Newly synthesized RNA encodes the exact information found in DNA. But it’s when later RNA editing occurs that RNA gets altered, and this change is most often catalyzed by ADARs.
“One thing we also know is that ADAR protein levels are not altered in disease, implying that other mechanisms are also at work regulating ADAR-mediated RNA editing,” Dr Hundley said. “By identifying this major regulator of noncoding editing in C elegans, we can now focus on dissecting the regulatory mechanism and determining the conservation of this regulatory protein in human cells.”

Credit: UC San Diego
School of Medicine
Scientists say they have identified a protein that broadly regulates how genetic information transcribed from DNA to messenger RNA (mRNA) is processed and ultimately translated into the myriad proteins necessary for life.
The group’s work shows that the protein ADR-1 binds to mRNA and then enhances RNA editing.
This process allows a gene to be present as multiple mRNAs that can each affect gene expression differently.
The research appears in Cell Reports.
“Problems with RNA editing show up in many human diseases, including those of neurodegeneration, cancer, and blood disorders,” said study author Gene Yeo, PhD, of the University of California, San Diego.
“This is the first time that a single protein has been identified that broadly regulates RNA editing. There are probably hundreds more. Our approach provides
a method to screen for them and opens up new ways to study human biology and disease.”
Using the model organism Caenorhabditis elegans, Dr Yeo and his colleagues identified more than 400 new mRNA editing sites. As the majority of them were regulated by ADR-1, the team declared the protein the first global regulator of RNA editing.
“What we’ve determined is that this protein’s ability to alter editing of mRNAs is not specific to just a few genes, but, instead, its ability to bind to mRNAs is required for proper RNA editing of most mRNAs,” said study author Michael C. Washburn, of Indiana University in Bloomington.
The group found that the region of ADR-1 protein that binds to target mRNAs in C elegans is required for regulating editing. This region is present in many human proteins, and a protein similar to ADR-1 is specifically expressed in human neurons.
“So it is likely that a similar mechanism exists to regulate editing in humans,” said study author Heather A. Hundley, PhD, also of Indiana University.
“Further work in our lab will be aimed at understanding the detailed mechanism of how these proteins regulate editing, in turn providing an inroad to developing therapeutics that modulate editing for the treatment of human diseases.”
C elegans, like humans, highly expresses a family of proteins in the nervous system called adenosine deaminases acting on RNA (ADARs), a family that includes ADR-1.
ADARs change specific nucleotides in RNA in a process called adenosine-to-inosine editing (A-to-I editing) that diversifies genetic information to specify different amino acids, splice sites, and structures.
Scientists currently estimate there are between 400,000 and 1 million A-to-I editing events in noncoding regions of the human transcriptome.
Newly synthesized RNA encodes the exact information found in DNA. But it’s when later RNA editing occurs that RNA gets altered, and this change is most often catalyzed by ADARs.
“One thing we also know is that ADAR protein levels are not altered in disease, implying that other mechanisms are also at work regulating ADAR-mediated RNA editing,” Dr Hundley said. “By identifying this major regulator of noncoding editing in C elegans, we can now focus on dissecting the regulatory mechanism and determining the conservation of this regulatory protein in human cells.”

Credit: UC San Diego
School of Medicine
Scientists say they have identified a protein that broadly regulates how genetic information transcribed from DNA to messenger RNA (mRNA) is processed and ultimately translated into the myriad proteins necessary for life.
The group’s work shows that the protein ADR-1 binds to mRNA and then enhances RNA editing.
This process allows a gene to be present as multiple mRNAs that can each affect gene expression differently.
The research appears in Cell Reports.
“Problems with RNA editing show up in many human diseases, including those of neurodegeneration, cancer, and blood disorders,” said study author Gene Yeo, PhD, of the University of California, San Diego.
“This is the first time that a single protein has been identified that broadly regulates RNA editing. There are probably hundreds more. Our approach provides
a method to screen for them and opens up new ways to study human biology and disease.”
Using the model organism Caenorhabditis elegans, Dr Yeo and his colleagues identified more than 400 new mRNA editing sites. As the majority of them were regulated by ADR-1, the team declared the protein the first global regulator of RNA editing.
“What we’ve determined is that this protein’s ability to alter editing of mRNAs is not specific to just a few genes, but, instead, its ability to bind to mRNAs is required for proper RNA editing of most mRNAs,” said study author Michael C. Washburn, of Indiana University in Bloomington.
The group found that the region of ADR-1 protein that binds to target mRNAs in C elegans is required for regulating editing. This region is present in many human proteins, and a protein similar to ADR-1 is specifically expressed in human neurons.
“So it is likely that a similar mechanism exists to regulate editing in humans,” said study author Heather A. Hundley, PhD, also of Indiana University.
“Further work in our lab will be aimed at understanding the detailed mechanism of how these proteins regulate editing, in turn providing an inroad to developing therapeutics that modulate editing for the treatment of human diseases.”
C elegans, like humans, highly expresses a family of proteins in the nervous system called adenosine deaminases acting on RNA (ADARs), a family that includes ADR-1.
ADARs change specific nucleotides in RNA in a process called adenosine-to-inosine editing (A-to-I editing) that diversifies genetic information to specify different amino acids, splice sites, and structures.
Scientists currently estimate there are between 400,000 and 1 million A-to-I editing events in noncoding regions of the human transcriptome.
Newly synthesized RNA encodes the exact information found in DNA. But it’s when later RNA editing occurs that RNA gets altered, and this change is most often catalyzed by ADARs.
“One thing we also know is that ADAR protein levels are not altered in disease, implying that other mechanisms are also at work regulating ADAR-mediated RNA editing,” Dr Hundley said. “By identifying this major regulator of noncoding editing in C elegans, we can now focus on dissecting the regulatory mechanism and determining the conservation of this regulatory protein in human cells.”
Leaders agree on SGR bill but funding still a question

Credit: Lawrence Jackson
Leaders from the US Senate and House of Representatives have agreed on a plan to replace the Medicare sustainable growth rate (SGR) formula, but they must still agree on how to pay for it.
The group’s bill, called the SGR Repeal and Medicare Provider Payment Modernization Act of 2014, would remove the threat of a 23.7% cut in Medicare payments that is set to take effect on April 1.
And it would provide annual payment updates of 0.5% for the next 5 years, as Medicare transitions to a payment system designed to reward physicians based on care quality, not quantity.
“This is a significant step forward in our long-standing effort to replace the flawed physician update formula with a 21st-century system focused on quality and value rather than the quantity of services,” said House Ways and Means Committee Ranking Member Sander Levin (D-Mich.).
“For the first time in many years, we have agreement among the bipartisan leadership of the 3 committees that oversee Medicare. Now, we have to turn to the thorny issue of offsets.”
The Congressional Budget Office has estimated that the new plan would increase direct spending by $150.4 billion from 2014 to 2023, although other sources have said the cost would be about $126 billion for the same time period.
Repealing the SGR
The SGR calls for annual, automatic cuts in Medicare payments to physicians. It was established in 1997 to control physician spending, but, over the years, the necessary cuts have not occurred.
Since 2003, Congress has spent about $150 billion to provide short-term fixes to spare physicians from the cuts. Without another short-term fix or legislation to permanently eliminate the SGR, physicians would see a 23.7% cut in Medicare payments starting April 1.
For years, Congress has been trying to eliminate the SGR, but agreeing on a plan to do so—and a way to pay for it—has proven difficult. Now, Congress says it has brought “renewed commitment to repealing and replacing the flawed SGR update mechanism.”
Provisions of the bill
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 would institute a 0.5% annual payment increase for physicians from 2014 through 2018. According to Congress, this would help the transition to a merit-based incentive payment system (MIPS), beginning in 2018.
The MIPS consolidates the 3 existing incentive programs—the Physician Quality Reporting System, the Value-Based Payment Modifier, and the Meaningful Use of Electronic Health Records—into a single program that would reward providers who meet performance thresholds.
The bill would also give a 5% bonus to providers who receive a significant portion of their revenue from an alternative payment model (APM) or patient-centered medical home (PCMH). Participants would need to receive at least 25% of their Medicare revenue through an APM in 2018-2019, and this threshold would increase over time.
The bill would provide incentives for participation in private-payer APMs as well. And it would establish a technical advisory committee to review and recommend physician-developed APMs based on criteria developed through an open-comment process.
Finally, the bill would expand the use of Medicare data. Quality and utilization data would be posted on the Physician Compare website with the goal of helping patients to make more informed decisions about their care.
Furthermore, “qualified entities” would be allowed to provide analyses and underlying data to providers, although this provision is subject to privacy and security laws. And qualified clinical data registries would be allowed to purchase claims data.
To read the bill in full, visit Congress.gov.

Credit: Lawrence Jackson
Leaders from the US Senate and House of Representatives have agreed on a plan to replace the Medicare sustainable growth rate (SGR) formula, but they must still agree on how to pay for it.
The group’s bill, called the SGR Repeal and Medicare Provider Payment Modernization Act of 2014, would remove the threat of a 23.7% cut in Medicare payments that is set to take effect on April 1.
And it would provide annual payment updates of 0.5% for the next 5 years, as Medicare transitions to a payment system designed to reward physicians based on care quality, not quantity.
“This is a significant step forward in our long-standing effort to replace the flawed physician update formula with a 21st-century system focused on quality and value rather than the quantity of services,” said House Ways and Means Committee Ranking Member Sander Levin (D-Mich.).
“For the first time in many years, we have agreement among the bipartisan leadership of the 3 committees that oversee Medicare. Now, we have to turn to the thorny issue of offsets.”
The Congressional Budget Office has estimated that the new plan would increase direct spending by $150.4 billion from 2014 to 2023, although other sources have said the cost would be about $126 billion for the same time period.
Repealing the SGR
The SGR calls for annual, automatic cuts in Medicare payments to physicians. It was established in 1997 to control physician spending, but, over the years, the necessary cuts have not occurred.
Since 2003, Congress has spent about $150 billion to provide short-term fixes to spare physicians from the cuts. Without another short-term fix or legislation to permanently eliminate the SGR, physicians would see a 23.7% cut in Medicare payments starting April 1.
For years, Congress has been trying to eliminate the SGR, but agreeing on a plan to do so—and a way to pay for it—has proven difficult. Now, Congress says it has brought “renewed commitment to repealing and replacing the flawed SGR update mechanism.”
Provisions of the bill
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 would institute a 0.5% annual payment increase for physicians from 2014 through 2018. According to Congress, this would help the transition to a merit-based incentive payment system (MIPS), beginning in 2018.
The MIPS consolidates the 3 existing incentive programs—the Physician Quality Reporting System, the Value-Based Payment Modifier, and the Meaningful Use of Electronic Health Records—into a single program that would reward providers who meet performance thresholds.
The bill would also give a 5% bonus to providers who receive a significant portion of their revenue from an alternative payment model (APM) or patient-centered medical home (PCMH). Participants would need to receive at least 25% of their Medicare revenue through an APM in 2018-2019, and this threshold would increase over time.
The bill would provide incentives for participation in private-payer APMs as well. And it would establish a technical advisory committee to review and recommend physician-developed APMs based on criteria developed through an open-comment process.
Finally, the bill would expand the use of Medicare data. Quality and utilization data would be posted on the Physician Compare website with the goal of helping patients to make more informed decisions about their care.
Furthermore, “qualified entities” would be allowed to provide analyses and underlying data to providers, although this provision is subject to privacy and security laws. And qualified clinical data registries would be allowed to purchase claims data.
To read the bill in full, visit Congress.gov.

Credit: Lawrence Jackson
Leaders from the US Senate and House of Representatives have agreed on a plan to replace the Medicare sustainable growth rate (SGR) formula, but they must still agree on how to pay for it.
The group’s bill, called the SGR Repeal and Medicare Provider Payment Modernization Act of 2014, would remove the threat of a 23.7% cut in Medicare payments that is set to take effect on April 1.
And it would provide annual payment updates of 0.5% for the next 5 years, as Medicare transitions to a payment system designed to reward physicians based on care quality, not quantity.
“This is a significant step forward in our long-standing effort to replace the flawed physician update formula with a 21st-century system focused on quality and value rather than the quantity of services,” said House Ways and Means Committee Ranking Member Sander Levin (D-Mich.).
“For the first time in many years, we have agreement among the bipartisan leadership of the 3 committees that oversee Medicare. Now, we have to turn to the thorny issue of offsets.”
The Congressional Budget Office has estimated that the new plan would increase direct spending by $150.4 billion from 2014 to 2023, although other sources have said the cost would be about $126 billion for the same time period.
Repealing the SGR
The SGR calls for annual, automatic cuts in Medicare payments to physicians. It was established in 1997 to control physician spending, but, over the years, the necessary cuts have not occurred.
Since 2003, Congress has spent about $150 billion to provide short-term fixes to spare physicians from the cuts. Without another short-term fix or legislation to permanently eliminate the SGR, physicians would see a 23.7% cut in Medicare payments starting April 1.
For years, Congress has been trying to eliminate the SGR, but agreeing on a plan to do so—and a way to pay for it—has proven difficult. Now, Congress says it has brought “renewed commitment to repealing and replacing the flawed SGR update mechanism.”
Provisions of the bill
The SGR Repeal and Medicare Provider Payment Modernization Act of 2014 would institute a 0.5% annual payment increase for physicians from 2014 through 2018. According to Congress, this would help the transition to a merit-based incentive payment system (MIPS), beginning in 2018.
The MIPS consolidates the 3 existing incentive programs—the Physician Quality Reporting System, the Value-Based Payment Modifier, and the Meaningful Use of Electronic Health Records—into a single program that would reward providers who meet performance thresholds.
The bill would also give a 5% bonus to providers who receive a significant portion of their revenue from an alternative payment model (APM) or patient-centered medical home (PCMH). Participants would need to receive at least 25% of their Medicare revenue through an APM in 2018-2019, and this threshold would increase over time.
The bill would provide incentives for participation in private-payer APMs as well. And it would establish a technical advisory committee to review and recommend physician-developed APMs based on criteria developed through an open-comment process.
Finally, the bill would expand the use of Medicare data. Quality and utilization data would be posted on the Physician Compare website with the goal of helping patients to make more informed decisions about their care.
Furthermore, “qualified entities” would be allowed to provide analyses and underlying data to providers, although this provision is subject to privacy and security laws. And qualified clinical data registries would be allowed to purchase claims data.
To read the bill in full, visit Congress.gov.
Climate change may increase malaria incidence

Credit: James Gathany
New research indicates that, toward the end of the century, climate change may make tropical highland regions suitable breeding grounds for malaria. And this could greatly increase the incidence of the disease.
Scientists compared the latest predictions for global warming with a range of statistical models commonly used to predict the spread of malaria.
And the models suggested that the changing climate will allow malaria to move into higher altitudes during warmer seasons and become permanently resident in larger areas.
This would mainly affect Africa but would also have an impact in Asia and South America.
In eastern Africa, the change could result in an additional 100 million people being exposed to malaria by the end of the 2080s, the researchers calculated.
However, they noted that the size of the impact is highly variable, as it can be affected by a number of factors.
“[W]e expect increased urbanization in these areas over the next 70 years,” said study author Cyril Caminade, PhD, of the University of Liverpool in the UK.
“Other developments, such as land use changes, population movements, and economic growth, will also have to be accounted for in future studies. What is clear is that diseases such as malaria are going to be moving, and this is a crucial element of how we prepare for the effects of climate change.”
The comparison of these models has not been carried out before, and by doing so, the researchers were able to find that malaria spreading to tropical highland areas was the one area in which the models agreed.
There was distinct variation in other parts of the world. Two of the models predicted that malaria would move northward, eventually spreading into Europe, Russia, and North America. Others showed that malaria would move north, but only as far as North Africa, where it was eliminated in the 20th century.

Credit: James Gathany
New research indicates that, toward the end of the century, climate change may make tropical highland regions suitable breeding grounds for malaria. And this could greatly increase the incidence of the disease.
Scientists compared the latest predictions for global warming with a range of statistical models commonly used to predict the spread of malaria.
And the models suggested that the changing climate will allow malaria to move into higher altitudes during warmer seasons and become permanently resident in larger areas.
This would mainly affect Africa but would also have an impact in Asia and South America.
In eastern Africa, the change could result in an additional 100 million people being exposed to malaria by the end of the 2080s, the researchers calculated.
However, they noted that the size of the impact is highly variable, as it can be affected by a number of factors.
“[W]e expect increased urbanization in these areas over the next 70 years,” said study author Cyril Caminade, PhD, of the University of Liverpool in the UK.
“Other developments, such as land use changes, population movements, and economic growth, will also have to be accounted for in future studies. What is clear is that diseases such as malaria are going to be moving, and this is a crucial element of how we prepare for the effects of climate change.”
The comparison of these models has not been carried out before, and by doing so, the researchers were able to find that malaria spreading to tropical highland areas was the one area in which the models agreed.
There was distinct variation in other parts of the world. Two of the models predicted that malaria would move northward, eventually spreading into Europe, Russia, and North America. Others showed that malaria would move north, but only as far as North Africa, where it was eliminated in the 20th century.

Credit: James Gathany
New research indicates that, toward the end of the century, climate change may make tropical highland regions suitable breeding grounds for malaria. And this could greatly increase the incidence of the disease.
Scientists compared the latest predictions for global warming with a range of statistical models commonly used to predict the spread of malaria.
And the models suggested that the changing climate will allow malaria to move into higher altitudes during warmer seasons and become permanently resident in larger areas.
This would mainly affect Africa but would also have an impact in Asia and South America.
In eastern Africa, the change could result in an additional 100 million people being exposed to malaria by the end of the 2080s, the researchers calculated.
However, they noted that the size of the impact is highly variable, as it can be affected by a number of factors.
“[W]e expect increased urbanization in these areas over the next 70 years,” said study author Cyril Caminade, PhD, of the University of Liverpool in the UK.
“Other developments, such as land use changes, population movements, and economic growth, will also have to be accounted for in future studies. What is clear is that diseases such as malaria are going to be moving, and this is a crucial element of how we prepare for the effects of climate change.”
The comparison of these models has not been carried out before, and by doing so, the researchers were able to find that malaria spreading to tropical highland areas was the one area in which the models agreed.
There was distinct variation in other parts of the world. Two of the models predicted that malaria would move northward, eventually spreading into Europe, Russia, and North America. Others showed that malaria would move north, but only as far as North Africa, where it was eliminated in the 20th century.