User login
Anaphylaxis risk with IV iron low, but varies with formulation
The results of the new retrospective cohort study were published online March 29 in Annals of Internal Medicine (doi: 10.7326/M21-4009).
“The rates of anaphylaxis were very low with all IV iron products but were three- to eightfold greater for iron dextran and ferumoxytol than for iron sucrose,” wrote Chintan V. Dave, PharmD, PhD, of Rutgers University, New Brunswick, N.J., and colleagues.
Using data from Medicare insurance claims, the researchers evaluated the incidence of anaphylaxis among patients 65 years or older receiving their first dose of one of five different IV iron formulations for the treatment of iron deficiency anemia. Patients were treated between July 2013 and December 2018 and the iron formulations were ferric carboxymaltose, ferumoxytol, ferric gluconate, iron dextran, or iron sucrose.
Overall, 167,925 patients were included and categorized based on the iron supplement they received. Dr. Dave and colleagues found that the adjusted incidence rates (IRs) for anaphylaxis per 10,000 first administrations were 9.8 cases for iron dextran (95% confidence interval [CI], 6.2 to 15.3 cases), 4.0 cases for ferumoxytol (95% CI, 2.5 to 6.6 cases), 1.5 cases for ferric gluconate (95% CI, 0.3 to 6.6 cases), 1.2 cases for iron sucrose (95% CI, 0.6 to 2.5 cases), and 0.8 cases for ferric carboxymaltose (95% CI, 0.3 to 2.6 cases).
Only those patients receiving iron dextran or ferumoxytol had anaphylactic reactions requiring hospitalization.
Using iron sucrose as the referent category, the researchers found that the odds ratios (ORs) for anaphylaxis were 8.3 for iron dextran (95% CI, 3.5-19.8) and 3.4 for ferumoxytol (95% CI, 1.4-8.3).
“Anaphylaxis is just one of many factors one should consider when deciding on the choice of IV iron therapy,” Dr. Dave noted in an interview, when asked whether he feels that these findings will change the use of parenteral iron in practice.
Acknowledging that anaphylaxis is a severe but rare complication, Dr. Dave stated that other factors such as “clinical indication, setting, dose, the number and duration of administrations required to replenish iron reserves, risk of other adverse reactions, and costs,” should also be considered when designing treatment plans using intravenous iron.
In the study, anaphylaxis was defined as reactions that occurred within 24 hours of IV iron administration and was restricted to the following:
- Anaphylaxis resulting in hospitalization.
- An outpatient or emergency department visit due to anaphylactic shock accompanied by codes relating to the administration of cardiopulmonary resuscitation or epinephrine or the occurrence of hypotension.
- Two separate encounters for anaphylactic shock within the same day representing different encounter types, that is, inpatient, outpatient, or emergency department visit.
Dr. Dave and colleagues acknowledged study limitations, such as the fact the anaphylaxis criteria included only the most severe cases and could therefore have missed milder cases of anaphylaxis secondary to IV iron. Further, they noted that these findings may not be applicable to a younger patient population.
Patients were excluded from the study if they had received IV iron between January 2007 and July 2013, had a diagnosis of HIV or end-stage renal disease, had a recent blood transfusion, or had a history of anaphylactic reactions.
The study authors disclosed no relevant financial relationships.
The results of the new retrospective cohort study were published online March 29 in Annals of Internal Medicine (doi: 10.7326/M21-4009).
“The rates of anaphylaxis were very low with all IV iron products but were three- to eightfold greater for iron dextran and ferumoxytol than for iron sucrose,” wrote Chintan V. Dave, PharmD, PhD, of Rutgers University, New Brunswick, N.J., and colleagues.
Using data from Medicare insurance claims, the researchers evaluated the incidence of anaphylaxis among patients 65 years or older receiving their first dose of one of five different IV iron formulations for the treatment of iron deficiency anemia. Patients were treated between July 2013 and December 2018 and the iron formulations were ferric carboxymaltose, ferumoxytol, ferric gluconate, iron dextran, or iron sucrose.
Overall, 167,925 patients were included and categorized based on the iron supplement they received. Dr. Dave and colleagues found that the adjusted incidence rates (IRs) for anaphylaxis per 10,000 first administrations were 9.8 cases for iron dextran (95% confidence interval [CI], 6.2 to 15.3 cases), 4.0 cases for ferumoxytol (95% CI, 2.5 to 6.6 cases), 1.5 cases for ferric gluconate (95% CI, 0.3 to 6.6 cases), 1.2 cases for iron sucrose (95% CI, 0.6 to 2.5 cases), and 0.8 cases for ferric carboxymaltose (95% CI, 0.3 to 2.6 cases).
Only those patients receiving iron dextran or ferumoxytol had anaphylactic reactions requiring hospitalization.
Using iron sucrose as the referent category, the researchers found that the odds ratios (ORs) for anaphylaxis were 8.3 for iron dextran (95% CI, 3.5-19.8) and 3.4 for ferumoxytol (95% CI, 1.4-8.3).
“Anaphylaxis is just one of many factors one should consider when deciding on the choice of IV iron therapy,” Dr. Dave noted in an interview, when asked whether he feels that these findings will change the use of parenteral iron in practice.
Acknowledging that anaphylaxis is a severe but rare complication, Dr. Dave stated that other factors such as “clinical indication, setting, dose, the number and duration of administrations required to replenish iron reserves, risk of other adverse reactions, and costs,” should also be considered when designing treatment plans using intravenous iron.
In the study, anaphylaxis was defined as reactions that occurred within 24 hours of IV iron administration and was restricted to the following:
- Anaphylaxis resulting in hospitalization.
- An outpatient or emergency department visit due to anaphylactic shock accompanied by codes relating to the administration of cardiopulmonary resuscitation or epinephrine or the occurrence of hypotension.
- Two separate encounters for anaphylactic shock within the same day representing different encounter types, that is, inpatient, outpatient, or emergency department visit.
Dr. Dave and colleagues acknowledged study limitations, such as the fact the anaphylaxis criteria included only the most severe cases and could therefore have missed milder cases of anaphylaxis secondary to IV iron. Further, they noted that these findings may not be applicable to a younger patient population.
Patients were excluded from the study if they had received IV iron between January 2007 and July 2013, had a diagnosis of HIV or end-stage renal disease, had a recent blood transfusion, or had a history of anaphylactic reactions.
The study authors disclosed no relevant financial relationships.
The results of the new retrospective cohort study were published online March 29 in Annals of Internal Medicine (doi: 10.7326/M21-4009).
“The rates of anaphylaxis were very low with all IV iron products but were three- to eightfold greater for iron dextran and ferumoxytol than for iron sucrose,” wrote Chintan V. Dave, PharmD, PhD, of Rutgers University, New Brunswick, N.J., and colleagues.
Using data from Medicare insurance claims, the researchers evaluated the incidence of anaphylaxis among patients 65 years or older receiving their first dose of one of five different IV iron formulations for the treatment of iron deficiency anemia. Patients were treated between July 2013 and December 2018 and the iron formulations were ferric carboxymaltose, ferumoxytol, ferric gluconate, iron dextran, or iron sucrose.
Overall, 167,925 patients were included and categorized based on the iron supplement they received. Dr. Dave and colleagues found that the adjusted incidence rates (IRs) for anaphylaxis per 10,000 first administrations were 9.8 cases for iron dextran (95% confidence interval [CI], 6.2 to 15.3 cases), 4.0 cases for ferumoxytol (95% CI, 2.5 to 6.6 cases), 1.5 cases for ferric gluconate (95% CI, 0.3 to 6.6 cases), 1.2 cases for iron sucrose (95% CI, 0.6 to 2.5 cases), and 0.8 cases for ferric carboxymaltose (95% CI, 0.3 to 2.6 cases).
Only those patients receiving iron dextran or ferumoxytol had anaphylactic reactions requiring hospitalization.
Using iron sucrose as the referent category, the researchers found that the odds ratios (ORs) for anaphylaxis were 8.3 for iron dextran (95% CI, 3.5-19.8) and 3.4 for ferumoxytol (95% CI, 1.4-8.3).
“Anaphylaxis is just one of many factors one should consider when deciding on the choice of IV iron therapy,” Dr. Dave noted in an interview, when asked whether he feels that these findings will change the use of parenteral iron in practice.
Acknowledging that anaphylaxis is a severe but rare complication, Dr. Dave stated that other factors such as “clinical indication, setting, dose, the number and duration of administrations required to replenish iron reserves, risk of other adverse reactions, and costs,” should also be considered when designing treatment plans using intravenous iron.
In the study, anaphylaxis was defined as reactions that occurred within 24 hours of IV iron administration and was restricted to the following:
- Anaphylaxis resulting in hospitalization.
- An outpatient or emergency department visit due to anaphylactic shock accompanied by codes relating to the administration of cardiopulmonary resuscitation or epinephrine or the occurrence of hypotension.
- Two separate encounters for anaphylactic shock within the same day representing different encounter types, that is, inpatient, outpatient, or emergency department visit.
Dr. Dave and colleagues acknowledged study limitations, such as the fact the anaphylaxis criteria included only the most severe cases and could therefore have missed milder cases of anaphylaxis secondary to IV iron. Further, they noted that these findings may not be applicable to a younger patient population.
Patients were excluded from the study if they had received IV iron between January 2007 and July 2013, had a diagnosis of HIV or end-stage renal disease, had a recent blood transfusion, or had a history of anaphylactic reactions.
The study authors disclosed no relevant financial relationships.
FROM ANNALS OF INTERNAL MEDICINE
Clinical clarity grows about toenail disorder, experts report
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
BOSTON – The main commonly leading to the wrong therapy and no resolution to the problem, according to an expert update at the annual meeting of the American Academy of Dermatology.
Misinterpretation of the yellow discoloration, a common feature of retronychia, means “many patients are maintained on antifungal therapy for years and years with no change in their condition,” reported Phoebe Rich, MD, director of the Nail Disorders Clinic, Oregon Health & Science University, Portland.
Infection is not commonly involved in retronychia, but importantly, antifungals and antibiotics “have no role in treating the underlying disorder,” Dr. Rich said.
The term retronychia and its description is only about 20 years old, according to Dr. Rich, who cited work by David A. de Berker, MBBS, PhD, a consultant dermatologist at University Hospitals in Bristol, England. His publication on this disorder appeared in 1999, with a more detailed description published about 10 years later.
Recently, the body of literature on this disorder has been growing, contributing to an increasing consensus about etiology, diagnosis, and treatments to consider in the context of causes and severity, Dr. Rich said.
Some but not all patients have abnormal formation of the nail bed, increasing susceptibility to retronychia, but trauma or microtrauma typically serve as a trigger in most cases. Dancing, high heels, steel-toed shoes, and other sources of trauma to the toes are implicated.
Whether or not patients have an inherent susceptibility, injury separates the existing nail from the matrix and nail bed so that newly forming nail begins to grow under the nail rather continuing to push out the old nail.
Susceptibility is increased substantially in individuals with a shortened nail bed, according to Dr. Rich. In severe cases, when there is simply inadequate nail bed for the nail growth to attach, recurrence is common or even inevitable. Even when the nail is removed and regrowth appears normal at the end of a year, those patients with very short nail beds cannot count on a cure.
“Due to the slow growth of nails, it might take 2 or 3 years for the problem to recur,” Dr. Rich cautioned. For this reason, cure rates reported for the various interventions at 1 year might not predict longer-term benefit.
Retronychia is usually a clinical diagnosis based on the presence of the increased bulk of the toenail when overlapping nails cannot be seen. This is not necessarily a single overgrowth. In some cases, multiple layers of nails are stacked one on top of the other. Xanthonychia (yellow nail) is usually present.
“The layering might not be visible without removing the nail,” said Dr. Rich, explaining one reason that the diagnosis is sometimes missed. Ultrasound is a noninvasive means to confirm the problem, although Rich warned that imaging is not necessarily reimbursed.
“There is no diagnosis by histopathology, so it cannot be confirmed with biopsy,” Dr. Rich said.
Treatments range from conservative strategies, particularly topical or intralesional steroids in mild cases, to more invasive procedures such as clipping of the nail plate or surgical avulsion. All can be effective when used appropriately, according to Dr. Rich.
“The more invasive procedures are the more effective, but the caveat is they are also associated with more complications,” said Dr. Rich, citing, for example, the risk of nail dystrophies. Because of the increasing number of studies, the relative benefits and risks of retronychia treatment have now been summarized in a recent review. Dr. Rich suggested the review is one of the most recent and detailed evaluations of the topic that “I encourage everyone to read.”
Despite progress in describing retronychia, Dr. Rich said that there might be more to learn about risk. In particular, she cited the work of Dana W. Stern, MD, a specialist in nail disorders who is in private practice in New York. Dr. Stern is pursuing a hypothesis that at least some cases are caused by potentially targetable biomechanical issues.
“I have observed that many of the younger patients in my practice with retronychia seem to have atypical foot anatomy,” Dr. Stern said in an interview. “I am collecting cases and hoping to explore this issue in more depth.”
She said that foot anatomy in relationship to retronychia has not been adequately evaluated.
“In my review of the literature, I could not find a single study that showed imagery of the feet,” she said. She is considering a collaboration with others, including Rich, to explore this as a factor in retronychia.
Asked about risk of misdiagnosis, Dr. Stern reiterated some of the points made by Dr. Rich. In particular, she agreed that discolored nails alone should not be a reason to initiate antimycotic therapy without considering the possibility of retronychia.
“So many providers are not familiar with the diagnosis, and only 50% of yellow thickened nails are in fact onychomycosis,” she said. “We end up seeing a plethora of patients [with retronychia] who are unfortunately misdiagnosed for years.”
Dr. Rich reported financial relationships with numerous pharmaceutical companies. Dr. Stern reported a financial relationship with Rare Beauty Brands. Neither Dr. Rich nor Dr. Stern said they had any disclosures related to this topic.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Sustained jawline definition from hyaluronic gel, study reports
BOSTON – After several promising early phase studies, from what study authors characterized as a “pivotal” randomized multicenter trial. The results were presented during a late-breaking research session at the annual meeting of the American Academy of Dermatology.
The primary outcome, assessed at 6 months, was at least a 1-point improvement in a photonumeric scale used to grade jawline sagging, reported Jeremy Green, MD, Skin Associates of South Florida, Coral Gables.
When those randomized to the hyaluronic filler gel VYC-25L (Vycross, Juvéderm) were compared with untreated controls, 68.5% versus 38.4% met the criterion for benefit at 6 months. Importantly, the effect in treated patients was sustained when reevaluated at 12 months. Green reported that the response is generally sustained at the maximum follow-up, now out to 17 months.
Most enrolled patients are severely affected
In this study, 208 patients with severe (74%) or moderate loss of jawline definition were randomized in a 3:1 ratio to receive the filler or serve as controls. The initially untreated controls received the gel after the primary outcome analysis at 6 months.
The hyaluronic gel was injected at five sites along the jawline. The mean age of participants was 58 years. The majority were women, and most were White.
Dermatologists blinded to treatment compared photos at 6 months with those taken at baseline using the photonumeric grading system of 1-5. Change in patient satisfaction at 6 months and again at 12 months relative to baseline was also evaluated.
From baseline, when 28.9% of participants reported satisfaction on the Global Aesthetic Improvement Scale (GAIS), rates rose to 89.0% at month 6. There was a decline at month 12, but 79.9% remained satisfied after this period of follow-up.
Most patients experienced injection site reactions that were mainly mild to moderate and all resolved within several days of treatment. Pain with mastication was initially reported by 1.9%, but again this complaint was also mild and transient. All complaints had largely resolved by day 3.
The results are consistent with several previous clinical studies of VYC-25L for the same indication. In a similarly designed trial conducted in Europe that also used a 3:1 randomization scheme, the primary outcome assessed at 3 months was change in facial angle. Relative to controls, the angle improved by 2.51 degrees (P < .0001).
Patient satisfaction supports filler benefit
In the similar European trial, the clinical significance of the objective primary outcome also was supported by patient satisfaction assessed with several instruments, including the GAIS. Some degree of swelling or tenderness was experienced by almost all patients after injection, but none were serious, and all resolved.
In another trial, 202 patients with chin retrusion were randomized in a 3:1 ratio to VYC-25L or a control group. In that study, the primary outcome was at least a 1-point improvement in the Allergan Chin Retrusion Scale at 6 months. This advantage for treatment (56.3% vs. 27.5%) was again supported by several instruments for evaluating patient satisfaction, including GAIS.
As in the other studies, most patients had injection site reactions. Although all resolved within days of treatment, one patient left the study after experiencing cellulitis and injection-site inflammation.
Dissatisfaction with jawline definition is a relatively common complaint in Dr. Green’s experience, who said that there is a need for more effective and well-tolerated treatments. Given the efficacy, tolerability, and safety of VYC-25L in this controlled study, he suggested this product has potential utility.
In the field of cosmetic dermatology, there appears to be incremental progress in fillers with favorable clinical characteristics, according to Sandy U. Tsao, MD, a dermatologic surgeon at Massachusetts General Hospital, Boston.
“We are seeing filler lasting longer and longer,” she said, commenting specifically about the results presented by Dr. Green. She called sustained aesthetic improvement at 12 months for the filler in this study “really exciting.”
Dr. Green has reported financial relationships with numerous pharmaceutical companies. Dr. Tsao has reported financial relationships with Epiphany Dermatology, Lazarus AI, and UpToDate.
A version of this article first appeared on Medscape.com.
BOSTON – After several promising early phase studies, from what study authors characterized as a “pivotal” randomized multicenter trial. The results were presented during a late-breaking research session at the annual meeting of the American Academy of Dermatology.
The primary outcome, assessed at 6 months, was at least a 1-point improvement in a photonumeric scale used to grade jawline sagging, reported Jeremy Green, MD, Skin Associates of South Florida, Coral Gables.
When those randomized to the hyaluronic filler gel VYC-25L (Vycross, Juvéderm) were compared with untreated controls, 68.5% versus 38.4% met the criterion for benefit at 6 months. Importantly, the effect in treated patients was sustained when reevaluated at 12 months. Green reported that the response is generally sustained at the maximum follow-up, now out to 17 months.
Most enrolled patients are severely affected
In this study, 208 patients with severe (74%) or moderate loss of jawline definition were randomized in a 3:1 ratio to receive the filler or serve as controls. The initially untreated controls received the gel after the primary outcome analysis at 6 months.
The hyaluronic gel was injected at five sites along the jawline. The mean age of participants was 58 years. The majority were women, and most were White.
Dermatologists blinded to treatment compared photos at 6 months with those taken at baseline using the photonumeric grading system of 1-5. Change in patient satisfaction at 6 months and again at 12 months relative to baseline was also evaluated.
From baseline, when 28.9% of participants reported satisfaction on the Global Aesthetic Improvement Scale (GAIS), rates rose to 89.0% at month 6. There was a decline at month 12, but 79.9% remained satisfied after this period of follow-up.
Most patients experienced injection site reactions that were mainly mild to moderate and all resolved within several days of treatment. Pain with mastication was initially reported by 1.9%, but again this complaint was also mild and transient. All complaints had largely resolved by day 3.
The results are consistent with several previous clinical studies of VYC-25L for the same indication. In a similarly designed trial conducted in Europe that also used a 3:1 randomization scheme, the primary outcome assessed at 3 months was change in facial angle. Relative to controls, the angle improved by 2.51 degrees (P < .0001).
Patient satisfaction supports filler benefit
In the similar European trial, the clinical significance of the objective primary outcome also was supported by patient satisfaction assessed with several instruments, including the GAIS. Some degree of swelling or tenderness was experienced by almost all patients after injection, but none were serious, and all resolved.
In another trial, 202 patients with chin retrusion were randomized in a 3:1 ratio to VYC-25L or a control group. In that study, the primary outcome was at least a 1-point improvement in the Allergan Chin Retrusion Scale at 6 months. This advantage for treatment (56.3% vs. 27.5%) was again supported by several instruments for evaluating patient satisfaction, including GAIS.
As in the other studies, most patients had injection site reactions. Although all resolved within days of treatment, one patient left the study after experiencing cellulitis and injection-site inflammation.
Dissatisfaction with jawline definition is a relatively common complaint in Dr. Green’s experience, who said that there is a need for more effective and well-tolerated treatments. Given the efficacy, tolerability, and safety of VYC-25L in this controlled study, he suggested this product has potential utility.
In the field of cosmetic dermatology, there appears to be incremental progress in fillers with favorable clinical characteristics, according to Sandy U. Tsao, MD, a dermatologic surgeon at Massachusetts General Hospital, Boston.
“We are seeing filler lasting longer and longer,” she said, commenting specifically about the results presented by Dr. Green. She called sustained aesthetic improvement at 12 months for the filler in this study “really exciting.”
Dr. Green has reported financial relationships with numerous pharmaceutical companies. Dr. Tsao has reported financial relationships with Epiphany Dermatology, Lazarus AI, and UpToDate.
A version of this article first appeared on Medscape.com.
BOSTON – After several promising early phase studies, from what study authors characterized as a “pivotal” randomized multicenter trial. The results were presented during a late-breaking research session at the annual meeting of the American Academy of Dermatology.
The primary outcome, assessed at 6 months, was at least a 1-point improvement in a photonumeric scale used to grade jawline sagging, reported Jeremy Green, MD, Skin Associates of South Florida, Coral Gables.
When those randomized to the hyaluronic filler gel VYC-25L (Vycross, Juvéderm) were compared with untreated controls, 68.5% versus 38.4% met the criterion for benefit at 6 months. Importantly, the effect in treated patients was sustained when reevaluated at 12 months. Green reported that the response is generally sustained at the maximum follow-up, now out to 17 months.
Most enrolled patients are severely affected
In this study, 208 patients with severe (74%) or moderate loss of jawline definition were randomized in a 3:1 ratio to receive the filler or serve as controls. The initially untreated controls received the gel after the primary outcome analysis at 6 months.
The hyaluronic gel was injected at five sites along the jawline. The mean age of participants was 58 years. The majority were women, and most were White.
Dermatologists blinded to treatment compared photos at 6 months with those taken at baseline using the photonumeric grading system of 1-5. Change in patient satisfaction at 6 months and again at 12 months relative to baseline was also evaluated.
From baseline, when 28.9% of participants reported satisfaction on the Global Aesthetic Improvement Scale (GAIS), rates rose to 89.0% at month 6. There was a decline at month 12, but 79.9% remained satisfied after this period of follow-up.
Most patients experienced injection site reactions that were mainly mild to moderate and all resolved within several days of treatment. Pain with mastication was initially reported by 1.9%, but again this complaint was also mild and transient. All complaints had largely resolved by day 3.
The results are consistent with several previous clinical studies of VYC-25L for the same indication. In a similarly designed trial conducted in Europe that also used a 3:1 randomization scheme, the primary outcome assessed at 3 months was change in facial angle. Relative to controls, the angle improved by 2.51 degrees (P < .0001).
Patient satisfaction supports filler benefit
In the similar European trial, the clinical significance of the objective primary outcome also was supported by patient satisfaction assessed with several instruments, including the GAIS. Some degree of swelling or tenderness was experienced by almost all patients after injection, but none were serious, and all resolved.
In another trial, 202 patients with chin retrusion were randomized in a 3:1 ratio to VYC-25L or a control group. In that study, the primary outcome was at least a 1-point improvement in the Allergan Chin Retrusion Scale at 6 months. This advantage for treatment (56.3% vs. 27.5%) was again supported by several instruments for evaluating patient satisfaction, including GAIS.
As in the other studies, most patients had injection site reactions. Although all resolved within days of treatment, one patient left the study after experiencing cellulitis and injection-site inflammation.
Dissatisfaction with jawline definition is a relatively common complaint in Dr. Green’s experience, who said that there is a need for more effective and well-tolerated treatments. Given the efficacy, tolerability, and safety of VYC-25L in this controlled study, he suggested this product has potential utility.
In the field of cosmetic dermatology, there appears to be incremental progress in fillers with favorable clinical characteristics, according to Sandy U. Tsao, MD, a dermatologic surgeon at Massachusetts General Hospital, Boston.
“We are seeing filler lasting longer and longer,” she said, commenting specifically about the results presented by Dr. Green. She called sustained aesthetic improvement at 12 months for the filler in this study “really exciting.”
Dr. Green has reported financial relationships with numerous pharmaceutical companies. Dr. Tsao has reported financial relationships with Epiphany Dermatology, Lazarus AI, and UpToDate.
A version of this article first appeared on Medscape.com.
AT AAD 2022
Angioimmunoblastic T-cell Lymphoma Mimicking DRESS Syndrome
Angioimmunoblastic T-cell lymphoma (AITL) is a rare and aggressive lymphoma arising from follicular T-helper cells that predominantly affects older adults and carries a 5-year overall survival rate of 32%.1 Notably, as many as 50% of AITL patients present with a skin rash in addition to the more common but nonspecific acute-onset generalized lymphadenopathy, hepatosplenomegaly, and anemia.2 At presentation, most AITL patients are already at an advanced (III/IV) stage of disease.
Formerly known as angioimmunoblastic lymphadenopathy with dysproteinemia, AITL was once considered a benign entity that carried a risk for malignant transformation. As more cases have been identified and explored, this entity has been recategorized as a frank lymphoma.3 Therefore, it is critical that AITL be diagnosed and treated as early as possible.
We present the case of a 65-year-old man with clinical features that resembled drug reaction with eosinophilia and systemic symptoms (DRESS syndrome). After extensive workup, he was found to have AITL. This atypical case highlights the importance of maintaining a flexible differential diagnosis in patients with a persistent rash that does not improve with appropriate drug withdrawal and therapy.
Case Report
A 65-year-old Filipino man whose medical history was notable for hepatitis B that had been treated with entecavir for years without issue was admitted to the internal medicine service with fever of unknown origin and malaise of approximately 6 weeks’ duration. Six days prior to admission and 5 days after completing courses of the antiviral oseltamivir phosphate and amoxicillin for an upper respiratory tract infection and sinusitis, he developed worsening of an intermittently pruritic rash of approximately 1 month's duration. The dermatology department was consulted the day of hospital admission for evaluation of the rash. Chronic home medications included entecavir, lisinopril/hydrochlorothiazide, amlodipine, atorvastatin, metformin, salsalate, and over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) as needed.
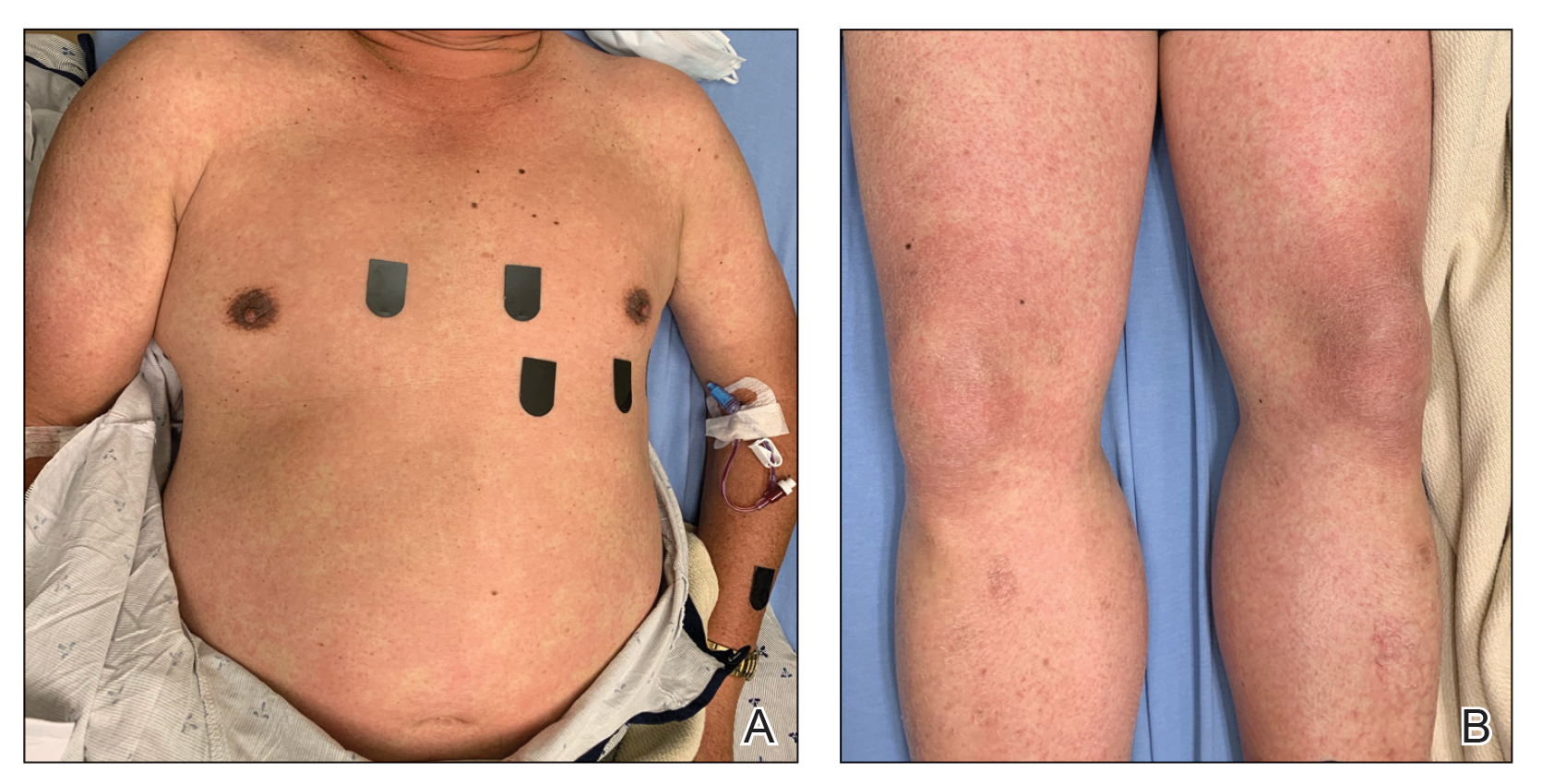
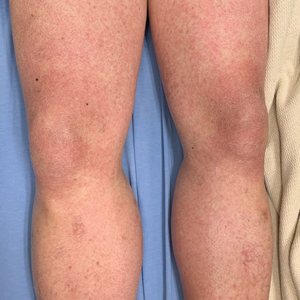
Physical examination was notable for mild erythema and scale distributed across the entire face; mild facial edema; and a blanchable, nonconfluent, macular erythema distributed across the trunk and upper and proximal lower extremities (Figure). In addition, the patient displayed conjunctival injection, pitting edema of the hands, and bilateral cervical and inguinal lymphadenopathy.
Laboratory tests revealed mild leukocytosis (11.6×109/L, [reference range, 4.0–10.5×109/L]), anemia (hemoglobin, 125 g/L (reference range, 138–170 g/L); hematocrit, 36.9%, [reference range, 40.0%–50.0%)], eosinophilia (1.07×109/L [reference range, 0.00–0.70×109/L)], hyponatremia, hypokalemia, and a mildly elevated creatinine level. Computed tomography and full-body positron-emission tomography (PET) scans during admission demonstrated diffuse lymphadenopathy. A skin biopsy from the left chest and a left inguinal lymph node biopsy also were performed.
Despite the lack of a clear medication trigger within the usual timeline for severe cutaneous drug-induced hypersensitivity reactions, DRESS syndrome was high on the differential diagnosis at the time of the initial presentation given the diffuse morbilliform eruption with pruritus, facial edema, eosinophilia, and lymphadenopathy.
Home medications were discontinued except for amlodipine, atorvastatin, and entecavir. The patient was treated symptomatically with topical steroids because it was believed that, if the clinical presentation represented DRESS syndrome, it was a mild variant that could be treated topically.4 His case was considered mild because of a lack of confirmed organ dysfunction and a mild protracted course.
After discharge following a 3-day inpatient stay, the patient was followed in the clinic weekly for 3 weeks without considerable change in the skin or laboratory findings. Discontinuation of entecavir was discussed and approved by his hepatologist.
Posthospitalization analysis of the punch biopsy specimen from the chest performed during the patient’s hospital stay revealed a superficial and deep dermal lymphoid infiltrate comprising CD3-, CD5-, and programmed cell death protein 1–positive cells with cytologic atypia in a perivascular distribution. Analysis of the lymph node biopsy specimen performed during the hospitalization showed effacement of the nodal architecture, a polymorphous lymphoid cell population with irregular nuclear contour, and abundant clear cytoplasm associated with high endothelial venules (HEVs). Cells of interest were positive for CD3, CD4, CD2, CD5, and CD7, with a subset staining positive for programmed cell death protein 1, inducible costimulator, CD10, and chemokine (C-X-C motif) ligand (CXCL) 13. CD21 demonstrated an expanded follicular dendritic cell meshwork in association with HEVs. Polymerase chain reaction revealed a clonal T-cell population. These findings of the skin and lymph node biopsies were consistent with AITL. Subsequent bone marrow biopsy with flow cytometry showed a normal CD4:CD8 ratio in T cells and no increase in natural killer cells.
Cyclophosphamide–hydroxydaunorubicin–Oncovin–prednisone (CHOP) chemotherapy was initiated; the patient completed a total of 6 cycles. He has had near resolution of the skin findings and is considered in remission based on a PET scan performed approximately 7 months after the initial presentation.
Comment
Angioimmunoblastic T-cell lymphoma is a rare peripheral T-cell lymphoma, part of a group of aggressive neoplasms that constitute approximately 15% of peripheral T-cell lymphomas and approximately 2% of non-Hodgkin lymphomas in adults worldwide.5 Cutaneous involvement occurs in approximately half of AITL cases and can be the first manifestation of disease.2 Skin findings are largely nonspecific, ranging from simple morbilliform rashes to erythroderma, at times manifesting with purpura.
Given this variability in the presentation of AITL, early diagnosis is challenging in the absence of more specific signs and symptoms.2 It can conceivably be mistaken for common entities such as viral exanthems or drug eruptions, depending on the history and context. DRESS syndrome, a T cell-mediated, delayed type-IV hypersensitivity drug reaction can present in a manner highly similar to that of AITL, with cutaneous involvement (diffuse morbilliform rash, fever, facial edema, and generalized lymphadenopathy) and variable systemic involvement. Laboratory findings of eosinophilia, atypical lymphocytes, and thrombocytopenia also might be seen in both entities.6 Furthermore, the AITL in our patient was accompanied by electrolyte disturbances that were concerning for syndrome of inappropriate antidiuretic hormone secretion, a rare complication of patients with DRESS syndrome complicated by encephalitis.7,8
Our patient met 4 RegiSCAR criteria for DRESS syndrome, warranting high clinical suspicion for an offending drug.9 DRESS syndrome can be caused by numerous medications—most commonly anticonvulsants, sulfonamides, antibiotics, allopurinol, and NSAIDs. A review of our patient’s medication list identified NSAIDs (including salsalate), entecavir, and amoxicillin, as possible culpable medications. Notably, the only new addition to the patient’s regimen was amoxicillin, which did not fit the typical 2- to 8-week timeline for a DRESS syndrome nidus.10 Our patient’s fever began well before the antibiotic was initiated, and skin findings appeared within 1 week after the course of amoxicillin was completed. Although there is documented variability in the latency of onset of DRESS syndrome following administration of a culprit medication,11 it is critical to maintain a broad differential diagnosis to allow for further diagnostic information to be obtained, especially when the medication timeline does not align with the clinical presentation.
DRESS syndrome is far more common than AITL. Similarities in their clinical presentation pose a substantial challenge and often cause a delay in the diagnosis of AITL, which is made by excisional tissue biopsy, most commonly of a lymph node, with assessment of morphology and immunophenotyping. Histologic assessment of tissue reveals a polymorphous infiltrate of variably sized atypical lymphocytes with prominent arborizing HEVs as well as expanded populations of follicular dendritic cells that can be detected by CD21 staining. Cells express CD3 and CD4, variably express BCL6 (B-cell lymphoma 6 antigen) and CD10, and also may have partial or complete loss of expression of a subset of pan T-cell antigens (CD2, CD3, CD5, and CD7).12-18
The treatment approach to AITL mirrors that of other nodal peripheral T-cell lymphomas, including chemotherapy and consideration of autologous stem-cell transplantation. Recent prospective trials of CHOP and CHOP-like chemotherapy have reported 3-year event-free survival and overall survival rates of 50% and 68%, respectively.19 Novel chemotherapeutic targets and gene-expression profiling are being investigated as potential therapeutic avenues.20
Conclusion
DRESS syndrome and AITL can have near-identical presentations. Clinicians should maintain a high index of suspicion for AITL in patients with presumed DRESS syndrome whose rash does not improve with appropriate drug withdrawal and steroid therapy or who lack a strong offending medication history. In such cases, skin and lymph node biopsies should be performed as early as possible to evaluate for AITL and so that appropriate therapy can be initiated.
- Federico M, Rudiger T, Bellei M, et al. Clinicopathologic characteristics of angioimmunoblastic T-cell lymphoma: analysis of the international peripheral T-cell lymphoma project. J Clin Oncol. 2013;31:240-246. doi:10.1200/JCO.2011.37.3647
- Botros N, Cerroni L, Shawwa A, et al. Cutaneous manifestations of angioimmunoblastic T-cell lymphoma: clinical and pathological characteristics. Am J Dermatopathol. 2015;37:274-283. doi:10.1097/DAD.0000000000000144
- Sachsida-Colombo E, Barbosa Mariano LC, Bastos FQ, et al. A difficult case of angioimmunoblastic T-cell lymphoma to diagnose. Rev Bras Hematol Hemoter. 2016;38:82-85. doi:10.1016/j.bjhh.2015.11.002
- Funck-Brentano E, Duong T-A, Bouvresse S, et al. Therapeutic management of DRESS: a retrospective study of 38 cases. J Am Acad Dermatol. 2015;72:246-252. doi:10.1016/j.jaad.2014.10.032
- Lunning MA, Vose JM. Angioimmunoblastic T-cell lymphoma: the many-faced lymphoma. Blood. 2017;129:1095-1102. doi:10.1182/blood-2016-09-692541
- Sato R, Itoh M, Suzuki H, et al. Pathological findings of lymphadenopathy in drug-induced hypersensitivity syndrome (DIHS)/drug reaction with eosinophilia and systemic syndrome (DRESS): similarities with angioimmunoblastic T-cell lymphoma. Eur J Dermatol. 2017;27:201-202. doi:10.1684/ejd.2016.2954
- Osizik L, Tanriover MD, Saka E. Autoimmune limbic encephalitis and syndrome of inappropriate antidiuretic hormone secretion associated with lamotrigine-induced drug rash with eosinophilia and systemic symptoms (DRESS) syndrome. Intern Med. 2015;55:1393-1396. doi:10.2169/internalmedicine.55.6035
- Sakuma K, Kano Y, Fukuhara M, et al. Syndrome of inappropriate secretion of antidiuretic hormone associated with limbic encephalitis in a patient with drug-induced hypersensitivity syndrome. Clin Exp Dermatol. 2008;33:287-290. doi:10.1111/j.1365-2230.2007.02645.x
- Pannu AK, Saroch A. Diagnostic criteria for drug rash and eosinophilia with systemic symptoms. J Family Med Prim Care. 2017;6:693-694. doi:10.4103/2249-4863.222050
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al; RegiSCAR study group. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080. doi:10.1111/bjd.12501
- Soria A, Bernier C, Veyrac G, et al. Drug reaction with eosinophilia and systemic symptoms may occur within 2 weeks of drug exposure: a retrospective study. J Am Acad Dermatol. 2020;82:606.
- Loghavi S, Wang SA, Medeiros LJ, et al. Immunophenotypic and diagnostic characterization of angioimmunoblastic T-cell lymphoma by advanced flow cytometric technology. Leuk Lymphoma. 2016;57:2804-2812. doi:10.3109/10428194.2016.1170827
- Lee S-S, R, Odenwald T, et al. Angioimmunoblastic T cell lymphoma is derived from mature T-helper cells with varying expression and loss of detectable CD4. Int J Cancer. 2003;103:12-20. doi:10.1002/ijc.10758
- Feller AC, Griesser H, Schilling CV, et al. Clonal gene rearrangement patterns correlate with immunophenotype and clinical parameters in patients with angioimmunoblastic lymphadenopathy. Am J Pathol. 1988;133:549-556.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. IARC Press; 2008.
- Attygalle A, Al-Jehani R, Diss TC, et al. Neoplastic T cells in angioimmunoblastic T-cell lymphoma express CD10. Blood. 2002;99:627-633. doi:10.1182/blood.v99.2.627
- Mourad N, Mounier N, J, et al; Groupe d’Etude des Lymphomes de l’Adulte. Clinical, biologic, and pathologic features in 157 patients with angioimmunoblastic T-cell lymphoma treated within the Groupe d’Etude des Lymphomes de l’Adulte (GELA) trials. Blood. 2008;111:4463-4470. doi:10.1182/blood-2007-08-105759
- Marafioti T, Paterson JC, Ballabio E, et al. The inducible T-cell co-stimulator molecule is expressed on subsets of T cells and is a new marker of lymphomas of T follicular helper cell-derivation. Haematologica. 2010;95:432-439. doi:10.3324/haematol.2009.010991
- Schmitz N, L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients withT-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116:3418-3425. doi:10.1182/blood-2010-02-270785
- Moskowitz AJ. Practical treatment approach for angioimmunoblastic T-cell lymphoma. J Oncol Pract. 2019;15:137-143. doi:10.1200/JOP.18.00511
Angioimmunoblastic T-cell lymphoma (AITL) is a rare and aggressive lymphoma arising from follicular T-helper cells that predominantly affects older adults and carries a 5-year overall survival rate of 32%.1 Notably, as many as 50% of AITL patients present with a skin rash in addition to the more common but nonspecific acute-onset generalized lymphadenopathy, hepatosplenomegaly, and anemia.2 At presentation, most AITL patients are already at an advanced (III/IV) stage of disease.
Formerly known as angioimmunoblastic lymphadenopathy with dysproteinemia, AITL was once considered a benign entity that carried a risk for malignant transformation. As more cases have been identified and explored, this entity has been recategorized as a frank lymphoma.3 Therefore, it is critical that AITL be diagnosed and treated as early as possible.
We present the case of a 65-year-old man with clinical features that resembled drug reaction with eosinophilia and systemic symptoms (DRESS syndrome). After extensive workup, he was found to have AITL. This atypical case highlights the importance of maintaining a flexible differential diagnosis in patients with a persistent rash that does not improve with appropriate drug withdrawal and therapy.
Case Report
A 65-year-old Filipino man whose medical history was notable for hepatitis B that had been treated with entecavir for years without issue was admitted to the internal medicine service with fever of unknown origin and malaise of approximately 6 weeks’ duration. Six days prior to admission and 5 days after completing courses of the antiviral oseltamivir phosphate and amoxicillin for an upper respiratory tract infection and sinusitis, he developed worsening of an intermittently pruritic rash of approximately 1 month's duration. The dermatology department was consulted the day of hospital admission for evaluation of the rash. Chronic home medications included entecavir, lisinopril/hydrochlorothiazide, amlodipine, atorvastatin, metformin, salsalate, and over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) as needed.
Physical examination was notable for mild erythema and scale distributed across the entire face; mild facial edema; and a blanchable, nonconfluent, macular erythema distributed across the trunk and upper and proximal lower extremities (Figure). In addition, the patient displayed conjunctival injection, pitting edema of the hands, and bilateral cervical and inguinal lymphadenopathy.
Laboratory tests revealed mild leukocytosis (11.6×109/L, [reference range, 4.0–10.5×109/L]), anemia (hemoglobin, 125 g/L (reference range, 138–170 g/L); hematocrit, 36.9%, [reference range, 40.0%–50.0%)], eosinophilia (1.07×109/L [reference range, 0.00–0.70×109/L)], hyponatremia, hypokalemia, and a mildly elevated creatinine level. Computed tomography and full-body positron-emission tomography (PET) scans during admission demonstrated diffuse lymphadenopathy. A skin biopsy from the left chest and a left inguinal lymph node biopsy also were performed.
Despite the lack of a clear medication trigger within the usual timeline for severe cutaneous drug-induced hypersensitivity reactions, DRESS syndrome was high on the differential diagnosis at the time of the initial presentation given the diffuse morbilliform eruption with pruritus, facial edema, eosinophilia, and lymphadenopathy.
Home medications were discontinued except for amlodipine, atorvastatin, and entecavir. The patient was treated symptomatically with topical steroids because it was believed that, if the clinical presentation represented DRESS syndrome, it was a mild variant that could be treated topically.4 His case was considered mild because of a lack of confirmed organ dysfunction and a mild protracted course.
After discharge following a 3-day inpatient stay, the patient was followed in the clinic weekly for 3 weeks without considerable change in the skin or laboratory findings. Discontinuation of entecavir was discussed and approved by his hepatologist.
Posthospitalization analysis of the punch biopsy specimen from the chest performed during the patient’s hospital stay revealed a superficial and deep dermal lymphoid infiltrate comprising CD3-, CD5-, and programmed cell death protein 1–positive cells with cytologic atypia in a perivascular distribution. Analysis of the lymph node biopsy specimen performed during the hospitalization showed effacement of the nodal architecture, a polymorphous lymphoid cell population with irregular nuclear contour, and abundant clear cytoplasm associated with high endothelial venules (HEVs). Cells of interest were positive for CD3, CD4, CD2, CD5, and CD7, with a subset staining positive for programmed cell death protein 1, inducible costimulator, CD10, and chemokine (C-X-C motif) ligand (CXCL) 13. CD21 demonstrated an expanded follicular dendritic cell meshwork in association with HEVs. Polymerase chain reaction revealed a clonal T-cell population. These findings of the skin and lymph node biopsies were consistent with AITL. Subsequent bone marrow biopsy with flow cytometry showed a normal CD4:CD8 ratio in T cells and no increase in natural killer cells.
Cyclophosphamide–hydroxydaunorubicin–Oncovin–prednisone (CHOP) chemotherapy was initiated; the patient completed a total of 6 cycles. He has had near resolution of the skin findings and is considered in remission based on a PET scan performed approximately 7 months after the initial presentation.
Comment
Angioimmunoblastic T-cell lymphoma is a rare peripheral T-cell lymphoma, part of a group of aggressive neoplasms that constitute approximately 15% of peripheral T-cell lymphomas and approximately 2% of non-Hodgkin lymphomas in adults worldwide.5 Cutaneous involvement occurs in approximately half of AITL cases and can be the first manifestation of disease.2 Skin findings are largely nonspecific, ranging from simple morbilliform rashes to erythroderma, at times manifesting with purpura.
Given this variability in the presentation of AITL, early diagnosis is challenging in the absence of more specific signs and symptoms.2 It can conceivably be mistaken for common entities such as viral exanthems or drug eruptions, depending on the history and context. DRESS syndrome, a T cell-mediated, delayed type-IV hypersensitivity drug reaction can present in a manner highly similar to that of AITL, with cutaneous involvement (diffuse morbilliform rash, fever, facial edema, and generalized lymphadenopathy) and variable systemic involvement. Laboratory findings of eosinophilia, atypical lymphocytes, and thrombocytopenia also might be seen in both entities.6 Furthermore, the AITL in our patient was accompanied by electrolyte disturbances that were concerning for syndrome of inappropriate antidiuretic hormone secretion, a rare complication of patients with DRESS syndrome complicated by encephalitis.7,8
Our patient met 4 RegiSCAR criteria for DRESS syndrome, warranting high clinical suspicion for an offending drug.9 DRESS syndrome can be caused by numerous medications—most commonly anticonvulsants, sulfonamides, antibiotics, allopurinol, and NSAIDs. A review of our patient’s medication list identified NSAIDs (including salsalate), entecavir, and amoxicillin, as possible culpable medications. Notably, the only new addition to the patient’s regimen was amoxicillin, which did not fit the typical 2- to 8-week timeline for a DRESS syndrome nidus.10 Our patient’s fever began well before the antibiotic was initiated, and skin findings appeared within 1 week after the course of amoxicillin was completed. Although there is documented variability in the latency of onset of DRESS syndrome following administration of a culprit medication,11 it is critical to maintain a broad differential diagnosis to allow for further diagnostic information to be obtained, especially when the medication timeline does not align with the clinical presentation.
DRESS syndrome is far more common than AITL. Similarities in their clinical presentation pose a substantial challenge and often cause a delay in the diagnosis of AITL, which is made by excisional tissue biopsy, most commonly of a lymph node, with assessment of morphology and immunophenotyping. Histologic assessment of tissue reveals a polymorphous infiltrate of variably sized atypical lymphocytes with prominent arborizing HEVs as well as expanded populations of follicular dendritic cells that can be detected by CD21 staining. Cells express CD3 and CD4, variably express BCL6 (B-cell lymphoma 6 antigen) and CD10, and also may have partial or complete loss of expression of a subset of pan T-cell antigens (CD2, CD3, CD5, and CD7).12-18
The treatment approach to AITL mirrors that of other nodal peripheral T-cell lymphomas, including chemotherapy and consideration of autologous stem-cell transplantation. Recent prospective trials of CHOP and CHOP-like chemotherapy have reported 3-year event-free survival and overall survival rates of 50% and 68%, respectively.19 Novel chemotherapeutic targets and gene-expression profiling are being investigated as potential therapeutic avenues.20
Conclusion
DRESS syndrome and AITL can have near-identical presentations. Clinicians should maintain a high index of suspicion for AITL in patients with presumed DRESS syndrome whose rash does not improve with appropriate drug withdrawal and steroid therapy or who lack a strong offending medication history. In such cases, skin and lymph node biopsies should be performed as early as possible to evaluate for AITL and so that appropriate therapy can be initiated.
Angioimmunoblastic T-cell lymphoma (AITL) is a rare and aggressive lymphoma arising from follicular T-helper cells that predominantly affects older adults and carries a 5-year overall survival rate of 32%.1 Notably, as many as 50% of AITL patients present with a skin rash in addition to the more common but nonspecific acute-onset generalized lymphadenopathy, hepatosplenomegaly, and anemia.2 At presentation, most AITL patients are already at an advanced (III/IV) stage of disease.
Formerly known as angioimmunoblastic lymphadenopathy with dysproteinemia, AITL was once considered a benign entity that carried a risk for malignant transformation. As more cases have been identified and explored, this entity has been recategorized as a frank lymphoma.3 Therefore, it is critical that AITL be diagnosed and treated as early as possible.
We present the case of a 65-year-old man with clinical features that resembled drug reaction with eosinophilia and systemic symptoms (DRESS syndrome). After extensive workup, he was found to have AITL. This atypical case highlights the importance of maintaining a flexible differential diagnosis in patients with a persistent rash that does not improve with appropriate drug withdrawal and therapy.
Case Report
A 65-year-old Filipino man whose medical history was notable for hepatitis B that had been treated with entecavir for years without issue was admitted to the internal medicine service with fever of unknown origin and malaise of approximately 6 weeks’ duration. Six days prior to admission and 5 days after completing courses of the antiviral oseltamivir phosphate and amoxicillin for an upper respiratory tract infection and sinusitis, he developed worsening of an intermittently pruritic rash of approximately 1 month's duration. The dermatology department was consulted the day of hospital admission for evaluation of the rash. Chronic home medications included entecavir, lisinopril/hydrochlorothiazide, amlodipine, atorvastatin, metformin, salsalate, and over-the-counter nonsteroidal anti-inflammatory drugs (NSAIDs) as needed.
Physical examination was notable for mild erythema and scale distributed across the entire face; mild facial edema; and a blanchable, nonconfluent, macular erythema distributed across the trunk and upper and proximal lower extremities (Figure). In addition, the patient displayed conjunctival injection, pitting edema of the hands, and bilateral cervical and inguinal lymphadenopathy.
Laboratory tests revealed mild leukocytosis (11.6×109/L, [reference range, 4.0–10.5×109/L]), anemia (hemoglobin, 125 g/L (reference range, 138–170 g/L); hematocrit, 36.9%, [reference range, 40.0%–50.0%)], eosinophilia (1.07×109/L [reference range, 0.00–0.70×109/L)], hyponatremia, hypokalemia, and a mildly elevated creatinine level. Computed tomography and full-body positron-emission tomography (PET) scans during admission demonstrated diffuse lymphadenopathy. A skin biopsy from the left chest and a left inguinal lymph node biopsy also were performed.
Despite the lack of a clear medication trigger within the usual timeline for severe cutaneous drug-induced hypersensitivity reactions, DRESS syndrome was high on the differential diagnosis at the time of the initial presentation given the diffuse morbilliform eruption with pruritus, facial edema, eosinophilia, and lymphadenopathy.
Home medications were discontinued except for amlodipine, atorvastatin, and entecavir. The patient was treated symptomatically with topical steroids because it was believed that, if the clinical presentation represented DRESS syndrome, it was a mild variant that could be treated topically.4 His case was considered mild because of a lack of confirmed organ dysfunction and a mild protracted course.
After discharge following a 3-day inpatient stay, the patient was followed in the clinic weekly for 3 weeks without considerable change in the skin or laboratory findings. Discontinuation of entecavir was discussed and approved by his hepatologist.
Posthospitalization analysis of the punch biopsy specimen from the chest performed during the patient’s hospital stay revealed a superficial and deep dermal lymphoid infiltrate comprising CD3-, CD5-, and programmed cell death protein 1–positive cells with cytologic atypia in a perivascular distribution. Analysis of the lymph node biopsy specimen performed during the hospitalization showed effacement of the nodal architecture, a polymorphous lymphoid cell population with irregular nuclear contour, and abundant clear cytoplasm associated with high endothelial venules (HEVs). Cells of interest were positive for CD3, CD4, CD2, CD5, and CD7, with a subset staining positive for programmed cell death protein 1, inducible costimulator, CD10, and chemokine (C-X-C motif) ligand (CXCL) 13. CD21 demonstrated an expanded follicular dendritic cell meshwork in association with HEVs. Polymerase chain reaction revealed a clonal T-cell population. These findings of the skin and lymph node biopsies were consistent with AITL. Subsequent bone marrow biopsy with flow cytometry showed a normal CD4:CD8 ratio in T cells and no increase in natural killer cells.
Cyclophosphamide–hydroxydaunorubicin–Oncovin–prednisone (CHOP) chemotherapy was initiated; the patient completed a total of 6 cycles. He has had near resolution of the skin findings and is considered in remission based on a PET scan performed approximately 7 months after the initial presentation.
Comment
Angioimmunoblastic T-cell lymphoma is a rare peripheral T-cell lymphoma, part of a group of aggressive neoplasms that constitute approximately 15% of peripheral T-cell lymphomas and approximately 2% of non-Hodgkin lymphomas in adults worldwide.5 Cutaneous involvement occurs in approximately half of AITL cases and can be the first manifestation of disease.2 Skin findings are largely nonspecific, ranging from simple morbilliform rashes to erythroderma, at times manifesting with purpura.
Given this variability in the presentation of AITL, early diagnosis is challenging in the absence of more specific signs and symptoms.2 It can conceivably be mistaken for common entities such as viral exanthems or drug eruptions, depending on the history and context. DRESS syndrome, a T cell-mediated, delayed type-IV hypersensitivity drug reaction can present in a manner highly similar to that of AITL, with cutaneous involvement (diffuse morbilliform rash, fever, facial edema, and generalized lymphadenopathy) and variable systemic involvement. Laboratory findings of eosinophilia, atypical lymphocytes, and thrombocytopenia also might be seen in both entities.6 Furthermore, the AITL in our patient was accompanied by electrolyte disturbances that were concerning for syndrome of inappropriate antidiuretic hormone secretion, a rare complication of patients with DRESS syndrome complicated by encephalitis.7,8
Our patient met 4 RegiSCAR criteria for DRESS syndrome, warranting high clinical suspicion for an offending drug.9 DRESS syndrome can be caused by numerous medications—most commonly anticonvulsants, sulfonamides, antibiotics, allopurinol, and NSAIDs. A review of our patient’s medication list identified NSAIDs (including salsalate), entecavir, and amoxicillin, as possible culpable medications. Notably, the only new addition to the patient’s regimen was amoxicillin, which did not fit the typical 2- to 8-week timeline for a DRESS syndrome nidus.10 Our patient’s fever began well before the antibiotic was initiated, and skin findings appeared within 1 week after the course of amoxicillin was completed. Although there is documented variability in the latency of onset of DRESS syndrome following administration of a culprit medication,11 it is critical to maintain a broad differential diagnosis to allow for further diagnostic information to be obtained, especially when the medication timeline does not align with the clinical presentation.
DRESS syndrome is far more common than AITL. Similarities in their clinical presentation pose a substantial challenge and often cause a delay in the diagnosis of AITL, which is made by excisional tissue biopsy, most commonly of a lymph node, with assessment of morphology and immunophenotyping. Histologic assessment of tissue reveals a polymorphous infiltrate of variably sized atypical lymphocytes with prominent arborizing HEVs as well as expanded populations of follicular dendritic cells that can be detected by CD21 staining. Cells express CD3 and CD4, variably express BCL6 (B-cell lymphoma 6 antigen) and CD10, and also may have partial or complete loss of expression of a subset of pan T-cell antigens (CD2, CD3, CD5, and CD7).12-18
The treatment approach to AITL mirrors that of other nodal peripheral T-cell lymphomas, including chemotherapy and consideration of autologous stem-cell transplantation. Recent prospective trials of CHOP and CHOP-like chemotherapy have reported 3-year event-free survival and overall survival rates of 50% and 68%, respectively.19 Novel chemotherapeutic targets and gene-expression profiling are being investigated as potential therapeutic avenues.20
Conclusion
DRESS syndrome and AITL can have near-identical presentations. Clinicians should maintain a high index of suspicion for AITL in patients with presumed DRESS syndrome whose rash does not improve with appropriate drug withdrawal and steroid therapy or who lack a strong offending medication history. In such cases, skin and lymph node biopsies should be performed as early as possible to evaluate for AITL and so that appropriate therapy can be initiated.
- Federico M, Rudiger T, Bellei M, et al. Clinicopathologic characteristics of angioimmunoblastic T-cell lymphoma: analysis of the international peripheral T-cell lymphoma project. J Clin Oncol. 2013;31:240-246. doi:10.1200/JCO.2011.37.3647
- Botros N, Cerroni L, Shawwa A, et al. Cutaneous manifestations of angioimmunoblastic T-cell lymphoma: clinical and pathological characteristics. Am J Dermatopathol. 2015;37:274-283. doi:10.1097/DAD.0000000000000144
- Sachsida-Colombo E, Barbosa Mariano LC, Bastos FQ, et al. A difficult case of angioimmunoblastic T-cell lymphoma to diagnose. Rev Bras Hematol Hemoter. 2016;38:82-85. doi:10.1016/j.bjhh.2015.11.002
- Funck-Brentano E, Duong T-A, Bouvresse S, et al. Therapeutic management of DRESS: a retrospective study of 38 cases. J Am Acad Dermatol. 2015;72:246-252. doi:10.1016/j.jaad.2014.10.032
- Lunning MA, Vose JM. Angioimmunoblastic T-cell lymphoma: the many-faced lymphoma. Blood. 2017;129:1095-1102. doi:10.1182/blood-2016-09-692541
- Sato R, Itoh M, Suzuki H, et al. Pathological findings of lymphadenopathy in drug-induced hypersensitivity syndrome (DIHS)/drug reaction with eosinophilia and systemic syndrome (DRESS): similarities with angioimmunoblastic T-cell lymphoma. Eur J Dermatol. 2017;27:201-202. doi:10.1684/ejd.2016.2954
- Osizik L, Tanriover MD, Saka E. Autoimmune limbic encephalitis and syndrome of inappropriate antidiuretic hormone secretion associated with lamotrigine-induced drug rash with eosinophilia and systemic symptoms (DRESS) syndrome. Intern Med. 2015;55:1393-1396. doi:10.2169/internalmedicine.55.6035
- Sakuma K, Kano Y, Fukuhara M, et al. Syndrome of inappropriate secretion of antidiuretic hormone associated with limbic encephalitis in a patient with drug-induced hypersensitivity syndrome. Clin Exp Dermatol. 2008;33:287-290. doi:10.1111/j.1365-2230.2007.02645.x
- Pannu AK, Saroch A. Diagnostic criteria for drug rash and eosinophilia with systemic symptoms. J Family Med Prim Care. 2017;6:693-694. doi:10.4103/2249-4863.222050
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al; RegiSCAR study group. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080. doi:10.1111/bjd.12501
- Soria A, Bernier C, Veyrac G, et al. Drug reaction with eosinophilia and systemic symptoms may occur within 2 weeks of drug exposure: a retrospective study. J Am Acad Dermatol. 2020;82:606.
- Loghavi S, Wang SA, Medeiros LJ, et al. Immunophenotypic and diagnostic characterization of angioimmunoblastic T-cell lymphoma by advanced flow cytometric technology. Leuk Lymphoma. 2016;57:2804-2812. doi:10.3109/10428194.2016.1170827
- Lee S-S, R, Odenwald T, et al. Angioimmunoblastic T cell lymphoma is derived from mature T-helper cells with varying expression and loss of detectable CD4. Int J Cancer. 2003;103:12-20. doi:10.1002/ijc.10758
- Feller AC, Griesser H, Schilling CV, et al. Clonal gene rearrangement patterns correlate with immunophenotype and clinical parameters in patients with angioimmunoblastic lymphadenopathy. Am J Pathol. 1988;133:549-556.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. IARC Press; 2008.
- Attygalle A, Al-Jehani R, Diss TC, et al. Neoplastic T cells in angioimmunoblastic T-cell lymphoma express CD10. Blood. 2002;99:627-633. doi:10.1182/blood.v99.2.627
- Mourad N, Mounier N, J, et al; Groupe d’Etude des Lymphomes de l’Adulte. Clinical, biologic, and pathologic features in 157 patients with angioimmunoblastic T-cell lymphoma treated within the Groupe d’Etude des Lymphomes de l’Adulte (GELA) trials. Blood. 2008;111:4463-4470. doi:10.1182/blood-2007-08-105759
- Marafioti T, Paterson JC, Ballabio E, et al. The inducible T-cell co-stimulator molecule is expressed on subsets of T cells and is a new marker of lymphomas of T follicular helper cell-derivation. Haematologica. 2010;95:432-439. doi:10.3324/haematol.2009.010991
- Schmitz N, L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients withT-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116:3418-3425. doi:10.1182/blood-2010-02-270785
- Moskowitz AJ. Practical treatment approach for angioimmunoblastic T-cell lymphoma. J Oncol Pract. 2019;15:137-143. doi:10.1200/JOP.18.00511
- Federico M, Rudiger T, Bellei M, et al. Clinicopathologic characteristics of angioimmunoblastic T-cell lymphoma: analysis of the international peripheral T-cell lymphoma project. J Clin Oncol. 2013;31:240-246. doi:10.1200/JCO.2011.37.3647
- Botros N, Cerroni L, Shawwa A, et al. Cutaneous manifestations of angioimmunoblastic T-cell lymphoma: clinical and pathological characteristics. Am J Dermatopathol. 2015;37:274-283. doi:10.1097/DAD.0000000000000144
- Sachsida-Colombo E, Barbosa Mariano LC, Bastos FQ, et al. A difficult case of angioimmunoblastic T-cell lymphoma to diagnose. Rev Bras Hematol Hemoter. 2016;38:82-85. doi:10.1016/j.bjhh.2015.11.002
- Funck-Brentano E, Duong T-A, Bouvresse S, et al. Therapeutic management of DRESS: a retrospective study of 38 cases. J Am Acad Dermatol. 2015;72:246-252. doi:10.1016/j.jaad.2014.10.032
- Lunning MA, Vose JM. Angioimmunoblastic T-cell lymphoma: the many-faced lymphoma. Blood. 2017;129:1095-1102. doi:10.1182/blood-2016-09-692541
- Sato R, Itoh M, Suzuki H, et al. Pathological findings of lymphadenopathy in drug-induced hypersensitivity syndrome (DIHS)/drug reaction with eosinophilia and systemic syndrome (DRESS): similarities with angioimmunoblastic T-cell lymphoma. Eur J Dermatol. 2017;27:201-202. doi:10.1684/ejd.2016.2954
- Osizik L, Tanriover MD, Saka E. Autoimmune limbic encephalitis and syndrome of inappropriate antidiuretic hormone secretion associated with lamotrigine-induced drug rash with eosinophilia and systemic symptoms (DRESS) syndrome. Intern Med. 2015;55:1393-1396. doi:10.2169/internalmedicine.55.6035
- Sakuma K, Kano Y, Fukuhara M, et al. Syndrome of inappropriate secretion of antidiuretic hormone associated with limbic encephalitis in a patient with drug-induced hypersensitivity syndrome. Clin Exp Dermatol. 2008;33:287-290. doi:10.1111/j.1365-2230.2007.02645.x
- Pannu AK, Saroch A. Diagnostic criteria for drug rash and eosinophilia with systemic symptoms. J Family Med Prim Care. 2017;6:693-694. doi:10.4103/2249-4863.222050
- Kardaun SH, Sekula P, Valeyrie-Allanore L, et al; RegiSCAR study group. Drug reaction with eosinophilia and systemic symptoms (DRESS): an original multisystem adverse drug reaction. results from the prospective RegiSCAR study. Br J Dermatol. 2013;169:1071-1080. doi:10.1111/bjd.12501
- Soria A, Bernier C, Veyrac G, et al. Drug reaction with eosinophilia and systemic symptoms may occur within 2 weeks of drug exposure: a retrospective study. J Am Acad Dermatol. 2020;82:606.
- Loghavi S, Wang SA, Medeiros LJ, et al. Immunophenotypic and diagnostic characterization of angioimmunoblastic T-cell lymphoma by advanced flow cytometric technology. Leuk Lymphoma. 2016;57:2804-2812. doi:10.3109/10428194.2016.1170827
- Lee S-S, R, Odenwald T, et al. Angioimmunoblastic T cell lymphoma is derived from mature T-helper cells with varying expression and loss of detectable CD4. Int J Cancer. 2003;103:12-20. doi:10.1002/ijc.10758
- Feller AC, Griesser H, Schilling CV, et al. Clonal gene rearrangement patterns correlate with immunophenotype and clinical parameters in patients with angioimmunoblastic lymphadenopathy. Am J Pathol. 1988;133:549-556.
- Swerdlow SH, Campo E, Harris NL, et al, eds. World Health Organization Classification of Tumours of Haematopoietic and Lymphoid Tissues. IARC Press; 2008.
- Attygalle A, Al-Jehani R, Diss TC, et al. Neoplastic T cells in angioimmunoblastic T-cell lymphoma express CD10. Blood. 2002;99:627-633. doi:10.1182/blood.v99.2.627
- Mourad N, Mounier N, J, et al; Groupe d’Etude des Lymphomes de l’Adulte. Clinical, biologic, and pathologic features in 157 patients with angioimmunoblastic T-cell lymphoma treated within the Groupe d’Etude des Lymphomes de l’Adulte (GELA) trials. Blood. 2008;111:4463-4470. doi:10.1182/blood-2007-08-105759
- Marafioti T, Paterson JC, Ballabio E, et al. The inducible T-cell co-stimulator molecule is expressed on subsets of T cells and is a new marker of lymphomas of T follicular helper cell-derivation. Haematologica. 2010;95:432-439. doi:10.3324/haematol.2009.010991
- Schmitz N, L, Ziepert M, et al. Treatment and prognosis of mature T-cell and NK-cell lymphoma: an analysis of patients withT-cell lymphoma treated in studies of the German High-Grade Non-Hodgkin Lymphoma Study Group. Blood. 2010;116:3418-3425. doi:10.1182/blood-2010-02-270785
- Moskowitz AJ. Practical treatment approach for angioimmunoblastic T-cell lymphoma. J Oncol Pract. 2019;15:137-143. doi:10.1200/JOP.18.00511
Practice Points
- It is important to maintain a high index of suspicion for angioimmunoblastic T-cell lymphoma in older patients with a longstanding rash and no clear culprit for drug reaction with eosinophilia and systemic symptoms (DRESS syndrome).
- Consider performing a lymph node biopsy early in the course of disease in patients with presumed DRESS syndrome who do not improve with drug withdrawal and steroid therapy.
Take action: Turn up the heat on prior auth
In our recent member survey, 99% of respondents expressed that prior authorization has a negative impact on patients’ access to clinically appropriate treatments. We need to continue to put pressure on legislators to eliminate prior authorization burdens.
AGA endorses the Improving Seniors Timely Access to Care Act, which would streamline the prior authorization process in Medicare Advantage by approving in real-time commonly approved services and implementing a standardized electronic prior authorization process.
Despite large bipartisan support, we need your help getting this bill across the finish line! Please take five minutes to ask your Representative to cosponsor this necessary bill by participating in our campaign.
Go to the AGA action center to contact your lawmakers!
In our recent member survey, 99% of respondents expressed that prior authorization has a negative impact on patients’ access to clinically appropriate treatments. We need to continue to put pressure on legislators to eliminate prior authorization burdens.
AGA endorses the Improving Seniors Timely Access to Care Act, which would streamline the prior authorization process in Medicare Advantage by approving in real-time commonly approved services and implementing a standardized electronic prior authorization process.
Despite large bipartisan support, we need your help getting this bill across the finish line! Please take five minutes to ask your Representative to cosponsor this necessary bill by participating in our campaign.
Go to the AGA action center to contact your lawmakers!
In our recent member survey, 99% of respondents expressed that prior authorization has a negative impact on patients’ access to clinically appropriate treatments. We need to continue to put pressure on legislators to eliminate prior authorization burdens.
AGA endorses the Improving Seniors Timely Access to Care Act, which would streamline the prior authorization process in Medicare Advantage by approving in real-time commonly approved services and implementing a standardized electronic prior authorization process.
Despite large bipartisan support, we need your help getting this bill across the finish line! Please take five minutes to ask your Representative to cosponsor this necessary bill by participating in our campaign.
Go to the AGA action center to contact your lawmakers!
2022 AGA recognition prize award recipients

“AGA is proud to officially announce the exceptional individuals selected for 2022 AGA Recognition Prizes. I wish to thank all the nominators and those who provided nomination letters, and the selection committees for the tough task they had to select among the many superb nominees,” said Bishr Omary, MD, PhD, AGAF, chair of the AGA. “Please join us in congratulating this year’s distinguished awardees and applauding their contributions to the field of gastroenterology that advance our profession and the patients we serve.”
AGA looks forward to celebrating the recipients during Digestive Disease Week® 2022, May 21-24, in San Diego, Calif.
Meet and learn more about our award recipients here.

“AGA is proud to officially announce the exceptional individuals selected for 2022 AGA Recognition Prizes. I wish to thank all the nominators and those who provided nomination letters, and the selection committees for the tough task they had to select among the many superb nominees,” said Bishr Omary, MD, PhD, AGAF, chair of the AGA. “Please join us in congratulating this year’s distinguished awardees and applauding their contributions to the field of gastroenterology that advance our profession and the patients we serve.”
AGA looks forward to celebrating the recipients during Digestive Disease Week® 2022, May 21-24, in San Diego, Calif.
Meet and learn more about our award recipients here.

“AGA is proud to officially announce the exceptional individuals selected for 2022 AGA Recognition Prizes. I wish to thank all the nominators and those who provided nomination letters, and the selection committees for the tough task they had to select among the many superb nominees,” said Bishr Omary, MD, PhD, AGAF, chair of the AGA. “Please join us in congratulating this year’s distinguished awardees and applauding their contributions to the field of gastroenterology that advance our profession and the patients we serve.”
AGA looks forward to celebrating the recipients during Digestive Disease Week® 2022, May 21-24, in San Diego, Calif.
Meet and learn more about our award recipients here.
New governing board members
M. Bishr Omary, MD, PhD, AGAF, chair of the AGA Nominating Committee, is pleased to announce that Maria T. Abreu, MD, AGAF, joins the presidential line-up for AGA.
Vice President
Maria T. Abreu, MD, AGAF
Director, Crohn’s and Colitis Center
University of Miami
Maria T. Abreu, MD, AGAF, has more than 20 years of leadership experience in basic, translational, and clinical research and mentoring. She is AGA’s current councillor at-large, past chair of the AGA Institute Council, and an AGA Institute Council Section Research Mentor Award recipient (2020) for the IMIBD section. Dr. Abreu is also a recipient of the 2019 Sherman Prize by The Bruce and Cynthia Sherman Charitable Foundation that recognizes outstanding achievements in intestinal bowel disease.
Read her bio from the University of Miami.
The nominating committee also appointed the following slate of councillors which is subject to membership vote.
At-Large Councillor
Kim Barrett, PhD, AGAF
Vice dean for research
University of California, Davis
Kim Barrett, PhD, AGAF, is the current chair of the AGA Publications Committee, former chair of the AGA Ethics And Audit Committees, and served twice as director of the Academic Skills Workshop. She was recognized with AGA’s top research award, the AGA Distinguished Achievement Award in Basic Science (2021).
Her research interests have centered on the physiology and pathophysiology of the intestinal epithelium and their relevance to inflammatory bowel diseases and diarrheal diseases and have resulted in more than 300 publications.
Read her bio from UC Davis.
Councillor For Development And Growth
Lawrence Kosinski, MD, MBA, AGAF
Chief medical officer
SonarMD
A serial entrepreneur and thought leader in the world of value-based payment, Larry Kosinski, MD, MBA, AGAF, currently serves as chief medical officer of SonarMD, the leading value-based care coordination solution for complex chronic diseases. He founded SonarMD in 2014 to make it easier for specialists and patients to work together to manage symptomatic chronic illness and prevent clinical deterioration, improving health outcomes, and lowering the cost of care.
In 2021, Dr. Kosinski was selected for his expertise in value-based payment to serve on the Centers for Medicare & Medicaid Services’ Physician-Focused Payment Model Technical Advisory Committee and help develop bold, new Medicare payment models.
Read his bio from the SonarMD website.
Education & Training Councillor
Sheryl Pfeil, MD, AGAF
Medical director and professor of clinical medicine, Clinical Skills Education and Assessment Center
The Ohio State University Wexner Medical Center
Sheryl Pfeil, MD, AGAF, has been an AGA member for 30 years, serving on the Education And Training Committee, as past chair of the Academy of Educators, as cochair of the AGA future leaders program, and on the editorial board for Gastro Hep Advances. Dr. Pfeil has 30 years of experience in medical education, leading medical students, residents, and fellow education.
Her educational research interests include professional development, training and assessment methods, and virtual education.
Read her bio from The Ohio State University.
Pending approval by the voting membership, all board members begin their terms after DDW 2022. The voting membership will be sent a ballot to approve the slate of councillors on or before March 28, 2022, with a response date of no later than April 29, 2022. Results will be announced at the AGA Annual Business Meeting on June 1, 2022.
M. Bishr Omary, MD, PhD, AGAF, chair of the AGA Nominating Committee, is pleased to announce that Maria T. Abreu, MD, AGAF, joins the presidential line-up for AGA.
Vice President
Maria T. Abreu, MD, AGAF
Director, Crohn’s and Colitis Center
University of Miami
Maria T. Abreu, MD, AGAF, has more than 20 years of leadership experience in basic, translational, and clinical research and mentoring. She is AGA’s current councillor at-large, past chair of the AGA Institute Council, and an AGA Institute Council Section Research Mentor Award recipient (2020) for the IMIBD section. Dr. Abreu is also a recipient of the 2019 Sherman Prize by The Bruce and Cynthia Sherman Charitable Foundation that recognizes outstanding achievements in intestinal bowel disease.
Read her bio from the University of Miami.
The nominating committee also appointed the following slate of councillors which is subject to membership vote.
At-Large Councillor
Kim Barrett, PhD, AGAF
Vice dean for research
University of California, Davis
Kim Barrett, PhD, AGAF, is the current chair of the AGA Publications Committee, former chair of the AGA Ethics And Audit Committees, and served twice as director of the Academic Skills Workshop. She was recognized with AGA’s top research award, the AGA Distinguished Achievement Award in Basic Science (2021).
Her research interests have centered on the physiology and pathophysiology of the intestinal epithelium and their relevance to inflammatory bowel diseases and diarrheal diseases and have resulted in more than 300 publications.
Read her bio from UC Davis.
Councillor For Development And Growth
Lawrence Kosinski, MD, MBA, AGAF
Chief medical officer
SonarMD
A serial entrepreneur and thought leader in the world of value-based payment, Larry Kosinski, MD, MBA, AGAF, currently serves as chief medical officer of SonarMD, the leading value-based care coordination solution for complex chronic diseases. He founded SonarMD in 2014 to make it easier for specialists and patients to work together to manage symptomatic chronic illness and prevent clinical deterioration, improving health outcomes, and lowering the cost of care.
In 2021, Dr. Kosinski was selected for his expertise in value-based payment to serve on the Centers for Medicare & Medicaid Services’ Physician-Focused Payment Model Technical Advisory Committee and help develop bold, new Medicare payment models.
Read his bio from the SonarMD website.
Education & Training Councillor
Sheryl Pfeil, MD, AGAF
Medical director and professor of clinical medicine, Clinical Skills Education and Assessment Center
The Ohio State University Wexner Medical Center
Sheryl Pfeil, MD, AGAF, has been an AGA member for 30 years, serving on the Education And Training Committee, as past chair of the Academy of Educators, as cochair of the AGA future leaders program, and on the editorial board for Gastro Hep Advances. Dr. Pfeil has 30 years of experience in medical education, leading medical students, residents, and fellow education.
Her educational research interests include professional development, training and assessment methods, and virtual education.
Read her bio from The Ohio State University.
Pending approval by the voting membership, all board members begin their terms after DDW 2022. The voting membership will be sent a ballot to approve the slate of councillors on or before March 28, 2022, with a response date of no later than April 29, 2022. Results will be announced at the AGA Annual Business Meeting on June 1, 2022.
M. Bishr Omary, MD, PhD, AGAF, chair of the AGA Nominating Committee, is pleased to announce that Maria T. Abreu, MD, AGAF, joins the presidential line-up for AGA.
Vice President
Maria T. Abreu, MD, AGAF
Director, Crohn’s and Colitis Center
University of Miami
Maria T. Abreu, MD, AGAF, has more than 20 years of leadership experience in basic, translational, and clinical research and mentoring. She is AGA’s current councillor at-large, past chair of the AGA Institute Council, and an AGA Institute Council Section Research Mentor Award recipient (2020) for the IMIBD section. Dr. Abreu is also a recipient of the 2019 Sherman Prize by The Bruce and Cynthia Sherman Charitable Foundation that recognizes outstanding achievements in intestinal bowel disease.
Read her bio from the University of Miami.
The nominating committee also appointed the following slate of councillors which is subject to membership vote.
At-Large Councillor
Kim Barrett, PhD, AGAF
Vice dean for research
University of California, Davis
Kim Barrett, PhD, AGAF, is the current chair of the AGA Publications Committee, former chair of the AGA Ethics And Audit Committees, and served twice as director of the Academic Skills Workshop. She was recognized with AGA’s top research award, the AGA Distinguished Achievement Award in Basic Science (2021).
Her research interests have centered on the physiology and pathophysiology of the intestinal epithelium and their relevance to inflammatory bowel diseases and diarrheal diseases and have resulted in more than 300 publications.
Read her bio from UC Davis.
Councillor For Development And Growth
Lawrence Kosinski, MD, MBA, AGAF
Chief medical officer
SonarMD
A serial entrepreneur and thought leader in the world of value-based payment, Larry Kosinski, MD, MBA, AGAF, currently serves as chief medical officer of SonarMD, the leading value-based care coordination solution for complex chronic diseases. He founded SonarMD in 2014 to make it easier for specialists and patients to work together to manage symptomatic chronic illness and prevent clinical deterioration, improving health outcomes, and lowering the cost of care.
In 2021, Dr. Kosinski was selected for his expertise in value-based payment to serve on the Centers for Medicare & Medicaid Services’ Physician-Focused Payment Model Technical Advisory Committee and help develop bold, new Medicare payment models.
Read his bio from the SonarMD website.
Education & Training Councillor
Sheryl Pfeil, MD, AGAF
Medical director and professor of clinical medicine, Clinical Skills Education and Assessment Center
The Ohio State University Wexner Medical Center
Sheryl Pfeil, MD, AGAF, has been an AGA member for 30 years, serving on the Education And Training Committee, as past chair of the Academy of Educators, as cochair of the AGA future leaders program, and on the editorial board for Gastro Hep Advances. Dr. Pfeil has 30 years of experience in medical education, leading medical students, residents, and fellow education.
Her educational research interests include professional development, training and assessment methods, and virtual education.
Read her bio from The Ohio State University.
Pending approval by the voting membership, all board members begin their terms after DDW 2022. The voting membership will be sent a ballot to approve the slate of councillors on or before March 28, 2022, with a response date of no later than April 29, 2022. Results will be announced at the AGA Annual Business Meeting on June 1, 2022.
Fertility after tubal ligation – It’s a matter of ‘AGE’
Despite the original intent of permanent contraception, tubal sterilization regret is experienced by 2%-26% of women as demonstrated by the United States Collaborative Review of Sterilization “CREST” 14-year study (Obstet Gynecol. 1999 Jun;93[6]:889-95). Regret appears to be higher in the United States than Europe and in resource-limited countries and is more common in women who are less than age 30, African-American, and unmarried. Nevertheless, requests for tubal reversal are estimated to be between 1% and 4% (Contraception. 1981 Jun;23[6]:579-89). The alternative option for fertility is in vitro fertilization (IVF) and this month’s column considers the pros and cons of both methods.
The procedure of tubal reanastomosis involves removing abnormal tissue and reapproximating the healthy tubal segments with attention to minimize adhesion formation through continued gentle irrigation. The surgery involves microsuturing using 6-0 to 10-0 sutures. Tubal patency can be confirmed during the procedure and with a subsequent hysterosalpingogram. While time from sterilization and the type of sterilization technique are factors that may influence the success rate of tubal reanastomosis, the age of the woman is the most predictive for pregnancy outcome.
In the original CREST study, the risk of ectopic pregnancy following tubal reanastomosis was contingent on the method of sterilization: Bipolar electrosurgery resulted in the highest probability of ectopic pregnancy (17.1 per 1,000 procedures at 10 years after permanent contraception), while postpartum partial salpingectomy resulted in the lowest (1.5 per 1,000 procedures at 10 years after permanent contraception) (N Engl J Med. 1997;336[11]:762). Comparatively, the ectopic pregnancy rate during an IVF cycle was 1.9% for pregnancies from transfers of fresh cleavage embryo, followed by transfers of frozen cleavage embryo (1.7%), transfers of fresh blastocyst (1.3%), and transfers of frozen blastocyst (0.8%) (Hum Reprod. 2015;30[9]:2048-54).
Reports vary regarding pregnancy rates from tubal reanastomosis. Prior use of rings and clips for sterilization appear to yield the highest outcomes as opposed to the use of electrocautery. In one large Canadian cohort study of over 300,000 women, those aged 15-30 years, 30-33 years, and 34-49 years had a conception rate of 73%, 64%, and 46%, respectively (Obstet Gynecol. 2003;101[4]:677-84). Most pregnancies were within 2 years after reversal and 48% of women achieved a delivery. Of interest, 23% of patients subsequently underwent another sterilization.
An Australian study of nearly 2,000 women found an overall cumulative live-delivery rate of 20% within the first year after reversal, 40% at 2 years, 51% at 5 years, and 52% at 10 years. As expected, the 5-year cumulative live-delivery rate was significantly lower in women who were aged 40-44 years (26%), compared with younger women. For all women below age 40 years, the live-delivery rate was approximately 50% within 5 years after tubal reanastomosis, while the rate halves after the age of 40 (Fertil Steril. 2015 Oct;104[4]:921-6).
To compare tubal reanastomosis with IVF, a retrospective cohort study of 163 patients demonstrated the cumulative delivery rate over 72 months was comparable for IVF vs. sterilization reversal (52% vs. 60%). The only significant difference was in a subset of patients aged <37 years (52% after IVF and 72% after reversal) and the lower cost of surgery. The authors advocated laparoscopic sterilization reversal in women younger than 37 years who have ≥4 cm of residual tube with IVF as the better alternative for all other women (Hum Reprod. 2007;22[10]:2660).
Indeed, tubal length is another important factor in successful reversal. The pregnancy rate after tubal anastomosis is 75% in women with tubal length of 4 cm or more, but only 19% in those with shorter tubes (Fertil Steril. 1987;48[1]:13-7). The literature does suggest equivalent pregnancy rates after laparoscopic tubal anastomosis and conventional microsurgical anastomosis. Although the laparoscopic approach may be more economical, it is more demanding technically than an open microsurgical procedure.
Tubal reanastomosis can also be performed using robot-assisted laparoscopy. In preliminary studies, robotic surgery appears to have a similar success rate and a shorter recovery time, but longer operative times and higher costs (Obstet Gynecol. 2007;109[6]:1375; Fertil Steril. 2008;90[4]:1175).
To educate women on the success of IVF based on individual characteristics, a valuable tool to approximate the cumulative outcome for a live birth following one cycle of IVF is offered by the Society for Assisted Reproductive Technology. To clarify, a cycle of IVF consists of one egg retrieval and the ultimate transfer of all embryos produced, i.e., fresh and frozen. The website also includes estimations of success following a second and third IVF cycle.
The woman’s age is a significant predictor of IVF success. Ovarian aging is currently best measured by combining chronologic age, antral follicle count (AFC) by transvaginal pelvic ultrasound, and serum anti-Müllerian hormone (AMH). Natural fecundity begins to decline, on average, above age 32-33 years. An AFC less than 11 reflects diminished ovarian reserve (DOR) and less than 6 is severe. AMH levels below 1.6 ng/mL have been shown to reduce the number of eggs retrieved with IVF, while levels below 0.4 ng/mL are very low. Very low AMH levels negatively affect the outcome of IVF cycles as demonstrated in the SART data study from a population of women with a mean age of 39.4 years: Cycle cancellation was 54%; of all retrieval attempts, no oocytes were obtained in 5.4%, and no embryo transfer occurred in 25.1% of cycles; the live birth rate per embryo transfer was 20.5% (9.5% per cycle start and 16.3% per retrieval) from a mean age of 36.8 years (Fertil Steril. 2016 Feb;105[2]:385-93.e3). The predictive ability of AMH on the live birth rate from IVF cycles was also shown in a study of over 85,000 women (Fertil Steril. 2018;109:258-65).
While low AMH has been shown to lessen a successful outcome from IVF, there appears to be no difference in natural pregnancy rates in women aged 30-44 years irrespective of AMH levels (JAMA. 2017;318[14]:1367-76). Of importance, the use of AMH in a population at low risk for DOR will yield a larger number of false-positive results (i.e., characterizing a woman as DOR when in fact she has normal ovarian reserve). Further, users of hormonal contraceptives have a 25.2% lower mean AMH level than nonusers.
When a patient is considering tubal reanastomosis vs. IVF, a useful acronym to remember is to check “AGE” – the A is for AMH because severely diminished ovarian reserve will reduce success with IVF as shown by the SART calculator; the G represents guy, i.e., ensuring a reasonably normal sperm analysis; and E stands for eggs representing ovulation function. In a woman who is anovulatory and who will require fertility medication, it would be reasonable to consider IVF given the need for ovarian stimulation. As in females, advanced paternal age has demonstrated a decline in fertility and sperm analysis parameters. Men above age 45 take approximately five times as long to achieve a pregnancy, compared with men less than 25 years of age. Further, there is evidence for advanced paternal age increasing risk of miscarriage, preterm birth, and birth defects. Men older than 40-45 years have twice the risk of an autistic child and five times the risk of having a child with schizophrenia (Transl Psychiatry 2017;7: e1019; Am J Psychiatry. 2002;159:1528-33).
To conclude, the data support consideration for sterilization reversal in women less than age 37 years with more than 4 cm of residual functional fallopian tube and the prior use of rings or clip sterilization. In other women, IVF may be the better option, particularly when ovulation dysfunction and/or male factor is present. IVF also offers the advantage of maintaining contraception and gender determination. However, given that AMH does not appear to reduce natural fertility, unlike during its effect during an IVF cycle, the option of tubal reversal may be more favorable in women with severe DOR.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Despite the original intent of permanent contraception, tubal sterilization regret is experienced by 2%-26% of women as demonstrated by the United States Collaborative Review of Sterilization “CREST” 14-year study (Obstet Gynecol. 1999 Jun;93[6]:889-95). Regret appears to be higher in the United States than Europe and in resource-limited countries and is more common in women who are less than age 30, African-American, and unmarried. Nevertheless, requests for tubal reversal are estimated to be between 1% and 4% (Contraception. 1981 Jun;23[6]:579-89). The alternative option for fertility is in vitro fertilization (IVF) and this month’s column considers the pros and cons of both methods.
The procedure of tubal reanastomosis involves removing abnormal tissue and reapproximating the healthy tubal segments with attention to minimize adhesion formation through continued gentle irrigation. The surgery involves microsuturing using 6-0 to 10-0 sutures. Tubal patency can be confirmed during the procedure and with a subsequent hysterosalpingogram. While time from sterilization and the type of sterilization technique are factors that may influence the success rate of tubal reanastomosis, the age of the woman is the most predictive for pregnancy outcome.
In the original CREST study, the risk of ectopic pregnancy following tubal reanastomosis was contingent on the method of sterilization: Bipolar electrosurgery resulted in the highest probability of ectopic pregnancy (17.1 per 1,000 procedures at 10 years after permanent contraception), while postpartum partial salpingectomy resulted in the lowest (1.5 per 1,000 procedures at 10 years after permanent contraception) (N Engl J Med. 1997;336[11]:762). Comparatively, the ectopic pregnancy rate during an IVF cycle was 1.9% for pregnancies from transfers of fresh cleavage embryo, followed by transfers of frozen cleavage embryo (1.7%), transfers of fresh blastocyst (1.3%), and transfers of frozen blastocyst (0.8%) (Hum Reprod. 2015;30[9]:2048-54).
Reports vary regarding pregnancy rates from tubal reanastomosis. Prior use of rings and clips for sterilization appear to yield the highest outcomes as opposed to the use of electrocautery. In one large Canadian cohort study of over 300,000 women, those aged 15-30 years, 30-33 years, and 34-49 years had a conception rate of 73%, 64%, and 46%, respectively (Obstet Gynecol. 2003;101[4]:677-84). Most pregnancies were within 2 years after reversal and 48% of women achieved a delivery. Of interest, 23% of patients subsequently underwent another sterilization.
An Australian study of nearly 2,000 women found an overall cumulative live-delivery rate of 20% within the first year after reversal, 40% at 2 years, 51% at 5 years, and 52% at 10 years. As expected, the 5-year cumulative live-delivery rate was significantly lower in women who were aged 40-44 years (26%), compared with younger women. For all women below age 40 years, the live-delivery rate was approximately 50% within 5 years after tubal reanastomosis, while the rate halves after the age of 40 (Fertil Steril. 2015 Oct;104[4]:921-6).
To compare tubal reanastomosis with IVF, a retrospective cohort study of 163 patients demonstrated the cumulative delivery rate over 72 months was comparable for IVF vs. sterilization reversal (52% vs. 60%). The only significant difference was in a subset of patients aged <37 years (52% after IVF and 72% after reversal) and the lower cost of surgery. The authors advocated laparoscopic sterilization reversal in women younger than 37 years who have ≥4 cm of residual tube with IVF as the better alternative for all other women (Hum Reprod. 2007;22[10]:2660).
Indeed, tubal length is another important factor in successful reversal. The pregnancy rate after tubal anastomosis is 75% in women with tubal length of 4 cm or more, but only 19% in those with shorter tubes (Fertil Steril. 1987;48[1]:13-7). The literature does suggest equivalent pregnancy rates after laparoscopic tubal anastomosis and conventional microsurgical anastomosis. Although the laparoscopic approach may be more economical, it is more demanding technically than an open microsurgical procedure.
Tubal reanastomosis can also be performed using robot-assisted laparoscopy. In preliminary studies, robotic surgery appears to have a similar success rate and a shorter recovery time, but longer operative times and higher costs (Obstet Gynecol. 2007;109[6]:1375; Fertil Steril. 2008;90[4]:1175).
To educate women on the success of IVF based on individual characteristics, a valuable tool to approximate the cumulative outcome for a live birth following one cycle of IVF is offered by the Society for Assisted Reproductive Technology. To clarify, a cycle of IVF consists of one egg retrieval and the ultimate transfer of all embryos produced, i.e., fresh and frozen. The website also includes estimations of success following a second and third IVF cycle.
The woman’s age is a significant predictor of IVF success. Ovarian aging is currently best measured by combining chronologic age, antral follicle count (AFC) by transvaginal pelvic ultrasound, and serum anti-Müllerian hormone (AMH). Natural fecundity begins to decline, on average, above age 32-33 years. An AFC less than 11 reflects diminished ovarian reserve (DOR) and less than 6 is severe. AMH levels below 1.6 ng/mL have been shown to reduce the number of eggs retrieved with IVF, while levels below 0.4 ng/mL are very low. Very low AMH levels negatively affect the outcome of IVF cycles as demonstrated in the SART data study from a population of women with a mean age of 39.4 years: Cycle cancellation was 54%; of all retrieval attempts, no oocytes were obtained in 5.4%, and no embryo transfer occurred in 25.1% of cycles; the live birth rate per embryo transfer was 20.5% (9.5% per cycle start and 16.3% per retrieval) from a mean age of 36.8 years (Fertil Steril. 2016 Feb;105[2]:385-93.e3). The predictive ability of AMH on the live birth rate from IVF cycles was also shown in a study of over 85,000 women (Fertil Steril. 2018;109:258-65).
While low AMH has been shown to lessen a successful outcome from IVF, there appears to be no difference in natural pregnancy rates in women aged 30-44 years irrespective of AMH levels (JAMA. 2017;318[14]:1367-76). Of importance, the use of AMH in a population at low risk for DOR will yield a larger number of false-positive results (i.e., characterizing a woman as DOR when in fact she has normal ovarian reserve). Further, users of hormonal contraceptives have a 25.2% lower mean AMH level than nonusers.
When a patient is considering tubal reanastomosis vs. IVF, a useful acronym to remember is to check “AGE” – the A is for AMH because severely diminished ovarian reserve will reduce success with IVF as shown by the SART calculator; the G represents guy, i.e., ensuring a reasonably normal sperm analysis; and E stands for eggs representing ovulation function. In a woman who is anovulatory and who will require fertility medication, it would be reasonable to consider IVF given the need for ovarian stimulation. As in females, advanced paternal age has demonstrated a decline in fertility and sperm analysis parameters. Men above age 45 take approximately five times as long to achieve a pregnancy, compared with men less than 25 years of age. Further, there is evidence for advanced paternal age increasing risk of miscarriage, preterm birth, and birth defects. Men older than 40-45 years have twice the risk of an autistic child and five times the risk of having a child with schizophrenia (Transl Psychiatry 2017;7: e1019; Am J Psychiatry. 2002;159:1528-33).
To conclude, the data support consideration for sterilization reversal in women less than age 37 years with more than 4 cm of residual functional fallopian tube and the prior use of rings or clip sterilization. In other women, IVF may be the better option, particularly when ovulation dysfunction and/or male factor is present. IVF also offers the advantage of maintaining contraception and gender determination. However, given that AMH does not appear to reduce natural fertility, unlike during its effect during an IVF cycle, the option of tubal reversal may be more favorable in women with severe DOR.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Despite the original intent of permanent contraception, tubal sterilization regret is experienced by 2%-26% of women as demonstrated by the United States Collaborative Review of Sterilization “CREST” 14-year study (Obstet Gynecol. 1999 Jun;93[6]:889-95). Regret appears to be higher in the United States than Europe and in resource-limited countries and is more common in women who are less than age 30, African-American, and unmarried. Nevertheless, requests for tubal reversal are estimated to be between 1% and 4% (Contraception. 1981 Jun;23[6]:579-89). The alternative option for fertility is in vitro fertilization (IVF) and this month’s column considers the pros and cons of both methods.
The procedure of tubal reanastomosis involves removing abnormal tissue and reapproximating the healthy tubal segments with attention to minimize adhesion formation through continued gentle irrigation. The surgery involves microsuturing using 6-0 to 10-0 sutures. Tubal patency can be confirmed during the procedure and with a subsequent hysterosalpingogram. While time from sterilization and the type of sterilization technique are factors that may influence the success rate of tubal reanastomosis, the age of the woman is the most predictive for pregnancy outcome.
In the original CREST study, the risk of ectopic pregnancy following tubal reanastomosis was contingent on the method of sterilization: Bipolar electrosurgery resulted in the highest probability of ectopic pregnancy (17.1 per 1,000 procedures at 10 years after permanent contraception), while postpartum partial salpingectomy resulted in the lowest (1.5 per 1,000 procedures at 10 years after permanent contraception) (N Engl J Med. 1997;336[11]:762). Comparatively, the ectopic pregnancy rate during an IVF cycle was 1.9% for pregnancies from transfers of fresh cleavage embryo, followed by transfers of frozen cleavage embryo (1.7%), transfers of fresh blastocyst (1.3%), and transfers of frozen blastocyst (0.8%) (Hum Reprod. 2015;30[9]:2048-54).
Reports vary regarding pregnancy rates from tubal reanastomosis. Prior use of rings and clips for sterilization appear to yield the highest outcomes as opposed to the use of electrocautery. In one large Canadian cohort study of over 300,000 women, those aged 15-30 years, 30-33 years, and 34-49 years had a conception rate of 73%, 64%, and 46%, respectively (Obstet Gynecol. 2003;101[4]:677-84). Most pregnancies were within 2 years after reversal and 48% of women achieved a delivery. Of interest, 23% of patients subsequently underwent another sterilization.
An Australian study of nearly 2,000 women found an overall cumulative live-delivery rate of 20% within the first year after reversal, 40% at 2 years, 51% at 5 years, and 52% at 10 years. As expected, the 5-year cumulative live-delivery rate was significantly lower in women who were aged 40-44 years (26%), compared with younger women. For all women below age 40 years, the live-delivery rate was approximately 50% within 5 years after tubal reanastomosis, while the rate halves after the age of 40 (Fertil Steril. 2015 Oct;104[4]:921-6).
To compare tubal reanastomosis with IVF, a retrospective cohort study of 163 patients demonstrated the cumulative delivery rate over 72 months was comparable for IVF vs. sterilization reversal (52% vs. 60%). The only significant difference was in a subset of patients aged <37 years (52% after IVF and 72% after reversal) and the lower cost of surgery. The authors advocated laparoscopic sterilization reversal in women younger than 37 years who have ≥4 cm of residual tube with IVF as the better alternative for all other women (Hum Reprod. 2007;22[10]:2660).
Indeed, tubal length is another important factor in successful reversal. The pregnancy rate after tubal anastomosis is 75% in women with tubal length of 4 cm or more, but only 19% in those with shorter tubes (Fertil Steril. 1987;48[1]:13-7). The literature does suggest equivalent pregnancy rates after laparoscopic tubal anastomosis and conventional microsurgical anastomosis. Although the laparoscopic approach may be more economical, it is more demanding technically than an open microsurgical procedure.
Tubal reanastomosis can also be performed using robot-assisted laparoscopy. In preliminary studies, robotic surgery appears to have a similar success rate and a shorter recovery time, but longer operative times and higher costs (Obstet Gynecol. 2007;109[6]:1375; Fertil Steril. 2008;90[4]:1175).
To educate women on the success of IVF based on individual characteristics, a valuable tool to approximate the cumulative outcome for a live birth following one cycle of IVF is offered by the Society for Assisted Reproductive Technology. To clarify, a cycle of IVF consists of one egg retrieval and the ultimate transfer of all embryos produced, i.e., fresh and frozen. The website also includes estimations of success following a second and third IVF cycle.
The woman’s age is a significant predictor of IVF success. Ovarian aging is currently best measured by combining chronologic age, antral follicle count (AFC) by transvaginal pelvic ultrasound, and serum anti-Müllerian hormone (AMH). Natural fecundity begins to decline, on average, above age 32-33 years. An AFC less than 11 reflects diminished ovarian reserve (DOR) and less than 6 is severe. AMH levels below 1.6 ng/mL have been shown to reduce the number of eggs retrieved with IVF, while levels below 0.4 ng/mL are very low. Very low AMH levels negatively affect the outcome of IVF cycles as demonstrated in the SART data study from a population of women with a mean age of 39.4 years: Cycle cancellation was 54%; of all retrieval attempts, no oocytes were obtained in 5.4%, and no embryo transfer occurred in 25.1% of cycles; the live birth rate per embryo transfer was 20.5% (9.5% per cycle start and 16.3% per retrieval) from a mean age of 36.8 years (Fertil Steril. 2016 Feb;105[2]:385-93.e3). The predictive ability of AMH on the live birth rate from IVF cycles was also shown in a study of over 85,000 women (Fertil Steril. 2018;109:258-65).
While low AMH has been shown to lessen a successful outcome from IVF, there appears to be no difference in natural pregnancy rates in women aged 30-44 years irrespective of AMH levels (JAMA. 2017;318[14]:1367-76). Of importance, the use of AMH in a population at low risk for DOR will yield a larger number of false-positive results (i.e., characterizing a woman as DOR when in fact she has normal ovarian reserve). Further, users of hormonal contraceptives have a 25.2% lower mean AMH level than nonusers.
When a patient is considering tubal reanastomosis vs. IVF, a useful acronym to remember is to check “AGE” – the A is for AMH because severely diminished ovarian reserve will reduce success with IVF as shown by the SART calculator; the G represents guy, i.e., ensuring a reasonably normal sperm analysis; and E stands for eggs representing ovulation function. In a woman who is anovulatory and who will require fertility medication, it would be reasonable to consider IVF given the need for ovarian stimulation. As in females, advanced paternal age has demonstrated a decline in fertility and sperm analysis parameters. Men above age 45 take approximately five times as long to achieve a pregnancy, compared with men less than 25 years of age. Further, there is evidence for advanced paternal age increasing risk of miscarriage, preterm birth, and birth defects. Men older than 40-45 years have twice the risk of an autistic child and five times the risk of having a child with schizophrenia (Transl Psychiatry 2017;7: e1019; Am J Psychiatry. 2002;159:1528-33).
To conclude, the data support consideration for sterilization reversal in women less than age 37 years with more than 4 cm of residual functional fallopian tube and the prior use of rings or clip sterilization. In other women, IVF may be the better option, particularly when ovulation dysfunction and/or male factor is present. IVF also offers the advantage of maintaining contraception and gender determination. However, given that AMH does not appear to reduce natural fertility, unlike during its effect during an IVF cycle, the option of tubal reversal may be more favorable in women with severe DOR.
Dr. Trolice is director of the IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando.
Five reasons to update your will
You have a will, so you can rest easy, right? Not necessarily. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
#1. Family changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
#2. Relocating to a new state
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
#3. Tax law changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
#4. You want to support a favorite cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
#5. Changes in your estate’s value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
Consider including a gift to the AGA Research Foundation in your will. You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at foundation@gastro.org.
You have a will, so you can rest easy, right? Not necessarily. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
#1. Family changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
#2. Relocating to a new state
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
#3. Tax law changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
#4. You want to support a favorite cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
#5. Changes in your estate’s value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
Consider including a gift to the AGA Research Foundation in your will. You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at foundation@gastro.org.
You have a will, so you can rest easy, right? Not necessarily. Even though it can provide for some contingencies, an old will can’t cover every change that may have occurred since it was first drawn. Professionals advise that you review your will every few years and more often if situations such as the following five have occurred since you last updated your will.
#1. Family changes
If you’ve had any changes in your family situation, you will probably need to update your will. Events such as marriage, divorce, death, birth, adoption, or a falling out with a loved one may affect how your estate will be distributed, who should act as guardian for your dependents, and who should be named as executor of your estate.
#2. Relocating to a new state
The laws among the states vary. Moving to a new state or purchasing property in another state can affect your estate plan and how property in that state will be taxed and distributed.
#3. Tax law changes
Federal and state legislatures are continually tinkering with federal estate and state inheritance tax laws. An old will may fail to take advantage of strategies that will minimize estate taxes.
#4. You want to support a favorite cause
If you have developed a connection to a cause, you may want to benefit a particular charity with a gift in your estate. Contact us for sample language you can share with your attorney to include a gift to us in your will.
#5. Changes in your estate’s value
When you made your will, your assets may have been relatively modest. Now the value may be larger and your will no longer reflects how you would like your estate divided.
Consider including a gift to the AGA Research Foundation in your will. You will help spark future discoveries in GI. Visit our website at https://gastro.planmylegacy.org or contact us at foundation@gastro.org.
Different variants may cause different long COVID symptoms: Study
Long COVID symptoms may differ depending on which SARS-CoV-2 variant is behind a person’s infection, a new study shows.
The data from Italy compared long COVID symptoms reported by patients infected with SARS-CoV-2 from March to December 2020 (when the original, or “Wuhan,” variant was dominant) with those reported by patients infected from January to April 2021 (B.1.1.7-, or Alpha variant-dominant). It showed a substantial change in the pattern of neurological and cognitive/emotional problems – the latter mostly seen with the Alpha variant.
Infectious disease specialist Michele Spinicci, MD, from the University of Florence and Careggi University Hospital, Italy, led the work. “Many of the symptoms reported in this study have been measured [before], but this is the first time they have been linked to different COVID-19 variants,” he told this news organization. “Findings in patients with long COVID were focused on neurological and psychological difficulties.”
However, he pointed out that much remains to be understood about long COVID in terms of symptoms, diagnosis, and treatment.
“Long COVID is a huge area that involves many different fields of medicine, so there is not one single piece of advice to give on management. There’s lots to consider when evaluating a long COVID patient,” he said.
Results showed that when the Alpha variant was the dominant variant, the prevalence of myalgia (10%), dyspnea (42%), brain fog/mental confusion (17%), and anxiety/depression (13%) significantly increased relative to the wild-type (original, Wuhan) variant, while anosmia (2%), dysgeusia (4%), and impaired hearing (1%) were less common.
When the wild-type (original, Wuhan) variant was dominant, fatigue (37%), insomnia (16%), dysgeusia (11%), and impaired hearing (5%) were all more common than with the Alpha variant. Dyspnea (33%), brain fog (10%), myalgia (4%), and anxiety/depression (6%) were less common.
Overall, 76% of the patients in the trial reported at least one persistent symptom, while the most common reported symptoms were dyspnea (37%) and chronic fatigue (36%), followed by insomnia (16%), visual disorders (13%), and brain fog (13%).
The findings come from an early-release abstract that will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, in a few weeks’ time.
‘The take-home point’
Michael A. Horberg, MD, associate medical director, Kaiser Permanente – Mid-Atlantic Permanente Medical Group, Rockville, Maryland, has recently presented data on symptoms seen with long COVID in over 28,000 people, as reported by this news organization, at the Conference on Retroviruses and Opportunistic Infections 2022. These people were infected with the wild-type virus.
Commenting on the study by Dr. Spinicci, he said: “The issue is that as we go along the COVID lifespan from acute to long COVID, what prompts patients to seek medical attention may change. If symptoms are not severe or were not well publicized previously, patients may not see the need to seek care or evaluation. As such, it doesn’t surprise me to find these changes over time, independent of any potential biological activity of the virus or its consequences.”
Dr. Horberg noted that their own study results are consistent with those of Dr. Spinicci et al. from March to December 2020 (original, Wuhan variant). “To me, the take-home point is long COVID is real, and physicians need to be on the lookout for it. However, not all symptoms are due to long COVID, and we need to keep the time course of symptoms during evaluation of such patients.”
Also providing comment on the findings was Debby Bogaert, MD, chair of Pediatric Medicine, University of Edinburgh. Reflecting on whether the symptoms were due to long COVID or another underlying disease, she said: “The number of patients with ongoing symptoms is very high, therefore [it is] unlikely that all of this is re-emergence of underlying or previous health problems. The type of symptoms reported are also as reported by other cohorts, so not unexpected. And irrespective of the root cause, they require care.”
Dr. Bogaert also noted that the data reiterate that COVID-19 is a new disease, and that “new variants might show shifting clinical pictures, not only regarding severity and symptoms of acute disease, but possibly also regarding sequela,” and that this, “underlines the importance of ongoing surveillance of variants, and ongoing evaluation of the acute and long-term clinical picture accompanying these, to ensure we adapt our public health approaches, clinical treatment plans, and long-term follow-up when and where needed.”
Dr. Bogaert stressed that only by keeping track of the changes in symptoms both acute and long-term – by patients and doctors – would the best patient care be provided.
“Patients need to know so they can report these back to their doctors, and doctors need to know over time that the picture of sequela might shift, so sequela are recognized early, and these patients receive the appropriate follow-up treatment,” she said. These shifting patterns might also apply to community patients as well as those hospitalized with COVID-19.
Study details
The retrospective, observational study included 428 patients, 59% men, with a mean age of 64 years, who had been treated at the Careggi University Hospital’s post-COVID outpatient service between June 2020 and June 2021, when the original form of SARS-CoV-2, and later the Alpha variant, were circulating, with some overlap.
All patients had been hospitalized with COVID-19 and discharged 4-12 weeks prior to attending the outpatient post-COVID service. They were asked to complete a questionnaire on persistent symptoms at the median of 53 days after being discharged from the hospital. In addition, data on medical history, microbiological and clinical COVID-19 course, self-reported symptoms (at the point of the follow-up visit), and patient demographics were obtained from electronic medical records.
Newer variants being studied
Upon analysis of long COVID symptoms according to treatment given during the acute phase using multivariate analysis, increasing oxygen support (odds ratio, 1.4; 95% confidence interval, 1.1-1.8), use of immunosuppressant drugs (OR, 6.4; 95% CI, 1.5-28), and female sex (OR, 1.8; 95% CI, 1.1-2.9) were associated with a higher risk for long COVID symptoms, while patients with type 2 diabetes (OR, 0.4; 95% CI, 0.2-0.7) had a lower risk of developing long COVID symptoms.
When asked whether the increased anxiety and depression seen with the Alpha variant might be also linked to the fact that people are living through hard times, with lockdowns, economic difficulties, possible illness, and even fatalities among family and friends due to COVID, Dr. Spinicci pointed out that “it’s a preliminary study, and there are lots of factors that we didn’t explore. It’s difficult to arrive at definite conclusions about long COVID because so much remains unknown. There are lots of external and environmental factors in the general population that might contribute to these findings.”
Dr. Spinicci has continued to enroll patients from later periods of the pandemic, including patients who were infected with the Delta and Omicron variants of SARS-CoV-2.
“We’re interested in finding out if these other variants are also associated with different phenotypes of long COVID. This study is part of our follow-up program here in the hospital where lots of different specialties are following patients for 20 months,” he said.
Dr. Horberg noted that one criticism of this study is that it was unclear whether the researchers accounted for pre-existing conditions. “They note the co-morbidities in the table 1, but don’t say how they accounted for that in their analyses. We found a lot of what patients were calling ‘long COVID’ were exacerbations of co-morbidities but not a new condition.”
Dr. Spinicci and his coauthors acknowledged that the study was observational. And, as such, it does not prove cause and effect, and they could not confirm which variant of the virus caused the infection in different patients, which may limit the conclusions that can be drawn.
“Future research should focus on the potential impacts of variants of concern and vaccination status on ongoing symptoms,” Spinicci said.
Early release of an abstract will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, April 23-26, 2022. Abstract 02768.
Dr. Spinicci and Dr. Horberg have disclosed no relevant financial relationships. Dr. Bogaert declared that she is on the program committee of ECCMID; she has been a member of SIGN/NICE COVID-19 rapid guideline: managing the long-term effects of COVID-19; and she is involved in multiple ongoing COVID-related studies, both acute and long-term sequela (funding MRC, CSO, ZonMw).
A version of this article first appeared on Medscape.com.
Long COVID symptoms may differ depending on which SARS-CoV-2 variant is behind a person’s infection, a new study shows.
The data from Italy compared long COVID symptoms reported by patients infected with SARS-CoV-2 from March to December 2020 (when the original, or “Wuhan,” variant was dominant) with those reported by patients infected from January to April 2021 (B.1.1.7-, or Alpha variant-dominant). It showed a substantial change in the pattern of neurological and cognitive/emotional problems – the latter mostly seen with the Alpha variant.
Infectious disease specialist Michele Spinicci, MD, from the University of Florence and Careggi University Hospital, Italy, led the work. “Many of the symptoms reported in this study have been measured [before], but this is the first time they have been linked to different COVID-19 variants,” he told this news organization. “Findings in patients with long COVID were focused on neurological and psychological difficulties.”
However, he pointed out that much remains to be understood about long COVID in terms of symptoms, diagnosis, and treatment.
“Long COVID is a huge area that involves many different fields of medicine, so there is not one single piece of advice to give on management. There’s lots to consider when evaluating a long COVID patient,” he said.
Results showed that when the Alpha variant was the dominant variant, the prevalence of myalgia (10%), dyspnea (42%), brain fog/mental confusion (17%), and anxiety/depression (13%) significantly increased relative to the wild-type (original, Wuhan) variant, while anosmia (2%), dysgeusia (4%), and impaired hearing (1%) were less common.
When the wild-type (original, Wuhan) variant was dominant, fatigue (37%), insomnia (16%), dysgeusia (11%), and impaired hearing (5%) were all more common than with the Alpha variant. Dyspnea (33%), brain fog (10%), myalgia (4%), and anxiety/depression (6%) were less common.
Overall, 76% of the patients in the trial reported at least one persistent symptom, while the most common reported symptoms were dyspnea (37%) and chronic fatigue (36%), followed by insomnia (16%), visual disorders (13%), and brain fog (13%).
The findings come from an early-release abstract that will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, in a few weeks’ time.
‘The take-home point’
Michael A. Horberg, MD, associate medical director, Kaiser Permanente – Mid-Atlantic Permanente Medical Group, Rockville, Maryland, has recently presented data on symptoms seen with long COVID in over 28,000 people, as reported by this news organization, at the Conference on Retroviruses and Opportunistic Infections 2022. These people were infected with the wild-type virus.
Commenting on the study by Dr. Spinicci, he said: “The issue is that as we go along the COVID lifespan from acute to long COVID, what prompts patients to seek medical attention may change. If symptoms are not severe or were not well publicized previously, patients may not see the need to seek care or evaluation. As such, it doesn’t surprise me to find these changes over time, independent of any potential biological activity of the virus or its consequences.”
Dr. Horberg noted that their own study results are consistent with those of Dr. Spinicci et al. from March to December 2020 (original, Wuhan variant). “To me, the take-home point is long COVID is real, and physicians need to be on the lookout for it. However, not all symptoms are due to long COVID, and we need to keep the time course of symptoms during evaluation of such patients.”
Also providing comment on the findings was Debby Bogaert, MD, chair of Pediatric Medicine, University of Edinburgh. Reflecting on whether the symptoms were due to long COVID or another underlying disease, she said: “The number of patients with ongoing symptoms is very high, therefore [it is] unlikely that all of this is re-emergence of underlying or previous health problems. The type of symptoms reported are also as reported by other cohorts, so not unexpected. And irrespective of the root cause, they require care.”
Dr. Bogaert also noted that the data reiterate that COVID-19 is a new disease, and that “new variants might show shifting clinical pictures, not only regarding severity and symptoms of acute disease, but possibly also regarding sequela,” and that this, “underlines the importance of ongoing surveillance of variants, and ongoing evaluation of the acute and long-term clinical picture accompanying these, to ensure we adapt our public health approaches, clinical treatment plans, and long-term follow-up when and where needed.”
Dr. Bogaert stressed that only by keeping track of the changes in symptoms both acute and long-term – by patients and doctors – would the best patient care be provided.
“Patients need to know so they can report these back to their doctors, and doctors need to know over time that the picture of sequela might shift, so sequela are recognized early, and these patients receive the appropriate follow-up treatment,” she said. These shifting patterns might also apply to community patients as well as those hospitalized with COVID-19.
Study details
The retrospective, observational study included 428 patients, 59% men, with a mean age of 64 years, who had been treated at the Careggi University Hospital’s post-COVID outpatient service between June 2020 and June 2021, when the original form of SARS-CoV-2, and later the Alpha variant, were circulating, with some overlap.
All patients had been hospitalized with COVID-19 and discharged 4-12 weeks prior to attending the outpatient post-COVID service. They were asked to complete a questionnaire on persistent symptoms at the median of 53 days after being discharged from the hospital. In addition, data on medical history, microbiological and clinical COVID-19 course, self-reported symptoms (at the point of the follow-up visit), and patient demographics were obtained from electronic medical records.
Newer variants being studied
Upon analysis of long COVID symptoms according to treatment given during the acute phase using multivariate analysis, increasing oxygen support (odds ratio, 1.4; 95% confidence interval, 1.1-1.8), use of immunosuppressant drugs (OR, 6.4; 95% CI, 1.5-28), and female sex (OR, 1.8; 95% CI, 1.1-2.9) were associated with a higher risk for long COVID symptoms, while patients with type 2 diabetes (OR, 0.4; 95% CI, 0.2-0.7) had a lower risk of developing long COVID symptoms.
When asked whether the increased anxiety and depression seen with the Alpha variant might be also linked to the fact that people are living through hard times, with lockdowns, economic difficulties, possible illness, and even fatalities among family and friends due to COVID, Dr. Spinicci pointed out that “it’s a preliminary study, and there are lots of factors that we didn’t explore. It’s difficult to arrive at definite conclusions about long COVID because so much remains unknown. There are lots of external and environmental factors in the general population that might contribute to these findings.”
Dr. Spinicci has continued to enroll patients from later periods of the pandemic, including patients who were infected with the Delta and Omicron variants of SARS-CoV-2.
“We’re interested in finding out if these other variants are also associated with different phenotypes of long COVID. This study is part of our follow-up program here in the hospital where lots of different specialties are following patients for 20 months,” he said.
Dr. Horberg noted that one criticism of this study is that it was unclear whether the researchers accounted for pre-existing conditions. “They note the co-morbidities in the table 1, but don’t say how they accounted for that in their analyses. We found a lot of what patients were calling ‘long COVID’ were exacerbations of co-morbidities but not a new condition.”
Dr. Spinicci and his coauthors acknowledged that the study was observational. And, as such, it does not prove cause and effect, and they could not confirm which variant of the virus caused the infection in different patients, which may limit the conclusions that can be drawn.
“Future research should focus on the potential impacts of variants of concern and vaccination status on ongoing symptoms,” Spinicci said.
Early release of an abstract will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, April 23-26, 2022. Abstract 02768.
Dr. Spinicci and Dr. Horberg have disclosed no relevant financial relationships. Dr. Bogaert declared that she is on the program committee of ECCMID; she has been a member of SIGN/NICE COVID-19 rapid guideline: managing the long-term effects of COVID-19; and she is involved in multiple ongoing COVID-related studies, both acute and long-term sequela (funding MRC, CSO, ZonMw).
A version of this article first appeared on Medscape.com.
Long COVID symptoms may differ depending on which SARS-CoV-2 variant is behind a person’s infection, a new study shows.
The data from Italy compared long COVID symptoms reported by patients infected with SARS-CoV-2 from March to December 2020 (when the original, or “Wuhan,” variant was dominant) with those reported by patients infected from January to April 2021 (B.1.1.7-, or Alpha variant-dominant). It showed a substantial change in the pattern of neurological and cognitive/emotional problems – the latter mostly seen with the Alpha variant.
Infectious disease specialist Michele Spinicci, MD, from the University of Florence and Careggi University Hospital, Italy, led the work. “Many of the symptoms reported in this study have been measured [before], but this is the first time they have been linked to different COVID-19 variants,” he told this news organization. “Findings in patients with long COVID were focused on neurological and psychological difficulties.”
However, he pointed out that much remains to be understood about long COVID in terms of symptoms, diagnosis, and treatment.
“Long COVID is a huge area that involves many different fields of medicine, so there is not one single piece of advice to give on management. There’s lots to consider when evaluating a long COVID patient,” he said.
Results showed that when the Alpha variant was the dominant variant, the prevalence of myalgia (10%), dyspnea (42%), brain fog/mental confusion (17%), and anxiety/depression (13%) significantly increased relative to the wild-type (original, Wuhan) variant, while anosmia (2%), dysgeusia (4%), and impaired hearing (1%) were less common.
When the wild-type (original, Wuhan) variant was dominant, fatigue (37%), insomnia (16%), dysgeusia (11%), and impaired hearing (5%) were all more common than with the Alpha variant. Dyspnea (33%), brain fog (10%), myalgia (4%), and anxiety/depression (6%) were less common.
Overall, 76% of the patients in the trial reported at least one persistent symptom, while the most common reported symptoms were dyspnea (37%) and chronic fatigue (36%), followed by insomnia (16%), visual disorders (13%), and brain fog (13%).
The findings come from an early-release abstract that will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, in a few weeks’ time.
‘The take-home point’
Michael A. Horberg, MD, associate medical director, Kaiser Permanente – Mid-Atlantic Permanente Medical Group, Rockville, Maryland, has recently presented data on symptoms seen with long COVID in over 28,000 people, as reported by this news organization, at the Conference on Retroviruses and Opportunistic Infections 2022. These people were infected with the wild-type virus.
Commenting on the study by Dr. Spinicci, he said: “The issue is that as we go along the COVID lifespan from acute to long COVID, what prompts patients to seek medical attention may change. If symptoms are not severe or were not well publicized previously, patients may not see the need to seek care or evaluation. As such, it doesn’t surprise me to find these changes over time, independent of any potential biological activity of the virus or its consequences.”
Dr. Horberg noted that their own study results are consistent with those of Dr. Spinicci et al. from March to December 2020 (original, Wuhan variant). “To me, the take-home point is long COVID is real, and physicians need to be on the lookout for it. However, not all symptoms are due to long COVID, and we need to keep the time course of symptoms during evaluation of such patients.”
Also providing comment on the findings was Debby Bogaert, MD, chair of Pediatric Medicine, University of Edinburgh. Reflecting on whether the symptoms were due to long COVID or another underlying disease, she said: “The number of patients with ongoing symptoms is very high, therefore [it is] unlikely that all of this is re-emergence of underlying or previous health problems. The type of symptoms reported are also as reported by other cohorts, so not unexpected. And irrespective of the root cause, they require care.”
Dr. Bogaert also noted that the data reiterate that COVID-19 is a new disease, and that “new variants might show shifting clinical pictures, not only regarding severity and symptoms of acute disease, but possibly also regarding sequela,” and that this, “underlines the importance of ongoing surveillance of variants, and ongoing evaluation of the acute and long-term clinical picture accompanying these, to ensure we adapt our public health approaches, clinical treatment plans, and long-term follow-up when and where needed.”
Dr. Bogaert stressed that only by keeping track of the changes in symptoms both acute and long-term – by patients and doctors – would the best patient care be provided.
“Patients need to know so they can report these back to their doctors, and doctors need to know over time that the picture of sequela might shift, so sequela are recognized early, and these patients receive the appropriate follow-up treatment,” she said. These shifting patterns might also apply to community patients as well as those hospitalized with COVID-19.
Study details
The retrospective, observational study included 428 patients, 59% men, with a mean age of 64 years, who had been treated at the Careggi University Hospital’s post-COVID outpatient service between June 2020 and June 2021, when the original form of SARS-CoV-2, and later the Alpha variant, were circulating, with some overlap.
All patients had been hospitalized with COVID-19 and discharged 4-12 weeks prior to attending the outpatient post-COVID service. They were asked to complete a questionnaire on persistent symptoms at the median of 53 days after being discharged from the hospital. In addition, data on medical history, microbiological and clinical COVID-19 course, self-reported symptoms (at the point of the follow-up visit), and patient demographics were obtained from electronic medical records.
Newer variants being studied
Upon analysis of long COVID symptoms according to treatment given during the acute phase using multivariate analysis, increasing oxygen support (odds ratio, 1.4; 95% confidence interval, 1.1-1.8), use of immunosuppressant drugs (OR, 6.4; 95% CI, 1.5-28), and female sex (OR, 1.8; 95% CI, 1.1-2.9) were associated with a higher risk for long COVID symptoms, while patients with type 2 diabetes (OR, 0.4; 95% CI, 0.2-0.7) had a lower risk of developing long COVID symptoms.
When asked whether the increased anxiety and depression seen with the Alpha variant might be also linked to the fact that people are living through hard times, with lockdowns, economic difficulties, possible illness, and even fatalities among family and friends due to COVID, Dr. Spinicci pointed out that “it’s a preliminary study, and there are lots of factors that we didn’t explore. It’s difficult to arrive at definite conclusions about long COVID because so much remains unknown. There are lots of external and environmental factors in the general population that might contribute to these findings.”
Dr. Spinicci has continued to enroll patients from later periods of the pandemic, including patients who were infected with the Delta and Omicron variants of SARS-CoV-2.
“We’re interested in finding out if these other variants are also associated with different phenotypes of long COVID. This study is part of our follow-up program here in the hospital where lots of different specialties are following patients for 20 months,” he said.
Dr. Horberg noted that one criticism of this study is that it was unclear whether the researchers accounted for pre-existing conditions. “They note the co-morbidities in the table 1, but don’t say how they accounted for that in their analyses. We found a lot of what patients were calling ‘long COVID’ were exacerbations of co-morbidities but not a new condition.”
Dr. Spinicci and his coauthors acknowledged that the study was observational. And, as such, it does not prove cause and effect, and they could not confirm which variant of the virus caused the infection in different patients, which may limit the conclusions that can be drawn.
“Future research should focus on the potential impacts of variants of concern and vaccination status on ongoing symptoms,” Spinicci said.
Early release of an abstract will be presented at the European Congress of Clinical Microbiology & Infectious Diseases (ECCMID) 2022, in Lisbon, Portugal, April 23-26, 2022. Abstract 02768.
Dr. Spinicci and Dr. Horberg have disclosed no relevant financial relationships. Dr. Bogaert declared that she is on the program committee of ECCMID; she has been a member of SIGN/NICE COVID-19 rapid guideline: managing the long-term effects of COVID-19; and she is involved in multiple ongoing COVID-related studies, both acute and long-term sequela (funding MRC, CSO, ZonMw).
A version of this article first appeared on Medscape.com.
CROI 2022