User login
For MD-IQ use only
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
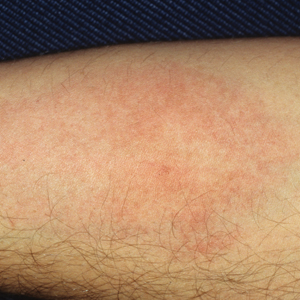
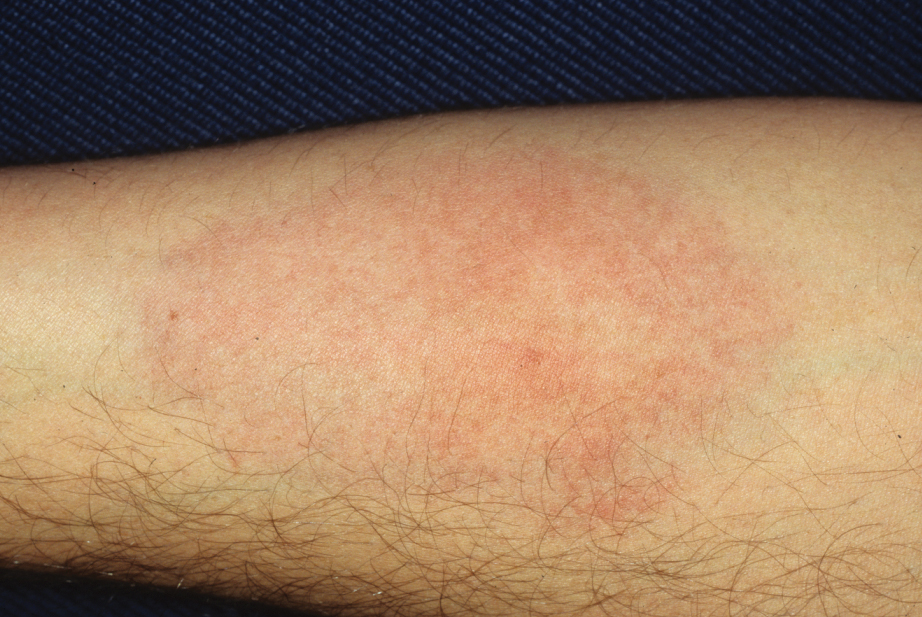
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3
Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
Triatome bugs cause painful bites and serve as vectors for Chagas disease. In this article, we will address diagnosis and vector identification.
Key Morphologic Features
Insects from the subfamily Triatominae are identifiable by their long legs and a shieldlike abdomen behind a platelike pronotum that covers the thorax. Their half-membranous wings overlap, covering the central abdomen but leaving the lateral portions visible. Tigerlike stripes are characteristically prominent on the visible portions of the lateral abdomen. The stalklike head has an articulated beaklike mouth that can be retracted and used to deliver a powerful bite (Figure 1).

Feeding Mechanisms and Host Reactions
Triatome bugs are blood-feeding arthropods that hide in cracks and crevices in domestic structures by day and feed at night. They are shy feeders, and laboratory colonies have been known to die rather than feed in daylight. They are particularly common in thatched or wattle-and-daub dwellings, where they can be present in great numbers and descend on sleeping inhabitants at night. Triatome bugs require regular blood meals throughout the 5 developmental nymph stages in order to undergo successful molting.
In the wild, triatome bugs feed on a range of animals with little specificity, but in domestic settings they feed largely on humans. Thermosensors in the antennae help them locate blood vessels under the skin, which they penetrate easily due to their long mouthparts. Like other blood-sucking arthropods, they release an anticoagulant that facilitates continuous blood flow while feeding, which accounts for many of the cutaneous reactions observed after the host sustains a triatomine bite.1
Triatomine bugs have trouble feeding through clothing and seek out exposed skin, particularly the eyelids, producing the characteristic unilateral eyelid swelling known as the Romaña sign. Other bite reactions include purpura; macular erythema; and vesiculobullous, papular, and urticarial lesions (Figure 2).2 Associated lymphangitis or lymphadenopathy may be noted, and anaphylaxis has been reported. Similar to those of cockroaches, triatome antigens have been associated with atopic dermatitis and asthma.3

Chagas Disease Risk and Transmission
Triatomine reduviids are the primary vector of Chagas disease, and the geographic range of both continues to expand, particularly in North America. The disease remains endemic in Latin America, with the highest incidence now reported in Brazil.4 An estimated 240,000 to 350,000 individuals in the United States are infected, primarily immigrants from Mexico, Central America, and South America; approximately 30% of those infected will develop cardiac and/or gastrointestinal complications.4 If left untreated, Chagas disease leads to autonomic ganglion destruction and subsequent gastrointestinal and cardiac complications, including megacolon, dilated cardiomyopathy, and heart failure.5
Trypanosoma cruzi, the microorganism responsible for Chagas disease, is spread to humans through triatomine fecal matter scratched into the bite wound.6 Triatomine bugs have a highly developed gastrocolic reflex and defecate liberally as they feed. Fecal volume is heavily dependent on species and sex, with fifth-stage female nymphs producing the highest volume of excrement and thereby acting as particularly adept disease vectors.6 Triatoma infestans and members of the genus Mepraia are key vectors of T cruzi.1 In areas of South America where populations of T infestans are controlled through public health measures, Mepraia emerge as a largely uncontrolled disease vector.1,7 While endemic to the southern United States and South America, T cruzi has spread to much of North America and Europe by way of Triatominae as naturalized or invasive species.8
There are 3 phases of Chagas disease: acute, indeterminate, and chronic. A chagoma is a localized erythematous swelling at the site of the bite. The acute phase often lacks systemic symptoms but may include fever, myalgia, and headache. The intermediate phase may include fatigue and recurrent fevers. The most serious manifestations occur in the chronic phase and include cardiomyopathy with signs of congestive heart failure, irregular heartbeat, cardiac arrest, abdominal pain, constipation, and dysphagia.
Deforestation has been identified as a driving factor in the spread of Chagas disease, as the disease vectors shift from wilderness areas and animal hosts to inhabited areas where humans are the most readily available food source. Triatome bugs in areas experiencing higher levels of development or forest harvesting are forced into human-populated areas. As a result, instances of Chagas disease are on the rise in these communities.7 Salvador, Bahia, Brazil, has been identified as one such target of increased vector presence due to heavy deforestation, and the hottest months were identified as having the greatest threat of vector exposure.9 Brazil became the leading geographic area for the disease partly because of heavy loss of forested land.10
Vector Control and Prevention Strategies
Elimination of cracks and crevices in walls; replacement of wattle and daub with stucco, plaster, and other solid building materials; and the use of insecticides with durability in the environment have been used to reduce triatome bug infestation in homes. However, highly persistent insecticides carry greater environmental risk and may drive resistance as declining concentrations select for resistant arthropods. Repellents have less environmental impact and play an important role in vector control. Citronella essential oil has been observed to repel several species of triatome bugs that are common in Arizona; specifically, the component alcohols geraniol and citronellol were found to be effective at inhibiting triatome feeding.11
Early detection of Chagas disease is essential, as end-stage cardiomyopathy and megacolon are difficult to treat. Newly developed multiantigen testing has shown promising results, suggesting a potential for more accurate testing for Chagas disease.8 Geospatial tracking and mapping of T cruzi vectors now are employed to track seasonal vector changes and disease patterns.9 Researchers also have developed a dedicated dichotomous key for the identification of triatome bugs endemic in Brazil with the hope of better identification and mapping of disease vector presence and density.10 The key consists of a series of statements with 2 choices in each step. It uses observable features of the arthropod to lead users to the correct identification.
Final Thoughts
Identification of triatome bugs can help with public health efforts to control the spread of disease. Patients with unilateral eyelid swelling should be evaluated for possible bedbug or triatome exposure.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
- Egaña C, Pinto R, Vergara F, et al. Fluctuations in Trypanosoma cruzi discrete typing unit composition in two naturally infected triatomines: Mepraia gajardoi and M. spinolai after laboratory feeding. Acta Trop. 2016;160:9-14. Erratum in: Acta Trop. 2016;162:248. doi:10.1016/j.actatropica.2016.04.008
- Moffitt JE, Venarske D, Goddard J, et al. Allergic reactions to Triatoma bites. Ann Allergy Asthma Immunol. 2003;91:122-128.
- Alonso A, Potenza M, Mouchián K, et al. Proteinase and gelatinolytic properties of a Triatoma infestans extract. Allergol Immunopathol (Madr). 2004;32:223-227.
- Hochberg NS, Montgomery SP. Chagas disease. Ann Intern Med. 2023;176:ITC17-ITC32. doi:10.7326/AITC202302210
- Pless M, Juranek D, Kozarsky P, et al. The epidemiology of Chagas’ disease in a hyperendemic area of Cochabamba, Bolivia: a clinical study including electrocardiography, seroreactivity to Trypanosoma cruzi, xenodiagnosis, and domiciliary triatomine distribution. Am J Trop Med Hyg. 1992;47:539-546.
- Piesman J, Sherlock IA. Factors controlling the volume of feces produced by triatomine vectors of Chagas’ disease. Acta Trop. 1983;40:351-358.
- Steverding D. The history of Chagas disease. Parasit Vectors. 2014;10:317.
- Granjon E, Dichtel-Danjoy ML, Saba E, et al. Development of a novel multiplex immunoassay multi-cruzi for the serological confirmation of Chagas disease. PLoS Negl Trop Dis. 2016;10:e0004596.
- Santana Kde S, Bavia ME, Lima AD, et al. Spatial distribution of triatomines (Reduviidae: Triatominae) in urban areas of the city of Salvador, Bahia, Brazil. Geospat Health. 2011;5:199-203.
- de Mello DV, Nhapulo EF, Cesaretto LP, et al. Dichotomous keys based on cytogenetic data for triatomines reported in Brazilian regions with outbreaks of orally transmitted Chagas disease (Pernambuco and Rio Grande Do Norte). Trop Med Infect Dis. 2023;8:196.
- Zamora D, Klotz SA, Meister EA, et al. Repellency of the components of the essential oil, citronella, to Triatoma rubida, Triatoma protracta, and Triatoma recurva (Hemiptera: Reduviidae: Triatominae). J Med Entomol. 2015;52:719-721.
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Cutaneous Reactions to Triatomine (Kissing Bug) Bites and the Risk for Chagas Disease
Practice Points
- Triatomine bugs, commonly known as kissing bugs, are widespread, especially in warmer climates, and their geographic range is expanding.
- The Romaña sign, characterized by unilateral swelling of the eyelid, is common in triatomine bites.
- Triatomine bugs are the primary vector for transmission of the parasite Trypanosoma cruzi, the causative agent of Chagas disease.
- In recent years, T cruzi has been detected in triatomine reduviids in suburban areas of the southwestern United States.
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.
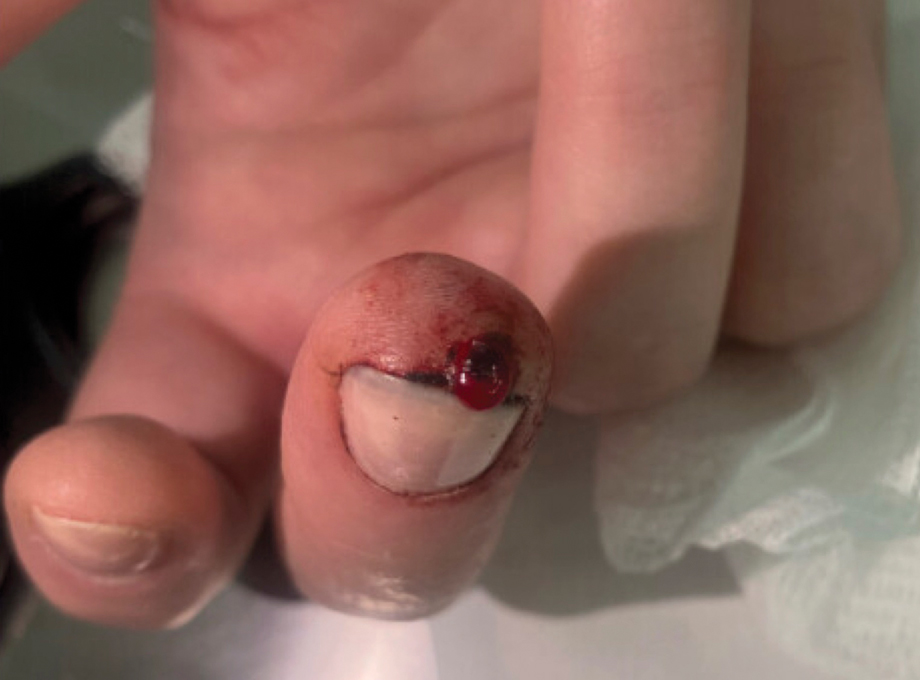
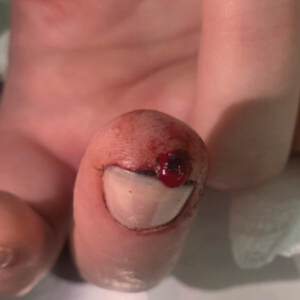
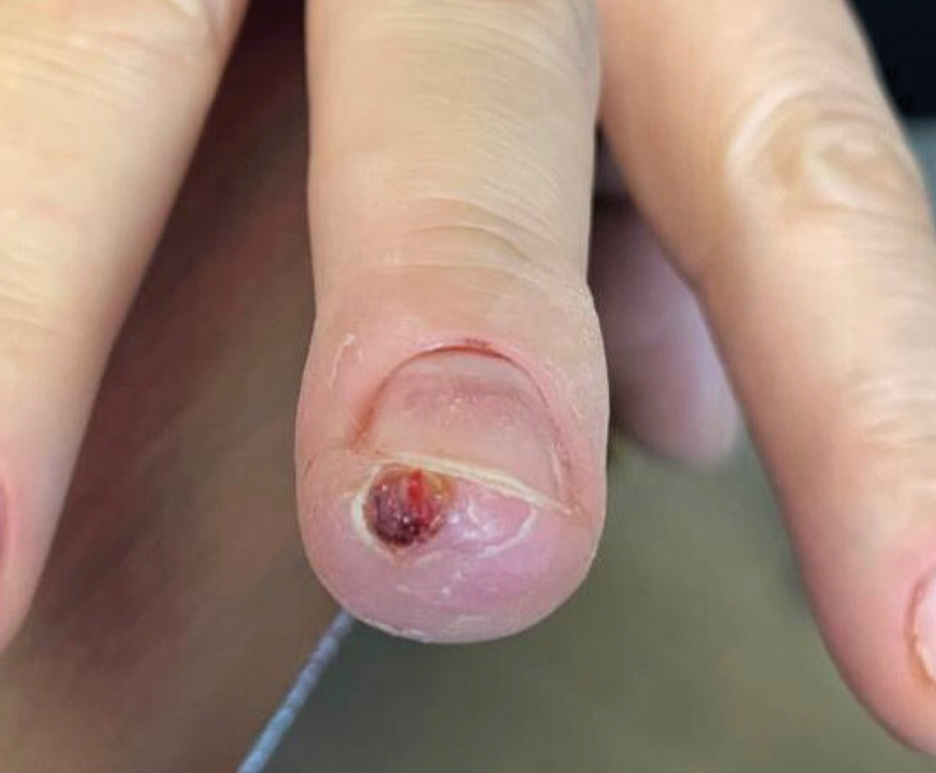
The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.
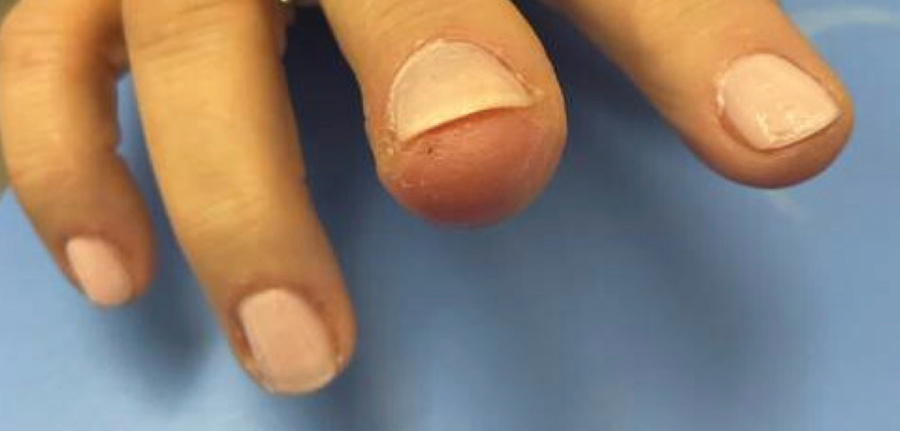
The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.
Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
Practice Gap
Pyogenic granulomas (PGs) are benign endothelial tumors of the skin or mucosae that frequently become ulcerated and may cause patients substantial discomfort or distress due to rapid enlargement and bleeding.1 These lesions often manifest as solitary red papules or polyps following localized trauma or irritation. They can grow up to 1 cm over a few weeks to several months. Pyogenic granulomas can develop at any age, but they most commonly are seen in children and young adults, with a slight male predominance.1,2 The differential diagnosis for PG includes amelanotic melanoma, bacillary angiomatosis, Kaposi sarcoma, glomus tumor, infantile hemangioma, and irritated melanocytic nevus.1 Histologically, PGs are well-circumscribed exophytic or pedunculated proliferations of small capillaries that often are arranged in a lobular pattern. Early lesions show packed endothelial cells, while advanced lesions display more ectatic vessels, erosion, and crusting.3 The term pyogenic granuloma is a misnomer, as these lesions display neither an infectious etiology nor granulomatous tissue on dermatopathologic examination.4 A more accurate clinical description for this lesion is a lobular capillary hemangioma.
Numerous surgical and laser techniques have been used to treat PGs, with varying degrees of success. Treatment often consists of either shave excision followed by electrosurgery at the base or full excision with suturing under local anesthesia for patients who can tolerate anesthetic injections.1 Pulsed dye laser has been proven to be a safe alternative treatment option, particularly in children who otherwise would not tolerate surgical procedures.5 Topical beta-blockers, silver nitrate cauterization, sclerotherapy, and liquid nitrogen all have been used as alternative treatment methods.1
Pyogenic granulomas often recur after first-line treatments, and patients may hesitate to try more invasive techniques when the first choice has failed. Children may not be amenable to any of these curative techniques, as they may not tolerate the pain associated with lidocaine injection and/or have a fear of needles or surgical intervention; even adults may be reluctant to have a procedure they perceive as painful. We present a less invasive technique for treatment of recurrent PGs using table salt and cryotherapy.
The Technique
A 51-year-old woman with no notable medical history presented to the emergency department for evaluation of a black dot on the pulp of the right third fingertip of 1 week’s duration. The patient reported rapid progression to an ulcerated red nodule with associated bleeding for the past 3 days (Figure 1). Direct pressure temporarily alleviated the bleeding, but it started again upon cessation of pressure. She denied any preceding trauma to the area or any associated systemic symptoms such as fever, chills, nausea, or vomiting.

The inpatient dermatology team recommended that the patient be discharged following silver nitrate cautery, with a referral sent to outpatient dermatology; however, the patient returned to the dermatology clinic 4 days later, at which time physical examination revealed a well-circumscribed, 5-mm, bright-red, erosive papule with overlying hemorrhagic crust that was not actively bleeding, as well as fissuring of the surrounding skin. The entire lesion was removed using tangential excision followed by electrodesiccation at the base. Pathology revealed small capillaries arranged in a lobular pattern, confirming the diagnosis of PG. At a 2-week follow-up visit, the patient noted that the lesion had recurred within 24 hours after the procedure and was larger (Figure 2). At this visit, management was switched to a single treatment of cryotherapy (3 cycles for 5 seconds per cycle), and the table salt method was recommended based on a literature review for alternative nonpainful approaches for PG.6-11 We used this technique in our patient as an adjuvant to cryotherapy with the goal of reducing the need for additional painful procedures, but table salt also can be used as a standalone treatment without prior cryotherapy.

The patient was instructed to apply table salt to the lesion once daily for 2 weeks by pressing the lesion into a small amount of salt placed on a clean plate and then applying an occlusive dressing such as surgical or paper tape. She also was advised to apply petroleum jelly around the periphery of the lesion prior to salt application to protect the unaffected skin from irritation. Complete resolution of the lesion was seen when the patient followed up 2 weeks later (Figure 3). At the most recent follow-up 2 months after treatment, no further recurrence of the PG was reported.

Practice Implication
Pyogenic granulomas can be distressing for both patients and providers because they are cosmetically bothersome and prone to spontaneous bleeding. Various medical and surgical options exist to treat PGs, but there is no clear consensus on a superior modality. A 2019 study by Daruwalla and Dhurat6 highlighted a less invasive treatment option for PGs using table salt applied once daily for 2 weeks under an occlusive dressing with good outcomes and without involving other treatments such as cryotherapy. Several other case reports have endorsed this approach, adding anecdotal evidence for its utility.7-11 Topical sodium chloride may treat PGs primarily through osmotic desiccation, drawing water out of the lesion and leading to endothelial cell shrinkage and collapse of its capillary network. This hyperosmolar environment also may induce microvascular thrombosis and ischemia, promoting lesion necrosis. Additionally, repeated application creates a dry, mildly irritative surface that may suppress angiogenesis and encourage regression of the vascular proliferation.
Consider topical application of table salt for treatment of PGs in certain subsets of patients; for example, patients who are not amenable to surgery or are too young for advanced surgical techniques may be good candidates for this method, as it does not require anesthetic injections and generally is pain free. Patients with resistant or recurrent PGs that did not respond to first-line treatments such as cryotherapy, tangential excision, or electrodesiccation may be more amenable to a less invasive secondary approach.
Importantly, we used a dual-therapy approach in our patient, initially using a single application of cryotherapy followed by the table salt method once daily for 2 weeks. This imposes limitations on the generalizability of table salt as a standalone approach for treating PGs. In this case, we did not have prior practical experience using table salt for this condition and only had small reports to justify its use. As a result, we attempted a more traditional treatment initially (cryotherapy) to avoid potential delays in resolution. The clinicians recommended table salt as an adjuvant prior to seeing the cryotherapy results because this treatment was benign and offered potential additive results, and therefore waiting was not necessary. However, various other cases have reported similar success using table salt as monotherapy.6-9,11 Patients should be advised of potential mild adverse events, such as irritation to the surrounding skin. Higher-level evidence studies are required to further vet the utility of the table salt method for treatment of PGs.
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
- Bolognia JL, Schaffer JV, Cerroni L. Vascular neoplasms and neoplastic‑like proliferations. In: Dermatology. Elsevier; 2018.
- Harris MN, Desai R, Chuang TY, et al. Lobular capillary hemangiomas: an epidemiologic report, with emphasis on cutaneous lesions. J Am Acad Dermatol. 2000;42:1012-1016.
- Ferringer TK, DiCaudo DJ, Elston D, et al. Dermatopathology. W.B. Saunders; 2008.
- Gomes SR, Shakir QJ, Thaker PV, et al. Pyogenic granuloma of the gingiva: a misnomer? - a case report and review of literature. J Indian Soc Periodontol. 2013;17:514-519. doi:10.4103/0972-124X.118327
- Sud AR, Tan ST. Pyogenic granuloma-treatment by shave-excision and/or pulsed-dye laser. J Plast Reconstr Aesthet Surg. 2010;63:1364-1368. doi:10.1016/j.bjps.2009.06.031
- Daruwalla SB, Dhurat RS. A pinch of salt is all it takes! the novel use of table salt for the effective treatment of pyogenic granuloma. J Am Acad Dermatol. 2020;83:E107-E108. doi:10.1016/j.jaad.2019.12.013
- Alhammad G, Albaraka M, Alotaibi H, et al. The use of common salt for the treatment of pyogenic granuloma. JAAD Case Rep. 2024;53:40-42. doi:10.1016/j.jdcr.2024.08.016
- Weiss ES, Wood D. Simple, safe, and effective treatment for pyogenic granuloma. Can Fam Physician. 2023;69:479-480. doi:10.46747/cfp.6907479
- Bernales Salinas A, Toro Sepúlveda A, Meier Pincheira H, et al. Case report: pyogenic granuloma-just salt, a simple and pain-free treatment. Dermatol Ther. 2022;35:E15194. doi:10.1111/dth.15194
- Martín-Nieto González J, Rodríguez-Sánchez B, Berna-Rico E, et al. Pyogenic granuloma resolved with timolol and table salt. An Pediatr (Engl Ed). 2025;102:503706. doi:10.1016/j.anpede.2025.503706
- Bin Rubaian NF. Complete resolution of a refractory pyogenic granuloma following topical salt treatment. Open Access Emerg Med. 2021;13:445-448. doi:10.2147/OAEM.S323793
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
Table Salt Method Following Cryotherapy for Recurrent Pyogenic Granuloma on the Fingertip
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
The American Academy of Dermatology’s 2026 Annual Meeting in Denver, Colorado, showcased advances in clinical practice and dermatology research. Selected key updates are summarized here for concise review of emerging dermatology data relevant to clinical practice.
AI Holds Promise in Dermatology, Issues Remain to be Addressed
Artificial intelligence (AI) is rapidly advancing in dermatology, improving image analysis, clinical decision support, and workflow efficiency; however, concerns remain about ethical use, training gaps, and potential skill loss among clinicians. While AI may enhance productivity and care, experts emphasize the need for cautious implementation, education, and ongoing evaluation of real-world performance.
Phase 2b Findings Support Novel Agent to Treat Alopecia Areata
A phase 2b trial of rezpegaldesleukin for severe alopecia areata showed considerably greater reductions in SALT scores vs placebo over 36 weeks, with higher response rates and no treatment plateau. The biologic, which enhances regulatory T-cell activity, demonstrated a favorable safety profile, with mainly mild injection-site reactions and no new safety signals.
JAK Inhibitors: Identifying Ideal Candidates and Putting Real-World Risks in Context
Emerging evidence suggests Janus kinase (JAK) inhibitors are safer in dermatology than early rheumatoid arthritis data indicated. Risks for cardiovascular events, thrombosis, and malignancy appear low and largely driven by baseline patient factors. With appropriate screening and monitoring, these agents can be used safely in most patients with inflammatory skin diseases.
Nemolizumab Phase 2 Findings Positive for Children 2-11 Years Old With Atopic Dermatitis
A phase 2 open-label study of nemolizumab in children aged 2 to 11 years with moderate to severe atopic dermatitis showed notable improvements in skin clearance, disease severity, and itch with weight-based dosing. Responses were rapid, durable through 52 weeks, and consistent with prior data, with no new safety signals identified in this population.
Melasma: A New Era of Topical Treatment Options Galore
Melasma treatment is rapidly expanding beyond traditional agents such as hydroquinone and triple combination therapy, with newer topicals including tranexamic acid, cysteamine, azelaic acid, thiamidol, and emerging compounds showing variable efficacy. While promising, evidence is still evolving, and combination regimens plus strict photoprotection remain the cornerstone of management.
Weight-Loss Drug–Biologic Combination Boosts Relief in Psoriatic Arthritis
In a phase 3b trial, combining tirzepatide with ixekizumab significantly improved joint and skin outcomes in patients with psoriatic arthritis and overweight/obesity (P<.05) compared with ixekizumab alone (P<.001). The combination yielded higher American College of Rheumatology and Psoriasis Area and Severity Index response rates, early symptom improvement, and meaningful weight loss, with safety profiles consistent with known effects.
Tips on Using Biologics for Psoriasis in Context of HIV
Evidence for biologic use in HIV-positive patients with moderate to severe psoriasis is limited, but available case reports suggest tumor necrosis factor inhibitors and newer IL-targeted biologics are generally effective without major impacts on viral load or CD4 counts. Experts recommend prioritizing nonimmunosuppressive options and coordinating care with HIV specialists due to potential infection risks.
Upadacitinib Results in Significant Improvements in Nonsegmental Vitiligo in Phase 3 Studies
Two phase 3 trials showed that the Janus kinase 1 inhibitor upadacitinib significantly improved repigmentation outcomes in adolescents and adults with nonsegmental vitiligo vs placebo over 48 weeks (P<.0001 for both), with a higher proportion achieving clinically meaningful reductions in Vitiligo Area and Severity Index scores. Benefits increased over time without plateau, and no new safety signals were identified.
AAD 2026 Annual Meeting Highlights
AAD 2026 Annual Meeting Highlights
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
How might AI enhance the detection of key histologic features in drug eruptions compared to traditional microscopy?
DR. BRIDGES: AI offers the potential to enhance detection of histologic features in drug eruptions by systematically analyzing entire whole-slide images. Convolutional neural networks and attention-based models can identify subtle or focal findings such as scattered dyskeratotic keratinocytes, focal spongiosis, early interface change, rare eosinophils, or microvascular injury, which may be overlooked during routine microscopy due to sampling limitations. This capability is particularly relevant in drug eruptions, where histologic changes often are heterogeneous and patchy.
AI-generated attention heatmaps can highlight diagnostically relevant regions across the slide, improving consistency and completeness of slide reviews. While AI has demonstrated high sensitivity and specificity in broader dermatopathology tasks, particularly neoplastic conditions, drug eruption–specific validation data are currently lacking. As such, the most realistic application at present is AI functioning as a sensitivity-enhancing adjunct or “second reader,” improving consistency and completeness of slide review while preserving expert human interpretation.
Which histologic patterns in drug eruptions are hardest to quantify, and how could AI help standardize their assessment?
DR. BRIDGES: AI-based image analysis can standardize the assessment of histologic patterns through objective reproducible quantification. Deep learning algorithms can segment epidermal and dermal compartments, identify inflammatory cell types, and calculate metrics such as eosinophil density per unit area, percentage of epidermis with vacuolar alteration, or number of affected vessels. Studies in quantitative immunohistochemistry demonstrate high accuracy for tissue segmentation and cell counting, suggesting feasibility for similar applications in inflammatory dermatopathology. While these tools would not replace diagnostic interpretation, they could provide standardized measurements that enhance reproducibility and improve clinicopathologic correlation.
What training challenges must be addressed in AI and drug eruption histology?
DR. BRIDGES: Training AI models for drug eruption histopathology faces several challenges, including the limited availability of high-quality, well-annotated datasets, as most existing AI dermatopathology research focuses on neoplastic conditions. Drug eruptions also exhibit marked histologic heterogeneity, ranging from spongiotic and lichenoid to vasculitic and cytotoxic patterns, often with significant overlap. Accurate labeling, therefore, requires robust clinicopathologic correlation, including medication history, timing, laboratory data, and clinical outcomes—information that is often incomplete or retrospective.
Inaccurate or inconsistent annotations can significantly degrade model performance, and expert disagreement in borderline cases further complicates the creation of reliable ground truth. Additionally, training data may reflect institutional or demographic biases, risking unequal performance across patient populations. Addressing these challenges will require multicenter collaboration, standardized annotation protocols, inclusion of diverse patient cohorts, and careful attention to bias mitigation. At present, these barriers place drug eruption AI firmly in the investigational rather than clinical domain.
How important is AI explainability in the interpretation of diagnostic suggestions?
DR. BRIDGES: Explainability is essential for trust, particularly in the evaluation of drug eruptions, where diagnostic decisions can have serious clinical consequences. Dermatopathologists must understand which histologic features are driving an AI model’s assessment to ensure that conclusions align with morphologic reality and clinicopathologic reasoning. Explainable AI tools (such as attention heatmaps, feature importance rankings, and methods like Shapley Additive Explanations or Local Interpretable Model-Agnostic Explanations) can help clarify which histologic features are driving the AI model’s assessment.
Without transparency, AI systems function as “black boxes,” limiting their utility in high-stakes settings where diagnostic accountability and clinical communication are paramount. Explainability also supports appropriate skepticism, allowing pathologists to recognize when model outputs may be unreliable due to artifacts, atypical patterns, or out-of-distribution cases. In cases of drug eruptions—where diagnosis relies on combining histology, clinical timing, and medication history—explainability is essential for proper use.
How could AI pattern recognition be integrated into your workflow to enhance diagnostic efficiency and accuracy? What safeguards would be required?
DR. BRIDGES: In the near term, AI pattern recognition can be useful as an assistive tool rather than a diagnostic authority. One potential application is pre-screening whole-slide images to flag cases with features such as prominent interface change, increased keratinocyte necrosis, eosinophil-rich infiltrates, or vascular injury, prompting expedited review in clinically concerning scenarios. During sign-out, AI overlays could aid efficiency by highlighting rare but relevant features and providing quantitative summaries that support standardized reporting.
Safeguards are essential. AI systems must be validated across diverse practice settings, staining protocols, and scanning platforms. Human oversight is mandatory, with the dermatopathologist retaining full diagnostic responsibility. AI involvement should be clearly documented for medicolegal transparency, and performance should be continuously monitored to detect algorithmic drift as new drug eruption patterns emerge. Given current limitations, AI is best viewed as a tool to refine and support expert judgment, not replace it.
What data-sharing or privacy challenges must be addressed to develop robust AI models for diverse drug-eruption histopathology?
DR. BRIDGES: Developing robust AI models for drug eruptions requires large diverse datasets, raising significant privacy and governance challenges. Rigorous de-identification protocols, clear informed consent frameworks, and strong institutional oversight are therefore essential. Multicenter collaborations must employ secure data-use agreements and governance structures that clearly define access, ownership, and downstream use of data.
Ensuring equitable representation is equally critical, as underrepresentation of certain populations may lead to biased performance and disparities in care. Standardized data formats and interoperable systems are needed to facilitate collaboration while preserving security. Transparent governance structures, clear rules regarding data use, and trust-building with patients and institutions will ultimately determine willingness to participate. Addressing these challenges is foundational to advancing AI research in drug eruptions responsibly and ethically.
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Evaluating Drug Eruptions Using AI: Tips From Alina G. Bridges, DO
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.
Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.
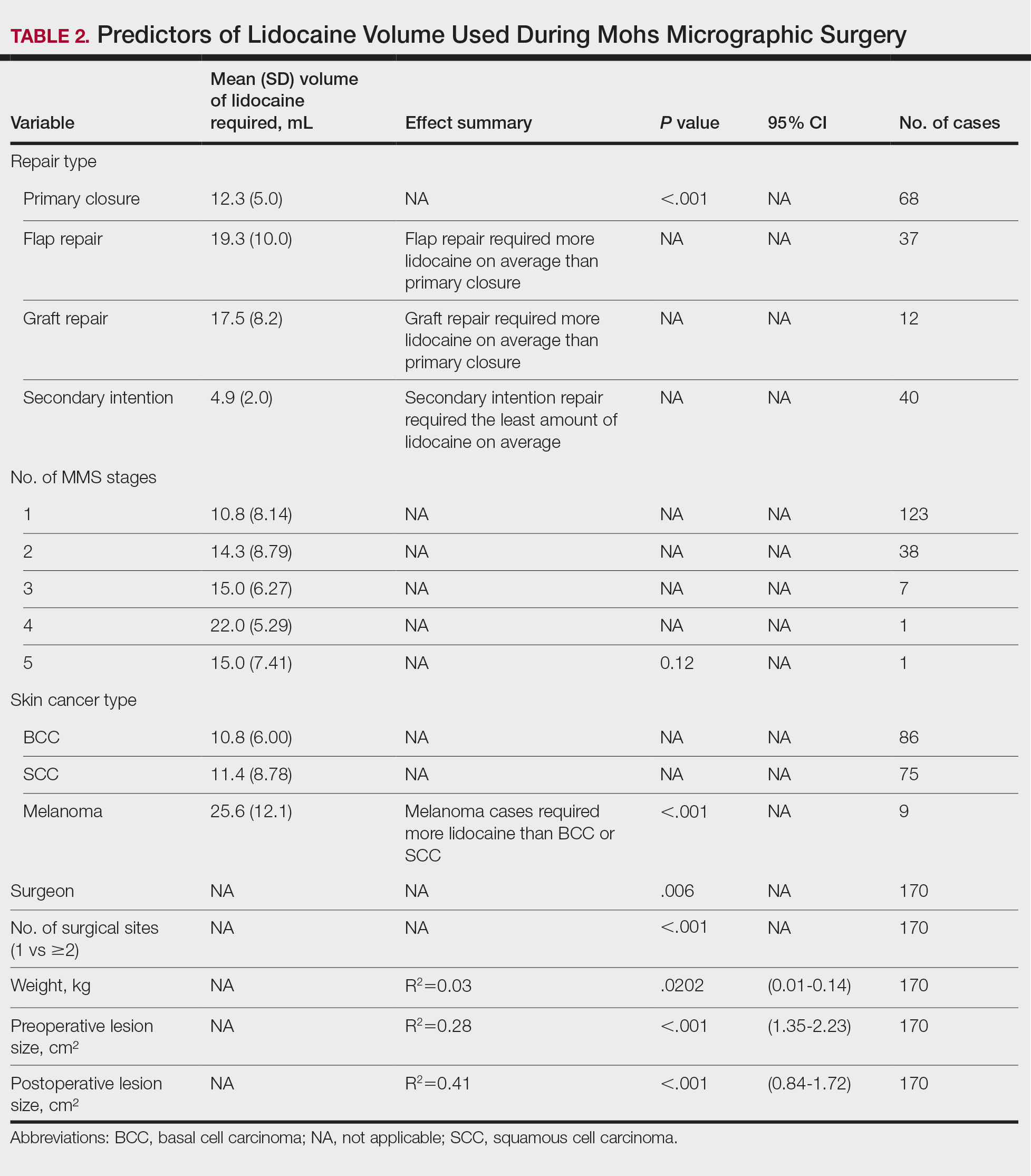
Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
To the Editor:
Mohs micrographic surgery (MMS) is performed in stages and often requires repeated administration of a local anesthetic, most commonly lidocaine. While generally safe, lidocaine administration carries the potential for cumulative toxicity, particularly in patients who have large or multiple lesions or medical comorbidities or who require extensive repair. Current safety guidelines suggest upper limits of 7 mg/kg (or 500 mg) of lidocaine with epinephrine and 4.5 mg/kg (or 300 mg) without epinephrine for adults.1 However, concerns have been raised about the relevance of these thresholds to MMS, in which anesthetic administration may be prolonged, cumulative, and influenced by surgical complexity.2-5 While clinical experience often guides anesthetic planning, limited data exist identifying predictors of lidocaine use during MMS.
We performed an institutional review board–approved retrospective chart review of 149 patients who underwent 170 MMS procedures at a single academic dermatologic surgery center between July 2022 and June 2023. The aim of our study was to identify clinical and surgical predictors of lidocaine volume used during MMS. All procedures were performed by board-certified dermatologic surgeons (including A.J.). All patients received 1% lidocaine with epinephrine as the primary anesthetic agent. We collected patient demographic variables (age, sex, race, weight), procedural characteristics (anatomic site, number of Mohs stages, skin cancer type, number of surgical sites treated in one day, preoperative and postoperative lesion size, surgeon, repair type), comorbid conditions (hypertension, diabetes), and time from diagnosis to surgery. Data were extracted from the institutional REDCap system. We used t tests and analysis of variance for categorical variables and linear regression for continuous predictors, with statistical significance set at P<.05.
Baseline characteristics of the study patients are outlined in Table 1. The mean (SD) age was 74.2 (9.4) years, and most patients (98.7% [147/149]) were White. The mean (SD) weight was 83.1 (19.1) kg. Most lesions were either basal cell carcinoma (BCC)(50.6%) or squamous cell carcinoma (SCC)(44.1%), with 5.3% of lesions representing melanoma. The mean (SD) total lidocaine volume administered was 11.8 (8.3) mL. The majority (123/149 [72.4%]) of cases required one Mohs stage, but a subset required multiple stages, with a maximum of 5.

Several procedural and patient factors were significantly associated with the volume of lidocaine used. As expected, lesion size strongly influenced lidocaine volume. Both preoperative and postoperative lesion sizes were highly significant linear predictors (R2=0.28 and 0.41, respectively; P<.001), and postoperative lesion size demonstrated the strongest correlation of all tested variables. Patient weight was also significantly associated with lidocaine use (R2=.03, P=.0202), though the proportion of explained variance was modest. The operating surgeon also was significantly associated with lidocaine use (P=.006), suggesting potential variation in anesthetic technique or threshold for reinfiltration. The number of surgical sites treated in a single session also was significantly associated with greater lidocaine volume (P<.001).
Skin cancer type was a notable categorical predictor. Melanomas required substantially more lidocaine than BCCs or SCCs, with a mean (SD) volume of 25.6 (12.1) mL compared with 10.8 (6.0) mL for BCC and 11.4 (8.8) mL for SCC (P<.001). This difference may reflect disparities in surgical margin requirements, tumor depth, or intraoperative technique. While lesion location and number of stages were not statistically significant overall, mean lidocaine volumes trended higher in lesions on the trunk (18.2 mL) and in procedures requiring 3 or more stages (up to 22.0 mL for a single 4-stage case), though small sample sizes limited the ability to detect statistically significant differences in these subgroups. Detailed comparisons are presented in Table 2.

Wound repair type also was significantly associated with lidocaine volume requirements. Primary closures required a mean (SD) volume of 12.3 (5.0) mL, whereas flap repairs required 19.3 (10.0) mL and graft repairs required 17.5 (8.2) mL. Secondary-intention healing used the lowest lidocaine volumes (mean [SD], 4.9 [2.0] mL). Differences across repair types were statistically significant (analysis of variance, P<.001). These findings indicate that more complex reconstructions, such as flaps and grafts, are associated with higher anesthetic needs when compared with primary closures or secondary-intention healing.
Several other predictors, including age, time from diagnosis to surgery, and comorbid conditions such as hypertension or diabetes, were not significantly associated with anesthetic volume in our cohort. Time from diagnosis to surgery ranged widely but did not correlate with lesion size or lidocaine use, possibly due to scheduling variability or biopsy technique.
These findings offer practical implications for clinical planning. While most MMS cases fall well within safe limits for lidocaine administration, some patients—particularly those with melanoma, large lesions, or multiple surgical sites—may approach thresholds at which further monitoring or dose tracking becomes relevant. Anticipating higher anesthetic requirements may help surgical teams plan procedure length, anesthesia restocking, or sequencing of multisite cases. Our analysis also showed that the type of wound repair meaningfully influences anesthetic use, with flap and graft repairs requiring substantially higher lidocaine volumes than primary closures and secondary-intention healing. Considering both tumor characteristics and the planned reconstruction may therefore improve the accuracy of anesthetic forecasting during preoperative planning.
We also observed surgeon-level variation in lidocaine volume despite standardized tumor types and case complexity. This suggests a role for individual technique (eg, depth of field block, number of reinfiltrations) and highlights the need for ongoing education around anesthetic optimization.
Our study was limited by its retrospective design, single-institution setting, and demographically homogeneous population. With 98.8% of patients identifying as White, generalizability to skin of color populations may be limited. In addition, lidocaine metabolism may vary across patient factors not captured here (eg, hepatic or renal function). Finally, although lidocaine volume was the outcome of interest, we did not measure patient-reported pain control, which may further clarify anesthetic adequacy. Nonetheless, our analysis demonstrated that routinely available clinical and procedural data can predict lidocaine volume requirements with reasonable reliability. Although no patient in our cohort approached the maximum recommended lidocaine dose, understanding these predictors may help anticipate scenarios nearing maximum dosing thresholds. In future studies, integrating weight-based thresholds (eg, mL/kg received) or serum lidocaine levels may improve safety monitoring and validate toxicity thresholds in complex cases.
In conclusion, we identified several key factors that predict lidocaine volume during MMS, including lesion size, melanoma diagnosis, number of surgical sites, patient weight, planned reconstruction type, and the operating surgeon. Among these factors, melanoma cases required more than twice the volume of lidocaine compared to BCC and SCC cases, and flap and graft repairs demonstrated the highest anesthetic requirements among closure types. Taken together, these findings reinforce the need for advanced anesthetic planning in aggressive, anatomically complex, or reconstruction-intensive cases and may support more informed intraoperative decision-making.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
- Kouba DJ, LoPiccolo MC, Alam M, et al. Guidelines for the use of local anesthesia in office-based dermatologic surgery. J Am Acad Dermatol. 2016;74:1201-1219. doi:10.1016/j.jaad.2016.01.022
- Wang A, Grushchak S, Kaul S, et al. Toxicity of infiltrative lidocaine in dermatologic surgery: are current limits valid? Dermatol Pract Concept. 2021;11:e2021120. doi:10.5826/dpc.1104a120
- Patrinely JR Jr, Darragh C, Frank N, et al. Risk of adverse events due to high volumes of local anesthesia during Mohs micrographic surgery. Arch Dermatol Res. 2021;313:679-684. doi:10.1007/s00403-020-02155-1
- Butterwick KJ, Goldman MP, Sriprachya-Anunt S. Lidocaine levels during the first two hours of infiltration of dilute anesthetic solution for tumescent liposuction: rapid versus slow delivery. Dermatol Surg. 1999;25:681-685. doi:10.1046/j.1524-4725.1999.98275.x
- Flanagan K, McLean R, Goldberg D. Is it time to redefine lidocaine administration guidelines in Mohs surgery? J Drugs Dermatol. 2020;19:433.
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Predictors of Lidocaine Volume Used During Mohs Micrographic Surgery
Practice Points
- Larger lesion size, melanoma diagnosis, and multiple surgical sites are associated with higher lidocaine volume requirements during Mohs micrographic surgery.
- Melanomas required more than twice the average lidocaine volume compared with basal cell carcinomas and squamous cell carcinomas.
- Flap and graft repairs require substantially more lidocaine than primary closures, while secondary-intention healing uses the least, making reconstruction type an important predictor of total anesthetic needs.
VA Invests in Transportation Aid for Rural Veterans
The US Department of Veterans Affairs (VA) recently announced plans to offer $7 million in new transportation services grants that could benefit 4.7 million veterans who live in rural areas. The grants would expand free transportation to medical appointments, something VA Secretary Doug Collins said is designed to “help break down the geographic barriers to health care some rural veterans face.”
Funding could be distributed later in 2026 to veteran service organizations, state agencies, and groups that transport veterans for health care. Eligible veterans would not need to do anything—the transportation is free for those living in qualifying areas.
Travel time and distance from health care facilities are significant barriers to receiving appropriate and timely care. The 2014 Veterans Access, Choice and Accountability Act (Choice) was intended to improve timely access to outpatient health care for veterans by allowing them to receive care from community facilities paid for by the VA. Under Choice, eligible veterans become eligible to receive community care if they have to drive > 40 miles to the nearest VA facility or wait > 30 days for care.
Even with this provision, many of the 2.7 million rural veterans enrolled in Veterans Health Administration (VHA) remained far from care. For instance, the VA Office of Rural Health says the closest facility for veterans in Hollis, Alaska, is > 1000 miles away.
Moreover, 56% of rural veterans enrolled in VHA care are aged > 65 years, and more likely to be diagnosed with diabetes, high blood pressure, and heart conditions than veterans living in more urban areas. Although studies comparing health outcomes between rural and urban veterans are sparse, research has long shown that lacking access to routine health care may worsen long-term outcomes.
The VA has also announced other initiatives aimed at improving health care for veterans, among them the opening of 34 new facilities. Other projects:
The Electronic Health Record (EHR) modernization project resumed April 11 with new deployments in Michigan. The VA says the new EHR system will result in more consistent medical records, fewer repeated tests, and better coordination between VA facilities and military health services.
In March, the VA announced a $112 million grant opportunity to strengthen community‑based suicide prevention programs, focusing on outreach outside traditional VA settings.
In February, the VA said it raised its spending cap for in‑home and community‑based services for veterans with complex medical needs, adding coverage for veterans with spinal cord injuries, Amyotrophic Lateral Sclerosis, and others.
In January, the VA announced plans to invest $4.8 billion in fiscal year 2026 to modernize, repair, and improve health care facilities nationwide via infrastructure upgrades, major building repairs, and improvements to EHR systems.
The US Department of Veterans Affairs (VA) recently announced plans to offer $7 million in new transportation services grants that could benefit 4.7 million veterans who live in rural areas. The grants would expand free transportation to medical appointments, something VA Secretary Doug Collins said is designed to “help break down the geographic barriers to health care some rural veterans face.”
Funding could be distributed later in 2026 to veteran service organizations, state agencies, and groups that transport veterans for health care. Eligible veterans would not need to do anything—the transportation is free for those living in qualifying areas.
Travel time and distance from health care facilities are significant barriers to receiving appropriate and timely care. The 2014 Veterans Access, Choice and Accountability Act (Choice) was intended to improve timely access to outpatient health care for veterans by allowing them to receive care from community facilities paid for by the VA. Under Choice, eligible veterans become eligible to receive community care if they have to drive > 40 miles to the nearest VA facility or wait > 30 days for care.
Even with this provision, many of the 2.7 million rural veterans enrolled in Veterans Health Administration (VHA) remained far from care. For instance, the VA Office of Rural Health says the closest facility for veterans in Hollis, Alaska, is > 1000 miles away.
Moreover, 56% of rural veterans enrolled in VHA care are aged > 65 years, and more likely to be diagnosed with diabetes, high blood pressure, and heart conditions than veterans living in more urban areas. Although studies comparing health outcomes between rural and urban veterans are sparse, research has long shown that lacking access to routine health care may worsen long-term outcomes.
The VA has also announced other initiatives aimed at improving health care for veterans, among them the opening of 34 new facilities. Other projects:
The Electronic Health Record (EHR) modernization project resumed April 11 with new deployments in Michigan. The VA says the new EHR system will result in more consistent medical records, fewer repeated tests, and better coordination between VA facilities and military health services.
In March, the VA announced a $112 million grant opportunity to strengthen community‑based suicide prevention programs, focusing on outreach outside traditional VA settings.
In February, the VA said it raised its spending cap for in‑home and community‑based services for veterans with complex medical needs, adding coverage for veterans with spinal cord injuries, Amyotrophic Lateral Sclerosis, and others.
In January, the VA announced plans to invest $4.8 billion in fiscal year 2026 to modernize, repair, and improve health care facilities nationwide via infrastructure upgrades, major building repairs, and improvements to EHR systems.
The US Department of Veterans Affairs (VA) recently announced plans to offer $7 million in new transportation services grants that could benefit 4.7 million veterans who live in rural areas. The grants would expand free transportation to medical appointments, something VA Secretary Doug Collins said is designed to “help break down the geographic barriers to health care some rural veterans face.”
Funding could be distributed later in 2026 to veteran service organizations, state agencies, and groups that transport veterans for health care. Eligible veterans would not need to do anything—the transportation is free for those living in qualifying areas.
Travel time and distance from health care facilities are significant barriers to receiving appropriate and timely care. The 2014 Veterans Access, Choice and Accountability Act (Choice) was intended to improve timely access to outpatient health care for veterans by allowing them to receive care from community facilities paid for by the VA. Under Choice, eligible veterans become eligible to receive community care if they have to drive > 40 miles to the nearest VA facility or wait > 30 days for care.
Even with this provision, many of the 2.7 million rural veterans enrolled in Veterans Health Administration (VHA) remained far from care. For instance, the VA Office of Rural Health says the closest facility for veterans in Hollis, Alaska, is > 1000 miles away.
Moreover, 56% of rural veterans enrolled in VHA care are aged > 65 years, and more likely to be diagnosed with diabetes, high blood pressure, and heart conditions than veterans living in more urban areas. Although studies comparing health outcomes between rural and urban veterans are sparse, research has long shown that lacking access to routine health care may worsen long-term outcomes.
The VA has also announced other initiatives aimed at improving health care for veterans, among them the opening of 34 new facilities. Other projects:
The Electronic Health Record (EHR) modernization project resumed April 11 with new deployments in Michigan. The VA says the new EHR system will result in more consistent medical records, fewer repeated tests, and better coordination between VA facilities and military health services.
In March, the VA announced a $112 million grant opportunity to strengthen community‑based suicide prevention programs, focusing on outreach outside traditional VA settings.
In February, the VA said it raised its spending cap for in‑home and community‑based services for veterans with complex medical needs, adding coverage for veterans with spinal cord injuries, Amyotrophic Lateral Sclerosis, and others.
In January, the VA announced plans to invest $4.8 billion in fiscal year 2026 to modernize, repair, and improve health care facilities nationwide via infrastructure upgrades, major building repairs, and improvements to EHR systems.
Male Vets Less Likely to Undergo Intimate Partner Violence Screening
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.
Male veterans are less likely than their female counterparts to be referred for follow-up questions when initial screening suggests they may be at risk of intimate partner violence (IPV), a recent large cross-sectional study finds.
Among 67,379 patients from 131 US Department of Veterans Affairs (VA) medical centers who screened positive for risk of IPV from October 2022 through September 2023, 17.7% failed to receive a mandated secondary screen to determine whether they were in danger of lethal violence, reported Galina A. Portnoy, PhD, of VA Connecticut Healthcare System and Yale School of Medicine, et al in JAMA Network Open. The rate was higher for men with initial positive screens than women (19.3% vs 12.1%, respectively, adjusted odds ratio [AOR], 1.42, P < .001).
Overall, women who underwent secondary screening were more likely to be considered in lethal danger from IPV than men (27.9% vs 13.3%, respectively, AOR 2.29, P < .001).
“While women face higher lethality risk, men’s IPV experiences are often overlooked, underscoring the need for consistent and reliable screening practices to identify all high-risk patients and connect them to life-saving services,” Portnoy told Federal Practitioner.
“IPV is one of the strongest predictors of homicide with risk escalating over time and especially high during periods of separation.”
“IPV among men is often underreported, unrecognized, and inadequately addressed in clinical settings,” Portnoy noted. “Men who experience IPV often face barriers to reporting—stigma, shame, and concerns about not being taken seriously.”
The VA has implemented annual screening of IPV in women of reproductive age using a modified version of the 5-question Hurt, Insult, Threaten, Scream (HITS) tool. HITS asks how often a woman’s partner had screamed, cursed, insulted, or talked down to them; threatened to harm or physically hurt them, or forced or pressured them to “have sexual contact against your will, or when you were unable to say no” in the last year.
If a patient answers yes to any of these questions, clinicians should follow up with a secondary lethality screen with 3 questions:
Has the IPV behavior increased in frequency/severity in the past 6 months?
Has your partner ever choked or strangled you? and
Do you believe your partner may kill you?
The test is considered positive if a patient answers yes to any question.
The study focused on 67,379 patients out of 1,265,115 at the VA who scored positive on HITS (mean age, 52.3 years; 23% women; 62.9% White; 8.2% Hispanic/Latino). More than two-thirds (69.0%) had a service-connected disability rating > 50%.
Portnoy said there are several possible reasons for the gender disparity in misclassification such as time constraints, discomfort, limited resources, and lack of training. Clinician bias can be a factor, too, “with IPV still widely seen as primarily a women’s issue.”
“We don’t know whether IPV screening tools work the same for men as they do for women,” Portnoy added. “The HITS tool was developed and validated using samples of women who experienced IPV, and research is needed to test whether it performs as effectively in men.”
Bethany L. Backes, PhD, associate professor and lead, Violence Against Women Faculty Cluster, University of Central Florida, Orlando, is familiar with the study findings and said in an interview that discomfort among clinicians is a significant factor in preventing follow-up IPV screening.
“When you’re asking about this and someone says ‘yes,’ how do you respond? You just go to the next thing, the next question: ‘How many drinks have you had in the last week?’” Backes told Federal Practitioner. “We’ve talked about creating some scripts for our student health clinicians on campus about how to talk to someone when they disclose, how to then engage or provide resources.”
This is especially important because “it’s hard for people to admit that they’re experiencing this, and then when they do and it’s brushed over, they’re less likely to tell someone again,” Backes added.
C. Nadine Wathen, PhD, a professor who studies IPV at Western University in London, is also familiar with the study findings, but critiqued the HITS, calling it a “terrible name.” The tool, she said, asks about very different behaviors–being screamed or cursed, for example, and forced sexual contact,” she explained to Federal Practitioner.
“If you’re a physician and you’re asking a man, ‘Does she scream or curse at you?’ and he says ‘Yeah, she screams all the time,’ a provider might say, ‘I’m not actually thinking that he’s experiencing intimate partner violence,” Wathen said. “He might be experiencing a bad relationship.’”
That could be true, Wathen said. Couples may scream and throw things at each other, and “you probably could benefit with some couples counseling on how to have a better relationship and manage stress and anger in your relationship. But that is different than ‘intimate partner terrorism,’ where there‘s a pattern of control.”
Wathen prefers a screening tool she helped develop called the Composite Abuse Scale, which she considers more sensitive and specific than HITS. It differentiates the types of abuse that people experience, and “it also recognizes that men in relationships with other men can experience those forms of intimate terrorism, and women can also be the perpetrator of those forms.”
Recognizing that VA clinicians may not have a choice of screening tool, Wathen suggested they follow up the question about screaming and cursing question this query: “Does that make you afraid?”
The study was funded by US Department of Veterans Affairs Quality Enhancement Research Initiative and the Veterans Health Administration’s Care Management and Social Work Service via the Intimate Partner Violence Center for Implementation, Research, and Evaluation.
Portnoy has no disclosures. One author discloses relationships with the National Council on Family Relations and Military Family Research Institute. Backes and Wathen have no disclosures.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)
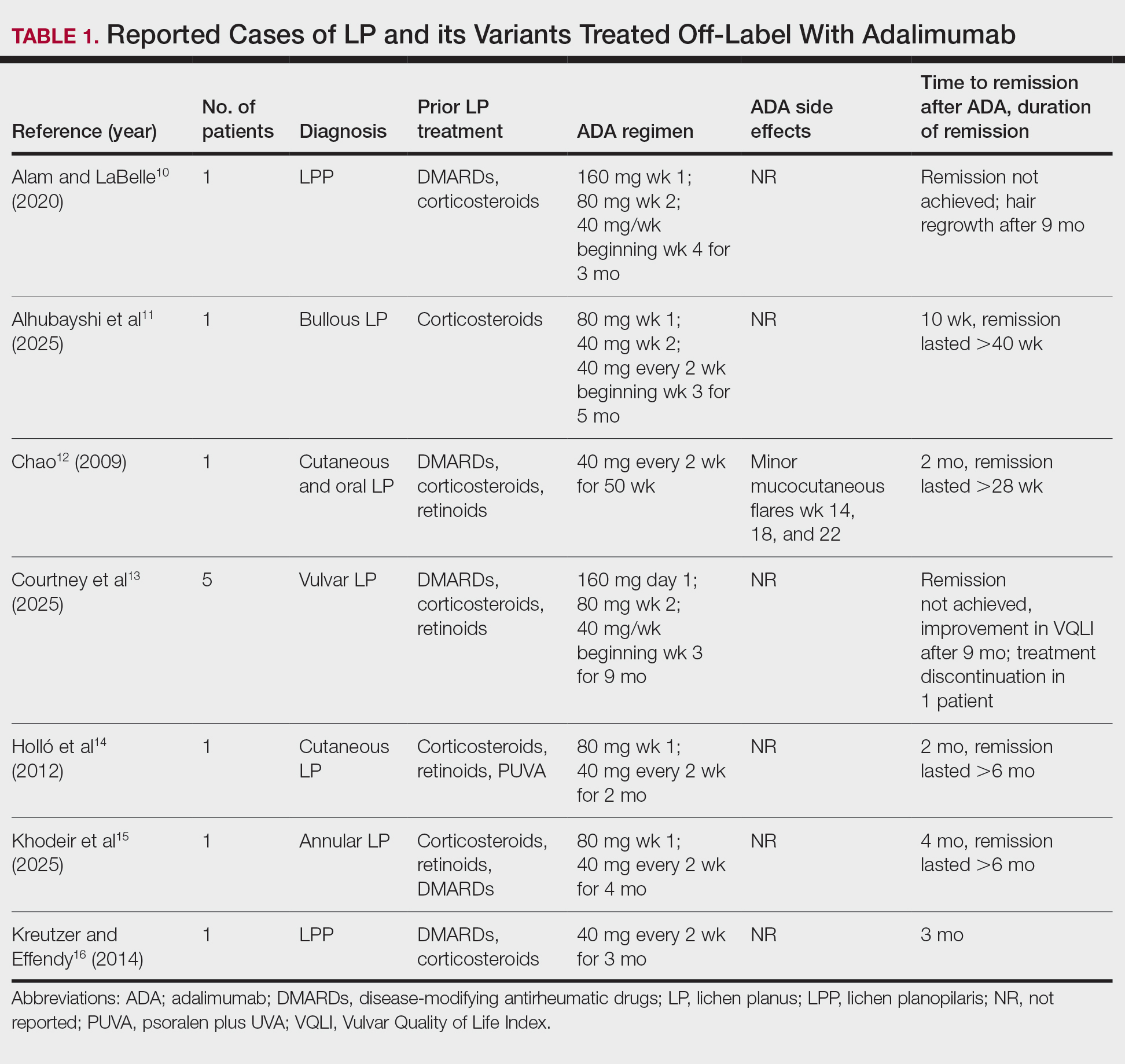
Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22
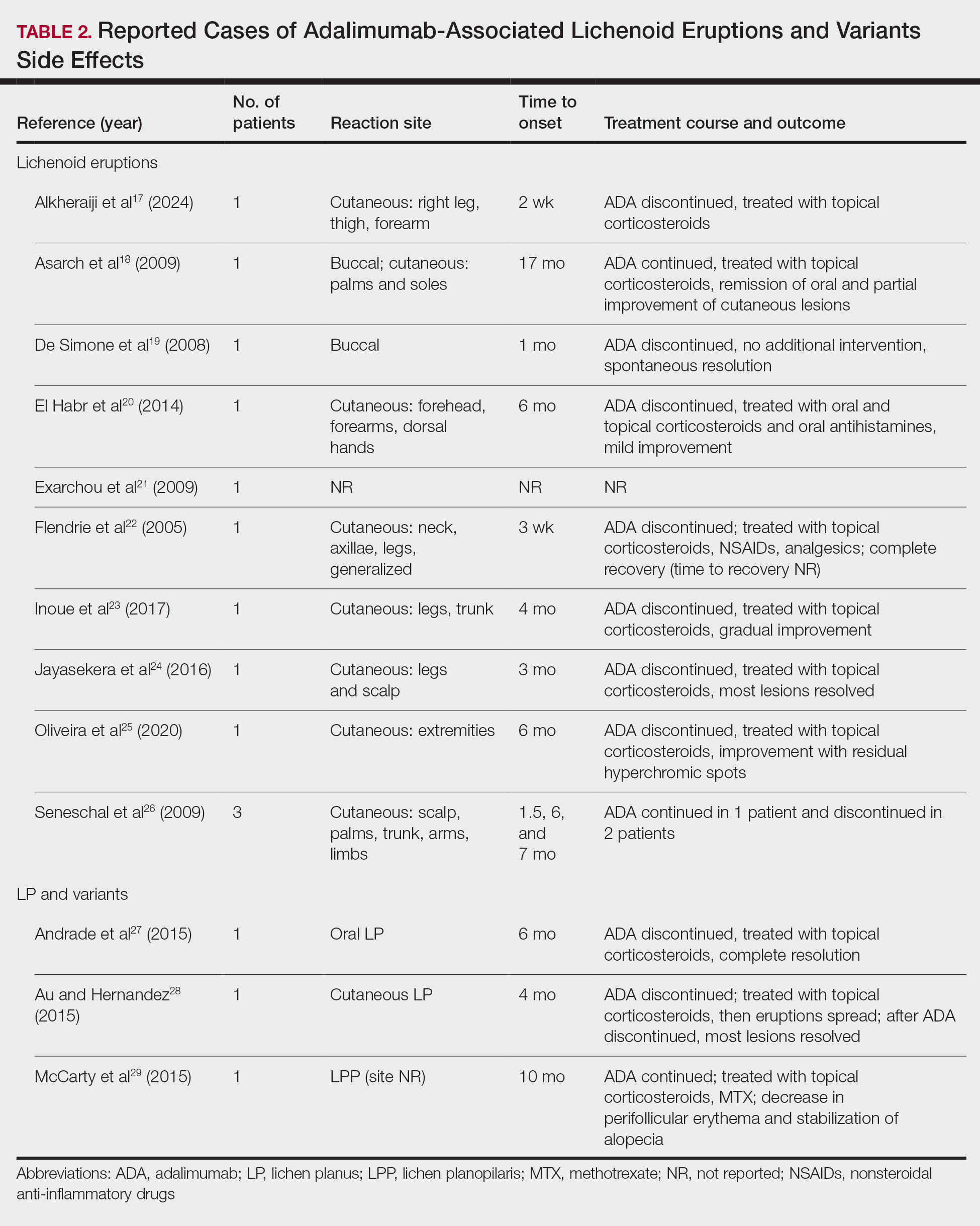
In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
Lichen planus (LP) is a chronic inflammatory condition affecting the skin (cutaneous LP), mucous membranes (oral, ocular, or vulvar LP), hair (lichen planopilaris [LPP]), and nails that predominantly occurs in middle-aged adults. Although the true etiology remains unknown, the pathogenesis of LP is thought to involve multiple factors. Several human leukocyte antigen (HLA) alleles have been associated with LP and its variants, including HLA-B27, HLA-B51, HLA-DR1 (cutaneous and oral LP), HLA-DRB1*11, and HLA-DQB1*03 (LPP). Additionally, HLA-Bw57 has been reported to be associated with oral LP in a cohort of British patients.1 In addition to HLA alleles, genetic polymorphisms in cytokines including IL-4, IL-6, IL-18, interferon (IFN) γ, and tumor necrosis factor (TNF) α and its receptor have been found to be associated with LP.2 Beyond genetics, chronic viral infection has been implicated in the development of LP. Systemic infection with the hepatitis C virus has been linked to the development of oral LP by promoting the recruitment of hepatitis C virus–specific CD8+ T cells from peripheral blood to the oral lesions, where they exhibit a terminally differentiated effector status.3 Another report found an association between human herpesvirus 7 (HHV-7) and cutaneous LP; in this study, HHV-7 RNA was detected in plasmacytoid dendritic cells but not T cells and diminished after treatment, providing evidence for dendritic cells being involved in the HHV-7–mediated pathogenesis of cutaneous LP.4 These findings were further corroborated by another study of oral LP patients that found enhanced infiltration of plasmacytoid and myeloid dendritic cells and upregulation in toll-like receptor and IFN-γ signaling.4
In addition to immune cell dysregulation, LP and its variants have been linked to neurogenic inflammation. In oral LP lesions, neurokinin 1 receptor and substance P were highly expressed and demonstrated a positive correlation with the expression of apoptotic marker caspase-3 and proliferation marker Ki-67.5 These results suggest that neuropeptides may be involved in cell proliferation and turnover in oral LP. Similarly, in patients with LPP, substance P was more abundant in affected areas, whereas another neuropeptide, calcitonin gene-related peptide, was more highly expressed in unaffected areas,6 further supporting the pathogenic role of neurogenic inflammation in LP.
A mucosal variant that often goes undiagnosed is vulvar LP. Although no distinct pathologic mechanism for vulvar LP has been established, prior reports found an association with autoantibodies.7,8 In patients with erosive vulvar LP, epidermal-binding basement membrane zone antibodies were detected in epidermal skin biopsies and in circulation with reactivity to bullous pemphigoid antigens 180 (9/11 [81.8%] patients) and 230 (2/11 [18.2%] patients).7 A similar study in patients with vulvar lichen sclerosus found similar proportions of circulating antibodies reactive to bullous pemphigoid antigens 180 (6/7 [85.7%] patients) and 230 (1/7 [14.3%] patients).8 Erosive vulvar LP has been shown to be associated with autoimmune disease (eg, alopecia areata, celiac disease and pernicious anemia),9 which suggests that the previously reported autoreactive antibodies7,8 are secondary to autoimmunity rather than primary drivers of vulvar LP pathogenesis.
Certain medications also have been reported to cause cutaneous lichenoid drug eruptions. Although they can clinically and histologically mimic classic LP, lichenoid drug eruptions are a distinct entity. Common inciting medications include thiazide diuretics, angiotensin-converting enzyme inhibitors, anti-inflammatory drugs, antimalarials, checkpoint inhibitors, antimicrobials, antihypertensives, antidiabetics, and psychiatric drugs. The exact pathologic mechanism of lichenoid drug eruptions currently is unclear but is thought to involve the binding of drug molecules to the cell-surface proteins of the epidermis, creating an antigenic hapten stimulus for CD8+T cells and triggering apoptosis of keratinocytes.1
The clinical severity of LP can range from mild localized disease to widespread and debilitating involvement. Multiple treatment modalities have been developed for management of LP, including topical and intralesional corticosteroids, phototherapy, Janus kinase inhibitors, phosphodiesterase-4 inhibitors, and anti–TNF-α inhibitors. Herein, we provide a narrative review and summary of the use of the TNF-α inhibitor adalimumab as a potential effective treatment for patients with LP.
Methods
We conducted a PubMed search of articles indexed for MEDLINE from 2005 to 2025 using the terms adalimumab AND lichen planus or adalimumab AND lichen. Articles that reported cases of oral LP, cutaneous LP, LPP, or lichenoid eruptions and adalimumab therapy were included in our review. Articles that used non-adalimumab TNF-α inhibitors were excluded. Using the search terms, 2 independent reviewers (M.G. and N.E.) conducted the literature review then screened the articles based on the inclusion and exclusion criteria. Our literature search yielded 40 articles, of which 20 met the criteria for inclusion in our narrative review.
Results
Our literature search yielded 11 patients with LP who were treated with adalimumab across studies (Table 1).10-16 Prior LP treatments included topical corticosteroids (11/11 [100%]), disease-modifying antirheumatic drugs (6/11 [54.5%]), retinoids (4/11 [36.4%]), and psoralen plus UVA (1/11 [36.4%]). Adalimumab was administered subcutaneously following 4 treatment regimens: (1)

Paradoxically, our review of the literature yielded 12 patients in whom adalimumab was associated with lichenoid-type eruptions across 9 studies (Table 2).17-29 The conditions for which these patients were undergoing treatment with adalimumab included ulcerative colitis,17 psoriasis,18,19 Crohn disease,20,26 rheumatoid arthritis,21-23,26 oligoarthritis,24 and ankylosing spondylitis.25 Lichenoid drug eruptions occurred on the legs (5/12 [41.7%]), arms (3/12 [25%]), oral mucosa (2/12 [16.7%]), and forehead or scalp (2/12 [16.7%]). Onset of time to these lichenoid eruptions ranged from 2 weeks to 17 months, with a median of 4 months. Adalimumab was discontinued in 9 (75.0%) patients and was continued in 3 (25.0%). One patient who had an onset of their lichenoid eruption after 17 months of treatment with adalimumab continued to receive adalimumab therapy with the addition of topical corticosteroids, which led to resolution of their oral lesions and partial remission of their cutaneous lesions. In 1 (8.3%) patient with localized buccal lichenoid eruptions, discontinuation of adalimumab on its own was sufficient to completely clear the lesions. Seven patients (7/12 [58.3%]) received topical corticosteroids with minimal (2/12 [16.7%]) or moderate (4/12 [33.3%]) improvement, and 1 (8.3%) patient did not have reported outcomes data. Eosinophils were detected within the adalimumab-associated lichenoid eruptions in 3 (25.0%) patients.17,20,22

In addition to its association with lichenoid drug eruptions, adalimumab also was reported to induce LPP in a patient who was being treated for Behçet disease,29 oral LP in a patient being treated for Crohn disease,27 and cutaneous LP in a patient being treated for Crohn disease (Table 2).28 Time to onset ranged from 4 to 10 months, with a median of 6 months. Adalimumab was discontinued in 2 of 3 (66.7%) patients and was continued in the other patient (33.3%). After cessation of adalimumab therapy, administration of topical steroids led to complete resolution in the case of associated oral LP. In contrast, in adalimumab-induced cutaneous LP, initial topical corticosteroid treatment led to progression of lesions, which mostly resolved after adalimumab cessation. In 1 patient with LPP in whom adalimumab therapy could not be discontinued, topical corticosteroid and methotrexate therapy reduced the perifollicular erythema and stabilized the alopecia without full remission.
Comment
Conventional treatment modalities for LP often include topical corticosteroids as first-line therapy, with systemic corticosteroids, phototherapy, retinoids, or immunosuppressants (eg, cyclosporine or methotrexate) reserved for more severe or widespread disease. Historically, these approaches primarily have aimed to control symptoms rather than achieve long-term resolution; however, novel therapies including biologics and targeted immunomodulators show potential to induce sustained remission and improve quality of life for patients with refractory or mucosal LP.
In all reports where adalimumab was used to treat LP, patients initially received topical corticosteroids. While corticosteroids and other immunosuppressive agents are standard therapies, they often provide only temporary relief and may have an unfavorable side effect profile. Our review highlights the emerging role of adalimumab, a TNF-α inhibitor, in off-label management of LP subtypes, including cutaneous, mucosal, and vulvar LP and LPP. In several small case series and reports, patients treated with adalimumab experienced clinical improvement, including symptom resolution and quality-of-life enhancement, as well as complete remission, indicating a durable response.
The potential benefit of adalimumab in treating LP must be balanced with its paradoxical risk for inducing lichenoid eruptions as well as LP and its variants, as identified in our narrative review that included reports of patients receiving this biologic for other indications.17-29 Since adalimumab is a fully humanized antibody, the development of neutralizing antibodies may not account for drug-induced LP and lichenoid eruptions. Given that it blocks TNF-α, adalimumab may induce these lesions through a cytokine imbalance. This is supported by data demonstrating enhanced type I IFN-related proteins in plaques of patients with psoriasiform lesions treated with TNF-α inhibitors.26 These drug-induced eruptions often resolved or improved with topical corticosteroids after discontinuation, but their occurrence underscores the complexity of therapeutically targeting TNF-α in the management of LP. Our literature review suggests that adalimumab may offer therapeutic benefit in select cases of LP refractory to conventional therapy, especially when systemic control is required. Nonetheless, the risk for LP and lichenoid reactions necessitates cautious use and further investigation.
Conclusion
While the current evidence is limited to case reports and series, adalimumab shows promise as an effective and tolerable off-label treatment for LP, particularly in patients who are unresponsive to conventional immunosuppressive therapies. Remission or clinically significant improvement was achieved in several cases; however, the potential for adalimumab to induce LP and lichenoid eruptions underscores the need for careful patient selection and monitoring. Further prospective studies and larger cohorts are warranted to better define the safety and efficacy of adalimumab in treating LP lesions.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
- Boch K, Langan EA, Kridin K, et al. Lichen planus. Front Med (Lausanne). 2021;8:737813.
- Gorouhi F, Davari P, Fazel N. Cutaneous and mucosal lichen planus: a comprehensive review of clinical subtypes, risk factors, diagnosis, and prognosis. ScientificWorldJournal. 2014;2014:742826.
- Pilli M, Penna A, Zerbini A, et al. Oral lichen planus pathogenesis: a role for the HCV-specific cellular immune response. Hepatology. 2002;36:1446-1452.
- Wang Y, Shang S, Sun Q, et al. Increased infiltration of CD11 c+/CD123+ dendritic cell subsets and upregulation of TLR/IFN-α signaling participate in pathogenesis of oral lichen planus. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125:459-467.E2.
- González Moles M, Esteban F, Ruiz-Ávila I, et al. A role for the substance P/NK-1 receptor complex in cell proliferation and apoptosis in oral lichen planus. Oral Dis. 2009;15:162-169.
- Doche I, Wilcox GL, Ericson M, et al. Evidence for neurogenic inflammation in lichen planopilaris and frontal fibrosing alopecia pathogenic mechanism. Exp Dermatol. 2020;29:282-285.
- Cooper SM, Dean D, Allen J, et al. Erosive lichen planus of the vulva: weak circulating basement membrane zone antibodies are present. Clin Exp Dermatol. 2005;30:551-556.
- Howard A, Dean D, Cooper S, et al. Circulating basement membrane zone antibodies are found in lichen sclerosus of the vulva. Australas J Dermatol. 2004;45:12-15.
- Cooper SM, Ali I, Baldo M, et al. The association of lichen sclerosus and erosive lichen planus of the vulva with autoimmune disease: a case-control study. Arch Dermatol. 2008;144:1432-1435.
- Alam MS, LaBelle B. Treatment of lichen planopilaris with adalimumab in a patient with hidradenitis suppurativa and rheumatoid arthritis. JAAD Case Rep. 2020;6:219-221.
- Alhubayshi BS, Alnoshan AA, Alhumidi AA, et al. Bullous lichen planus treated with adalimumab: a case report. Case Rep Dermatol. 2025;17:42-47.
- Chao TJ. Adalimumab in the management of cutaneous and oral lichen planus. Cutis. 2009;84:325-328.
- Courtney A, Adamson SR, Veysey E. Adalimumab use in severe recalcitrant vulval lichen sclerosus and vulval lichen planus. J Low Genit Tract Dis. 2025;29:190-194.
- Holló P, Szakonyi J, Kiss D, et al. Successful treatment of lichen planus with adalimumab. Acta Derm Venereol. 2012;92:385-386.
- Khodeir J, Ohanian P, Ohanian M. Successful treatment of annular atrophic lichen planus with adalimumab. Clin Case Rep. 2025;13:E70036.
- Kreutzer K, Effendy I. Therapy-resistant folliculitis decalvans and lichen planopilaris successfully treated with adalimumab. J Dtsch Dermatol Ges. 2014;12:74-76.
- Alkheraiji A, Alotaibi H, Irfan Thalib H. Lichenoid drug eruption secondary to adalimumab: a case report. Cureus. 2024;16:E64013.
- Asarch A, Gottlieb AB, Lee J, et al. Lichen planus-like eruptions: an emerging side effect of tumor necrosis factor-alpha antagonists. J Am Acad Dermatol. 2009;61:104-111.
- De Simone C, Caldarola G, D’Agostino M, et al. Lichenoid reaction induced by adalimumab. J Eur Acad Dermatol Venereol. 2008;22:626-627.
- El Habr C, Meguerian Z, Sammour R. Adalimumab-induced lichenoid drug eruption. J Med Liban. 2014;62:238-240.
- Exarchou SA, Voulgari PV, Markatseli TE, et al. Immune-mediated skin lesions in patients treated with anti-tumour necrosis factor alpha inhibitors. Scand J Rheumatol. 2009;38:328-331.
- Flendrie M, Vissers WH, Creemers MC, et al. Dermatological conditions during TNF-α-blocking therapy in patients with rheumatoid arthritis: a prospective study. Arthritis Res Ther. 2005;7:R666-R676.
- Inoue A, Sawada Y, Yamaguchi T, et al. Lichenoid drug eruption caused by adalimumab: a case report and literature review. Eur J Dermatol. 2017;27:69-70.
- Jayasekera PSA, Walsh ML, Hurrell D, et al. Case report of lichen planopilaris occurring in a pediatric patient receiving a tumor necrosis factor α inhibitor and a review of the literature. Pediatr Dermatol. 2016;33:E143-E146.
- Oliveira SCD, Vasconcelos AHC, Magalhães EPB, et al. Clinical, histopathological and outcome analysis of five patients with lichenoid eruption following anti-tumor necrosis factor-alpha therapy for ankylosing spondylitis: report of one case and review of the literature. Cureus. 2020;12:E10598.
- Seneschal J, Milpied B, Vergier B, et al. Cytokine imbalance with increased production of interferon-alpha in psoriasiform eruptions associated with antitumour necrosis factor-alpha treatments. Br J Dermatol. 2009;161:1081-1088.
- Andrade P, Lopes S, Albuquerque A, et al. Oral lichen planus in IBD patients: a paradoxical adverse effect of anti-TNF-α therapy. Dig Dis Sci. 2015;60:2746-2749.
- Au S, Hernandez C. Paradoxical induction of psoriasis and lichen planus by tumor necrosis factor-α inhibitors. Skinmed. 2015;13:403-405.
- McCarty M, Basile A, Bair B, et al. Lichenoid reactions in association with tumor necrosis factor alpha inhibitors. J Clin Aesthet Dermatol. 2015;8:45-49.
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Adalimumab in Lichen Planus: A Narrative Review of Treatment and Paradoxical Reactions
Practice Points
- Adalimumab can be beneficial when used off label for treatment of lichen planus in patients who do not respond to conventional therapies, including corticosteroids and immunosuppressants.
- Clinicians should be aware that adalimumab could potentially lead to paradoxical lichenoid eruptions and should monitor patients closely during treatment.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
To the Editor:
Severe cutaneous adverse reactions (SCARs) are rare, life-threatening reactions that include acute generalized exanthematous pustulosis (AGEP), drug reaction with eosinophilia and systemic symptoms (DRESS), and Stevens-Johnson syndrome (SJS)/toxic epidermal necrolysis (TEN).1 In addition to being associated with commonly implicated medications, SCARs also may occur in the setting of antineoplastic therapy.2,3 Although antineoplastic-associated SCARs have been described, diagnosis can be difficult due to varying latency periods and atypical clinical features, such as those observed with BRAF inhibitor–related DRESS during immunotherapy.4 Severe cutaneous adverse reactions can increase morbidity and mortality in the oncologic patient population due to both the clinical sequelae from the cutaneous reaction and the potential to interrupt cancer treatment.
The aim of this study was to evaluate the clinical characteristics, outcomes, and impact on cancer treatment among patients diagnosed with a SCAR while receiving active therapy for malignancy. We conducted a retrospective chart review of electronic medical records at Yale New Haven Hospital (New Haven, Connecticut) from 2013 to 2023, identifying patients receiving antineoplastic therapy who were diagnosed with a SCAR. Cases were identified through a search of the electronic medical record performed by the joint data analytics team using the keywords DRESS, SJS, TEN, AGEP, and generalized bullous fixed drug eruption, along with spelling variations (both abbreviations and full terms), in addition to manual review by one of the authors (K.V.) of the inpatient dermatology consultation log and dermatopathology database. Only patients for whom an antineoplastic agent was identified as a high-probability culprit by the dermatology and/or oncology teams were included.
In total, 20 patients (11 female, 9 male) were identified as having an antineoplastic-associated SCAR. All patients had metastatic or advanced disease. We identified 2 (10%) cases of AGEP, 16 (80%) cases of DRESS, and 3 (15%) cases of SJS/TEN. One patient on immunotherapy had 2 distinct SCARs (AGEP, DRESS) at different time points. Table 1 describes patient and SCAR characteristics as well as impact on cancer treatment. The median (interquartile range [IQR]) latency period for AGEP was 7.5 (4-11) days. The median (IQR) latency period for 13 of the 16 (81%) DRESS cases was 14 (10-32) days. For 3 DRESS cases with a potential second-hit phenomenon in the setting of current or antecedent immunotherapy,5 the median (IQR) latency period was 122 (96-426) days for the immunotherapy drug and 28 (21-52) days for the drug culprit. The median (IQR) latency period for SJS/TEN was 23 (20-27) days.

Patients received treatment with combination systemic corticosteroids and topical corticosteroids in 13 (65%) cases, systemic corticosteroid monotherapy in 6 (30%) cases, or combination systemic corticosteroids and etanercept in 1 (5%) case. All patients experienced resolution of the SCAR and survived to hospital discharge. Most (17/20 [85%]) patients experienced interruption or discontinuation of cancer treatment. Table 2 describes the implicated antineoplastic therapies, which included chemotherapy (3 DRESS, 1 SJS/TEN), hormonal therapy (1 DRESS), immunotherapy (1 AGEP, 4 DRESS), and targeted therapy (1 AGEP, 8 DRESS, 2 SJS/TEN).

Limitations of this study include the retrospective study design, the small sample size, and the challenge of drug culprit identification in oncologic patients on multiple high-probability medications.
Though rare, SCARs can be encountered in patients on antineoplastic therapy with a wide range of drug culprits. In our cohort, SCARs occurred with various antineoplastic agents, including chemotherapy, hormonal therapy, immunotherapy, and targeted therapy. The most common antineoplastic-associated SCAR was DRESS, which had the widest latency period in the setting of a potential second-hit phenomenon with another drug culprit. Although we did not observe any cases of SJS/TEN in the immunotherapy category, it is important to consider progressive immunotherapy-related mucocutaneous eruption in the differential diagnosis. Fortunately, all patients survived to hospital discharge and experienced SCAR resolution with systemic treatment; however, most patients experienced interruption of cancer therapy, which has the potential to affect oncologic outcomes. This interruption is not uncommon, as rechallenge of an antineoplastic agent in patients with a therapy-related SCAR generally is not recommended. The awareness and prompt management of SCARs in a patient on treatment for malignancy are critical in order to minimize negative outcomes in this vulnerable patient population.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
- Duong TA, Valeyrie-Allanore L, Wolkenstein P, et al. Severe cutaneous adverse reactions to drugs. Lancet. 2017;390: 1996-2011.
- Chen CB, Wu MY, Ng CY, et al. Severe cutaneous adverse reactions induced by targeted anticancer therapies and immunotherapies. Cancer Manag Res. 2018;10:1259-1273.
- Ng CY, Chen CB, Wu MY, et al. Anticancer drugs induced severe adverse cutaneous drug reactions: an updated review on the risks associated with anticancer targeted therapy or immunotherapies. J Immunol Res. 2018;2018:5376476.
- Maloney NJ, Rana J, Yang JJ, et al. Clinical features of druginduced hypersensitivity syndrome to BRAF inhibitors with and without previous immune checkpoint inhibition: a review. Support Care Cancer. 2022;30:2839-2851.
- Hammond S, Olsson-Brown A, Grice S, et al. Does immune checkpoint inhibitor therapy increase the frequency of adverse reactions to concomitant medications? Clin Exp Allergy. 2022;52:600-603.
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Severe Cutaneous Adverse Reactions in the Setting of Antineoplastic Therapy: A Single-Institution Retrospective Study
Practice Points
- Clinicians should be aware of the occurrence of severe cutaneous adverse reactions (SCARs) in patients on antineoplastic therapy to prevent delays in treatment and improve patient outcomes.
- Rapid initiation of treatment can be effective in resolving SCARs and ensuring full recovery.
- Close coordination between dermatology and oncology teams is crucial to manage SCARs while minimizing cancer treatment interruptions.
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
To the Editor:
At-home microcurrent facial devices have gained rapid popularity for cosmetic rejuvenation, promising improvements in skin tone, contour, and collagen production.¹ In particular, the post–COVID-19 era has seen a surge in at-home beauty practices driven by social media influence, with the global microcurrent facial market estimated at $372.9 million in 2022 and projected to grow at a compound annual growth rate of 7.3% through 2030.1 Microcurrent devices deliver low-level electrical currents to the skin and underlying muscles. Given the limited exploration of the long-term safety, we aimed to collate existing data and identify trends in reports of adverse events (AEs) associated with these microcurrent devices.
On April 15, 2025, the US Food and Drug Administration’s Manufacturer and User Facility Device Experience (MAUDE) database was queried for medical device reports from January 1, 2013, through March 31, 2025, using product names and keywords including NuFACE, TheraFace, FOREO, and microcurrent device. Search terms were limited to brands for which complaint data existed in the MAUDE database at the time of query. To ensure accuracy, reports were manually reviewed to eliminate duplicates and irrelevant entries.
A total of 28 unique AE reports associated with at-home microcurrent devices were identified (eTable). The majority involved NuFACE devices (ie, NuFACE Trinity, NuFACE Mini, and NuFACE Trinity+)(NuFACE)(n=25), followed by the TheraFace PRO (Therabody, Inc)(n=2) and the FOREO BEAR (FOREO)(n=1). The most frequently documented AEs associated with the NuFACE devices included arrhythmia (7/25 [28%]), pain (6/25 [24%]), dizziness (4/25 [16%]), headache (4/25 [16%]), and inflammation (4/25 [16%]). There was 1 (4%) case of retinal detachment. The TheraFace PRO was associated with device overheating (2/2 [100%]), and the FOREO BEAR was associated with facial deformity/disfigurement (1/1 [100%]).

While microcurrent therapy is widely marketed to consumers through social media influencers and at-home beauty platforms,1 randomized controlled trials (RCTs) evaluating AEs related to use of this technology are lacking, possibly due to nonstringent regulation of nonprescription cosmetic devices.² Contrary to our findings, RCTs of microcurrent devices have reported minimal or no AEs; for instance, an RCT evaluating 56 participants treated 5 times weekly for 12 weeks with a microcurrent device that was not included in our analysis reported only mild erythema in all experimental group participants.2 In another RCT of 30 participants, 15 of whom were treated with a microcurrent device and 15 with placebo for 30 minutes once daily over a period of 10 days, no AEs were reported.3 A cohort analysis of 34 patients also provided preliminary evidence supporting the use of microcurrent therapy for chronic back and neck pain, beyond its cosmetic applications.4 Despite the lack of reported AEs in the literature, there is a notable absence of large-scale, rigorous studies on this topic.
Our analysis was subject to the limitations of the MAUDE database, in which reports of severe AEs are more likely to be reported than transient ones. Additionally, the small sample size and lack of a known denominator make it difficult to compare frequencies of AEs among different microcurrent tools. The products chosen for this study were the select few that reported complaint data, but there is a large existing market of devices that may be associated with AEs that have yet to be reported, potentially because of their novelty.
Our findings suggest that, despite their over-the-counter availability, microcurrent facial devices may carry major risks—particularly in at-home settings. While short-term studies have highlighted potential benefits, the small sample sizes and limited follow-up make it difficult to comprehensively characterize long-term safety risks. Among available studies on microcurrent beauty treatments, the longest follow-up was only 12 weeks.2 Our findings support the need for further large-scale and longitudinal studies to evaluate both the efficacy and safety of at-home microcurrent therapy, especially with increasing consumer interest. The diversity of the products available adds to the challenge of broad safety guidelines, in addition to the lack of long-term clinical studies.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
- Microcurrent Facial Market Size, Growth & Trends Report 2030. Grand View Research. Published 2023. Accessed March 3, 2026. https://www.grandviewresearch.com/industry-analysis/microcurrent-facial-market-report
- Bu P, Duan R, Luo J, et al. Development of home beauty devices for facial rejuvenation: establishment of efficacy evaluation system. Clin Cosmet Investig Dermatol. 2024;17:553-563.
- Jain S, Arora M. Effect of microcurrent facial muscle toning on fine wrinkles & firmness of face. IAMR J Physiother. 2012;1:13-19.
- Armstrong K, Gokal R, Chevalier A, et al. Microcurrent point stimulation applied to lower back acupuncture points for the treatment of nonspecific neck pain. J Altern Complement Med. 2017;23:295-299.
Adverse Events Associated With At-Home Microcurrent Facial Devices
Adverse Events Associated With At-Home Microcurrent Facial Devices
PRACTICE POINTS
- At-home microcurrent facial devices have been associated with serious adverse events, including arrhythmia, pain, dizziness, and retinal detachment, based on US Food and Drug Administration Manufacturer and User Facility Device Experience database reports, underscoring the importance of counseling patients about potential risks prior to use.
- Existing randomized controlled trials of microcurrent devices are limited by small sample sizes and short follow-up periods (maximum 12 weeks), making it difficult to characterize the long-term safety profile of these increasingly popular devices.
- Dermatologists should be aware that the largely unregulated at-home microcurrent device market lacks robust, large-scale safety data. Patients, particularly those with cardiac conditions or implanted electrical devices, should be advised to consult a physician before use.