User login
FDA clears new blood-draw device
Photo courtesy of
Velano Vascular
The US Food and Drug Administration (FDA) has cleared for marketing a device that reduces the need for venipunctures for in-hospital blood draws.
Velano Vascular’s blood-draw device resembles a common syringe.
It allows peripheral intravenous catheters to be repurposed to draw blood from patients, thereby reducing the need for additional needle sticks among patients receiving medications and hydration via intravenous delivery.
The single-use device will soon be used for clinical evaluation in select hospitals, including the University of Pennsylvania in Philadelphia and University Hospitals Case Medical Center of Cleveland in Ohio.
“A fundamental benefit of this technology is reducing the ‘pin cushion effect,’ in which hospitalized patients are ‘stuck’ several times daily to obtain blood tests,” said Eric M. Stone, co-founder and chief executive officer of Velano Vascular, the company developing the blood-draw device.
“Oftentimes, the draw procedure is plagued by multiple failed attempts. The FDA’s clearance of this novel technology validates the existing clinical need and will allow us to expedite our efforts to bring this innovation to patients, healthcare providers, and hospitals around the world.”
According to research conducted by Velano Vascular, 1 of every 3 hospital patients is stuck 2 or more times daily for blood draws, with a significant subset of these patients receiving 3 or more blood draws, along with numerous needle sticks.
Twenty-eight percent of adult venipunctures and 44% of pediatric venipunctures require more than one stick to successfully draw blood.
“Traditional blood draws are one of the most common and most problematic healthcare procedures,” said Karen Daley, PhD, RN, past president of the American Nurses Association and a healthcare worker safety advocate.
“It is an antiquated technology that creates pain and anxiety for many patients, a significant safety risk for healthcare professionals, and a real inefficiency in our healthcare system. Velano Vascular has developed a common-sense solution to this pervasive, long-standing problem.” ![]()

Photo courtesy of
Velano Vascular
The US Food and Drug Administration (FDA) has cleared for marketing a device that reduces the need for venipunctures for in-hospital blood draws.
Velano Vascular’s blood-draw device resembles a common syringe.
It allows peripheral intravenous catheters to be repurposed to draw blood from patients, thereby reducing the need for additional needle sticks among patients receiving medications and hydration via intravenous delivery.
The single-use device will soon be used for clinical evaluation in select hospitals, including the University of Pennsylvania in Philadelphia and University Hospitals Case Medical Center of Cleveland in Ohio.
“A fundamental benefit of this technology is reducing the ‘pin cushion effect,’ in which hospitalized patients are ‘stuck’ several times daily to obtain blood tests,” said Eric M. Stone, co-founder and chief executive officer of Velano Vascular, the company developing the blood-draw device.
“Oftentimes, the draw procedure is plagued by multiple failed attempts. The FDA’s clearance of this novel technology validates the existing clinical need and will allow us to expedite our efforts to bring this innovation to patients, healthcare providers, and hospitals around the world.”
According to research conducted by Velano Vascular, 1 of every 3 hospital patients is stuck 2 or more times daily for blood draws, with a significant subset of these patients receiving 3 or more blood draws, along with numerous needle sticks.
Twenty-eight percent of adult venipunctures and 44% of pediatric venipunctures require more than one stick to successfully draw blood.
“Traditional blood draws are one of the most common and most problematic healthcare procedures,” said Karen Daley, PhD, RN, past president of the American Nurses Association and a healthcare worker safety advocate.
“It is an antiquated technology that creates pain and anxiety for many patients, a significant safety risk for healthcare professionals, and a real inefficiency in our healthcare system. Velano Vascular has developed a common-sense solution to this pervasive, long-standing problem.” ![]()

Photo courtesy of
Velano Vascular
The US Food and Drug Administration (FDA) has cleared for marketing a device that reduces the need for venipunctures for in-hospital blood draws.
Velano Vascular’s blood-draw device resembles a common syringe.
It allows peripheral intravenous catheters to be repurposed to draw blood from patients, thereby reducing the need for additional needle sticks among patients receiving medications and hydration via intravenous delivery.
The single-use device will soon be used for clinical evaluation in select hospitals, including the University of Pennsylvania in Philadelphia and University Hospitals Case Medical Center of Cleveland in Ohio.
“A fundamental benefit of this technology is reducing the ‘pin cushion effect,’ in which hospitalized patients are ‘stuck’ several times daily to obtain blood tests,” said Eric M. Stone, co-founder and chief executive officer of Velano Vascular, the company developing the blood-draw device.
“Oftentimes, the draw procedure is plagued by multiple failed attempts. The FDA’s clearance of this novel technology validates the existing clinical need and will allow us to expedite our efforts to bring this innovation to patients, healthcare providers, and hospitals around the world.”
According to research conducted by Velano Vascular, 1 of every 3 hospital patients is stuck 2 or more times daily for blood draws, with a significant subset of these patients receiving 3 or more blood draws, along with numerous needle sticks.
Twenty-eight percent of adult venipunctures and 44% of pediatric venipunctures require more than one stick to successfully draw blood.
“Traditional blood draws are one of the most common and most problematic healthcare procedures,” said Karen Daley, PhD, RN, past president of the American Nurses Association and a healthcare worker safety advocate.
“It is an antiquated technology that creates pain and anxiety for many patients, a significant safety risk for healthcare professionals, and a real inefficiency in our healthcare system. Velano Vascular has developed a common-sense solution to this pervasive, long-standing problem.” ![]()
Team creates new cells for modeling malaria

infection in iPSC-derived liver
cells 8 days after infection
Credit: Shengyong Ng et al.
Researchers say they’ve found a way to grow liver-like cells from induced pluripotent stem cells (iPSCs).
The liver-like cells can be infected with several strains of the malaria parasite and respond to existing drugs the same way mature human liver cells do.
The new cells, described in Stem Cell Reports, could allow scientists to test drugs on cells from people with different genetic backgrounds, who may respond differently to malaria infection and treatment.
Modeling infection
Until now, malaria researchers have not had many reliable ways to test new drugs in liver tissue.
“What’s historically been done is people have tried to make do with the systems that were available,” said study author Sangeeta Bhatia, MD, PhD, of the Massachusetts Institute of Technology in Cambridge.
In 2013, Dr Bhatia and her colleagues showed they could model malaria infection in hepatocytes from human donors. However, this generates only a limited supply from each donor, and not all of the cells work well for drug studies.
The researchers then turned to iPSCs, which can be generated from human skin cells by adding reprogramming factors. To create liver cells, the researchers added a series of growth factors, including hepatocyte growth factor, to the iPSCs.
The team generated these cells in 2012 and used them to model infection of hepatitis C. However, these cells, known as hepatocyte-like cells, did not seem to be as mature as real adult liver cells.
In the current study, the researchers found these cells could be infected with several strains of malaria. But, initially, the cells did not respond to drugs in the same way as adult liver cells.
In particular, they were not sensitive to primaquine, which works only if cells have a certain set of drug-metabolism enzymes found in mature liver cells.
To induce the cells to become more mature and turn on these metabolic enzymes, the researchers added a molecule they had identified in a previous study. This compound, which the researchers call a “maturin,” stimulated the cells to turn on those enzymes, which made them sensitive to primaquine.
Toward better drugs
The team is now working with the nonprofit foundation Medical Malaria Ventures to test about 10 potential malaria drugs that are in the pipeline, first using adult donor liver cells and then the hepatocyte-like cells generated in this study.
These cells could also prove useful to help identify new drug targets, the researchers said. In this study, they found the liver-like cells can be infected with malaria when they are still in the equivalent of fetal stages of development, when they become hepatoblasts, which are precursors to hepatocytes.
In future studies, the researchers plan to investigate which genes get turned on when the cells become susceptible to infection, which may suggest new targets for malaria drugs.
They also hope to compare the genes needed for malaria infection with those needed for hepatitis infection, in hopes of identifying common pathways to target for both diseases. ![]()

infection in iPSC-derived liver
cells 8 days after infection
Credit: Shengyong Ng et al.
Researchers say they’ve found a way to grow liver-like cells from induced pluripotent stem cells (iPSCs).
The liver-like cells can be infected with several strains of the malaria parasite and respond to existing drugs the same way mature human liver cells do.
The new cells, described in Stem Cell Reports, could allow scientists to test drugs on cells from people with different genetic backgrounds, who may respond differently to malaria infection and treatment.
Modeling infection
Until now, malaria researchers have not had many reliable ways to test new drugs in liver tissue.
“What’s historically been done is people have tried to make do with the systems that were available,” said study author Sangeeta Bhatia, MD, PhD, of the Massachusetts Institute of Technology in Cambridge.
In 2013, Dr Bhatia and her colleagues showed they could model malaria infection in hepatocytes from human donors. However, this generates only a limited supply from each donor, and not all of the cells work well for drug studies.
The researchers then turned to iPSCs, which can be generated from human skin cells by adding reprogramming factors. To create liver cells, the researchers added a series of growth factors, including hepatocyte growth factor, to the iPSCs.
The team generated these cells in 2012 and used them to model infection of hepatitis C. However, these cells, known as hepatocyte-like cells, did not seem to be as mature as real adult liver cells.
In the current study, the researchers found these cells could be infected with several strains of malaria. But, initially, the cells did not respond to drugs in the same way as adult liver cells.
In particular, they were not sensitive to primaquine, which works only if cells have a certain set of drug-metabolism enzymes found in mature liver cells.
To induce the cells to become more mature and turn on these metabolic enzymes, the researchers added a molecule they had identified in a previous study. This compound, which the researchers call a “maturin,” stimulated the cells to turn on those enzymes, which made them sensitive to primaquine.
Toward better drugs
The team is now working with the nonprofit foundation Medical Malaria Ventures to test about 10 potential malaria drugs that are in the pipeline, first using adult donor liver cells and then the hepatocyte-like cells generated in this study.
These cells could also prove useful to help identify new drug targets, the researchers said. In this study, they found the liver-like cells can be infected with malaria when they are still in the equivalent of fetal stages of development, when they become hepatoblasts, which are precursors to hepatocytes.
In future studies, the researchers plan to investigate which genes get turned on when the cells become susceptible to infection, which may suggest new targets for malaria drugs.
They also hope to compare the genes needed for malaria infection with those needed for hepatitis infection, in hopes of identifying common pathways to target for both diseases. ![]()

infection in iPSC-derived liver
cells 8 days after infection
Credit: Shengyong Ng et al.
Researchers say they’ve found a way to grow liver-like cells from induced pluripotent stem cells (iPSCs).
The liver-like cells can be infected with several strains of the malaria parasite and respond to existing drugs the same way mature human liver cells do.
The new cells, described in Stem Cell Reports, could allow scientists to test drugs on cells from people with different genetic backgrounds, who may respond differently to malaria infection and treatment.
Modeling infection
Until now, malaria researchers have not had many reliable ways to test new drugs in liver tissue.
“What’s historically been done is people have tried to make do with the systems that were available,” said study author Sangeeta Bhatia, MD, PhD, of the Massachusetts Institute of Technology in Cambridge.
In 2013, Dr Bhatia and her colleagues showed they could model malaria infection in hepatocytes from human donors. However, this generates only a limited supply from each donor, and not all of the cells work well for drug studies.
The researchers then turned to iPSCs, which can be generated from human skin cells by adding reprogramming factors. To create liver cells, the researchers added a series of growth factors, including hepatocyte growth factor, to the iPSCs.
The team generated these cells in 2012 and used them to model infection of hepatitis C. However, these cells, known as hepatocyte-like cells, did not seem to be as mature as real adult liver cells.
In the current study, the researchers found these cells could be infected with several strains of malaria. But, initially, the cells did not respond to drugs in the same way as adult liver cells.
In particular, they were not sensitive to primaquine, which works only if cells have a certain set of drug-metabolism enzymes found in mature liver cells.
To induce the cells to become more mature and turn on these metabolic enzymes, the researchers added a molecule they had identified in a previous study. This compound, which the researchers call a “maturin,” stimulated the cells to turn on those enzymes, which made them sensitive to primaquine.
Toward better drugs
The team is now working with the nonprofit foundation Medical Malaria Ventures to test about 10 potential malaria drugs that are in the pipeline, first using adult donor liver cells and then the hepatocyte-like cells generated in this study.
These cells could also prove useful to help identify new drug targets, the researchers said. In this study, they found the liver-like cells can be infected with malaria when they are still in the equivalent of fetal stages of development, when they become hepatoblasts, which are precursors to hepatocytes.
In future studies, the researchers plan to investigate which genes get turned on when the cells become susceptible to infection, which may suggest new targets for malaria drugs.
They also hope to compare the genes needed for malaria infection with those needed for hepatitis infection, in hopes of identifying common pathways to target for both diseases. ![]()
Vitamin A as malaria prophylaxis

Photo by Sarah Mattison
New research suggests vitamin A may reduce the risk of malaria in young children.
The study showed that children under 5 living in sub-Saharan Africa were 54% less likely to develop malaria if they had been given a single, large dose of vitamin A.
The finding, published in eLife, indicates that vitamin A may be able to protect children from the malaria parasite, especially if administered during the wet season, when malaria-infected mosquitos are most prevalent.
“Now, we need to test vitamin A in a randomized, controlled, clinical trial to better understand whether this could really be an effective way to prevent this disease,” said study author Maria-Graciela Hollm-Delgado, PhD, of the Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland.
Dr Hollm-Delgado and her colleagues analyzed national survey data from 4 sub-Saharan countries—Burkina Faso, Mozambique, Rwanda, and Senegal—on children between the ages of 6 months and 59 months.
The goal was to determine the risk of Plasmodium parasitemia (n=8390) and Plasmodium falciparum-specific antigenemia (n=6121) following vitamin A supplementation and vaccinations.
The researchers found the measles and polio vaccines were not associated with malaria. And Bacille Calmette Guerin vaccination was associated with an increased risk of antigenemia (relative risk [RR]=4.06) but not parasitemia.
Only vitamin A was protective against malaria. Children who received vitamin A were less likely to present with parasitemia (RR=0.46) and antigenemia (RR=0.23).
Vitamin A appeared to be more protective under certain circumstances, including when administered during the rainy season, as well as when given to older children and when more time had passed since supplementation.
The researchers aren’t certain why vitamin A would reduce the rate of malaria infection, but they suspect it is because vitamin A, which is known to boost immunity and improve the ability to fight off infection, may help the body clear out the malaria parasite more quickly.
Only 62% of children in the study had received vitamin A supplementation, even though World Health Organization guidelines recommend that all children in sub-Saharan Africa receive a single, large dose of vitamin A.
Rates were higher for many vaccinations, Dr Hollm-Delgado said, noting that the guidelines for vitamin A aren’t as specific as they are for most vaccinations. ![]()

Photo by Sarah Mattison
New research suggests vitamin A may reduce the risk of malaria in young children.
The study showed that children under 5 living in sub-Saharan Africa were 54% less likely to develop malaria if they had been given a single, large dose of vitamin A.
The finding, published in eLife, indicates that vitamin A may be able to protect children from the malaria parasite, especially if administered during the wet season, when malaria-infected mosquitos are most prevalent.
“Now, we need to test vitamin A in a randomized, controlled, clinical trial to better understand whether this could really be an effective way to prevent this disease,” said study author Maria-Graciela Hollm-Delgado, PhD, of the Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland.
Dr Hollm-Delgado and her colleagues analyzed national survey data from 4 sub-Saharan countries—Burkina Faso, Mozambique, Rwanda, and Senegal—on children between the ages of 6 months and 59 months.
The goal was to determine the risk of Plasmodium parasitemia (n=8390) and Plasmodium falciparum-specific antigenemia (n=6121) following vitamin A supplementation and vaccinations.
The researchers found the measles and polio vaccines were not associated with malaria. And Bacille Calmette Guerin vaccination was associated with an increased risk of antigenemia (relative risk [RR]=4.06) but not parasitemia.
Only vitamin A was protective against malaria. Children who received vitamin A were less likely to present with parasitemia (RR=0.46) and antigenemia (RR=0.23).
Vitamin A appeared to be more protective under certain circumstances, including when administered during the rainy season, as well as when given to older children and when more time had passed since supplementation.
The researchers aren’t certain why vitamin A would reduce the rate of malaria infection, but they suspect it is because vitamin A, which is known to boost immunity and improve the ability to fight off infection, may help the body clear out the malaria parasite more quickly.
Only 62% of children in the study had received vitamin A supplementation, even though World Health Organization guidelines recommend that all children in sub-Saharan Africa receive a single, large dose of vitamin A.
Rates were higher for many vaccinations, Dr Hollm-Delgado said, noting that the guidelines for vitamin A aren’t as specific as they are for most vaccinations. ![]()

Photo by Sarah Mattison
New research suggests vitamin A may reduce the risk of malaria in young children.
The study showed that children under 5 living in sub-Saharan Africa were 54% less likely to develop malaria if they had been given a single, large dose of vitamin A.
The finding, published in eLife, indicates that vitamin A may be able to protect children from the malaria parasite, especially if administered during the wet season, when malaria-infected mosquitos are most prevalent.
“Now, we need to test vitamin A in a randomized, controlled, clinical trial to better understand whether this could really be an effective way to prevent this disease,” said study author Maria-Graciela Hollm-Delgado, PhD, of the Johns Hopkins Bloomberg School of Public Health in Baltimore, Maryland.
Dr Hollm-Delgado and her colleagues analyzed national survey data from 4 sub-Saharan countries—Burkina Faso, Mozambique, Rwanda, and Senegal—on children between the ages of 6 months and 59 months.
The goal was to determine the risk of Plasmodium parasitemia (n=8390) and Plasmodium falciparum-specific antigenemia (n=6121) following vitamin A supplementation and vaccinations.
The researchers found the measles and polio vaccines were not associated with malaria. And Bacille Calmette Guerin vaccination was associated with an increased risk of antigenemia (relative risk [RR]=4.06) but not parasitemia.
Only vitamin A was protective against malaria. Children who received vitamin A were less likely to present with parasitemia (RR=0.46) and antigenemia (RR=0.23).
Vitamin A appeared to be more protective under certain circumstances, including when administered during the rainy season, as well as when given to older children and when more time had passed since supplementation.
The researchers aren’t certain why vitamin A would reduce the rate of malaria infection, but they suspect it is because vitamin A, which is known to boost immunity and improve the ability to fight off infection, may help the body clear out the malaria parasite more quickly.
Only 62% of children in the study had received vitamin A supplementation, even though World Health Organization guidelines recommend that all children in sub-Saharan Africa receive a single, large dose of vitamin A.
Rates were higher for many vaccinations, Dr Hollm-Delgado said, noting that the guidelines for vitamin A aren’t as specific as they are for most vaccinations. ![]()
Team touts a new, improved hydrogel
Louis Heiser & Robert Ackland
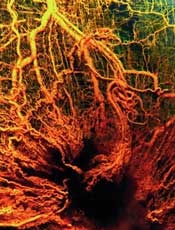
A new hydrogel improves on previous models by enabling the generation of more mature blood vessels, according to research published in ACS Nano.
The hydrogel also overcomes several other issues that have kept previous hydrogels from reaching their potential to treat injuries and forming new vasculature to treat heart attack, stroke, and ischemic tissue diseases.
Like earlier versions, the new hydrogel can be injected in liquid form and turns into a nanofiber-infused gel at the site of the injury. The difference with this hydrogel, according to researchers, is the quality of the blood vessels that are formed.
This hydrogel is made of self-assembling synthetic peptides that form nanofiber scaffolds. And the peptides incorporate a mimic of vascular endothelial growth factor, a signal protein that promotes angiogenesis.
Furthermore, the hydrogel can be easily delivered by syringe, is quickly infiltrated by hematopoietic and mesenchymal cells, and quickly forms a mature vascular network.
“In a lot of the published literature, you see rings that only have the endothelial cell lining, and that indicates a very immature blood vessel,” said study author Jeffrey Hartgerink, PhD, of Rice University in Houston, Texas.
“These types of vessels usually don’t persist and disappear shortly after they show up. In ours, you see that same endothelial cell layer, but surrounding it is a smooth muscle cell layer that indicates a much more mature vessel that’s likely to persist.”
Furthermore, the scaffolds the hydrogel forms show no signs of fibrous encapsulation. After 3 weeks, they are resorbed into the native tissue.
In previous studies, implanted synthetic materials tended to become encapsulated by fibrous barriers that kept cells and blood vessels from infiltrating the scaffold, Dr Hartgerink said.
“That is an extremely common problem in synthetic materials put into the body,” he explained. “Some avoid this problem, but if the body doesn’t like a material and isn’t able to destroy it, the solution is to wall it off.”
“As soon as that happens, the flow of nutrients across that barrier decreases to almost nothing. So the fact that we’ve developed syringe-directed delivery of a material that doesn’t develop fibrous encapsulation is really important.”
Other negative characteristics of earlier hydrogels—unwanted immune responses, surface degradation preceding their integration into biological systems, and the release of artificial degradation byproducts—have been eliminated as well, Dr Hartgerink said.
“There are a lot of features about this hydrogel that come together to make it a unique system,” he added. “If you look through the literature at what other people have done, each concept that is involved in our system probably exists somewhere already. The difference is that we have all these features in one place working together.” ![]()

Louis Heiser & Robert Ackland
A new hydrogel improves on previous models by enabling the generation of more mature blood vessels, according to research published in ACS Nano.
The hydrogel also overcomes several other issues that have kept previous hydrogels from reaching their potential to treat injuries and forming new vasculature to treat heart attack, stroke, and ischemic tissue diseases.
Like earlier versions, the new hydrogel can be injected in liquid form and turns into a nanofiber-infused gel at the site of the injury. The difference with this hydrogel, according to researchers, is the quality of the blood vessels that are formed.
This hydrogel is made of self-assembling synthetic peptides that form nanofiber scaffolds. And the peptides incorporate a mimic of vascular endothelial growth factor, a signal protein that promotes angiogenesis.
Furthermore, the hydrogel can be easily delivered by syringe, is quickly infiltrated by hematopoietic and mesenchymal cells, and quickly forms a mature vascular network.
“In a lot of the published literature, you see rings that only have the endothelial cell lining, and that indicates a very immature blood vessel,” said study author Jeffrey Hartgerink, PhD, of Rice University in Houston, Texas.
“These types of vessels usually don’t persist and disappear shortly after they show up. In ours, you see that same endothelial cell layer, but surrounding it is a smooth muscle cell layer that indicates a much more mature vessel that’s likely to persist.”
Furthermore, the scaffolds the hydrogel forms show no signs of fibrous encapsulation. After 3 weeks, they are resorbed into the native tissue.
In previous studies, implanted synthetic materials tended to become encapsulated by fibrous barriers that kept cells and blood vessels from infiltrating the scaffold, Dr Hartgerink said.
“That is an extremely common problem in synthetic materials put into the body,” he explained. “Some avoid this problem, but if the body doesn’t like a material and isn’t able to destroy it, the solution is to wall it off.”
“As soon as that happens, the flow of nutrients across that barrier decreases to almost nothing. So the fact that we’ve developed syringe-directed delivery of a material that doesn’t develop fibrous encapsulation is really important.”
Other negative characteristics of earlier hydrogels—unwanted immune responses, surface degradation preceding their integration into biological systems, and the release of artificial degradation byproducts—have been eliminated as well, Dr Hartgerink said.
“There are a lot of features about this hydrogel that come together to make it a unique system,” he added. “If you look through the literature at what other people have done, each concept that is involved in our system probably exists somewhere already. The difference is that we have all these features in one place working together.” ![]()

Louis Heiser & Robert Ackland
A new hydrogel improves on previous models by enabling the generation of more mature blood vessels, according to research published in ACS Nano.
The hydrogel also overcomes several other issues that have kept previous hydrogels from reaching their potential to treat injuries and forming new vasculature to treat heart attack, stroke, and ischemic tissue diseases.
Like earlier versions, the new hydrogel can be injected in liquid form and turns into a nanofiber-infused gel at the site of the injury. The difference with this hydrogel, according to researchers, is the quality of the blood vessels that are formed.
This hydrogel is made of self-assembling synthetic peptides that form nanofiber scaffolds. And the peptides incorporate a mimic of vascular endothelial growth factor, a signal protein that promotes angiogenesis.
Furthermore, the hydrogel can be easily delivered by syringe, is quickly infiltrated by hematopoietic and mesenchymal cells, and quickly forms a mature vascular network.
“In a lot of the published literature, you see rings that only have the endothelial cell lining, and that indicates a very immature blood vessel,” said study author Jeffrey Hartgerink, PhD, of Rice University in Houston, Texas.
“These types of vessels usually don’t persist and disappear shortly after they show up. In ours, you see that same endothelial cell layer, but surrounding it is a smooth muscle cell layer that indicates a much more mature vessel that’s likely to persist.”
Furthermore, the scaffolds the hydrogel forms show no signs of fibrous encapsulation. After 3 weeks, they are resorbed into the native tissue.
In previous studies, implanted synthetic materials tended to become encapsulated by fibrous barriers that kept cells and blood vessels from infiltrating the scaffold, Dr Hartgerink said.
“That is an extremely common problem in synthetic materials put into the body,” he explained. “Some avoid this problem, but if the body doesn’t like a material and isn’t able to destroy it, the solution is to wall it off.”
“As soon as that happens, the flow of nutrients across that barrier decreases to almost nothing. So the fact that we’ve developed syringe-directed delivery of a material that doesn’t develop fibrous encapsulation is really important.”
Other negative characteristics of earlier hydrogels—unwanted immune responses, surface degradation preceding their integration into biological systems, and the release of artificial degradation byproducts—have been eliminated as well, Dr Hartgerink said.
“There are a lot of features about this hydrogel that come together to make it a unique system,” he added. “If you look through the literature at what other people have done, each concept that is involved in our system probably exists somewhere already. The difference is that we have all these features in one place working together.” ![]()
Group uncovers structure of receptor-chemokine complex
in complex with a chemokine
(purple surface)
Credit: Katya Kadyshevskaya
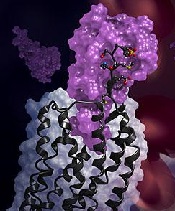
Researchers have reported the first crystal structure of the cellular receptor CXCR4 bound to the viral chemokine antagonist vMIP-II.
The structure, published in Science, answers longstanding questions about a molecular interaction that plays an important role in human development, immune responses, cancer spread, and HIV infections.
“This new information could ultimately aid the development of better small molecular inhibitors of CXCR4-chemokine interactions—inhibitors that have the potential to block cancer metastasis or viral infections,” said study author Tracy M. Handel, PhD, of the University of California, San Diego.
Dr Handel and her colleagues knew that CXCR4 binds chemokines to transmit messages to the inside of the cell. This signal relay helps cells migrate normally during development and inflammation.
But CXCR4 signaling can also play a role in abnormal cell migration, such as when cancer cells metastasize. And CXCR4 is infamous for another reason: HIV uses it to bind and infect human immune cells.
Despite its far-reaching consequences, researchers have long lacked data to show how exactly the CXCR4-chemokine interaction occurs, or even how many CXCR4 receptors a single chemokine molecule might simultaneously engage.
This is because membrane receptors like CXCR4 are exceptionally challenging structural targets. And the difficulty dramatically increases when studying such receptors in complexes with the proteins they bind.
To overcome these experimental challenges, Dr Handel’s team used a novel approach. They combined computational modeling and a technique known as disulfide trapping to stabilize the complex.
Once it was stabilized, the researchers were able to use X-ray crystallography to determine the CXCR4-chemokine complex’s 3D atomic structure.
This is the first time a receptor like CXCR4 has been crystallized with a protein binding partner, and the results revealed several new insights. First, the new crystal structure shows that one chemokine binds to just one receptor.
Additionally, the structure reveals that the contacts between the receptor and its binding partner are more extensive than previously thought. It is one very large, contiguous surface of interaction rather than two separate binding sites.
“The plasticity of the CXCR4 receptor—its ability to bind many unrelated small molecules, peptides, and proteins—is remarkable,” said Irina Kufareva, PhD, also of UC San Diego.
“Our understanding of this plasticity may impact the design of therapeutics with better inhibition and safety profiles.”
“With more than 800 members, 7-transmembrane receptors like CXCR4 are the largest protein family in the human genome,” added Raymond Stevens, PhD, of the Bridge Institute at the University of Southern California in Los Angeles. “Each new structure opens up so many doors to understanding different aspects of human biology, and this time it is about chemokine signaling.” ![]()

in complex with a chemokine
(purple surface)
Credit: Katya Kadyshevskaya
Researchers have reported the first crystal structure of the cellular receptor CXCR4 bound to the viral chemokine antagonist vMIP-II.
The structure, published in Science, answers longstanding questions about a molecular interaction that plays an important role in human development, immune responses, cancer spread, and HIV infections.
“This new information could ultimately aid the development of better small molecular inhibitors of CXCR4-chemokine interactions—inhibitors that have the potential to block cancer metastasis or viral infections,” said study author Tracy M. Handel, PhD, of the University of California, San Diego.
Dr Handel and her colleagues knew that CXCR4 binds chemokines to transmit messages to the inside of the cell. This signal relay helps cells migrate normally during development and inflammation.
But CXCR4 signaling can also play a role in abnormal cell migration, such as when cancer cells metastasize. And CXCR4 is infamous for another reason: HIV uses it to bind and infect human immune cells.
Despite its far-reaching consequences, researchers have long lacked data to show how exactly the CXCR4-chemokine interaction occurs, or even how many CXCR4 receptors a single chemokine molecule might simultaneously engage.
This is because membrane receptors like CXCR4 are exceptionally challenging structural targets. And the difficulty dramatically increases when studying such receptors in complexes with the proteins they bind.
To overcome these experimental challenges, Dr Handel’s team used a novel approach. They combined computational modeling and a technique known as disulfide trapping to stabilize the complex.
Once it was stabilized, the researchers were able to use X-ray crystallography to determine the CXCR4-chemokine complex’s 3D atomic structure.
This is the first time a receptor like CXCR4 has been crystallized with a protein binding partner, and the results revealed several new insights. First, the new crystal structure shows that one chemokine binds to just one receptor.
Additionally, the structure reveals that the contacts between the receptor and its binding partner are more extensive than previously thought. It is one very large, contiguous surface of interaction rather than two separate binding sites.
“The plasticity of the CXCR4 receptor—its ability to bind many unrelated small molecules, peptides, and proteins—is remarkable,” said Irina Kufareva, PhD, also of UC San Diego.
“Our understanding of this plasticity may impact the design of therapeutics with better inhibition and safety profiles.”
“With more than 800 members, 7-transmembrane receptors like CXCR4 are the largest protein family in the human genome,” added Raymond Stevens, PhD, of the Bridge Institute at the University of Southern California in Los Angeles. “Each new structure opens up so many doors to understanding different aspects of human biology, and this time it is about chemokine signaling.” ![]()

in complex with a chemokine
(purple surface)
Credit: Katya Kadyshevskaya
Researchers have reported the first crystal structure of the cellular receptor CXCR4 bound to the viral chemokine antagonist vMIP-II.
The structure, published in Science, answers longstanding questions about a molecular interaction that plays an important role in human development, immune responses, cancer spread, and HIV infections.
“This new information could ultimately aid the development of better small molecular inhibitors of CXCR4-chemokine interactions—inhibitors that have the potential to block cancer metastasis or viral infections,” said study author Tracy M. Handel, PhD, of the University of California, San Diego.
Dr Handel and her colleagues knew that CXCR4 binds chemokines to transmit messages to the inside of the cell. This signal relay helps cells migrate normally during development and inflammation.
But CXCR4 signaling can also play a role in abnormal cell migration, such as when cancer cells metastasize. And CXCR4 is infamous for another reason: HIV uses it to bind and infect human immune cells.
Despite its far-reaching consequences, researchers have long lacked data to show how exactly the CXCR4-chemokine interaction occurs, or even how many CXCR4 receptors a single chemokine molecule might simultaneously engage.
This is because membrane receptors like CXCR4 are exceptionally challenging structural targets. And the difficulty dramatically increases when studying such receptors in complexes with the proteins they bind.
To overcome these experimental challenges, Dr Handel’s team used a novel approach. They combined computational modeling and a technique known as disulfide trapping to stabilize the complex.
Once it was stabilized, the researchers were able to use X-ray crystallography to determine the CXCR4-chemokine complex’s 3D atomic structure.
This is the first time a receptor like CXCR4 has been crystallized with a protein binding partner, and the results revealed several new insights. First, the new crystal structure shows that one chemokine binds to just one receptor.
Additionally, the structure reveals that the contacts between the receptor and its binding partner are more extensive than previously thought. It is one very large, contiguous surface of interaction rather than two separate binding sites.
“The plasticity of the CXCR4 receptor—its ability to bind many unrelated small molecules, peptides, and proteins—is remarkable,” said Irina Kufareva, PhD, also of UC San Diego.
“Our understanding of this plasticity may impact the design of therapeutics with better inhibition and safety profiles.”
“With more than 800 members, 7-transmembrane receptors like CXCR4 are the largest protein family in the human genome,” added Raymond Stevens, PhD, of the Bridge Institute at the University of Southern California in Los Angeles. “Each new structure opens up so many doors to understanding different aspects of human biology, and this time it is about chemokine signaling.” ![]()
Cell imaging gets colorful

Credit: Rhoda Baer
The detection and imaging of protein-protein interactions in live cells just got a lot more colorful, researchers have reported in Nature Methods.
The team created a technique that converts biochemical processes into color changes that are easily visualized.
The group said this provides a new tool scientists can use to answer questions about fundamental mechanisms in cell biology, aid the discovery of novel therapeutics, and more.
Robert E. Campbell, PhD, of the University of Alberta in Edmonton, Alberta, Canada, and his colleagues conducted this research.
They developed the technique, dubbed FPX, that employs genetically encoded fluorescent proteins to image dynamic biochemical events in live cells and tissues. The FPX method converts a change in protein-protein interactions into a dramatic green to red (or vice versa) color change that is immediately visible.
“Strategies for converting fluorescent proteins into active biosensors of intracellular biochemistry are few in number and technically challenging,” Dr Campbell said. “With this development, we can immediately image activity happening at the cellular level, offering an alternative to existing methods for detecting and imaging of protein-protein interactions in live cells.”
The FPX method is based on green and red dimerization-dependent fluorescent proteins (ddFPs) that Dr Campbell and his colleagues first reported in 2012.
Yidan Ding, PhD, a research assistant at the University of Alberta and the primary contributor to this work, found she could combine the use of both green and red ddFPs in single cells, such that the proteins could be green or red, but not both, at the same time.
By introducing modified versions of the proteins into live cells, and taking advantage of the fact that green and red fluorescence are mutually exclusive, Dr Ding was able to construct a wide variety of biosensors that underwent dramatic changes in fluorescence in response to biochemical processes of interest.
By adding this new dimension to fluorescent proteins and engineering them to be biosensors that change their color in response to specific biological events, Drs Ding and Campbell and their colleagues have provided a tool for researchers to immediately pinpoint a major change at the cellular level.
This minimizes the need for extensive biosensor optimization and provides a versatile new approach to building the next generation of biosensors.
“This allows for a wide scope of applications,” Dr Campbell said. “It will be immediately relevant to many areas of fundamental cell biology research and practical applications such as drug discovery. Ultimately, it will help researchers achieve breakthroughs in a wide variety of areas in the life sciences, such as neuroscience, diabetes, and cancer.”
Dr Campbell has a patent pending on the technology. ![]()

Credit: Rhoda Baer
The detection and imaging of protein-protein interactions in live cells just got a lot more colorful, researchers have reported in Nature Methods.
The team created a technique that converts biochemical processes into color changes that are easily visualized.
The group said this provides a new tool scientists can use to answer questions about fundamental mechanisms in cell biology, aid the discovery of novel therapeutics, and more.
Robert E. Campbell, PhD, of the University of Alberta in Edmonton, Alberta, Canada, and his colleagues conducted this research.
They developed the technique, dubbed FPX, that employs genetically encoded fluorescent proteins to image dynamic biochemical events in live cells and tissues. The FPX method converts a change in protein-protein interactions into a dramatic green to red (or vice versa) color change that is immediately visible.
“Strategies for converting fluorescent proteins into active biosensors of intracellular biochemistry are few in number and technically challenging,” Dr Campbell said. “With this development, we can immediately image activity happening at the cellular level, offering an alternative to existing methods for detecting and imaging of protein-protein interactions in live cells.”
The FPX method is based on green and red dimerization-dependent fluorescent proteins (ddFPs) that Dr Campbell and his colleagues first reported in 2012.
Yidan Ding, PhD, a research assistant at the University of Alberta and the primary contributor to this work, found she could combine the use of both green and red ddFPs in single cells, such that the proteins could be green or red, but not both, at the same time.
By introducing modified versions of the proteins into live cells, and taking advantage of the fact that green and red fluorescence are mutually exclusive, Dr Ding was able to construct a wide variety of biosensors that underwent dramatic changes in fluorescence in response to biochemical processes of interest.
By adding this new dimension to fluorescent proteins and engineering them to be biosensors that change their color in response to specific biological events, Drs Ding and Campbell and their colleagues have provided a tool for researchers to immediately pinpoint a major change at the cellular level.
This minimizes the need for extensive biosensor optimization and provides a versatile new approach to building the next generation of biosensors.
“This allows for a wide scope of applications,” Dr Campbell said. “It will be immediately relevant to many areas of fundamental cell biology research and practical applications such as drug discovery. Ultimately, it will help researchers achieve breakthroughs in a wide variety of areas in the life sciences, such as neuroscience, diabetes, and cancer.”
Dr Campbell has a patent pending on the technology. ![]()

Credit: Rhoda Baer
The detection and imaging of protein-protein interactions in live cells just got a lot more colorful, researchers have reported in Nature Methods.
The team created a technique that converts biochemical processes into color changes that are easily visualized.
The group said this provides a new tool scientists can use to answer questions about fundamental mechanisms in cell biology, aid the discovery of novel therapeutics, and more.
Robert E. Campbell, PhD, of the University of Alberta in Edmonton, Alberta, Canada, and his colleagues conducted this research.
They developed the technique, dubbed FPX, that employs genetically encoded fluorescent proteins to image dynamic biochemical events in live cells and tissues. The FPX method converts a change in protein-protein interactions into a dramatic green to red (or vice versa) color change that is immediately visible.
“Strategies for converting fluorescent proteins into active biosensors of intracellular biochemistry are few in number and technically challenging,” Dr Campbell said. “With this development, we can immediately image activity happening at the cellular level, offering an alternative to existing methods for detecting and imaging of protein-protein interactions in live cells.”
The FPX method is based on green and red dimerization-dependent fluorescent proteins (ddFPs) that Dr Campbell and his colleagues first reported in 2012.
Yidan Ding, PhD, a research assistant at the University of Alberta and the primary contributor to this work, found she could combine the use of both green and red ddFPs in single cells, such that the proteins could be green or red, but not both, at the same time.
By introducing modified versions of the proteins into live cells, and taking advantage of the fact that green and red fluorescence are mutually exclusive, Dr Ding was able to construct a wide variety of biosensors that underwent dramatic changes in fluorescence in response to biochemical processes of interest.
By adding this new dimension to fluorescent proteins and engineering them to be biosensors that change their color in response to specific biological events, Drs Ding and Campbell and their colleagues have provided a tool for researchers to immediately pinpoint a major change at the cellular level.
This minimizes the need for extensive biosensor optimization and provides a versatile new approach to building the next generation of biosensors.
“This allows for a wide scope of applications,” Dr Campbell said. “It will be immediately relevant to many areas of fundamental cell biology research and practical applications such as drug discovery. Ultimately, it will help researchers achieve breakthroughs in a wide variety of areas in the life sciences, such as neuroscience, diabetes, and cancer.”
Dr Campbell has a patent pending on the technology. ![]()
Gene variation explains drug toxicity in ALL

Credit: Peter Barta
Inherited variations in the NUDT15 gene can reduce tolerance of the drug mercaptopurine in children with acute lymphoblastic leukemia (ALL), according to research published in the Journal of Clinical Oncology.
The study showed that patients who inherited one or two copies of the newly identified variation in the NUDT15 gene were extremely sensitive to mercaptopurine.
The patients required dose reductions of as much as 92%.
And when mercaptopurine was given at standard doses, the patients developed side effects that caused treatment delays.
These findings should aid efforts to improve the identification and treatment of patients who need reduced doses of mercaptopurine, according to researchers.
“Mercaptopurine intolerance has been suspected to be a problem for young ALL patients of East Asian ancestry,” said study author Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee.
“Even at very low doses, the patients often develop toxicity that delays treatment. But, until now, the genetic basis of the problem was unknown.”
With that in mind, Dr Yang and his colleagues performed a genome-wide association study in children with ALL who received mercaptopurine treatment regimens. The discovery and replication cohorts included 657 and 371 children, respectively, from two prospective trials.
The research revealed that patients of East Asian and Hispanic background were more likely to inherit the NUDT15 variant than patients from other racial and ethnic groups.
Among patients of East Asian ancestry, 9.8% carried at least one copy of the NUDT15 variant, compared to 3.9% of Hispanic patients. (East Asia includes China, Japan, and Korea.)
The NUDT15 variant was rarer among patients of European or African ancestry.
This study also confirmed previous research that showed variations in another gene, TPMT, are associated with an increased risk of mercaptopurine toxicity.
TPMT carries instructions for assembling an enzyme of the same name that inactivates mercaptopurine and related drugs. The TPMT variants are less able to inactivate the drug, which can lead to a dangerous build-up of mercaptopurine and suppression of the immune system.
The researchers suspect the NUDT15 variant acts in a similar fashion.
Regardless, the team found that 100% of children who were homozygous for either TPMT or NUDT15 variants or heterozygous for both required at least a 50% reduction in mercaptopurine dose. Only 7.7% of the other patients required similar reductions.
“The results of this study confirm that TPMT genetic variation is one of the most critical determinants of mercaptopurine tolerance, particularly in non-East Asian populations,” said senior study author Mary Relling, PharmD, of St Jude.
“But we also found that TPMT variants do not completely explain mercaptopurine intolerance, particularly in patients of East Asian ancestry. Other factors, both genetic and non-genetic, are still to be discovered to improve the safety and effectiveness of mercaptopurine treatment for children with ALL.” ![]()

Credit: Peter Barta
Inherited variations in the NUDT15 gene can reduce tolerance of the drug mercaptopurine in children with acute lymphoblastic leukemia (ALL), according to research published in the Journal of Clinical Oncology.
The study showed that patients who inherited one or two copies of the newly identified variation in the NUDT15 gene were extremely sensitive to mercaptopurine.
The patients required dose reductions of as much as 92%.
And when mercaptopurine was given at standard doses, the patients developed side effects that caused treatment delays.
These findings should aid efforts to improve the identification and treatment of patients who need reduced doses of mercaptopurine, according to researchers.
“Mercaptopurine intolerance has been suspected to be a problem for young ALL patients of East Asian ancestry,” said study author Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee.
“Even at very low doses, the patients often develop toxicity that delays treatment. But, until now, the genetic basis of the problem was unknown.”
With that in mind, Dr Yang and his colleagues performed a genome-wide association study in children with ALL who received mercaptopurine treatment regimens. The discovery and replication cohorts included 657 and 371 children, respectively, from two prospective trials.
The research revealed that patients of East Asian and Hispanic background were more likely to inherit the NUDT15 variant than patients from other racial and ethnic groups.
Among patients of East Asian ancestry, 9.8% carried at least one copy of the NUDT15 variant, compared to 3.9% of Hispanic patients. (East Asia includes China, Japan, and Korea.)
The NUDT15 variant was rarer among patients of European or African ancestry.
This study also confirmed previous research that showed variations in another gene, TPMT, are associated with an increased risk of mercaptopurine toxicity.
TPMT carries instructions for assembling an enzyme of the same name that inactivates mercaptopurine and related drugs. The TPMT variants are less able to inactivate the drug, which can lead to a dangerous build-up of mercaptopurine and suppression of the immune system.
The researchers suspect the NUDT15 variant acts in a similar fashion.
Regardless, the team found that 100% of children who were homozygous for either TPMT or NUDT15 variants or heterozygous for both required at least a 50% reduction in mercaptopurine dose. Only 7.7% of the other patients required similar reductions.
“The results of this study confirm that TPMT genetic variation is one of the most critical determinants of mercaptopurine tolerance, particularly in non-East Asian populations,” said senior study author Mary Relling, PharmD, of St Jude.
“But we also found that TPMT variants do not completely explain mercaptopurine intolerance, particularly in patients of East Asian ancestry. Other factors, both genetic and non-genetic, are still to be discovered to improve the safety and effectiveness of mercaptopurine treatment for children with ALL.” ![]()

Credit: Peter Barta
Inherited variations in the NUDT15 gene can reduce tolerance of the drug mercaptopurine in children with acute lymphoblastic leukemia (ALL), according to research published in the Journal of Clinical Oncology.
The study showed that patients who inherited one or two copies of the newly identified variation in the NUDT15 gene were extremely sensitive to mercaptopurine.
The patients required dose reductions of as much as 92%.
And when mercaptopurine was given at standard doses, the patients developed side effects that caused treatment delays.
These findings should aid efforts to improve the identification and treatment of patients who need reduced doses of mercaptopurine, according to researchers.
“Mercaptopurine intolerance has been suspected to be a problem for young ALL patients of East Asian ancestry,” said study author Jun J. Yang, PhD, of St Jude Children’s Research Hospital in Memphis, Tennessee.
“Even at very low doses, the patients often develop toxicity that delays treatment. But, until now, the genetic basis of the problem was unknown.”
With that in mind, Dr Yang and his colleagues performed a genome-wide association study in children with ALL who received mercaptopurine treatment regimens. The discovery and replication cohorts included 657 and 371 children, respectively, from two prospective trials.
The research revealed that patients of East Asian and Hispanic background were more likely to inherit the NUDT15 variant than patients from other racial and ethnic groups.
Among patients of East Asian ancestry, 9.8% carried at least one copy of the NUDT15 variant, compared to 3.9% of Hispanic patients. (East Asia includes China, Japan, and Korea.)
The NUDT15 variant was rarer among patients of European or African ancestry.
This study also confirmed previous research that showed variations in another gene, TPMT, are associated with an increased risk of mercaptopurine toxicity.
TPMT carries instructions for assembling an enzyme of the same name that inactivates mercaptopurine and related drugs. The TPMT variants are less able to inactivate the drug, which can lead to a dangerous build-up of mercaptopurine and suppression of the immune system.
The researchers suspect the NUDT15 variant acts in a similar fashion.
Regardless, the team found that 100% of children who were homozygous for either TPMT or NUDT15 variants or heterozygous for both required at least a 50% reduction in mercaptopurine dose. Only 7.7% of the other patients required similar reductions.
“The results of this study confirm that TPMT genetic variation is one of the most critical determinants of mercaptopurine tolerance, particularly in non-East Asian populations,” said senior study author Mary Relling, PharmD, of St Jude.
“But we also found that TPMT variants do not completely explain mercaptopurine intolerance, particularly in patients of East Asian ancestry. Other factors, both genetic and non-genetic, are still to be discovered to improve the safety and effectiveness of mercaptopurine treatment for children with ALL.”
Why EBV-positive lymphomas resist IFN therapy

Credit: Ed Uthman
New research has revealed how Epstein Barr virus (EBV) and other herpes viruses outwit the body’s immune response.
It seems these viruses carry microRNAs (miRNAs) that block the interferon (IFN) response—when immune cells release IFN to prevent viral replication, which often kills or slows the growth of infected host cells.
This appears to explain why patients with EBV-positive lymphomas and other viral cancers may resist treatment with IFN.
Jennifer Cox, a graduate student at the University of Texas Austin, and her colleagues recounted these findings in PNAS.
The team noted that many viruses, including EBV, carry miRNAs they use to hijack natural processes in a host’s cells during an infection.
Viral miRNAs are known to prevent host cell death, promote host cell growth, and dampen the host cell’s viral defenses. However, scientists don’t yet know which viral miRNAs perform which functions.
To gain some insight, Cox and her colleagues screened a library of more than 70 human viral miRNAs. This revealed 3 unrelated miRNAs from distantly related herpes viruses that significantly inhibited IFN signaling.
The 5’ and 3’ derivatives from EBV-encoded miR-BART-18 precursor miRNA and the orthologous precursor miRNA from Rhesus lymphocryptovirus all reduced expression of the cyclic AMP-responsive element-binding protein (CBP), which, as part of the p300-CBP complex, mediates IFN signaling.
When the researchers restored miR-BART-18 to cells infected with an EBV miRNA mutant, they observed a cellular growth advantage upon IFN treatment. And they found that miRNAs from other herpes viruses were able to complement this activity.
The team also showed that blocking miR-BART-18 function in an EBV-positive tumor cell line rendered cells more susceptible to IFN-mediated effects.
“[These findings] could explain the variability seen in the success of previous interferon-based cancer treatments,” Cox said. “While this work does not immediately identify new drugs, the fact that such different tumor viruses have converged on the same strategy makes this an exciting pursuit for future therapies against viral cancers.”

Credit: Ed Uthman
New research has revealed how Epstein Barr virus (EBV) and other herpes viruses outwit the body’s immune response.
It seems these viruses carry microRNAs (miRNAs) that block the interferon (IFN) response—when immune cells release IFN to prevent viral replication, which often kills or slows the growth of infected host cells.
This appears to explain why patients with EBV-positive lymphomas and other viral cancers may resist treatment with IFN.
Jennifer Cox, a graduate student at the University of Texas Austin, and her colleagues recounted these findings in PNAS.
The team noted that many viruses, including EBV, carry miRNAs they use to hijack natural processes in a host’s cells during an infection.
Viral miRNAs are known to prevent host cell death, promote host cell growth, and dampen the host cell’s viral defenses. However, scientists don’t yet know which viral miRNAs perform which functions.
To gain some insight, Cox and her colleagues screened a library of more than 70 human viral miRNAs. This revealed 3 unrelated miRNAs from distantly related herpes viruses that significantly inhibited IFN signaling.
The 5’ and 3’ derivatives from EBV-encoded miR-BART-18 precursor miRNA and the orthologous precursor miRNA from Rhesus lymphocryptovirus all reduced expression of the cyclic AMP-responsive element-binding protein (CBP), which, as part of the p300-CBP complex, mediates IFN signaling.
When the researchers restored miR-BART-18 to cells infected with an EBV miRNA mutant, they observed a cellular growth advantage upon IFN treatment. And they found that miRNAs from other herpes viruses were able to complement this activity.
The team also showed that blocking miR-BART-18 function in an EBV-positive tumor cell line rendered cells more susceptible to IFN-mediated effects.
“[These findings] could explain the variability seen in the success of previous interferon-based cancer treatments,” Cox said. “While this work does not immediately identify new drugs, the fact that such different tumor viruses have converged on the same strategy makes this an exciting pursuit for future therapies against viral cancers.”

Credit: Ed Uthman
New research has revealed how Epstein Barr virus (EBV) and other herpes viruses outwit the body’s immune response.
It seems these viruses carry microRNAs (miRNAs) that block the interferon (IFN) response—when immune cells release IFN to prevent viral replication, which often kills or slows the growth of infected host cells.
This appears to explain why patients with EBV-positive lymphomas and other viral cancers may resist treatment with IFN.
Jennifer Cox, a graduate student at the University of Texas Austin, and her colleagues recounted these findings in PNAS.
The team noted that many viruses, including EBV, carry miRNAs they use to hijack natural processes in a host’s cells during an infection.
Viral miRNAs are known to prevent host cell death, promote host cell growth, and dampen the host cell’s viral defenses. However, scientists don’t yet know which viral miRNAs perform which functions.
To gain some insight, Cox and her colleagues screened a library of more than 70 human viral miRNAs. This revealed 3 unrelated miRNAs from distantly related herpes viruses that significantly inhibited IFN signaling.
The 5’ and 3’ derivatives from EBV-encoded miR-BART-18 precursor miRNA and the orthologous precursor miRNA from Rhesus lymphocryptovirus all reduced expression of the cyclic AMP-responsive element-binding protein (CBP), which, as part of the p300-CBP complex, mediates IFN signaling.
When the researchers restored miR-BART-18 to cells infected with an EBV miRNA mutant, they observed a cellular growth advantage upon IFN treatment. And they found that miRNAs from other herpes viruses were able to complement this activity.
The team also showed that blocking miR-BART-18 function in an EBV-positive tumor cell line rendered cells more susceptible to IFN-mediated effects.
“[These findings] could explain the variability seen in the success of previous interferon-based cancer treatments,” Cox said. “While this work does not immediately identify new drugs, the fact that such different tumor viruses have converged on the same strategy makes this an exciting pursuit for future therapies against viral cancers.”
Drug on the fast track to treat HAE

within pancreatic tissue
Credit: Louisa Howard
The US Food and Drug Administration (FDA) has granted fast track designation for BCX4161, an oral inhibitor of plasma kallikrein intended to treat hereditary angioedema (HAE).
Uncontrolled activation of plasma kallikrein, caused by deficiency of its physiological inhibitor (C1 inhibitor) in HAE, results in acute systemic edema.
By inhibiting plasma kallikrein, BCX4161 suppresses the production of bradykinin, the mediator of acute swelling attacks in HAE patients.
HAE is a severely debilitating and potentially fatal condition that occurs in approximately 1 in 50,000 people. Symptoms include recurrent episodes of edema in various locations, as well as bouts of excruciating abdominal pain, nausea, and vomiting that are caused by swelling in the intestinal walls.
HAE patients have a defect in the gene that controls C1 inhibitor, and this results in the production of inadequate or non-functioning C1 inhibitor protein.
Normal C1 inhibitor helps regulate the biochemical interactions of blood-based systems involved in disease-fighting, inflammatory response, and coagulation.
Because defective C1 inhibitor does not adequately perform its regulatory function, a biochemical imbalance can occur and produce unwanted peptides that induce the capillaries to release fluids into surrounding tissue, causing edema.
BCX4161 trials
In May 2014, BioCryst Pharmaceuticals, the company developing BCX4161, announced results from the phase 2a OPuS-1 trial.
OPuS-1 investigators evaluated 400 mg of BCX4161 administered 3 times a day for 28 days in HAE patients with a high angioedema attack frequency (≥ 1 per week), in a randomized, placebo-controlled, 2-period cross-over design.
BCX4161 demonstrated a significant reduction in mean attack rate compared to placebo. The mean attack rate per patient-week was 0.82 on BCX4161 treatment and 1.27 on placebo (P<0.001).
The mean number of attack-free days during each treatment period improved from 19 for placebo to 22 for BCX4161 (P=0.008). Three subjects were attack-free during the BCX4161 period, compared to none during the placebo period.
BCX4161 was generally well-tolerated, BioCryst reported, with an adverse event profile similar to that observed for placebo. There was one serious adverse event reported, an abdominal HAE attack during the placebo period.
In December, the first patient was dosed in the OPuS-2 trial, a double-blind, randomized, placebo- controlled trial conducted in the US and European Union.
Study investigators will evaluate the efficacy and safety of BCX4161 treatment for 12 weeks in patients with HAE. BioCryst expects to report results from OPuS-2 by the end of 2015.
About fast track designation
The FDA’s fast track process is designed to facilitate the development and expedite the review and approval of drugs intended to treat serious or life-threatening conditions that also address unmet medical needs.
A drug that receives fast track designation is usually eligible for more frequent written communication and meetings with the FDA to discuss the drug’s development plan and the collection of appropriate data supporting drug approval.
Priority review and rolling review may be granted if relevant criteria are met. Rolling review allows a drug company to submit completed sections of its new drug application on an ongoing basis, rather than wait until the entire application is complete.

within pancreatic tissue
Credit: Louisa Howard
The US Food and Drug Administration (FDA) has granted fast track designation for BCX4161, an oral inhibitor of plasma kallikrein intended to treat hereditary angioedema (HAE).
Uncontrolled activation of plasma kallikrein, caused by deficiency of its physiological inhibitor (C1 inhibitor) in HAE, results in acute systemic edema.
By inhibiting plasma kallikrein, BCX4161 suppresses the production of bradykinin, the mediator of acute swelling attacks in HAE patients.
HAE is a severely debilitating and potentially fatal condition that occurs in approximately 1 in 50,000 people. Symptoms include recurrent episodes of edema in various locations, as well as bouts of excruciating abdominal pain, nausea, and vomiting that are caused by swelling in the intestinal walls.
HAE patients have a defect in the gene that controls C1 inhibitor, and this results in the production of inadequate or non-functioning C1 inhibitor protein.
Normal C1 inhibitor helps regulate the biochemical interactions of blood-based systems involved in disease-fighting, inflammatory response, and coagulation.
Because defective C1 inhibitor does not adequately perform its regulatory function, a biochemical imbalance can occur and produce unwanted peptides that induce the capillaries to release fluids into surrounding tissue, causing edema.
BCX4161 trials
In May 2014, BioCryst Pharmaceuticals, the company developing BCX4161, announced results from the phase 2a OPuS-1 trial.
OPuS-1 investigators evaluated 400 mg of BCX4161 administered 3 times a day for 28 days in HAE patients with a high angioedema attack frequency (≥ 1 per week), in a randomized, placebo-controlled, 2-period cross-over design.
BCX4161 demonstrated a significant reduction in mean attack rate compared to placebo. The mean attack rate per patient-week was 0.82 on BCX4161 treatment and 1.27 on placebo (P<0.001).
The mean number of attack-free days during each treatment period improved from 19 for placebo to 22 for BCX4161 (P=0.008). Three subjects were attack-free during the BCX4161 period, compared to none during the placebo period.
BCX4161 was generally well-tolerated, BioCryst reported, with an adverse event profile similar to that observed for placebo. There was one serious adverse event reported, an abdominal HAE attack during the placebo period.
In December, the first patient was dosed in the OPuS-2 trial, a double-blind, randomized, placebo- controlled trial conducted in the US and European Union.
Study investigators will evaluate the efficacy and safety of BCX4161 treatment for 12 weeks in patients with HAE. BioCryst expects to report results from OPuS-2 by the end of 2015.
About fast track designation
The FDA’s fast track process is designed to facilitate the development and expedite the review and approval of drugs intended to treat serious or life-threatening conditions that also address unmet medical needs.
A drug that receives fast track designation is usually eligible for more frequent written communication and meetings with the FDA to discuss the drug’s development plan and the collection of appropriate data supporting drug approval.
Priority review and rolling review may be granted if relevant criteria are met. Rolling review allows a drug company to submit completed sections of its new drug application on an ongoing basis, rather than wait until the entire application is complete.

within pancreatic tissue
Credit: Louisa Howard
The US Food and Drug Administration (FDA) has granted fast track designation for BCX4161, an oral inhibitor of plasma kallikrein intended to treat hereditary angioedema (HAE).
Uncontrolled activation of plasma kallikrein, caused by deficiency of its physiological inhibitor (C1 inhibitor) in HAE, results in acute systemic edema.
By inhibiting plasma kallikrein, BCX4161 suppresses the production of bradykinin, the mediator of acute swelling attacks in HAE patients.
HAE is a severely debilitating and potentially fatal condition that occurs in approximately 1 in 50,000 people. Symptoms include recurrent episodes of edema in various locations, as well as bouts of excruciating abdominal pain, nausea, and vomiting that are caused by swelling in the intestinal walls.
HAE patients have a defect in the gene that controls C1 inhibitor, and this results in the production of inadequate or non-functioning C1 inhibitor protein.
Normal C1 inhibitor helps regulate the biochemical interactions of blood-based systems involved in disease-fighting, inflammatory response, and coagulation.
Because defective C1 inhibitor does not adequately perform its regulatory function, a biochemical imbalance can occur and produce unwanted peptides that induce the capillaries to release fluids into surrounding tissue, causing edema.
BCX4161 trials
In May 2014, BioCryst Pharmaceuticals, the company developing BCX4161, announced results from the phase 2a OPuS-1 trial.
OPuS-1 investigators evaluated 400 mg of BCX4161 administered 3 times a day for 28 days in HAE patients with a high angioedema attack frequency (≥ 1 per week), in a randomized, placebo-controlled, 2-period cross-over design.
BCX4161 demonstrated a significant reduction in mean attack rate compared to placebo. The mean attack rate per patient-week was 0.82 on BCX4161 treatment and 1.27 on placebo (P<0.001).
The mean number of attack-free days during each treatment period improved from 19 for placebo to 22 for BCX4161 (P=0.008). Three subjects were attack-free during the BCX4161 period, compared to none during the placebo period.
BCX4161 was generally well-tolerated, BioCryst reported, with an adverse event profile similar to that observed for placebo. There was one serious adverse event reported, an abdominal HAE attack during the placebo period.
In December, the first patient was dosed in the OPuS-2 trial, a double-blind, randomized, placebo- controlled trial conducted in the US and European Union.
Study investigators will evaluate the efficacy and safety of BCX4161 treatment for 12 weeks in patients with HAE. BioCryst expects to report results from OPuS-2 by the end of 2015.
About fast track designation
The FDA’s fast track process is designed to facilitate the development and expedite the review and approval of drugs intended to treat serious or life-threatening conditions that also address unmet medical needs.
A drug that receives fast track designation is usually eligible for more frequent written communication and meetings with the FDA to discuss the drug’s development plan and the collection of appropriate data supporting drug approval.
Priority review and rolling review may be granted if relevant criteria are met. Rolling review allows a drug company to submit completed sections of its new drug application on an ongoing basis, rather than wait until the entire application is complete.
Why neutrophils have conflicting roles in cancer

Credit: Volker Brinkmann
New research has revealed distinct subpopulations of neutrophils that have conflicting functions when it comes to cancer.
Investigators identified a population of “normal,” high-density neutrophils (HDNs) that have anti-tumor properties and a population of low-density
neutrophils (LDNs) that exhibit pro-tumor activity.
They also found that the ratio of HDNs to LDNs determines whether the neutrophils have a pro- or anti-cancer effect overall.
This suggests we may be able to fight cancers by increasing the proportion of the anti-tumor HDNs while limiting the pro-tumor LDNs, according to Zvika Granot, PhD, of Hebrew University Medical School in Jerusalem, Israel.
“The novel distinction between harmful and beneficial neutrophils opens up new diagnostic and therapeutic opportunities,” Dr Granot said. “We are currently evaluating the effects of boosting the helpful anti-tumor neutrophil population, while limiting the tumor-promoting neutrophil population, on progression of the disease. If successful, this therapeutic strategy may take us closer to developing effective new therapies for cancer.”
Dr Granot and his colleagues described their discovery of LDNs and HDNs in Cell Reports.
The investigators found that LDNs were rare in healthy control subjects, but they accumulated in tumor-bearing mice and in cancer patients. HDNs exhibited cytotoxicity toward cancer cells and retarded tumor growth, but LDNs did not.
LDNs had impaired neutrophil function and immunosuppressive properties. They consisted of immature myeloid-derived suppressor cells and mature cells that were derived from HDNs.
HDNs transformed into LDNs in a TGF-β-dependent fashion, losing their anti-tumor properties and gaining immunosuppressive properties.
In the early stages of cancer development, HDNs prevailed. But as cancer progressed, the LDNs took over, and the overall neutrophil contribution became tumor-promoting.
The investigators noted that the expansion of LDNs was a relatively late event. So although these neutrophils likely contribute to tumor growth and progression, it’s less likely that they contribute to tumor initiation.
Regardless, this research challenges the concept that mature neutrophils are limited in their ability to change. It shows that neutrophils are not a homogeneous population of cells and may consist of multiple subtypes. And it provides a mechanistic explanation for the controversy surrounding neutrophil function in cancer.

Credit: Volker Brinkmann
New research has revealed distinct subpopulations of neutrophils that have conflicting functions when it comes to cancer.
Investigators identified a population of “normal,” high-density neutrophils (HDNs) that have anti-tumor properties and a population of low-density
neutrophils (LDNs) that exhibit pro-tumor activity.
They also found that the ratio of HDNs to LDNs determines whether the neutrophils have a pro- or anti-cancer effect overall.
This suggests we may be able to fight cancers by increasing the proportion of the anti-tumor HDNs while limiting the pro-tumor LDNs, according to Zvika Granot, PhD, of Hebrew University Medical School in Jerusalem, Israel.
“The novel distinction between harmful and beneficial neutrophils opens up new diagnostic and therapeutic opportunities,” Dr Granot said. “We are currently evaluating the effects of boosting the helpful anti-tumor neutrophil population, while limiting the tumor-promoting neutrophil population, on progression of the disease. If successful, this therapeutic strategy may take us closer to developing effective new therapies for cancer.”
Dr Granot and his colleagues described their discovery of LDNs and HDNs in Cell Reports.
The investigators found that LDNs were rare in healthy control subjects, but they accumulated in tumor-bearing mice and in cancer patients. HDNs exhibited cytotoxicity toward cancer cells and retarded tumor growth, but LDNs did not.
LDNs had impaired neutrophil function and immunosuppressive properties. They consisted of immature myeloid-derived suppressor cells and mature cells that were derived from HDNs.
HDNs transformed into LDNs in a TGF-β-dependent fashion, losing their anti-tumor properties and gaining immunosuppressive properties.
In the early stages of cancer development, HDNs prevailed. But as cancer progressed, the LDNs took over, and the overall neutrophil contribution became tumor-promoting.
The investigators noted that the expansion of LDNs was a relatively late event. So although these neutrophils likely contribute to tumor growth and progression, it’s less likely that they contribute to tumor initiation.
Regardless, this research challenges the concept that mature neutrophils are limited in their ability to change. It shows that neutrophils are not a homogeneous population of cells and may consist of multiple subtypes. And it provides a mechanistic explanation for the controversy surrounding neutrophil function in cancer.

Credit: Volker Brinkmann
New research has revealed distinct subpopulations of neutrophils that have conflicting functions when it comes to cancer.
Investigators identified a population of “normal,” high-density neutrophils (HDNs) that have anti-tumor properties and a population of low-density
neutrophils (LDNs) that exhibit pro-tumor activity.
They also found that the ratio of HDNs to LDNs determines whether the neutrophils have a pro- or anti-cancer effect overall.
This suggests we may be able to fight cancers by increasing the proportion of the anti-tumor HDNs while limiting the pro-tumor LDNs, according to Zvika Granot, PhD, of Hebrew University Medical School in Jerusalem, Israel.
“The novel distinction between harmful and beneficial neutrophils opens up new diagnostic and therapeutic opportunities,” Dr Granot said. “We are currently evaluating the effects of boosting the helpful anti-tumor neutrophil population, while limiting the tumor-promoting neutrophil population, on progression of the disease. If successful, this therapeutic strategy may take us closer to developing effective new therapies for cancer.”
Dr Granot and his colleagues described their discovery of LDNs and HDNs in Cell Reports.
The investigators found that LDNs were rare in healthy control subjects, but they accumulated in tumor-bearing mice and in cancer patients. HDNs exhibited cytotoxicity toward cancer cells and retarded tumor growth, but LDNs did not.
LDNs had impaired neutrophil function and immunosuppressive properties. They consisted of immature myeloid-derived suppressor cells and mature cells that were derived from HDNs.
HDNs transformed into LDNs in a TGF-β-dependent fashion, losing their anti-tumor properties and gaining immunosuppressive properties.
In the early stages of cancer development, HDNs prevailed. But as cancer progressed, the LDNs took over, and the overall neutrophil contribution became tumor-promoting.
The investigators noted that the expansion of LDNs was a relatively late event. So although these neutrophils likely contribute to tumor growth and progression, it’s less likely that they contribute to tumor initiation.
Regardless, this research challenges the concept that mature neutrophils are limited in their ability to change. It shows that neutrophils are not a homogeneous population of cells and may consist of multiple subtypes. And it provides a mechanistic explanation for the controversy surrounding neutrophil function in cancer.