User login
A step toward safer, effective malaria treatment

Walter and Eliza Hall Institute
In uncovering how an antibiotic fights malaria, investigators may have enabled the development of safer and more effective antimalarial drugs.
The group has determined how the antibiotic emetine blocks the molecular machinery that produces the proteins required for malaria parasite survival.
Although emetine is known to be effective against malaria, the drug is not used for this purpose because it confers significant side effects.
By gaining new insight into how emetine works, the investigators have forged the way ahead for designing drugs that can provide the efficacy of emetine without the adverse effects.
Wilson Wong, PhD, of the Walter and Eliza Hall Institute in Victoria, Australia, and his colleagues described this research in eLife.
For this work, the team used Cryo-EM, a technique that allows researchers to visualize the structure of protein complexes from cellular material without having to crystallize them. This method revealed how emetine binds to the ribosome of the malaria parasite Plasmodium falciparum.
“The ribosome is responsible for constructing all proteins inside the cell, based on the DNA ‘blueprint,’” Dr Wong said. “Antibiotics such as emetine kill the malaria parasite by binding to its ribosome and preventing the parasite from building the proteins it needs to produce energy, grow, reproduce, and evade the immune system.”
The investigators discovered that emetine interacts with the E-site of the ribosomal small subunit and shares a similar binding site with the antibiotic pactamycin. And emetine delivers its antimalaria effect by blocking mRNA/tRNA translocation.
“Our structure is an exciting discovery, as it gives a clear path forward in developing new drugs to tackle this deadly disease,” Dr Wong said. “We have found features of the parasitic ribosome that are not found in the human form. Drug makers could exploit these features in order to specifically target the production of proteins within the malaria parasite.”
“We are now working with our colleagues from the institute’s ACRF Chemical Biology division to develop new molecules based on emetine and pactamycin. Knowing exactly how these antibiotics work will enable development of new antimalarial drugs that replicate the active component of these antibiotics, while changing the parts that make it toxic to patients.” ![]()

Walter and Eliza Hall Institute
In uncovering how an antibiotic fights malaria, investigators may have enabled the development of safer and more effective antimalarial drugs.
The group has determined how the antibiotic emetine blocks the molecular machinery that produces the proteins required for malaria parasite survival.
Although emetine is known to be effective against malaria, the drug is not used for this purpose because it confers significant side effects.
By gaining new insight into how emetine works, the investigators have forged the way ahead for designing drugs that can provide the efficacy of emetine without the adverse effects.
Wilson Wong, PhD, of the Walter and Eliza Hall Institute in Victoria, Australia, and his colleagues described this research in eLife.
For this work, the team used Cryo-EM, a technique that allows researchers to visualize the structure of protein complexes from cellular material without having to crystallize them. This method revealed how emetine binds to the ribosome of the malaria parasite Plasmodium falciparum.
“The ribosome is responsible for constructing all proteins inside the cell, based on the DNA ‘blueprint,’” Dr Wong said. “Antibiotics such as emetine kill the malaria parasite by binding to its ribosome and preventing the parasite from building the proteins it needs to produce energy, grow, reproduce, and evade the immune system.”
The investigators discovered that emetine interacts with the E-site of the ribosomal small subunit and shares a similar binding site with the antibiotic pactamycin. And emetine delivers its antimalaria effect by blocking mRNA/tRNA translocation.
“Our structure is an exciting discovery, as it gives a clear path forward in developing new drugs to tackle this deadly disease,” Dr Wong said. “We have found features of the parasitic ribosome that are not found in the human form. Drug makers could exploit these features in order to specifically target the production of proteins within the malaria parasite.”
“We are now working with our colleagues from the institute’s ACRF Chemical Biology division to develop new molecules based on emetine and pactamycin. Knowing exactly how these antibiotics work will enable development of new antimalarial drugs that replicate the active component of these antibiotics, while changing the parts that make it toxic to patients.” ![]()

Walter and Eliza Hall Institute
In uncovering how an antibiotic fights malaria, investigators may have enabled the development of safer and more effective antimalarial drugs.
The group has determined how the antibiotic emetine blocks the molecular machinery that produces the proteins required for malaria parasite survival.
Although emetine is known to be effective against malaria, the drug is not used for this purpose because it confers significant side effects.
By gaining new insight into how emetine works, the investigators have forged the way ahead for designing drugs that can provide the efficacy of emetine without the adverse effects.
Wilson Wong, PhD, of the Walter and Eliza Hall Institute in Victoria, Australia, and his colleagues described this research in eLife.
For this work, the team used Cryo-EM, a technique that allows researchers to visualize the structure of protein complexes from cellular material without having to crystallize them. This method revealed how emetine binds to the ribosome of the malaria parasite Plasmodium falciparum.
“The ribosome is responsible for constructing all proteins inside the cell, based on the DNA ‘blueprint,’” Dr Wong said. “Antibiotics such as emetine kill the malaria parasite by binding to its ribosome and preventing the parasite from building the proteins it needs to produce energy, grow, reproduce, and evade the immune system.”
The investigators discovered that emetine interacts with the E-site of the ribosomal small subunit and shares a similar binding site with the antibiotic pactamycin. And emetine delivers its antimalaria effect by blocking mRNA/tRNA translocation.
“Our structure is an exciting discovery, as it gives a clear path forward in developing new drugs to tackle this deadly disease,” Dr Wong said. “We have found features of the parasitic ribosome that are not found in the human form. Drug makers could exploit these features in order to specifically target the production of proteins within the malaria parasite.”
“We are now working with our colleagues from the institute’s ACRF Chemical Biology division to develop new molecules based on emetine and pactamycin. Knowing exactly how these antibiotics work will enable development of new antimalarial drugs that replicate the active component of these antibiotics, while changing the parts that make it toxic to patients.” ![]()
Patch solves problem plaguing malaria vaccination

patches on her hand
Credit: Tómas Tyner
Delivering malaria vaccines via a skin patch can streamline the vaccination process, according to preclinical research published in Scientific Reports.
The patch has arrays of tiny silicon microneedles that painlessly create temporary pores in the outermost layer of the skin, permitting the vaccine to flow into the skin.
Researchers used the patch to vaccinate mice with a live adenovirus engineered to deliver a protein from the malaria parasite Plasmodium yoelii.
The team found the patch could overcome one of the main problems with this type of vaccine—namely, although it induces high levels of immunity to malaria, it can also induce a strong immune response to the adenovirus itself.
This anti-adenovirus immunity prevents its repeated use as a vaccine, as the immune system recognizes the adenovirus and prevents it from delivering the malaria protein. So another vaccine type or adenovirus strain needs to be used in the booster immunization.
“What’s exciting from this work is that administration of this vaccine with the microneedle patch did not induce this strong anti-adenovirus immunity, even though very potent immunity to the malaria antigen is generated,” said study author Anne Moore, PhD, of University College Cork in Ireland.
This suggests the patch can facilitate the repeated use of the same adenovirus vaccine, thereby potentially reducing manufacturing costs of multiple vaccines.
In their experiments with mice, Dr Moore and her colleagues demonstrated that using the microneedle patch in the primary immunization does indeed permit repeated use of the same adenovirus vaccine. And this immunization method induced potent and highly protective immune responses against malaria.
Specifically, the researchers delivered a vaccine known as HAdV5-PyMSP142 to mice. They found the patch induced equivalent or enhanced antibody responses but decreased anti-vector responses when compared to intradermal delivery of the vaccine.
The addition of a heterologous vaccine known as MVA-PyMSP142 also produced greater antibody responses in mice primed with HAdV5-PyMSP142 via the patch, compared to those vaccinated intradermally.
The researchers observed the highest protection against blood-stage malaria when mice were vaccinated first with the patch and then intradermally.
In an attempt to commercialize this research, Dr Moore is heading to Silicon Valley next week to meet with venture capitalists and technology companies. ![]()

patches on her hand
Credit: Tómas Tyner
Delivering malaria vaccines via a skin patch can streamline the vaccination process, according to preclinical research published in Scientific Reports.
The patch has arrays of tiny silicon microneedles that painlessly create temporary pores in the outermost layer of the skin, permitting the vaccine to flow into the skin.
Researchers used the patch to vaccinate mice with a live adenovirus engineered to deliver a protein from the malaria parasite Plasmodium yoelii.
The team found the patch could overcome one of the main problems with this type of vaccine—namely, although it induces high levels of immunity to malaria, it can also induce a strong immune response to the adenovirus itself.
This anti-adenovirus immunity prevents its repeated use as a vaccine, as the immune system recognizes the adenovirus and prevents it from delivering the malaria protein. So another vaccine type or adenovirus strain needs to be used in the booster immunization.
“What’s exciting from this work is that administration of this vaccine with the microneedle patch did not induce this strong anti-adenovirus immunity, even though very potent immunity to the malaria antigen is generated,” said study author Anne Moore, PhD, of University College Cork in Ireland.
This suggests the patch can facilitate the repeated use of the same adenovirus vaccine, thereby potentially reducing manufacturing costs of multiple vaccines.
In their experiments with mice, Dr Moore and her colleagues demonstrated that using the microneedle patch in the primary immunization does indeed permit repeated use of the same adenovirus vaccine. And this immunization method induced potent and highly protective immune responses against malaria.
Specifically, the researchers delivered a vaccine known as HAdV5-PyMSP142 to mice. They found the patch induced equivalent or enhanced antibody responses but decreased anti-vector responses when compared to intradermal delivery of the vaccine.
The addition of a heterologous vaccine known as MVA-PyMSP142 also produced greater antibody responses in mice primed with HAdV5-PyMSP142 via the patch, compared to those vaccinated intradermally.
The researchers observed the highest protection against blood-stage malaria when mice were vaccinated first with the patch and then intradermally.
In an attempt to commercialize this research, Dr Moore is heading to Silicon Valley next week to meet with venture capitalists and technology companies. ![]()

patches on her hand
Credit: Tómas Tyner
Delivering malaria vaccines via a skin patch can streamline the vaccination process, according to preclinical research published in Scientific Reports.
The patch has arrays of tiny silicon microneedles that painlessly create temporary pores in the outermost layer of the skin, permitting the vaccine to flow into the skin.
Researchers used the patch to vaccinate mice with a live adenovirus engineered to deliver a protein from the malaria parasite Plasmodium yoelii.
The team found the patch could overcome one of the main problems with this type of vaccine—namely, although it induces high levels of immunity to malaria, it can also induce a strong immune response to the adenovirus itself.
This anti-adenovirus immunity prevents its repeated use as a vaccine, as the immune system recognizes the adenovirus and prevents it from delivering the malaria protein. So another vaccine type or adenovirus strain needs to be used in the booster immunization.
“What’s exciting from this work is that administration of this vaccine with the microneedle patch did not induce this strong anti-adenovirus immunity, even though very potent immunity to the malaria antigen is generated,” said study author Anne Moore, PhD, of University College Cork in Ireland.
This suggests the patch can facilitate the repeated use of the same adenovirus vaccine, thereby potentially reducing manufacturing costs of multiple vaccines.
In their experiments with mice, Dr Moore and her colleagues demonstrated that using the microneedle patch in the primary immunization does indeed permit repeated use of the same adenovirus vaccine. And this immunization method induced potent and highly protective immune responses against malaria.
Specifically, the researchers delivered a vaccine known as HAdV5-PyMSP142 to mice. They found the patch induced equivalent or enhanced antibody responses but decreased anti-vector responses when compared to intradermal delivery of the vaccine.
The addition of a heterologous vaccine known as MVA-PyMSP142 also produced greater antibody responses in mice primed with HAdV5-PyMSP142 via the patch, compared to those vaccinated intradermally.
The researchers observed the highest protection against blood-stage malaria when mice were vaccinated first with the patch and then intradermally.
In an attempt to commercialize this research, Dr Moore is heading to Silicon Valley next week to meet with venture capitalists and technology companies. ![]()
Journals may fail to correct major errors in articles

Credit: CDC/James Gathany
A review of errata reports from medical publications revealed that nearly a quarter of the errors may have changed the way study data were interpreted.
And about half of the errors were not corrected in the original text, or the errata report did not specify whether a correction was made.
This suggests authors and journals must be more vigilant and consistent in identifying and reporting errors, said Paul Hauptman, MD, of Saint Louis University in Missouri.
He and his colleagues conducted this research and reported the results in The American Journal of Medicine.
The researchers reviewed 20 prominent English language journals in the fields of general medicine and cardiology. And they identified 577 error reports over an 18-month period.
More than 24% of these reports included an error the team rated as major. Major errors were associated with material changes in the interpretation of data in text, figures, or tables, or with significant alterations in the article’s conclusions.
One of the examples of a major error report in the study comes from a paper about depression. In the original article, the incidence of new depression cases was misquoted by a factor of 10. The article initially reported 15.8% of women with new depression, rather than the real figure of 1.58%, which was later corrected.
“As the volume of research publications continues to rise, the scientific community needs to examine how it manages its mistakes,” said Eric Armbrecht, PhD, of the Saint Louis University Center for Outcomes Research.
“Transparency, consistency, and clarity are essential. Our study found that these are not common among some of the top medical journals.”
In fact, the researchers found that 51% of the errors they identified were not corrected in the original text, or the errata report did not specify whether a correction was made.
The team noted that this study did not provide any definitive explanations for why such errors appear in publications, although one possibility is that most authors don’t read and edit the final version of their manuscripts prior to publication.
“It’s noteworthy that although final approval of an article may fall to the first or corresponding author, the criteria put forth by [the International Committee of Medical Journal Editors] specifies that each author must provide final approval of the version to be published,” Dr Hauptman said.
He added that, at this point, it’s not possible to measure how frequently a journal reader incorporates errata into clinical care or the extent to which patient outcomes may be affected. ![]()

Credit: CDC/James Gathany
A review of errata reports from medical publications revealed that nearly a quarter of the errors may have changed the way study data were interpreted.
And about half of the errors were not corrected in the original text, or the errata report did not specify whether a correction was made.
This suggests authors and journals must be more vigilant and consistent in identifying and reporting errors, said Paul Hauptman, MD, of Saint Louis University in Missouri.
He and his colleagues conducted this research and reported the results in The American Journal of Medicine.
The researchers reviewed 20 prominent English language journals in the fields of general medicine and cardiology. And they identified 577 error reports over an 18-month period.
More than 24% of these reports included an error the team rated as major. Major errors were associated with material changes in the interpretation of data in text, figures, or tables, or with significant alterations in the article’s conclusions.
One of the examples of a major error report in the study comes from a paper about depression. In the original article, the incidence of new depression cases was misquoted by a factor of 10. The article initially reported 15.8% of women with new depression, rather than the real figure of 1.58%, which was later corrected.
“As the volume of research publications continues to rise, the scientific community needs to examine how it manages its mistakes,” said Eric Armbrecht, PhD, of the Saint Louis University Center for Outcomes Research.
“Transparency, consistency, and clarity are essential. Our study found that these are not common among some of the top medical journals.”
In fact, the researchers found that 51% of the errors they identified were not corrected in the original text, or the errata report did not specify whether a correction was made.
The team noted that this study did not provide any definitive explanations for why such errors appear in publications, although one possibility is that most authors don’t read and edit the final version of their manuscripts prior to publication.
“It’s noteworthy that although final approval of an article may fall to the first or corresponding author, the criteria put forth by [the International Committee of Medical Journal Editors] specifies that each author must provide final approval of the version to be published,” Dr Hauptman said.
He added that, at this point, it’s not possible to measure how frequently a journal reader incorporates errata into clinical care or the extent to which patient outcomes may be affected. ![]()

Credit: CDC/James Gathany
A review of errata reports from medical publications revealed that nearly a quarter of the errors may have changed the way study data were interpreted.
And about half of the errors were not corrected in the original text, or the errata report did not specify whether a correction was made.
This suggests authors and journals must be more vigilant and consistent in identifying and reporting errors, said Paul Hauptman, MD, of Saint Louis University in Missouri.
He and his colleagues conducted this research and reported the results in The American Journal of Medicine.
The researchers reviewed 20 prominent English language journals in the fields of general medicine and cardiology. And they identified 577 error reports over an 18-month period.
More than 24% of these reports included an error the team rated as major. Major errors were associated with material changes in the interpretation of data in text, figures, or tables, or with significant alterations in the article’s conclusions.
One of the examples of a major error report in the study comes from a paper about depression. In the original article, the incidence of new depression cases was misquoted by a factor of 10. The article initially reported 15.8% of women with new depression, rather than the real figure of 1.58%, which was later corrected.
“As the volume of research publications continues to rise, the scientific community needs to examine how it manages its mistakes,” said Eric Armbrecht, PhD, of the Saint Louis University Center for Outcomes Research.
“Transparency, consistency, and clarity are essential. Our study found that these are not common among some of the top medical journals.”
In fact, the researchers found that 51% of the errors they identified were not corrected in the original text, or the errata report did not specify whether a correction was made.
The team noted that this study did not provide any definitive explanations for why such errors appear in publications, although one possibility is that most authors don’t read and edit the final version of their manuscripts prior to publication.
“It’s noteworthy that although final approval of an article may fall to the first or corresponding author, the criteria put forth by [the International Committee of Medical Journal Editors] specifies that each author must provide final approval of the version to be published,” Dr Hauptman said.
He added that, at this point, it’s not possible to measure how frequently a journal reader incorporates errata into clinical care or the extent to which patient outcomes may be affected. ![]()
Cellular RNA can template DNA repair in yeast
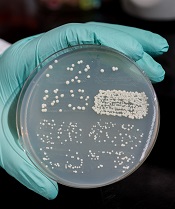
DNA has been repaired by
transcript RNA within the cells
Credit: Georgia Tech/Rob Felt
Scientists have shown that RNA produced within yeast cells can serve as a template for repairing the most devastating DNA damage—a break in both strands of a DNA helix.
The group believes their study is the first to show that a cell’s own RNA can be used for DNA recombination and repair.
If the phenomenon extends to human cells, it could potentially lead to new therapeutic or preventative strategies for genetic diseases.
The scientists described the phenomenon in Nature.
“We have found that genetic information can flow from RNA to DNA in a homology-driven manner, from cellular RNA to a homologous DNA sequence,” said study author Francesca Storici, PhD, of the Georgia Institute of Technology in Atlanta.
“This process is moving the genetic information in the opposite direction from which it normally flows. We have shown that when an endogenous RNA molecule can anneal to broken homologous DNA without being removed, the RNA can repair the damaged DNA. This finding reveals the existence of a novel mechanism of genetic recombination.”
Dr Storici’s team previously showed that synthetic RNA introduced into cells—including human cells—could repair DNA damage. But the process was inefficient, and there were questions about whether it could occur naturally.
To find out whether cells could use endogenous RNA transcripts to repair DNA damage, she and her colleagues devised experiments using the yeast Saccharomyces cerevisiae.
The team developed a strategy for distinguishing repair by endogenous RNA from repair by the normal DNA-based mechanisms in the budding yeast cells, including using mutants that lacked the ability to convert the RNA into a DNA copy.
They then induced a DNA double-strand break in the yeast genome and observed whether the organism could survive and grow by repairing the damage using only transcript RNA within the cells.
The DNA region that generates the transcript was constructed to contain a marker gene interrupted by an intron. Following intron removal during transcription, the transcript RNA sequence has no intron, while the DNA region that generates the transcript retains the intron.
Only the repair templated by the transcript devoid of the intron can restore the function of a homologous marker gene in which the DNA double-strand break is induced.
Dr Storici and her colleagues measured success by counting the number of yeast colonies growing on a Petri dish, indicating that the repair had been made by endogenous RNA.
They conducted testing on two types of breaks, one in the DNA from which the RNA transcript had been made, and the other in a homologous sequence from a different location in the DNA.
The team found that proximity of the RNA to the broken DNA increased the efficiency of the repair, and the repair occurred via a homologous recombination process. Dr Storici believes the repair mechanism may operate in cells beyond yeast, and many types of RNA can be used.
“We are showing that the flow of genetic information from RNA to DNA is not restricted to retro-elements and telomeres but occurs with a generic cellular transcript, making it more of a general phenomenon than had been anticipated,” she explained. “Potentially, any RNA in the cell could have this function.”
For the future, Dr Storici hopes to learn more about the mechanism, including what regulates it. She also wants to determine whether it takes place in human cells. If so, that could have implications for treating or preventing diseases caused by genetic damage.
“Cells synthesize lots of RNA transcripts during their life spans,” Dr Storici said. “Therefore, RNA may have an unanticipated impact on genomic stability and plasticity. We need to understand in which situations cells would activate RNA-DNA recombination. Better understanding this molecular process could also help us manipulate mechanisms for therapy, allowing us to treat a disease or prevent it altogether.” ![]()

DNA has been repaired by
transcript RNA within the cells
Credit: Georgia Tech/Rob Felt
Scientists have shown that RNA produced within yeast cells can serve as a template for repairing the most devastating DNA damage—a break in both strands of a DNA helix.
The group believes their study is the first to show that a cell’s own RNA can be used for DNA recombination and repair.
If the phenomenon extends to human cells, it could potentially lead to new therapeutic or preventative strategies for genetic diseases.
The scientists described the phenomenon in Nature.
“We have found that genetic information can flow from RNA to DNA in a homology-driven manner, from cellular RNA to a homologous DNA sequence,” said study author Francesca Storici, PhD, of the Georgia Institute of Technology in Atlanta.
“This process is moving the genetic information in the opposite direction from which it normally flows. We have shown that when an endogenous RNA molecule can anneal to broken homologous DNA without being removed, the RNA can repair the damaged DNA. This finding reveals the existence of a novel mechanism of genetic recombination.”
Dr Storici’s team previously showed that synthetic RNA introduced into cells—including human cells—could repair DNA damage. But the process was inefficient, and there were questions about whether it could occur naturally.
To find out whether cells could use endogenous RNA transcripts to repair DNA damage, she and her colleagues devised experiments using the yeast Saccharomyces cerevisiae.
The team developed a strategy for distinguishing repair by endogenous RNA from repair by the normal DNA-based mechanisms in the budding yeast cells, including using mutants that lacked the ability to convert the RNA into a DNA copy.
They then induced a DNA double-strand break in the yeast genome and observed whether the organism could survive and grow by repairing the damage using only transcript RNA within the cells.
The DNA region that generates the transcript was constructed to contain a marker gene interrupted by an intron. Following intron removal during transcription, the transcript RNA sequence has no intron, while the DNA region that generates the transcript retains the intron.
Only the repair templated by the transcript devoid of the intron can restore the function of a homologous marker gene in which the DNA double-strand break is induced.
Dr Storici and her colleagues measured success by counting the number of yeast colonies growing on a Petri dish, indicating that the repair had been made by endogenous RNA.
They conducted testing on two types of breaks, one in the DNA from which the RNA transcript had been made, and the other in a homologous sequence from a different location in the DNA.
The team found that proximity of the RNA to the broken DNA increased the efficiency of the repair, and the repair occurred via a homologous recombination process. Dr Storici believes the repair mechanism may operate in cells beyond yeast, and many types of RNA can be used.
“We are showing that the flow of genetic information from RNA to DNA is not restricted to retro-elements and telomeres but occurs with a generic cellular transcript, making it more of a general phenomenon than had been anticipated,” she explained. “Potentially, any RNA in the cell could have this function.”
For the future, Dr Storici hopes to learn more about the mechanism, including what regulates it. She also wants to determine whether it takes place in human cells. If so, that could have implications for treating or preventing diseases caused by genetic damage.
“Cells synthesize lots of RNA transcripts during their life spans,” Dr Storici said. “Therefore, RNA may have an unanticipated impact on genomic stability and plasticity. We need to understand in which situations cells would activate RNA-DNA recombination. Better understanding this molecular process could also help us manipulate mechanisms for therapy, allowing us to treat a disease or prevent it altogether.” ![]()

DNA has been repaired by
transcript RNA within the cells
Credit: Georgia Tech/Rob Felt
Scientists have shown that RNA produced within yeast cells can serve as a template for repairing the most devastating DNA damage—a break in both strands of a DNA helix.
The group believes their study is the first to show that a cell’s own RNA can be used for DNA recombination and repair.
If the phenomenon extends to human cells, it could potentially lead to new therapeutic or preventative strategies for genetic diseases.
The scientists described the phenomenon in Nature.
“We have found that genetic information can flow from RNA to DNA in a homology-driven manner, from cellular RNA to a homologous DNA sequence,” said study author Francesca Storici, PhD, of the Georgia Institute of Technology in Atlanta.
“This process is moving the genetic information in the opposite direction from which it normally flows. We have shown that when an endogenous RNA molecule can anneal to broken homologous DNA without being removed, the RNA can repair the damaged DNA. This finding reveals the existence of a novel mechanism of genetic recombination.”
Dr Storici’s team previously showed that synthetic RNA introduced into cells—including human cells—could repair DNA damage. But the process was inefficient, and there were questions about whether it could occur naturally.
To find out whether cells could use endogenous RNA transcripts to repair DNA damage, she and her colleagues devised experiments using the yeast Saccharomyces cerevisiae.
The team developed a strategy for distinguishing repair by endogenous RNA from repair by the normal DNA-based mechanisms in the budding yeast cells, including using mutants that lacked the ability to convert the RNA into a DNA copy.
They then induced a DNA double-strand break in the yeast genome and observed whether the organism could survive and grow by repairing the damage using only transcript RNA within the cells.
The DNA region that generates the transcript was constructed to contain a marker gene interrupted by an intron. Following intron removal during transcription, the transcript RNA sequence has no intron, while the DNA region that generates the transcript retains the intron.
Only the repair templated by the transcript devoid of the intron can restore the function of a homologous marker gene in which the DNA double-strand break is induced.
Dr Storici and her colleagues measured success by counting the number of yeast colonies growing on a Petri dish, indicating that the repair had been made by endogenous RNA.
They conducted testing on two types of breaks, one in the DNA from which the RNA transcript had been made, and the other in a homologous sequence from a different location in the DNA.
The team found that proximity of the RNA to the broken DNA increased the efficiency of the repair, and the repair occurred via a homologous recombination process. Dr Storici believes the repair mechanism may operate in cells beyond yeast, and many types of RNA can be used.
“We are showing that the flow of genetic information from RNA to DNA is not restricted to retro-elements and telomeres but occurs with a generic cellular transcript, making it more of a general phenomenon than had been anticipated,” she explained. “Potentially, any RNA in the cell could have this function.”
For the future, Dr Storici hopes to learn more about the mechanism, including what regulates it. She also wants to determine whether it takes place in human cells. If so, that could have implications for treating or preventing diseases caused by genetic damage.
“Cells synthesize lots of RNA transcripts during their life spans,” Dr Storici said. “Therefore, RNA may have an unanticipated impact on genomic stability and plasticity. We need to understand in which situations cells would activate RNA-DNA recombination. Better understanding this molecular process could also help us manipulate mechanisms for therapy, allowing us to treat a disease or prevent it altogether.” ![]()
Method could improve malaria diagnosis

red blood cell; Credit: St Jude
Children’s Research Hospital
Researchers have found they can diagnose malaria using magnetic fields to detect a byproduct of malarial metabolism.
They used magnetic resonance relaxometry (MRR) to detect a parasitic waste product called hemozoin in malaria-infected red blood cells from mice and humans.
The team said MRR is more sensitive than other methods of detecting malaria, can be carried out using a portable benchtop system, and costs less than 10 cents per test.
Jongyoon Han, PhD, of the Massachusetts Institute of Technology in Cambridge, and his colleagues described the technique in Nature Medicine.
When malaria parasites infect red blood cells, they feed on the nutrient-rich hemoglobin. As hemoglobin breaks down, it releases iron, which can be toxic, so the parasite converts the iron into hemozoin—a weakly paramagnetic crystallite.
Those crystals interfere with the normal magnetic spins of hydrogen atoms. When exposed to a powerful magnetic field, hydrogen atoms align their spins in the same direction.
When a second, smaller field perturbs the atoms, they should all change their spins in synchrony. But if another magnetic particle, such as hemozoin, is present, this synchrony is disrupted through a process called relaxation. The more magnetic particles present, the more quickly the synchrony is disrupted.
“What we are trying to really measure is how the hydrogen’s nuclear magnetic resonance is affected by the proximity of other magnetic particles,” Dr Han said.
This MRR technique enables malaria diagnosis because hemozoin crystals are produced in all 4 stages of malaria infection and are generated by all known species of the Plasmodium parasite. Furthermore, the amount of hemozoin can reveal how severe the infection is, or whether it is responding to treatment.
Dr Han and his colleagues found they could use MRR to detect Plasmodium falciparum infection to as low as 0.0002% parasitemia in 750 nl of cultured blood in less than 5 minutes.
They also detected Plasmodium berghei in mice, allowing for reliable estimation of parasitemia to as low as 0.0001%.
The device the researchers used in this study is small enough to sit on a table or lab bench, but they are working on a portable version the size of a small electronic tablet.
“This system can be built at a very low cost, relative to the million-dollar MRI machines used in a hospital,” said study author Weng Kung Peng, PhD, of the Singapore-MIT Alliance for Research and Technology Centre in Singapore.
“Furthermore, since this technique does not rely on expensive labeling with chemical reagents, we are able to get each diagnostic test done at a cost of less than 10 cents.”
The researchers are launching a company to make this technology available at an affordable price. The team is also running field tests in Southeast Asia and exploring powering the device on solar energy. ![]()

red blood cell; Credit: St Jude
Children’s Research Hospital
Researchers have found they can diagnose malaria using magnetic fields to detect a byproduct of malarial metabolism.
They used magnetic resonance relaxometry (MRR) to detect a parasitic waste product called hemozoin in malaria-infected red blood cells from mice and humans.
The team said MRR is more sensitive than other methods of detecting malaria, can be carried out using a portable benchtop system, and costs less than 10 cents per test.
Jongyoon Han, PhD, of the Massachusetts Institute of Technology in Cambridge, and his colleagues described the technique in Nature Medicine.
When malaria parasites infect red blood cells, they feed on the nutrient-rich hemoglobin. As hemoglobin breaks down, it releases iron, which can be toxic, so the parasite converts the iron into hemozoin—a weakly paramagnetic crystallite.
Those crystals interfere with the normal magnetic spins of hydrogen atoms. When exposed to a powerful magnetic field, hydrogen atoms align their spins in the same direction.
When a second, smaller field perturbs the atoms, they should all change their spins in synchrony. But if another magnetic particle, such as hemozoin, is present, this synchrony is disrupted through a process called relaxation. The more magnetic particles present, the more quickly the synchrony is disrupted.
“What we are trying to really measure is how the hydrogen’s nuclear magnetic resonance is affected by the proximity of other magnetic particles,” Dr Han said.
This MRR technique enables malaria diagnosis because hemozoin crystals are produced in all 4 stages of malaria infection and are generated by all known species of the Plasmodium parasite. Furthermore, the amount of hemozoin can reveal how severe the infection is, or whether it is responding to treatment.
Dr Han and his colleagues found they could use MRR to detect Plasmodium falciparum infection to as low as 0.0002% parasitemia in 750 nl of cultured blood in less than 5 minutes.
They also detected Plasmodium berghei in mice, allowing for reliable estimation of parasitemia to as low as 0.0001%.
The device the researchers used in this study is small enough to sit on a table or lab bench, but they are working on a portable version the size of a small electronic tablet.
“This system can be built at a very low cost, relative to the million-dollar MRI machines used in a hospital,” said study author Weng Kung Peng, PhD, of the Singapore-MIT Alliance for Research and Technology Centre in Singapore.
“Furthermore, since this technique does not rely on expensive labeling with chemical reagents, we are able to get each diagnostic test done at a cost of less than 10 cents.”
The researchers are launching a company to make this technology available at an affordable price. The team is also running field tests in Southeast Asia and exploring powering the device on solar energy. ![]()

red blood cell; Credit: St Jude
Children’s Research Hospital
Researchers have found they can diagnose malaria using magnetic fields to detect a byproduct of malarial metabolism.
They used magnetic resonance relaxometry (MRR) to detect a parasitic waste product called hemozoin in malaria-infected red blood cells from mice and humans.
The team said MRR is more sensitive than other methods of detecting malaria, can be carried out using a portable benchtop system, and costs less than 10 cents per test.
Jongyoon Han, PhD, of the Massachusetts Institute of Technology in Cambridge, and his colleagues described the technique in Nature Medicine.
When malaria parasites infect red blood cells, they feed on the nutrient-rich hemoglobin. As hemoglobin breaks down, it releases iron, which can be toxic, so the parasite converts the iron into hemozoin—a weakly paramagnetic crystallite.
Those crystals interfere with the normal magnetic spins of hydrogen atoms. When exposed to a powerful magnetic field, hydrogen atoms align their spins in the same direction.
When a second, smaller field perturbs the atoms, they should all change their spins in synchrony. But if another magnetic particle, such as hemozoin, is present, this synchrony is disrupted through a process called relaxation. The more magnetic particles present, the more quickly the synchrony is disrupted.
“What we are trying to really measure is how the hydrogen’s nuclear magnetic resonance is affected by the proximity of other magnetic particles,” Dr Han said.
This MRR technique enables malaria diagnosis because hemozoin crystals are produced in all 4 stages of malaria infection and are generated by all known species of the Plasmodium parasite. Furthermore, the amount of hemozoin can reveal how severe the infection is, or whether it is responding to treatment.
Dr Han and his colleagues found they could use MRR to detect Plasmodium falciparum infection to as low as 0.0002% parasitemia in 750 nl of cultured blood in less than 5 minutes.
They also detected Plasmodium berghei in mice, allowing for reliable estimation of parasitemia to as low as 0.0001%.
The device the researchers used in this study is small enough to sit on a table or lab bench, but they are working on a portable version the size of a small electronic tablet.
“This system can be built at a very low cost, relative to the million-dollar MRI machines used in a hospital,” said study author Weng Kung Peng, PhD, of the Singapore-MIT Alliance for Research and Technology Centre in Singapore.
“Furthermore, since this technique does not rely on expensive labeling with chemical reagents, we are able to get each diagnostic test done at a cost of less than 10 cents.”
The researchers are launching a company to make this technology available at an affordable price. The team is also running field tests in Southeast Asia and exploring powering the device on solar energy. ![]()
Program improves depression treatment in cancer

Credit: NIH
Results of a large study suggest major depression is common—but largely untreated—among cancer patients in Scotland.
And 2 additional studies of Scottish patients showed that a program specifically designed for individuals with cancer can treat depression and improve quality of life more effectively than current methods of care.
These studies appear in The Lancet, The Lancet Oncology, and The Lancet Psychiatry.
In The Lancet Psychiatry, researchers recounted their analysis of data from 21,151 patients treated at cancer clinics in Scotland. The team found that major depression was substantially more common in cancer patients than in the general population.
Major depression was most common in patients with lung cancer (13%) and lowest in those with genitourinary cancer (6%). Moreover, nearly three-quarters (73%) of depressed cancer patients were not receiving treatment.
To address the problem of inadequate treatment, researchers initiated the SMaRT Oncology-2 trial. They reported the results in The Lancet.
The team evaluated a new treatment program called “Depression Care for People with Cancer” (DCPC). DCPC is delivered by specially trained cancer nurses and psychiatrists, working in collaboration with the patient’s cancer team and general practitioner, and is given as part of cancer care. It is a systematic treatment program that includes both antidepressants and psychological therapy.
The trial included 500 adults with major depression and a cancer with a good prognosis (predicted survival of more than 12 months).
Patients were randomized to receive either DCPC or “usual care,” which was provided by a patient’s general practitioner and might have included prescribing antidepressants or referring the patient to mental health services for assessment or psychological treatment.
Results showed that DCPC was more effective than usual care in reducing depression. At 6 months, 62% of patients who received DCPC responded to treatment (experiencing at least a 50% reduction in the severity of their depression), compared with 17% of those who received the usual care (P<0.0001). This benefit was sustained at 12 months.
In addition, DCPC improved anxiety, pain, fatigue, functioning, and overall quality of life (all P<0.05). The researchers also noted that the cost of providing DCPC was modest (£613 per patient).
“The huge benefit that DCPC delivers for patients with cancer and depression shows what we can achieve for patients if we take as much care with the treatment of their depression as we do with the treatment of their cancer,” said study author Michael Sharpe, MD, of the University of Oxford in the UK.
To see if patients with a poor-prognosis cancer could also benefit from DCPC, researchers initiated the SMaRT Oncology-3 trial. They reported the results in The Lancet Oncology.
The team tested a version of DCPC adapted for cancer patients with a poor prognosis. The trial included 142 patients with lung cancer and major depression.
Patients who received the modified version of DCPC had a significantly greater improvement in depression than those who received the usual care during 32 weeks of follow-up (P=0.0003). DCPC also improved patients’ anxiety (P=0.046), functioning (P=0.0019), and quality of life (P=0.018).
“Patients with lung cancer often have a poor prognosis,” said study author Jane Walker, MBChB, PhD, of the University of Oxford and Sobell House Hospice in Oxford, UK.
“If they also have major depression, that can blight the time they have left to live. This trial shows that we can effectively treat depression in patients with poor-prognosis cancers, like lung cancer, and really improve patients’ lives.” ![]()

Credit: NIH
Results of a large study suggest major depression is common—but largely untreated—among cancer patients in Scotland.
And 2 additional studies of Scottish patients showed that a program specifically designed for individuals with cancer can treat depression and improve quality of life more effectively than current methods of care.
These studies appear in The Lancet, The Lancet Oncology, and The Lancet Psychiatry.
In The Lancet Psychiatry, researchers recounted their analysis of data from 21,151 patients treated at cancer clinics in Scotland. The team found that major depression was substantially more common in cancer patients than in the general population.
Major depression was most common in patients with lung cancer (13%) and lowest in those with genitourinary cancer (6%). Moreover, nearly three-quarters (73%) of depressed cancer patients were not receiving treatment.
To address the problem of inadequate treatment, researchers initiated the SMaRT Oncology-2 trial. They reported the results in The Lancet.
The team evaluated a new treatment program called “Depression Care for People with Cancer” (DCPC). DCPC is delivered by specially trained cancer nurses and psychiatrists, working in collaboration with the patient’s cancer team and general practitioner, and is given as part of cancer care. It is a systematic treatment program that includes both antidepressants and psychological therapy.
The trial included 500 adults with major depression and a cancer with a good prognosis (predicted survival of more than 12 months).
Patients were randomized to receive either DCPC or “usual care,” which was provided by a patient’s general practitioner and might have included prescribing antidepressants or referring the patient to mental health services for assessment or psychological treatment.
Results showed that DCPC was more effective than usual care in reducing depression. At 6 months, 62% of patients who received DCPC responded to treatment (experiencing at least a 50% reduction in the severity of their depression), compared with 17% of those who received the usual care (P<0.0001). This benefit was sustained at 12 months.
In addition, DCPC improved anxiety, pain, fatigue, functioning, and overall quality of life (all P<0.05). The researchers also noted that the cost of providing DCPC was modest (£613 per patient).
“The huge benefit that DCPC delivers for patients with cancer and depression shows what we can achieve for patients if we take as much care with the treatment of their depression as we do with the treatment of their cancer,” said study author Michael Sharpe, MD, of the University of Oxford in the UK.
To see if patients with a poor-prognosis cancer could also benefit from DCPC, researchers initiated the SMaRT Oncology-3 trial. They reported the results in The Lancet Oncology.
The team tested a version of DCPC adapted for cancer patients with a poor prognosis. The trial included 142 patients with lung cancer and major depression.
Patients who received the modified version of DCPC had a significantly greater improvement in depression than those who received the usual care during 32 weeks of follow-up (P=0.0003). DCPC also improved patients’ anxiety (P=0.046), functioning (P=0.0019), and quality of life (P=0.018).
“Patients with lung cancer often have a poor prognosis,” said study author Jane Walker, MBChB, PhD, of the University of Oxford and Sobell House Hospice in Oxford, UK.
“If they also have major depression, that can blight the time they have left to live. This trial shows that we can effectively treat depression in patients with poor-prognosis cancers, like lung cancer, and really improve patients’ lives.” ![]()

Credit: NIH
Results of a large study suggest major depression is common—but largely untreated—among cancer patients in Scotland.
And 2 additional studies of Scottish patients showed that a program specifically designed for individuals with cancer can treat depression and improve quality of life more effectively than current methods of care.
These studies appear in The Lancet, The Lancet Oncology, and The Lancet Psychiatry.
In The Lancet Psychiatry, researchers recounted their analysis of data from 21,151 patients treated at cancer clinics in Scotland. The team found that major depression was substantially more common in cancer patients than in the general population.
Major depression was most common in patients with lung cancer (13%) and lowest in those with genitourinary cancer (6%). Moreover, nearly three-quarters (73%) of depressed cancer patients were not receiving treatment.
To address the problem of inadequate treatment, researchers initiated the SMaRT Oncology-2 trial. They reported the results in The Lancet.
The team evaluated a new treatment program called “Depression Care for People with Cancer” (DCPC). DCPC is delivered by specially trained cancer nurses and psychiatrists, working in collaboration with the patient’s cancer team and general practitioner, and is given as part of cancer care. It is a systematic treatment program that includes both antidepressants and psychological therapy.
The trial included 500 adults with major depression and a cancer with a good prognosis (predicted survival of more than 12 months).
Patients were randomized to receive either DCPC or “usual care,” which was provided by a patient’s general practitioner and might have included prescribing antidepressants or referring the patient to mental health services for assessment or psychological treatment.
Results showed that DCPC was more effective than usual care in reducing depression. At 6 months, 62% of patients who received DCPC responded to treatment (experiencing at least a 50% reduction in the severity of their depression), compared with 17% of those who received the usual care (P<0.0001). This benefit was sustained at 12 months.
In addition, DCPC improved anxiety, pain, fatigue, functioning, and overall quality of life (all P<0.05). The researchers also noted that the cost of providing DCPC was modest (£613 per patient).
“The huge benefit that DCPC delivers for patients with cancer and depression shows what we can achieve for patients if we take as much care with the treatment of their depression as we do with the treatment of their cancer,” said study author Michael Sharpe, MD, of the University of Oxford in the UK.
To see if patients with a poor-prognosis cancer could also benefit from DCPC, researchers initiated the SMaRT Oncology-3 trial. They reported the results in The Lancet Oncology.
The team tested a version of DCPC adapted for cancer patients with a poor prognosis. The trial included 142 patients with lung cancer and major depression.
Patients who received the modified version of DCPC had a significantly greater improvement in depression than those who received the usual care during 32 weeks of follow-up (P=0.0003). DCPC also improved patients’ anxiety (P=0.046), functioning (P=0.0019), and quality of life (P=0.018).
“Patients with lung cancer often have a poor prognosis,” said study author Jane Walker, MBChB, PhD, of the University of Oxford and Sobell House Hospice in Oxford, UK.
“If they also have major depression, that can blight the time they have left to live. This trial shows that we can effectively treat depression in patients with poor-prognosis cancers, like lung cancer, and really improve patients’ lives.” ![]()
T cells play role in clinical tolerance of malaria, team says

Credit: Malayaka house
Children repeatedly infected with malaria have been known to become asymptomatic, and researchers have found evidence suggesting a subset of γδ T cells play a role in this phenomenon.
Studying young children in Uganda, the team discovered that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells.
This appeared to facilitate immunological tolerance of the malaria parasite and, therefore, a reduction in clinical symptoms.
“These inflammatory immune cells are depleted in children with repeated malaria exposure, and those that remain behave differently than the same cell types in children who have not previously been infected,” said Prasanna Jagannathan, MD, of the University of California, San Francisco (UCSF).
He and his colleagues reported these findings in Science Translational Medicine.
The researchers collected data on malaria infections, disease symptoms, and immune responses in 78 children who were monitored from infancy as part of a research collaboration between UCSF and Makarere University in Kampala, Africa. The study was conducted in Tororo, Uganda.
At 1 year of age, all children showed clinical symptoms of malaria with each infection. At 4 years, fewer than 10% were symptom-free upon infection. But 1 year later, more than 20% were symptom-free when infected.
The researchers found that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells. They observed a decrease in cell proliferation and in the production of inflammatory cytokines (IFN-γ and TNF-α) in response to malaria antigens.
Repeated malaria infection was also associated with the upregulation of immunoregulatory pathways—increased expression of genes such as HAVCR2, FCRL6, LYN, BATF, and B3GAT1—that dampen the immune response.
Children with these characteristics were less likely than their peers to exhibit clinical symptoms upon subsequent malaria infection.
So it seems the depletion of Vδ2+ γδ T cells is beneficial in some ways and detrimental in others, said Margaret Feeney, MD, of UCSF. Individuals may no longer suffer symptoms, but they might not clear the parasite and could remain infectious.
Although this discovery has not provided a disease-fighting strategy as of yet, it does point to further avenues of study, according to Dr Feeney.
“We want to understand whether this is a generalizable phenomenon that also occurs among those who are first exposed to malaria as adults and in regions where malaria incidence is lower,” she said.
Dr Feeney speculates that malaria infection, by reshaping immune responses, might influence a person’s susceptibility to, and protection from, other infectious diseases. ![]()

Credit: Malayaka house
Children repeatedly infected with malaria have been known to become asymptomatic, and researchers have found evidence suggesting a subset of γδ T cells play a role in this phenomenon.
Studying young children in Uganda, the team discovered that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells.
This appeared to facilitate immunological tolerance of the malaria parasite and, therefore, a reduction in clinical symptoms.
“These inflammatory immune cells are depleted in children with repeated malaria exposure, and those that remain behave differently than the same cell types in children who have not previously been infected,” said Prasanna Jagannathan, MD, of the University of California, San Francisco (UCSF).
He and his colleagues reported these findings in Science Translational Medicine.
The researchers collected data on malaria infections, disease symptoms, and immune responses in 78 children who were monitored from infancy as part of a research collaboration between UCSF and Makarere University in Kampala, Africa. The study was conducted in Tororo, Uganda.
At 1 year of age, all children showed clinical symptoms of malaria with each infection. At 4 years, fewer than 10% were symptom-free upon infection. But 1 year later, more than 20% were symptom-free when infected.
The researchers found that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells. They observed a decrease in cell proliferation and in the production of inflammatory cytokines (IFN-γ and TNF-α) in response to malaria antigens.
Repeated malaria infection was also associated with the upregulation of immunoregulatory pathways—increased expression of genes such as HAVCR2, FCRL6, LYN, BATF, and B3GAT1—that dampen the immune response.
Children with these characteristics were less likely than their peers to exhibit clinical symptoms upon subsequent malaria infection.
So it seems the depletion of Vδ2+ γδ T cells is beneficial in some ways and detrimental in others, said Margaret Feeney, MD, of UCSF. Individuals may no longer suffer symptoms, but they might not clear the parasite and could remain infectious.
Although this discovery has not provided a disease-fighting strategy as of yet, it does point to further avenues of study, according to Dr Feeney.
“We want to understand whether this is a generalizable phenomenon that also occurs among those who are first exposed to malaria as adults and in regions where malaria incidence is lower,” she said.
Dr Feeney speculates that malaria infection, by reshaping immune responses, might influence a person’s susceptibility to, and protection from, other infectious diseases. ![]()

Credit: Malayaka house
Children repeatedly infected with malaria have been known to become asymptomatic, and researchers have found evidence suggesting a subset of γδ T cells play a role in this phenomenon.
Studying young children in Uganda, the team discovered that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells.
This appeared to facilitate immunological tolerance of the malaria parasite and, therefore, a reduction in clinical symptoms.
“These inflammatory immune cells are depleted in children with repeated malaria exposure, and those that remain behave differently than the same cell types in children who have not previously been infected,” said Prasanna Jagannathan, MD, of the University of California, San Francisco (UCSF).
He and his colleagues reported these findings in Science Translational Medicine.
The researchers collected data on malaria infections, disease symptoms, and immune responses in 78 children who were monitored from infancy as part of a research collaboration between UCSF and Makarere University in Kampala, Africa. The study was conducted in Tororo, Uganda.
At 1 year of age, all children showed clinical symptoms of malaria with each infection. At 4 years, fewer than 10% were symptom-free upon infection. But 1 year later, more than 20% were symptom-free when infected.
The researchers found that repeated malaria infection was associated with the loss and dysfunction of Vδ2+ γδ T cells. They observed a decrease in cell proliferation and in the production of inflammatory cytokines (IFN-γ and TNF-α) in response to malaria antigens.
Repeated malaria infection was also associated with the upregulation of immunoregulatory pathways—increased expression of genes such as HAVCR2, FCRL6, LYN, BATF, and B3GAT1—that dampen the immune response.
Children with these characteristics were less likely than their peers to exhibit clinical symptoms upon subsequent malaria infection.
So it seems the depletion of Vδ2+ γδ T cells is beneficial in some ways and detrimental in others, said Margaret Feeney, MD, of UCSF. Individuals may no longer suffer symptoms, but they might not clear the parasite and could remain infectious.
Although this discovery has not provided a disease-fighting strategy as of yet, it does point to further avenues of study, according to Dr Feeney.
“We want to understand whether this is a generalizable phenomenon that also occurs among those who are first exposed to malaria as adults and in regions where malaria incidence is lower,” she said.
Dr Feeney speculates that malaria infection, by reshaping immune responses, might influence a person’s susceptibility to, and protection from, other infectious diseases.
Nanoparticle may have multiple cancer applications

Credit: PNAS
A new type of nanoparticle (NP) could aid the diagnosis and treatment of cancers, according to research published in Nature Communications.
Built on an easy-to-make polymer, these particles can be used as contrast agents to light up tumors for MRI and PET scans or to deliver chemotherapy and other treatments to cancer cells.
Furthermore, in vivo experiments showed the particles are biocompatible and elicit minimal side effects.
“These are amazingly useful particles,” said study author Yuanpei Li, PhD, of the UC Davis Comprehensive Cancer Center in Sacramento, California.
“As a contrast agent, they make tumors easier to see on MRI and other scans. We can also use them as vehicles to deliver chemotherapy directly to tumors, apply light to make the nanoparticles release singlet oxygen (photodynamic therapy), or use a laser to heat them (photothermal therapy)—all proven ways to destroy tumors.”
These NPs are built on a porphyrin/cholic acid polymer. Porphyrins are common organic compounds, and cholic acid is produced by the liver.
To further stabilize the particles, the researchers added the amino acid cysteine (creating CNPs), which prevents them from prematurely releasing their therapeutic payload when exposed to blood proteins and other barriers.
Therapeutic applications
The researchers tested the CNPs, both in vitro and in vivo, for a wide range of tasks. On the therapeutic side, the particles effectively transported anticancer drugs, such as doxorubicin.
CNPs carrying doxorubicin provided excellent cancer control in animals, with minimal side effects.
Even when kept in the blood for many hours, CNPs only released small amounts of the drug. However, when exposed to light or agents such as glutathione, they readily released their payloads.
The researchers showed that, when exposed to a single wavelength of light, the CNPs could generate heat or produce singlet oxygen to destroy tumor cells.
Imaging applications
CNPs offer a number of advantages to enhance imaging, according to the researchers. The particles readily chelate imaging agents and can remain in the body for long periods.
In animal studies, CNPs largely accumulated in tumors, rather than in normal tissue. So they dramatically enhanced tumor contrast for MRI and may also be promising for PET-MRI scans, the researchers said.
“These particles can combine imaging and therapeutics,” Dr Li noted. “We could potentially use them to simultaneously deliver treatment and monitor treatment efficacy.”
The researchers are now conducting additional preclinical studies with the CNPs. If all goes well, they will proceed to human trials. In the meantime, the team is excited about these capabilities.
“This is the first nanoparticle to perform so many different jobs,” Dr Li said. “From delivering chemo, photodynamic, and photothermal therapies, to enhancing diagnostic imaging, it’s the complete package.”

Credit: PNAS
A new type of nanoparticle (NP) could aid the diagnosis and treatment of cancers, according to research published in Nature Communications.
Built on an easy-to-make polymer, these particles can be used as contrast agents to light up tumors for MRI and PET scans or to deliver chemotherapy and other treatments to cancer cells.
Furthermore, in vivo experiments showed the particles are biocompatible and elicit minimal side effects.
“These are amazingly useful particles,” said study author Yuanpei Li, PhD, of the UC Davis Comprehensive Cancer Center in Sacramento, California.
“As a contrast agent, they make tumors easier to see on MRI and other scans. We can also use them as vehicles to deliver chemotherapy directly to tumors, apply light to make the nanoparticles release singlet oxygen (photodynamic therapy), or use a laser to heat them (photothermal therapy)—all proven ways to destroy tumors.”
These NPs are built on a porphyrin/cholic acid polymer. Porphyrins are common organic compounds, and cholic acid is produced by the liver.
To further stabilize the particles, the researchers added the amino acid cysteine (creating CNPs), which prevents them from prematurely releasing their therapeutic payload when exposed to blood proteins and other barriers.
Therapeutic applications
The researchers tested the CNPs, both in vitro and in vivo, for a wide range of tasks. On the therapeutic side, the particles effectively transported anticancer drugs, such as doxorubicin.
CNPs carrying doxorubicin provided excellent cancer control in animals, with minimal side effects.
Even when kept in the blood for many hours, CNPs only released small amounts of the drug. However, when exposed to light or agents such as glutathione, they readily released their payloads.
The researchers showed that, when exposed to a single wavelength of light, the CNPs could generate heat or produce singlet oxygen to destroy tumor cells.
Imaging applications
CNPs offer a number of advantages to enhance imaging, according to the researchers. The particles readily chelate imaging agents and can remain in the body for long periods.
In animal studies, CNPs largely accumulated in tumors, rather than in normal tissue. So they dramatically enhanced tumor contrast for MRI and may also be promising for PET-MRI scans, the researchers said.
“These particles can combine imaging and therapeutics,” Dr Li noted. “We could potentially use them to simultaneously deliver treatment and monitor treatment efficacy.”
The researchers are now conducting additional preclinical studies with the CNPs. If all goes well, they will proceed to human trials. In the meantime, the team is excited about these capabilities.
“This is the first nanoparticle to perform so many different jobs,” Dr Li said. “From delivering chemo, photodynamic, and photothermal therapies, to enhancing diagnostic imaging, it’s the complete package.”

Credit: PNAS
A new type of nanoparticle (NP) could aid the diagnosis and treatment of cancers, according to research published in Nature Communications.
Built on an easy-to-make polymer, these particles can be used as contrast agents to light up tumors for MRI and PET scans or to deliver chemotherapy and other treatments to cancer cells.
Furthermore, in vivo experiments showed the particles are biocompatible and elicit minimal side effects.
“These are amazingly useful particles,” said study author Yuanpei Li, PhD, of the UC Davis Comprehensive Cancer Center in Sacramento, California.
“As a contrast agent, they make tumors easier to see on MRI and other scans. We can also use them as vehicles to deliver chemotherapy directly to tumors, apply light to make the nanoparticles release singlet oxygen (photodynamic therapy), or use a laser to heat them (photothermal therapy)—all proven ways to destroy tumors.”
These NPs are built on a porphyrin/cholic acid polymer. Porphyrins are common organic compounds, and cholic acid is produced by the liver.
To further stabilize the particles, the researchers added the amino acid cysteine (creating CNPs), which prevents them from prematurely releasing their therapeutic payload when exposed to blood proteins and other barriers.
Therapeutic applications
The researchers tested the CNPs, both in vitro and in vivo, for a wide range of tasks. On the therapeutic side, the particles effectively transported anticancer drugs, such as doxorubicin.
CNPs carrying doxorubicin provided excellent cancer control in animals, with minimal side effects.
Even when kept in the blood for many hours, CNPs only released small amounts of the drug. However, when exposed to light or agents such as glutathione, they readily released their payloads.
The researchers showed that, when exposed to a single wavelength of light, the CNPs could generate heat or produce singlet oxygen to destroy tumor cells.
Imaging applications
CNPs offer a number of advantages to enhance imaging, according to the researchers. The particles readily chelate imaging agents and can remain in the body for long periods.
In animal studies, CNPs largely accumulated in tumors, rather than in normal tissue. So they dramatically enhanced tumor contrast for MRI and may also be promising for PET-MRI scans, the researchers said.
“These particles can combine imaging and therapeutics,” Dr Li noted. “We could potentially use them to simultaneously deliver treatment and monitor treatment efficacy.”
The researchers are now conducting additional preclinical studies with the CNPs. If all goes well, they will proceed to human trials. In the meantime, the team is excited about these capabilities.
“This is the first nanoparticle to perform so many different jobs,” Dr Li said. “From delivering chemo, photodynamic, and photothermal therapies, to enhancing diagnostic imaging, it’s the complete package.”
RIKEN’s initial STAP cell experiments failed

Investigators at the Japanese research institute RIKEN have failed to create STAP (stimulus-triggered acquisition of pluripotency) cells in the experiments they’ve conducted thus far.
However, the group plans to continue its attempts to replicate the STAP cell phenomenon—inducing pluripotency in somatic cells by exposing them to stress—until next March.
So far, the group has failed to create STAP cells by exposing cells from newborn C57BL/6 mice to a low-pH environment.
Going forward, the researchers plan to conduct their experiments in another mouse strain. They also intend to alter the methods of stressing the cells.
RIKEN investigator Haruko Obokata, PhD, and her colleagues initially reported the STAP cell phenomenon in an article and a letter published in Nature last January.
Not long after the papers were published, members of the scientific community began to question the validity of the research. They voiced concerns about published images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation, ultimately concluding that Dr Obokata was guilty of misconduct, and some of her colleagues—including the recently deceased Yoshiki Sasai, MD, PhD—were guilty of negligence.
RIKEN also called for the papers to be retracted, and, in July, they were.
Throughout these proceedings, Dr Obokata insisted the STAP cell phenomenon is real. To investigate this claim, RIKEN organized a group of researchers to recreate Dr Obokata’s experiments.
The group has performed 22 experiments using different types of stress and cells from different tissues in C57BL/6 mice, but they have not reproduced the STAP cell phenomenon described in the Nature papers. (A report on these attempts is available in Japanese.)
Still, the investigators are continuing with their experiments and hope to have definitive results by March.

Investigators at the Japanese research institute RIKEN have failed to create STAP (stimulus-triggered acquisition of pluripotency) cells in the experiments they’ve conducted thus far.
However, the group plans to continue its attempts to replicate the STAP cell phenomenon—inducing pluripotency in somatic cells by exposing them to stress—until next March.
So far, the group has failed to create STAP cells by exposing cells from newborn C57BL/6 mice to a low-pH environment.
Going forward, the researchers plan to conduct their experiments in another mouse strain. They also intend to alter the methods of stressing the cells.
RIKEN investigator Haruko Obokata, PhD, and her colleagues initially reported the STAP cell phenomenon in an article and a letter published in Nature last January.
Not long after the papers were published, members of the scientific community began to question the validity of the research. They voiced concerns about published images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation, ultimately concluding that Dr Obokata was guilty of misconduct, and some of her colleagues—including the recently deceased Yoshiki Sasai, MD, PhD—were guilty of negligence.
RIKEN also called for the papers to be retracted, and, in July, they were.
Throughout these proceedings, Dr Obokata insisted the STAP cell phenomenon is real. To investigate this claim, RIKEN organized a group of researchers to recreate Dr Obokata’s experiments.
The group has performed 22 experiments using different types of stress and cells from different tissues in C57BL/6 mice, but they have not reproduced the STAP cell phenomenon described in the Nature papers. (A report on these attempts is available in Japanese.)
Still, the investigators are continuing with their experiments and hope to have definitive results by March.

Investigators at the Japanese research institute RIKEN have failed to create STAP (stimulus-triggered acquisition of pluripotency) cells in the experiments they’ve conducted thus far.
However, the group plans to continue its attempts to replicate the STAP cell phenomenon—inducing pluripotency in somatic cells by exposing them to stress—until next March.
So far, the group has failed to create STAP cells by exposing cells from newborn C57BL/6 mice to a low-pH environment.
Going forward, the researchers plan to conduct their experiments in another mouse strain. They also intend to alter the methods of stressing the cells.
RIKEN investigator Haruko Obokata, PhD, and her colleagues initially reported the STAP cell phenomenon in an article and a letter published in Nature last January.
Not long after the papers were published, members of the scientific community began to question the validity of the research. They voiced concerns about published images, possible plagiarism, and an inability to replicate the experiments described.
So RIKEN launched an investigation, ultimately concluding that Dr Obokata was guilty of misconduct, and some of her colleagues—including the recently deceased Yoshiki Sasai, MD, PhD—were guilty of negligence.
RIKEN also called for the papers to be retracted, and, in July, they were.
Throughout these proceedings, Dr Obokata insisted the STAP cell phenomenon is real. To investigate this claim, RIKEN organized a group of researchers to recreate Dr Obokata’s experiments.
The group has performed 22 experiments using different types of stress and cells from different tissues in C57BL/6 mice, but they have not reproduced the STAP cell phenomenon described in the Nature papers. (A report on these attempts is available in Japanese.)
Still, the investigators are continuing with their experiments and hope to have definitive results by March.
EPO may cut risk of brain abnormalities in preterm infants
Credit: Bertrand Devouard
Erythropoietin (EPO) can reduce the risk of brain injury in preterm infants, a new study suggests.
Infants who received 3 doses of EPO within 42 hours of birth were less likely than their untreated peers to have abnormal scores for white matter injury, white matter signal intensity, periventricular white matter loss, and gray matter injury.
Russia Ha-Vinh Leuchter, MD, of the University Hospital of Geneva in Switzerland, and her colleagues recounted these findings in JAMA.
The researchers evaluated 495 infants who were born in Switzerland between 2005 and 2012—at 26 weeks of gestation to 31 weeks and 6 days of gestation.
The children were randomized to receive EPO (n=256) or placebo (n=239) intravenously at 3 time points: before they reached 3 hours of age, at 12 to 18 hours after birth, and at 36 to 42 hours after birth.
Due to the limited availability of MRI, the researchers were only able to assess brain injury in a nonrandomized subset of 165 infants. Seventy-seven of these children received EPO, and 88 received placebo.
The patients underwent MRI at a median gestation age of 40 weeks and 5 days (range, 35 weeks and 5 days to 44 weeks and 6 days).
“We found that the brains of the children who had received the treatment had much less damage than those in the control group,” Dr Ha-Vinh Leuchter said. “This is the first time that the beneficial effect of the EPO hormone on the brains of premature babies has been shown.”
Specifically, the results showed that, at term-equivalent age, infants treated with EPO were less likely than controls to have abnormal scores for white matter injury (22% vs 36%), white matter signal intensity (3% vs 11%), periventricular white matter loss (18% vs 33%), and gray matter injury (7% vs 19%).
These results are only the first part of this study, according to the researchers. The second—and main—part of the work will focus on the neurocognitive development of these children, who will take part in various developmental tests at 2 and 5 years of age.
“[The tests] should confirm the effect that EPO treatment has on the neurodevelopmental disabilities that very premature babies often show during their infancy,” said Petra Hüppi, MD, also of the University Hospital of Geneva.
“If this does turn out to be the case, we will have taken an important step in preventing brain damage and its long-term consequences in premature babies.”

Credit: Bertrand Devouard
Erythropoietin (EPO) can reduce the risk of brain injury in preterm infants, a new study suggests.
Infants who received 3 doses of EPO within 42 hours of birth were less likely than their untreated peers to have abnormal scores for white matter injury, white matter signal intensity, periventricular white matter loss, and gray matter injury.
Russia Ha-Vinh Leuchter, MD, of the University Hospital of Geneva in Switzerland, and her colleagues recounted these findings in JAMA.
The researchers evaluated 495 infants who were born in Switzerland between 2005 and 2012—at 26 weeks of gestation to 31 weeks and 6 days of gestation.
The children were randomized to receive EPO (n=256) or placebo (n=239) intravenously at 3 time points: before they reached 3 hours of age, at 12 to 18 hours after birth, and at 36 to 42 hours after birth.
Due to the limited availability of MRI, the researchers were only able to assess brain injury in a nonrandomized subset of 165 infants. Seventy-seven of these children received EPO, and 88 received placebo.
The patients underwent MRI at a median gestation age of 40 weeks and 5 days (range, 35 weeks and 5 days to 44 weeks and 6 days).
“We found that the brains of the children who had received the treatment had much less damage than those in the control group,” Dr Ha-Vinh Leuchter said. “This is the first time that the beneficial effect of the EPO hormone on the brains of premature babies has been shown.”
Specifically, the results showed that, at term-equivalent age, infants treated with EPO were less likely than controls to have abnormal scores for white matter injury (22% vs 36%), white matter signal intensity (3% vs 11%), periventricular white matter loss (18% vs 33%), and gray matter injury (7% vs 19%).
These results are only the first part of this study, according to the researchers. The second—and main—part of the work will focus on the neurocognitive development of these children, who will take part in various developmental tests at 2 and 5 years of age.
“[The tests] should confirm the effect that EPO treatment has on the neurodevelopmental disabilities that very premature babies often show during their infancy,” said Petra Hüppi, MD, also of the University Hospital of Geneva.
“If this does turn out to be the case, we will have taken an important step in preventing brain damage and its long-term consequences in premature babies.”

Credit: Bertrand Devouard
Erythropoietin (EPO) can reduce the risk of brain injury in preterm infants, a new study suggests.
Infants who received 3 doses of EPO within 42 hours of birth were less likely than their untreated peers to have abnormal scores for white matter injury, white matter signal intensity, periventricular white matter loss, and gray matter injury.
Russia Ha-Vinh Leuchter, MD, of the University Hospital of Geneva in Switzerland, and her colleagues recounted these findings in JAMA.
The researchers evaluated 495 infants who were born in Switzerland between 2005 and 2012—at 26 weeks of gestation to 31 weeks and 6 days of gestation.
The children were randomized to receive EPO (n=256) or placebo (n=239) intravenously at 3 time points: before they reached 3 hours of age, at 12 to 18 hours after birth, and at 36 to 42 hours after birth.
Due to the limited availability of MRI, the researchers were only able to assess brain injury in a nonrandomized subset of 165 infants. Seventy-seven of these children received EPO, and 88 received placebo.
The patients underwent MRI at a median gestation age of 40 weeks and 5 days (range, 35 weeks and 5 days to 44 weeks and 6 days).
“We found that the brains of the children who had received the treatment had much less damage than those in the control group,” Dr Ha-Vinh Leuchter said. “This is the first time that the beneficial effect of the EPO hormone on the brains of premature babies has been shown.”
Specifically, the results showed that, at term-equivalent age, infants treated with EPO were less likely than controls to have abnormal scores for white matter injury (22% vs 36%), white matter signal intensity (3% vs 11%), periventricular white matter loss (18% vs 33%), and gray matter injury (7% vs 19%).
These results are only the first part of this study, according to the researchers. The second—and main—part of the work will focus on the neurocognitive development of these children, who will take part in various developmental tests at 2 and 5 years of age.
“[The tests] should confirm the effect that EPO treatment has on the neurodevelopmental disabilities that very premature babies often show during their infancy,” said Petra Hüppi, MD, also of the University Hospital of Geneva.
“If this does turn out to be the case, we will have taken an important step in preventing brain damage and its long-term consequences in premature babies.”