User login
Study reveals potential targets for MYC-dependent cancers

Credit: Juha Klefstrom
New research suggests the MYC protein drives cell growth by inhibiting a handful of genes involved in DNA packaging and cell death.
The study showed that MYC works through a microRNA to suppress the genes’ expression.
This marks the first time that a subset of MYC-controlled genes have been identified as critical players in the protein’s cancer-causing function, and it points to new therapeutic targets for MYC-dependent cancers.
“This is a different way of thinking about the roles of microRNA and chromatin packaging in cancer,” said Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
“We were very surprised to learn that the overexpression of one microRNA can mimic the cancerous effect of MYC.”
Dr Felsher and his colleagues reported this discovery in Cancer Cell.
The team noted that MYC overexpression has been known to prompt an increase in the levels of a microRNA family called miR-17-92.
“People have known for several years that MYC regulates the expression of microRNAs,” Dr Felsher said. “But it wasn’t clear how this was related to MYC’s oncogenic function.”
To gain some insight, Dr Felsher and his colleagues analyzed MYC-dependent cancer cells in vitro and in vivo.
The cells in which miR-17-92 expression was locked in the “on” position kept dividing even when MYC expression was blocked. This suggested that MYC works through the microRNA family to exert its cancer-causing effects.
The researchers then looked for an overlap among genes affected by MYC overexpression and those affected by miR-17-92. There were about 401 genes whose expression was either increased or suppressed by both MYC and miR-17-92.
The team chose to focus on genes that were suppressed because these genes exhibited, on average, many more binding sites for the microRNAs. They further narrowed their panel down to 15 genes regulated by more than one miR-17-92 binding site.
Of these genes, 5 stood out. Four of them—Sin3b, Hbp1, Suv420h1, and Btg1—encode proteins known to regulate chromatin packaging.
These 4 proteins affect cell proliferation and senescence by regulating gene accessibility within the chromatin. They had never before been identified as MYC or miR-17-92 targets.
The fifth gene encodes the apoptotic protein Bim. Previous research suggested that Bim expression is affected by miR-17-92.
All 5 of the proteins are known to affect either cellular proliferation, entry into senescence, or apoptosis, in part by granting or prohibiting access to genes in tightly packaged stretches of DNA in the chromatin.
“MYC is still a general amplifier of gene transcription and expression,” Dr Felsher said. “But our study shows that the maintenance of the cancerous state relies on a more focused mechanism.”
Lastly, the researchers showed that suppressing the expression of the 5 target genes, effectively mimicking MYC overexpression, partially mitigates the effect of MYC deactivation.
Up to 30% of MYC-dependent cancer cells in culture continued to grow—compared to 11% of control cells—in the absence of MYC expression. And tumors in mice either failed to regress or recurred within a few weeks.
“One of the biggest unanswered questions in oncology is how oncogenes cause cancer, and whether you can replace an oncogene with another gene product,” Dr Felsher said.
“These experiments begin to reveal how MYC affects the self-renewal decisions of cells. They may also help us target those aspects of MYC overexpression that contribute to the cancer phenotype.” ![]()

Credit: Juha Klefstrom
New research suggests the MYC protein drives cell growth by inhibiting a handful of genes involved in DNA packaging and cell death.
The study showed that MYC works through a microRNA to suppress the genes’ expression.
This marks the first time that a subset of MYC-controlled genes have been identified as critical players in the protein’s cancer-causing function, and it points to new therapeutic targets for MYC-dependent cancers.
“This is a different way of thinking about the roles of microRNA and chromatin packaging in cancer,” said Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
“We were very surprised to learn that the overexpression of one microRNA can mimic the cancerous effect of MYC.”
Dr Felsher and his colleagues reported this discovery in Cancer Cell.
The team noted that MYC overexpression has been known to prompt an increase in the levels of a microRNA family called miR-17-92.
“People have known for several years that MYC regulates the expression of microRNAs,” Dr Felsher said. “But it wasn’t clear how this was related to MYC’s oncogenic function.”
To gain some insight, Dr Felsher and his colleagues analyzed MYC-dependent cancer cells in vitro and in vivo.
The cells in which miR-17-92 expression was locked in the “on” position kept dividing even when MYC expression was blocked. This suggested that MYC works through the microRNA family to exert its cancer-causing effects.
The researchers then looked for an overlap among genes affected by MYC overexpression and those affected by miR-17-92. There were about 401 genes whose expression was either increased or suppressed by both MYC and miR-17-92.
The team chose to focus on genes that were suppressed because these genes exhibited, on average, many more binding sites for the microRNAs. They further narrowed their panel down to 15 genes regulated by more than one miR-17-92 binding site.
Of these genes, 5 stood out. Four of them—Sin3b, Hbp1, Suv420h1, and Btg1—encode proteins known to regulate chromatin packaging.
These 4 proteins affect cell proliferation and senescence by regulating gene accessibility within the chromatin. They had never before been identified as MYC or miR-17-92 targets.
The fifth gene encodes the apoptotic protein Bim. Previous research suggested that Bim expression is affected by miR-17-92.
All 5 of the proteins are known to affect either cellular proliferation, entry into senescence, or apoptosis, in part by granting or prohibiting access to genes in tightly packaged stretches of DNA in the chromatin.
“MYC is still a general amplifier of gene transcription and expression,” Dr Felsher said. “But our study shows that the maintenance of the cancerous state relies on a more focused mechanism.”
Lastly, the researchers showed that suppressing the expression of the 5 target genes, effectively mimicking MYC overexpression, partially mitigates the effect of MYC deactivation.
Up to 30% of MYC-dependent cancer cells in culture continued to grow—compared to 11% of control cells—in the absence of MYC expression. And tumors in mice either failed to regress or recurred within a few weeks.
“One of the biggest unanswered questions in oncology is how oncogenes cause cancer, and whether you can replace an oncogene with another gene product,” Dr Felsher said.
“These experiments begin to reveal how MYC affects the self-renewal decisions of cells. They may also help us target those aspects of MYC overexpression that contribute to the cancer phenotype.” ![]()

Credit: Juha Klefstrom
New research suggests the MYC protein drives cell growth by inhibiting a handful of genes involved in DNA packaging and cell death.
The study showed that MYC works through a microRNA to suppress the genes’ expression.
This marks the first time that a subset of MYC-controlled genes have been identified as critical players in the protein’s cancer-causing function, and it points to new therapeutic targets for MYC-dependent cancers.
“This is a different way of thinking about the roles of microRNA and chromatin packaging in cancer,” said Dean Felsher, MD, PhD, of the Stanford University School of Medicine in California.
“We were very surprised to learn that the overexpression of one microRNA can mimic the cancerous effect of MYC.”
Dr Felsher and his colleagues reported this discovery in Cancer Cell.
The team noted that MYC overexpression has been known to prompt an increase in the levels of a microRNA family called miR-17-92.
“People have known for several years that MYC regulates the expression of microRNAs,” Dr Felsher said. “But it wasn’t clear how this was related to MYC’s oncogenic function.”
To gain some insight, Dr Felsher and his colleagues analyzed MYC-dependent cancer cells in vitro and in vivo.
The cells in which miR-17-92 expression was locked in the “on” position kept dividing even when MYC expression was blocked. This suggested that MYC works through the microRNA family to exert its cancer-causing effects.
The researchers then looked for an overlap among genes affected by MYC overexpression and those affected by miR-17-92. There were about 401 genes whose expression was either increased or suppressed by both MYC and miR-17-92.
The team chose to focus on genes that were suppressed because these genes exhibited, on average, many more binding sites for the microRNAs. They further narrowed their panel down to 15 genes regulated by more than one miR-17-92 binding site.
Of these genes, 5 stood out. Four of them—Sin3b, Hbp1, Suv420h1, and Btg1—encode proteins known to regulate chromatin packaging.
These 4 proteins affect cell proliferation and senescence by regulating gene accessibility within the chromatin. They had never before been identified as MYC or miR-17-92 targets.
The fifth gene encodes the apoptotic protein Bim. Previous research suggested that Bim expression is affected by miR-17-92.
All 5 of the proteins are known to affect either cellular proliferation, entry into senescence, or apoptosis, in part by granting or prohibiting access to genes in tightly packaged stretches of DNA in the chromatin.
“MYC is still a general amplifier of gene transcription and expression,” Dr Felsher said. “But our study shows that the maintenance of the cancerous state relies on a more focused mechanism.”
Lastly, the researchers showed that suppressing the expression of the 5 target genes, effectively mimicking MYC overexpression, partially mitigates the effect of MYC deactivation.
Up to 30% of MYC-dependent cancer cells in culture continued to grow—compared to 11% of control cells—in the absence of MYC expression. And tumors in mice either failed to regress or recurred within a few weeks.
“One of the biggest unanswered questions in oncology is how oncogenes cause cancer, and whether you can replace an oncogene with another gene product,” Dr Felsher said.
“These experiments begin to reveal how MYC affects the self-renewal decisions of cells. They may also help us target those aspects of MYC overexpression that contribute to the cancer phenotype.” ![]()
Antibiotic recalled due to presence of particulates

Cubist Pharmaceuticals, Inc. is recalling 9 lots of the antibiotic Cubicin (daptomycin for injection), following complaints of foreign particulate matter in reconstituted vials.
Particulate matter in an intravenous drug poses a risk of thromboembolism and pulmonary embolism.
Other risks include phlebitis, the mechanical blocking of the capillaries or arterioles, the activation of platelets, the generation of microthrombi, and the formation of granulomas.
To date, there have been no adverse events associated with complaints of particulate matter from the 9 lots of Cubicin being recalled.
Cubicin is an intravenous product indicated for the treatment of skin infections and certain blood stream infections. The drug was distributed throughout the US, so the recall is nationwide.
The recall includes the following lots of Cubicin 500 mg (NDC 67919-011-01, UPC 3 67919-011-01 6):
Lot # Expiration date Ship dates
CDC203 DEC 2015 9/2/13 through 9/24/13
CDC207 JAN 2016 9/16/13 through 10/15/13
CDC213 FEB 2016 10/1/13 through 10/7/13
CDC217 MAR 2016 12/2/13 through 12/11/13
CDC226 APR 2016 7/29/13 through 8/26/13
CDC234 MAY 2016 8/26/13 through 9/19/13
CDC235 MAY 2016 9/19/13 through 10/17/13
CDC243 JUL 2016 10/17/13 through 11/12/13
CDC246 JUL 2016 11/12/13 through 12/2/13
Cubist Pharmaceuticals is notifying customers of this recall by letter and phone. Customers with product from the recalled lots should quarantine the product and discontinue distribution.
To arrange for the return and replacement of product, call Cubist at (855) 534-8309 between the hours of 9 am and 7 pm EDT, Monday through Friday.
Healthcare professionals and pharmacists with medical questions regarding this recall can contact Cubist Medical Information at (877) 282-4786 between the hours of 8 am and 5:30 pm EDT, Monday through Friday.
Adverse events or quality problems associated with the use of this product can be reported to the Food and Drug Administration’s MedWatch Adverse Events Program. ![]()

Cubist Pharmaceuticals, Inc. is recalling 9 lots of the antibiotic Cubicin (daptomycin for injection), following complaints of foreign particulate matter in reconstituted vials.
Particulate matter in an intravenous drug poses a risk of thromboembolism and pulmonary embolism.
Other risks include phlebitis, the mechanical blocking of the capillaries or arterioles, the activation of platelets, the generation of microthrombi, and the formation of granulomas.
To date, there have been no adverse events associated with complaints of particulate matter from the 9 lots of Cubicin being recalled.
Cubicin is an intravenous product indicated for the treatment of skin infections and certain blood stream infections. The drug was distributed throughout the US, so the recall is nationwide.
The recall includes the following lots of Cubicin 500 mg (NDC 67919-011-01, UPC 3 67919-011-01 6):
Lot # Expiration date Ship dates
CDC203 DEC 2015 9/2/13 through 9/24/13
CDC207 JAN 2016 9/16/13 through 10/15/13
CDC213 FEB 2016 10/1/13 through 10/7/13
CDC217 MAR 2016 12/2/13 through 12/11/13
CDC226 APR 2016 7/29/13 through 8/26/13
CDC234 MAY 2016 8/26/13 through 9/19/13
CDC235 MAY 2016 9/19/13 through 10/17/13
CDC243 JUL 2016 10/17/13 through 11/12/13
CDC246 JUL 2016 11/12/13 through 12/2/13
Cubist Pharmaceuticals is notifying customers of this recall by letter and phone. Customers with product from the recalled lots should quarantine the product and discontinue distribution.
To arrange for the return and replacement of product, call Cubist at (855) 534-8309 between the hours of 9 am and 7 pm EDT, Monday through Friday.
Healthcare professionals and pharmacists with medical questions regarding this recall can contact Cubist Medical Information at (877) 282-4786 between the hours of 8 am and 5:30 pm EDT, Monday through Friday.
Adverse events or quality problems associated with the use of this product can be reported to the Food and Drug Administration’s MedWatch Adverse Events Program. ![]()

Cubist Pharmaceuticals, Inc. is recalling 9 lots of the antibiotic Cubicin (daptomycin for injection), following complaints of foreign particulate matter in reconstituted vials.
Particulate matter in an intravenous drug poses a risk of thromboembolism and pulmonary embolism.
Other risks include phlebitis, the mechanical blocking of the capillaries or arterioles, the activation of platelets, the generation of microthrombi, and the formation of granulomas.
To date, there have been no adverse events associated with complaints of particulate matter from the 9 lots of Cubicin being recalled.
Cubicin is an intravenous product indicated for the treatment of skin infections and certain blood stream infections. The drug was distributed throughout the US, so the recall is nationwide.
The recall includes the following lots of Cubicin 500 mg (NDC 67919-011-01, UPC 3 67919-011-01 6):
Lot # Expiration date Ship dates
CDC203 DEC 2015 9/2/13 through 9/24/13
CDC207 JAN 2016 9/16/13 through 10/15/13
CDC213 FEB 2016 10/1/13 through 10/7/13
CDC217 MAR 2016 12/2/13 through 12/11/13
CDC226 APR 2016 7/29/13 through 8/26/13
CDC234 MAY 2016 8/26/13 through 9/19/13
CDC235 MAY 2016 9/19/13 through 10/17/13
CDC243 JUL 2016 10/17/13 through 11/12/13
CDC246 JUL 2016 11/12/13 through 12/2/13
Cubist Pharmaceuticals is notifying customers of this recall by letter and phone. Customers with product from the recalled lots should quarantine the product and discontinue distribution.
To arrange for the return and replacement of product, call Cubist at (855) 534-8309 between the hours of 9 am and 7 pm EDT, Monday through Friday.
Healthcare professionals and pharmacists with medical questions regarding this recall can contact Cubist Medical Information at (877) 282-4786 between the hours of 8 am and 5:30 pm EDT, Monday through Friday.
Adverse events or quality problems associated with the use of this product can be reported to the Food and Drug Administration’s MedWatch Adverse Events Program. ![]()
Method reveals unexpected hematopoiesis discovery

in the bone marrow
A new epigenetic profiling technique has allowed researchers to chart histone dynamics during hematopoiesis, and it has produced some surprising results.
The researchers developed a high-sensitivity, indexing-first chromatin immunoprecipitation approach (iChIP) that requires as few as 500 cells for accurate analysis.
The techniques currently in use require millions of cells for accurate detection and analysis, but iCHIP overcomes this limitation.
David Lara-Astiaso, of the Weizmann Institute of Science in Rehovot, Israel, and his colleagues described the new method in Science.
The team used iCHIP to profile the dynamics of 4 chromatin modifications across 16 stages of hematopoietic differentiation.
“Using this powerful approach, we were able to identify the exact DNA regulatory sequences, as well as the various regulatory proteins, that are involved in controlling stem cell fate, casting light on previously unseen parts of the basic program of life,” said Nir Friedman, PhD, of the Hebrew University of Jerusalem in Israel.
The research also suggested that as many as 50% of these regulatory sequences are established and opened during intermediate stages of cell development. This means epigenetics are active at stages in which we thought cell destiny was already set.
“This changes our whole understanding of the process of blood stem cell fate decisions,” Lara-Astiaso said.
“[The discovery suggests] the process is more dynamic and flexible than previously thought, giving the cell slightly more leeway at the later stages in deciding what type of cell to turn into, in case its circumstances change.”
Although this research was conducted on mouse HSCs, the researchers believe the mechanism may hold true for other types of cells as well.
“This research creates a lot of excitement in the field, as it sets the groundwork to study these regulatory elements in humans,” said Assaf Weiner, of the Hebrew University of Jerusalem.
Discovering the exact regulatory DNA sequence controlling the fate of hematopoietic stem cells, as well as understanding the mechanism, holds promise for the development of diagnostic tools and therapeutic interventions, the researchers noted. ![]()

in the bone marrow
A new epigenetic profiling technique has allowed researchers to chart histone dynamics during hematopoiesis, and it has produced some surprising results.
The researchers developed a high-sensitivity, indexing-first chromatin immunoprecipitation approach (iChIP) that requires as few as 500 cells for accurate analysis.
The techniques currently in use require millions of cells for accurate detection and analysis, but iCHIP overcomes this limitation.
David Lara-Astiaso, of the Weizmann Institute of Science in Rehovot, Israel, and his colleagues described the new method in Science.
The team used iCHIP to profile the dynamics of 4 chromatin modifications across 16 stages of hematopoietic differentiation.
“Using this powerful approach, we were able to identify the exact DNA regulatory sequences, as well as the various regulatory proteins, that are involved in controlling stem cell fate, casting light on previously unseen parts of the basic program of life,” said Nir Friedman, PhD, of the Hebrew University of Jerusalem in Israel.
The research also suggested that as many as 50% of these regulatory sequences are established and opened during intermediate stages of cell development. This means epigenetics are active at stages in which we thought cell destiny was already set.
“This changes our whole understanding of the process of blood stem cell fate decisions,” Lara-Astiaso said.
“[The discovery suggests] the process is more dynamic and flexible than previously thought, giving the cell slightly more leeway at the later stages in deciding what type of cell to turn into, in case its circumstances change.”
Although this research was conducted on mouse HSCs, the researchers believe the mechanism may hold true for other types of cells as well.
“This research creates a lot of excitement in the field, as it sets the groundwork to study these regulatory elements in humans,” said Assaf Weiner, of the Hebrew University of Jerusalem.
Discovering the exact regulatory DNA sequence controlling the fate of hematopoietic stem cells, as well as understanding the mechanism, holds promise for the development of diagnostic tools and therapeutic interventions, the researchers noted. ![]()

in the bone marrow
A new epigenetic profiling technique has allowed researchers to chart histone dynamics during hematopoiesis, and it has produced some surprising results.
The researchers developed a high-sensitivity, indexing-first chromatin immunoprecipitation approach (iChIP) that requires as few as 500 cells for accurate analysis.
The techniques currently in use require millions of cells for accurate detection and analysis, but iCHIP overcomes this limitation.
David Lara-Astiaso, of the Weizmann Institute of Science in Rehovot, Israel, and his colleagues described the new method in Science.
The team used iCHIP to profile the dynamics of 4 chromatin modifications across 16 stages of hematopoietic differentiation.
“Using this powerful approach, we were able to identify the exact DNA regulatory sequences, as well as the various regulatory proteins, that are involved in controlling stem cell fate, casting light on previously unseen parts of the basic program of life,” said Nir Friedman, PhD, of the Hebrew University of Jerusalem in Israel.
The research also suggested that as many as 50% of these regulatory sequences are established and opened during intermediate stages of cell development. This means epigenetics are active at stages in which we thought cell destiny was already set.
“This changes our whole understanding of the process of blood stem cell fate decisions,” Lara-Astiaso said.
“[The discovery suggests] the process is more dynamic and flexible than previously thought, giving the cell slightly more leeway at the later stages in deciding what type of cell to turn into, in case its circumstances change.”
Although this research was conducted on mouse HSCs, the researchers believe the mechanism may hold true for other types of cells as well.
“This research creates a lot of excitement in the field, as it sets the groundwork to study these regulatory elements in humans,” said Assaf Weiner, of the Hebrew University of Jerusalem.
Discovering the exact regulatory DNA sequence controlling the fate of hematopoietic stem cells, as well as understanding the mechanism, holds promise for the development of diagnostic tools and therapeutic interventions, the researchers noted. ![]()
New insight into stem cell differentiation
Adam Engler, UC San Diego
Jacobs School of Engineering
The stiffness of the extracellular matrix may play a larger role in stem cell differentiation than we thought, according to a new study.
Scientists have recently suggested that protein tethering and matrix porosity, as well as matrix stiffness and ligand type, regulate stem cell differentiation.
However, new research published in Nature Materials indicates that matrix stiffness regulates stem cell differentiation independently of tethering and porosity.
Adam Engler, PhD, of the University of California, San Diego, and his colleagues discovered that human adipose stromal cells and mesenchymal stromal cells underwent osteogenic differentiation if placed in a stiff hydrogel. But the cells underwent adipogenesis if placed in a soft hydrogel.
The protein binding the stem cell to the hydrogel was not a factor in the differentiation process. Results suggested the protein layer was merely an adhesive.
The researchers found that stem cell differentiation is a response to the mechanical deformation of the hydrogel from the force exerted by the cell. With a series of experiments, the team showed that this happens whether the protein tethering the cell to the matrix is tight, loose, or nonexistent.
Across multiple samples using a stiff matrix, varying the degree of tethering made no significant difference in the rate of osteogenic or adipogenic differentiation.
Likewise, the size of the pores in the matrix had no effect on stem cell differentiation, as long as the stiffness of the hydrogel remained the same.
However, Dr Engler pointed out that matrix stiffness is only “one cue out of dozens that are important in stem cell differentiation.”
“That doesn’t mean the other cues are irrelevant,” he noted. “They may still push the cells into a specific cell type. We have just ruled out porosity and tethering, and further emphasized stiffness in this process.” ![]()

Adam Engler, UC San Diego
Jacobs School of Engineering
The stiffness of the extracellular matrix may play a larger role in stem cell differentiation than we thought, according to a new study.
Scientists have recently suggested that protein tethering and matrix porosity, as well as matrix stiffness and ligand type, regulate stem cell differentiation.
However, new research published in Nature Materials indicates that matrix stiffness regulates stem cell differentiation independently of tethering and porosity.
Adam Engler, PhD, of the University of California, San Diego, and his colleagues discovered that human adipose stromal cells and mesenchymal stromal cells underwent osteogenic differentiation if placed in a stiff hydrogel. But the cells underwent adipogenesis if placed in a soft hydrogel.
The protein binding the stem cell to the hydrogel was not a factor in the differentiation process. Results suggested the protein layer was merely an adhesive.
The researchers found that stem cell differentiation is a response to the mechanical deformation of the hydrogel from the force exerted by the cell. With a series of experiments, the team showed that this happens whether the protein tethering the cell to the matrix is tight, loose, or nonexistent.
Across multiple samples using a stiff matrix, varying the degree of tethering made no significant difference in the rate of osteogenic or adipogenic differentiation.
Likewise, the size of the pores in the matrix had no effect on stem cell differentiation, as long as the stiffness of the hydrogel remained the same.
However, Dr Engler pointed out that matrix stiffness is only “one cue out of dozens that are important in stem cell differentiation.”
“That doesn’t mean the other cues are irrelevant,” he noted. “They may still push the cells into a specific cell type. We have just ruled out porosity and tethering, and further emphasized stiffness in this process.” ![]()

Adam Engler, UC San Diego
Jacobs School of Engineering
The stiffness of the extracellular matrix may play a larger role in stem cell differentiation than we thought, according to a new study.
Scientists have recently suggested that protein tethering and matrix porosity, as well as matrix stiffness and ligand type, regulate stem cell differentiation.
However, new research published in Nature Materials indicates that matrix stiffness regulates stem cell differentiation independently of tethering and porosity.
Adam Engler, PhD, of the University of California, San Diego, and his colleagues discovered that human adipose stromal cells and mesenchymal stromal cells underwent osteogenic differentiation if placed in a stiff hydrogel. But the cells underwent adipogenesis if placed in a soft hydrogel.
The protein binding the stem cell to the hydrogel was not a factor in the differentiation process. Results suggested the protein layer was merely an adhesive.
The researchers found that stem cell differentiation is a response to the mechanical deformation of the hydrogel from the force exerted by the cell. With a series of experiments, the team showed that this happens whether the protein tethering the cell to the matrix is tight, loose, or nonexistent.
Across multiple samples using a stiff matrix, varying the degree of tethering made no significant difference in the rate of osteogenic or adipogenic differentiation.
Likewise, the size of the pores in the matrix had no effect on stem cell differentiation, as long as the stiffness of the hydrogel remained the same.
However, Dr Engler pointed out that matrix stiffness is only “one cue out of dozens that are important in stem cell differentiation.”
“That doesn’t mean the other cues are irrelevant,” he noted. “They may still push the cells into a specific cell type. We have just ruled out porosity and tethering, and further emphasized stiffness in this process.” ![]()
Method could speed up cancer diagnosis

Credit: NIGMS
A new technique could enable faster diagnosis of cancer and various prenatal conditions, according to a paper published in Proceedings of the National Academy of Sciences.
The method, known as convex lens-induced confinement (CLIC), allows researchers to load long strands of DNA into a tunable, nanoscale imaging chamber in ways that maintain their structural identity and under conditions that are similar to those in the human body.
CLIC lets researchers map large genomes rapidly and identify specific gene sequences from single cells with single-molecule resolution, a process that is critical to diagnosing diseases like cancer.
“Current practices of genomic analysis typically require tens of thousands of cells worth of genomic material to obtain the information we need, but this new approach works with single cells,” said study author Rob Sladek, MD, of McGill University in Montreal, Canada.
“CLIC will allow researchers to avoid having to spend time stitching together maps of entire genomes, as we do under current techniques, and promises to make genomic analysis a much simpler and more efficient process.”
The CLIC imaging chamber can sit on top of a standard inverted fluorescence microscope, and strands of DNA can be loaded into the chamber from above, which allows the strands to maintain their integrity.
Existing tools used for genomic analysis rely on side-loading DNA under pressure into nanochannels in the imaging chamber. This breaks the DNA molecules into small pieces, making it a challenge to reconstruct the genome.
CLIC, on the other hand, is “like squeezing many soft spaghetti noodles into long, narrow tubes without breaking them,” according to study author Sabrina Leslie, PhD, also of McGill University.
“Once these long strands of DNA are gently squeezed down into nanochannels from a nanoscale bath above, they become effectively rigid, which means that we can map positions along uniformly stretched strands of DNA, while holding them still,” she said.
“This means diagnostics can be performed quickly, one cell at a time, which is critical for diagnosing many prenatal conditions and the onset of cancer.” ![]()

Credit: NIGMS
A new technique could enable faster diagnosis of cancer and various prenatal conditions, according to a paper published in Proceedings of the National Academy of Sciences.
The method, known as convex lens-induced confinement (CLIC), allows researchers to load long strands of DNA into a tunable, nanoscale imaging chamber in ways that maintain their structural identity and under conditions that are similar to those in the human body.
CLIC lets researchers map large genomes rapidly and identify specific gene sequences from single cells with single-molecule resolution, a process that is critical to diagnosing diseases like cancer.
“Current practices of genomic analysis typically require tens of thousands of cells worth of genomic material to obtain the information we need, but this new approach works with single cells,” said study author Rob Sladek, MD, of McGill University in Montreal, Canada.
“CLIC will allow researchers to avoid having to spend time stitching together maps of entire genomes, as we do under current techniques, and promises to make genomic analysis a much simpler and more efficient process.”
The CLIC imaging chamber can sit on top of a standard inverted fluorescence microscope, and strands of DNA can be loaded into the chamber from above, which allows the strands to maintain their integrity.
Existing tools used for genomic analysis rely on side-loading DNA under pressure into nanochannels in the imaging chamber. This breaks the DNA molecules into small pieces, making it a challenge to reconstruct the genome.
CLIC, on the other hand, is “like squeezing many soft spaghetti noodles into long, narrow tubes without breaking them,” according to study author Sabrina Leslie, PhD, also of McGill University.
“Once these long strands of DNA are gently squeezed down into nanochannels from a nanoscale bath above, they become effectively rigid, which means that we can map positions along uniformly stretched strands of DNA, while holding them still,” she said.
“This means diagnostics can be performed quickly, one cell at a time, which is critical for diagnosing many prenatal conditions and the onset of cancer.” ![]()

Credit: NIGMS
A new technique could enable faster diagnosis of cancer and various prenatal conditions, according to a paper published in Proceedings of the National Academy of Sciences.
The method, known as convex lens-induced confinement (CLIC), allows researchers to load long strands of DNA into a tunable, nanoscale imaging chamber in ways that maintain their structural identity and under conditions that are similar to those in the human body.
CLIC lets researchers map large genomes rapidly and identify specific gene sequences from single cells with single-molecule resolution, a process that is critical to diagnosing diseases like cancer.
“Current practices of genomic analysis typically require tens of thousands of cells worth of genomic material to obtain the information we need, but this new approach works with single cells,” said study author Rob Sladek, MD, of McGill University in Montreal, Canada.
“CLIC will allow researchers to avoid having to spend time stitching together maps of entire genomes, as we do under current techniques, and promises to make genomic analysis a much simpler and more efficient process.”
The CLIC imaging chamber can sit on top of a standard inverted fluorescence microscope, and strands of DNA can be loaded into the chamber from above, which allows the strands to maintain their integrity.
Existing tools used for genomic analysis rely on side-loading DNA under pressure into nanochannels in the imaging chamber. This breaks the DNA molecules into small pieces, making it a challenge to reconstruct the genome.
CLIC, on the other hand, is “like squeezing many soft spaghetti noodles into long, narrow tubes without breaking them,” according to study author Sabrina Leslie, PhD, also of McGill University.
“Once these long strands of DNA are gently squeezed down into nanochannels from a nanoscale bath above, they become effectively rigid, which means that we can map positions along uniformly stretched strands of DNA, while holding them still,” she said.
“This means diagnostics can be performed quickly, one cell at a time, which is critical for diagnosing many prenatal conditions and the onset of cancer.” ![]()
Study challenges traditional cancer classification

a tumor sample in a test tube
Credit: Rhoda Baer
Defining cancers by molecular criteria rather than their tissue of origin can provide patients with more accurate diagnoses, researchers have reported in Cell.
The group analyzed the molecular characteristics of more than 3500 samples of 12 different cancers and reclassified them according to the new information.
For 5 of the cancer types, including acute myeloid leukemia (AML), the molecular classification largely matched the tissue-of-origin classification.
For the remaining malignancies, that was not the case.
“This genomic study not only challenges our existing system of classifying cancers based on tissue type, but also provides a massive new data resource for further exploration, as well as a comprehensive list of the molecular features distinguishing each of the newly described cancer classes,” said study author Christopher Benz, MD, of the University of California, San Francisco.
The researchers said each molecular subtype they identified may reflect tumors arising from distinct cell types. For example, the data showed a marked difference between cancers of epithelial and non-epithelial origins.
“We think the subtypes reflect, primarily, the cell of origin,” said study author Joshua Stuart, PhD, of the University of California, Santa Cruz.
“Another factor is the nature of the genomic lesion, and third is the microenvironment of the cell and how surrounding cells influence it. We are disentangling the signals from these different factors so we can gauge each one for its prognostic power.”
Identifying molecular subtypes
The researchers performed an integrative analysis using 5 genome-wide platforms and 1 proteomic platform on 3527 specimens from 12 cancer types.
This included AML, glioblastoma multiforme, serous ovarian carcinoma, colon and rectal adenocarcinomas, lung squamous cell carcinoma, breast cancer, endometrial cancer, renal cell carcinoma, bladder urothelial adenocarcinoma, lung adenocarcinoma, and head and neck squamous cell carcinoma.
The group’s analyses allowed them to classify these cancer types into 11 major cellular/molecular subtypes. Two of the initial 13 subtypes (numbers 11 and 12) were eliminated from further analysis because they included fewer than 10 samples.
Five of the classification types—C5-renal cell carcinoma, C6-endometrial cancer, C9-serous ovarian carcinoma, C10-glioblastoma multiforme, and C13-AML—showed near 1-to-1 relationships with the tissue site of origin. However, there were a few cases of reclassification here and there, such as a case of breast cancer that fell in the AML subtype.
Another subtype stayed pretty true to its tissues of origin. C7-colon adenocarcinoma/rectal adenocarcinoma was composed mainly of colon and rectal adenocarcinomas but also included a case of endometrial cancer.
The C1-lung adenocarcinoma-enriched subtype was predominantly composed of non-small cell lung adenocarcinoma samples. But it also included cases of bladder cancer, breast cancer, colon adenocarcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, renal cell carcinoma, lung squamous cell carcinoma , serous ovarian carcinoma, and endometrial cancer.
The C2-squamous-like subtype consisted largely of head and neck squamous cell carcinoma and lung squamous cell carcinoma but also included bladder urothelial adenocarcinoma and breast cancer.
Breast cancers were further divided into the C3-breast cancer/luminal subtype and the C4-breast cancer/basal subtype. The C4 subtype also included lung adenocarcinoma and lung squamous cell carcinoma.
The researchers noted that breast cancers were present in 7 of the subtype classifications. And while this study confirmed known differences between the subtypes of breast cancer, the team was surprised to discover that basal-like breast cancers actually constitute their own cancer class.
“Even though these basal-like cancers arise in the breast, on the molecular level, they have more in common with ovarian cancers and cancers of squamous-cell origin than with other subtypes of breast cancer,” said study author Christina Yau, PhD, of the University of California, San Francisco.
Like breast cancers, bladder cancers were present in 7 of the subtype classifications. There were 1 or 2 cases in C5, C10, C11, and C12. But most bladder cancer samples fell into 1 of 3 categories: C1-lung adenocarcinoma-enriched, C2-squamous-like, and C8-bladder urothelial adenocarcinoma.
Although the C8-bladder urothelial adenocarcinoma subtype consisted largely of bladder cancer, it also included breast cancer, head and neck squamous cell carcinoma, lung adenocarcinoma, and lung squamous cell carcinoma.
These findings may help explain why patients with bladder cancer “often respond very differently when treated with the same systemic therapy for their seemingly identical cancer type,” Dr Benz said.
In fact, the researchers found the bladder cancers that clustered with other tumor types had a worse prognosis.
Next steps
The researchers noted that follow-up studies are needed to validate these findings, but this analysis lays the groundwork for classifying tumors into molecularly defined subtypes. The new classification scheme could be used to enroll patients in clinical trials and could lead to different treatment options based on molecular subtypes.
“We can now say what the telltale signatures of the subtypes are, so you can classify a patient’s tumor just based on the gene expression data, or just based on mutation data, if that’s what you have,” Dr Stuart said. “Having a molecular map like this could help get a patient into the right clinical trial.”
The researchers believe the percentage of tumors that should be reclassified based on molecular signatures is likely to grow as more samples and tumor types are analyzed. This study suggested that 1 in 10 cancers could be reclassified in clinically meaningful ways, but the researchers said their next analysis will include 21 tumor types instead of 12.
“We’re just appreciating the tip of the iceberg when considering the potential of this multiplatform type of genomic analysis,” Dr Benz said. “It could be that as many as 30% or 50% of cancers need to be reclassified.”
The data sets and results from this study have been made available to other researchers through the Synapse website. ![]()

a tumor sample in a test tube
Credit: Rhoda Baer
Defining cancers by molecular criteria rather than their tissue of origin can provide patients with more accurate diagnoses, researchers have reported in Cell.
The group analyzed the molecular characteristics of more than 3500 samples of 12 different cancers and reclassified them according to the new information.
For 5 of the cancer types, including acute myeloid leukemia (AML), the molecular classification largely matched the tissue-of-origin classification.
For the remaining malignancies, that was not the case.
“This genomic study not only challenges our existing system of classifying cancers based on tissue type, but also provides a massive new data resource for further exploration, as well as a comprehensive list of the molecular features distinguishing each of the newly described cancer classes,” said study author Christopher Benz, MD, of the University of California, San Francisco.
The researchers said each molecular subtype they identified may reflect tumors arising from distinct cell types. For example, the data showed a marked difference between cancers of epithelial and non-epithelial origins.
“We think the subtypes reflect, primarily, the cell of origin,” said study author Joshua Stuart, PhD, of the University of California, Santa Cruz.
“Another factor is the nature of the genomic lesion, and third is the microenvironment of the cell and how surrounding cells influence it. We are disentangling the signals from these different factors so we can gauge each one for its prognostic power.”
Identifying molecular subtypes
The researchers performed an integrative analysis using 5 genome-wide platforms and 1 proteomic platform on 3527 specimens from 12 cancer types.
This included AML, glioblastoma multiforme, serous ovarian carcinoma, colon and rectal adenocarcinomas, lung squamous cell carcinoma, breast cancer, endometrial cancer, renal cell carcinoma, bladder urothelial adenocarcinoma, lung adenocarcinoma, and head and neck squamous cell carcinoma.
The group’s analyses allowed them to classify these cancer types into 11 major cellular/molecular subtypes. Two of the initial 13 subtypes (numbers 11 and 12) were eliminated from further analysis because they included fewer than 10 samples.
Five of the classification types—C5-renal cell carcinoma, C6-endometrial cancer, C9-serous ovarian carcinoma, C10-glioblastoma multiforme, and C13-AML—showed near 1-to-1 relationships with the tissue site of origin. However, there were a few cases of reclassification here and there, such as a case of breast cancer that fell in the AML subtype.
Another subtype stayed pretty true to its tissues of origin. C7-colon adenocarcinoma/rectal adenocarcinoma was composed mainly of colon and rectal adenocarcinomas but also included a case of endometrial cancer.
The C1-lung adenocarcinoma-enriched subtype was predominantly composed of non-small cell lung adenocarcinoma samples. But it also included cases of bladder cancer, breast cancer, colon adenocarcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, renal cell carcinoma, lung squamous cell carcinoma , serous ovarian carcinoma, and endometrial cancer.
The C2-squamous-like subtype consisted largely of head and neck squamous cell carcinoma and lung squamous cell carcinoma but also included bladder urothelial adenocarcinoma and breast cancer.
Breast cancers were further divided into the C3-breast cancer/luminal subtype and the C4-breast cancer/basal subtype. The C4 subtype also included lung adenocarcinoma and lung squamous cell carcinoma.
The researchers noted that breast cancers were present in 7 of the subtype classifications. And while this study confirmed known differences between the subtypes of breast cancer, the team was surprised to discover that basal-like breast cancers actually constitute their own cancer class.
“Even though these basal-like cancers arise in the breast, on the molecular level, they have more in common with ovarian cancers and cancers of squamous-cell origin than with other subtypes of breast cancer,” said study author Christina Yau, PhD, of the University of California, San Francisco.
Like breast cancers, bladder cancers were present in 7 of the subtype classifications. There were 1 or 2 cases in C5, C10, C11, and C12. But most bladder cancer samples fell into 1 of 3 categories: C1-lung adenocarcinoma-enriched, C2-squamous-like, and C8-bladder urothelial adenocarcinoma.
Although the C8-bladder urothelial adenocarcinoma subtype consisted largely of bladder cancer, it also included breast cancer, head and neck squamous cell carcinoma, lung adenocarcinoma, and lung squamous cell carcinoma.
These findings may help explain why patients with bladder cancer “often respond very differently when treated with the same systemic therapy for their seemingly identical cancer type,” Dr Benz said.
In fact, the researchers found the bladder cancers that clustered with other tumor types had a worse prognosis.
Next steps
The researchers noted that follow-up studies are needed to validate these findings, but this analysis lays the groundwork for classifying tumors into molecularly defined subtypes. The new classification scheme could be used to enroll patients in clinical trials and could lead to different treatment options based on molecular subtypes.
“We can now say what the telltale signatures of the subtypes are, so you can classify a patient’s tumor just based on the gene expression data, or just based on mutation data, if that’s what you have,” Dr Stuart said. “Having a molecular map like this could help get a patient into the right clinical trial.”
The researchers believe the percentage of tumors that should be reclassified based on molecular signatures is likely to grow as more samples and tumor types are analyzed. This study suggested that 1 in 10 cancers could be reclassified in clinically meaningful ways, but the researchers said their next analysis will include 21 tumor types instead of 12.
“We’re just appreciating the tip of the iceberg when considering the potential of this multiplatform type of genomic analysis,” Dr Benz said. “It could be that as many as 30% or 50% of cancers need to be reclassified.”
The data sets and results from this study have been made available to other researchers through the Synapse website. ![]()

a tumor sample in a test tube
Credit: Rhoda Baer
Defining cancers by molecular criteria rather than their tissue of origin can provide patients with more accurate diagnoses, researchers have reported in Cell.
The group analyzed the molecular characteristics of more than 3500 samples of 12 different cancers and reclassified them according to the new information.
For 5 of the cancer types, including acute myeloid leukemia (AML), the molecular classification largely matched the tissue-of-origin classification.
For the remaining malignancies, that was not the case.
“This genomic study not only challenges our existing system of classifying cancers based on tissue type, but also provides a massive new data resource for further exploration, as well as a comprehensive list of the molecular features distinguishing each of the newly described cancer classes,” said study author Christopher Benz, MD, of the University of California, San Francisco.
The researchers said each molecular subtype they identified may reflect tumors arising from distinct cell types. For example, the data showed a marked difference between cancers of epithelial and non-epithelial origins.
“We think the subtypes reflect, primarily, the cell of origin,” said study author Joshua Stuart, PhD, of the University of California, Santa Cruz.
“Another factor is the nature of the genomic lesion, and third is the microenvironment of the cell and how surrounding cells influence it. We are disentangling the signals from these different factors so we can gauge each one for its prognostic power.”
Identifying molecular subtypes
The researchers performed an integrative analysis using 5 genome-wide platforms and 1 proteomic platform on 3527 specimens from 12 cancer types.
This included AML, glioblastoma multiforme, serous ovarian carcinoma, colon and rectal adenocarcinomas, lung squamous cell carcinoma, breast cancer, endometrial cancer, renal cell carcinoma, bladder urothelial adenocarcinoma, lung adenocarcinoma, and head and neck squamous cell carcinoma.
The group’s analyses allowed them to classify these cancer types into 11 major cellular/molecular subtypes. Two of the initial 13 subtypes (numbers 11 and 12) were eliminated from further analysis because they included fewer than 10 samples.
Five of the classification types—C5-renal cell carcinoma, C6-endometrial cancer, C9-serous ovarian carcinoma, C10-glioblastoma multiforme, and C13-AML—showed near 1-to-1 relationships with the tissue site of origin. However, there were a few cases of reclassification here and there, such as a case of breast cancer that fell in the AML subtype.
Another subtype stayed pretty true to its tissues of origin. C7-colon adenocarcinoma/rectal adenocarcinoma was composed mainly of colon and rectal adenocarcinomas but also included a case of endometrial cancer.
The C1-lung adenocarcinoma-enriched subtype was predominantly composed of non-small cell lung adenocarcinoma samples. But it also included cases of bladder cancer, breast cancer, colon adenocarcinoma, glioblastoma multiforme, head and neck squamous cell carcinoma, renal cell carcinoma, lung squamous cell carcinoma , serous ovarian carcinoma, and endometrial cancer.
The C2-squamous-like subtype consisted largely of head and neck squamous cell carcinoma and lung squamous cell carcinoma but also included bladder urothelial adenocarcinoma and breast cancer.
Breast cancers were further divided into the C3-breast cancer/luminal subtype and the C4-breast cancer/basal subtype. The C4 subtype also included lung adenocarcinoma and lung squamous cell carcinoma.
The researchers noted that breast cancers were present in 7 of the subtype classifications. And while this study confirmed known differences between the subtypes of breast cancer, the team was surprised to discover that basal-like breast cancers actually constitute their own cancer class.
“Even though these basal-like cancers arise in the breast, on the molecular level, they have more in common with ovarian cancers and cancers of squamous-cell origin than with other subtypes of breast cancer,” said study author Christina Yau, PhD, of the University of California, San Francisco.
Like breast cancers, bladder cancers were present in 7 of the subtype classifications. There were 1 or 2 cases in C5, C10, C11, and C12. But most bladder cancer samples fell into 1 of 3 categories: C1-lung adenocarcinoma-enriched, C2-squamous-like, and C8-bladder urothelial adenocarcinoma.
Although the C8-bladder urothelial adenocarcinoma subtype consisted largely of bladder cancer, it also included breast cancer, head and neck squamous cell carcinoma, lung adenocarcinoma, and lung squamous cell carcinoma.
These findings may help explain why patients with bladder cancer “often respond very differently when treated with the same systemic therapy for their seemingly identical cancer type,” Dr Benz said.
In fact, the researchers found the bladder cancers that clustered with other tumor types had a worse prognosis.
Next steps
The researchers noted that follow-up studies are needed to validate these findings, but this analysis lays the groundwork for classifying tumors into molecularly defined subtypes. The new classification scheme could be used to enroll patients in clinical trials and could lead to different treatment options based on molecular subtypes.
“We can now say what the telltale signatures of the subtypes are, so you can classify a patient’s tumor just based on the gene expression data, or just based on mutation data, if that’s what you have,” Dr Stuart said. “Having a molecular map like this could help get a patient into the right clinical trial.”
The researchers believe the percentage of tumors that should be reclassified based on molecular signatures is likely to grow as more samples and tumor types are analyzed. This study suggested that 1 in 10 cancers could be reclassified in clinically meaningful ways, but the researchers said their next analysis will include 21 tumor types instead of 12.
“We’re just appreciating the tip of the iceberg when considering the potential of this multiplatform type of genomic analysis,” Dr Benz said. “It could be that as many as 30% or 50% of cancers need to be reclassified.”
The data sets and results from this study have been made available to other researchers through the Synapse website. ![]()
Two new tests can detect CJD

Credit: Elise Amendola
Two groups of scientists have developed new tests to diagnose Creutzfeldt-Jakob disease (CJD).
One test uses samples collected from nasal passages to detect sporadic CJD, and the other uses urine samples to identify variant CJD.
The researchers said these tests provide simple methods for differentiating CJD from other diseases and could help prevent the transmission of CJD via blood
transfusions, transplants, or contaminated surgical instruments.
Both tests are described in The New England Journal of Medicine.
Nasal test for sporadic CJD
In one NEJM article, Byron Caughey, PhD, of the National Institute of Allergy and Infectious Diseases in Rockville, Maryland, and his colleagues detailed their results with the nasal test.
The researchers collected 31 nasal samples from patients with sporadic CJD and 43 samples from patients who had other neurologic diseases or no neurologic disease. The team brushed the inside of a subject’s nose to collect olfactory neurons connected to the brain.
Testing these samples allowed the researchers to correctly identify 30 of the 31 sporadic CJD patients (97% sensitivity). The tests also correctly showed negative results for all 43 of the non-CJD patients (100% specificity).
By comparison, tests using cerebral spinal fluid, which is currently used to detect sporadic CJD, were 77% sensitive and 100% specific. And these results took twice as long to obtain.
While continuing to validate the new testing method in CJD patients, the scientists are looking to expand their research to diagnose forms of prion diseases in sheep, cattle, and wildlife. The team also hopes to replace the nasal brush with an even simpler swabbing approach.
Urine test for variant CJD
In another NEJM article, Fabio Moda, PhD, of the University of Texas Medical School at Houston, and his colleagues described results observed with their urine test.
The team noted that the infectious agent in transmissible spongiform encephalopathies appears to be composed exclusively of the misfolded form of the prion protein, PrPSc. So they set out to determine if they could detect PrPSc in the urine of patients with CJD.
The researchers analyzed urine samples from healthy individuals (n=52) and patients with variant CJD (n=68), sporadic CJD (n=14), genetic forms of prion disease (n=4), other neurodegenerative disorders (n=50), and nondegenerative neurologic disorders (n=50).
The group found they could only detect PrPSc in samples from patients with variant CJD. They found “minute quantities” of PrPSc in 13 of the 14 urine samples from variant CJD patients, but PrPSc was not present in any of the samples from the other patients or the healthy individuals.
This suggests the test has a sensitivity of 92.9% and a specificity of 100%. ![]()

Credit: Elise Amendola
Two groups of scientists have developed new tests to diagnose Creutzfeldt-Jakob disease (CJD).
One test uses samples collected from nasal passages to detect sporadic CJD, and the other uses urine samples to identify variant CJD.
The researchers said these tests provide simple methods for differentiating CJD from other diseases and could help prevent the transmission of CJD via blood
transfusions, transplants, or contaminated surgical instruments.
Both tests are described in The New England Journal of Medicine.
Nasal test for sporadic CJD
In one NEJM article, Byron Caughey, PhD, of the National Institute of Allergy and Infectious Diseases in Rockville, Maryland, and his colleagues detailed their results with the nasal test.
The researchers collected 31 nasal samples from patients with sporadic CJD and 43 samples from patients who had other neurologic diseases or no neurologic disease. The team brushed the inside of a subject’s nose to collect olfactory neurons connected to the brain.
Testing these samples allowed the researchers to correctly identify 30 of the 31 sporadic CJD patients (97% sensitivity). The tests also correctly showed negative results for all 43 of the non-CJD patients (100% specificity).
By comparison, tests using cerebral spinal fluid, which is currently used to detect sporadic CJD, were 77% sensitive and 100% specific. And these results took twice as long to obtain.
While continuing to validate the new testing method in CJD patients, the scientists are looking to expand their research to diagnose forms of prion diseases in sheep, cattle, and wildlife. The team also hopes to replace the nasal brush with an even simpler swabbing approach.
Urine test for variant CJD
In another NEJM article, Fabio Moda, PhD, of the University of Texas Medical School at Houston, and his colleagues described results observed with their urine test.
The team noted that the infectious agent in transmissible spongiform encephalopathies appears to be composed exclusively of the misfolded form of the prion protein, PrPSc. So they set out to determine if they could detect PrPSc in the urine of patients with CJD.
The researchers analyzed urine samples from healthy individuals (n=52) and patients with variant CJD (n=68), sporadic CJD (n=14), genetic forms of prion disease (n=4), other neurodegenerative disorders (n=50), and nondegenerative neurologic disorders (n=50).
The group found they could only detect PrPSc in samples from patients with variant CJD. They found “minute quantities” of PrPSc in 13 of the 14 urine samples from variant CJD patients, but PrPSc was not present in any of the samples from the other patients or the healthy individuals.
This suggests the test has a sensitivity of 92.9% and a specificity of 100%. ![]()

Credit: Elise Amendola
Two groups of scientists have developed new tests to diagnose Creutzfeldt-Jakob disease (CJD).
One test uses samples collected from nasal passages to detect sporadic CJD, and the other uses urine samples to identify variant CJD.
The researchers said these tests provide simple methods for differentiating CJD from other diseases and could help prevent the transmission of CJD via blood
transfusions, transplants, or contaminated surgical instruments.
Both tests are described in The New England Journal of Medicine.
Nasal test for sporadic CJD
In one NEJM article, Byron Caughey, PhD, of the National Institute of Allergy and Infectious Diseases in Rockville, Maryland, and his colleagues detailed their results with the nasal test.
The researchers collected 31 nasal samples from patients with sporadic CJD and 43 samples from patients who had other neurologic diseases or no neurologic disease. The team brushed the inside of a subject’s nose to collect olfactory neurons connected to the brain.
Testing these samples allowed the researchers to correctly identify 30 of the 31 sporadic CJD patients (97% sensitivity). The tests also correctly showed negative results for all 43 of the non-CJD patients (100% specificity).
By comparison, tests using cerebral spinal fluid, which is currently used to detect sporadic CJD, were 77% sensitive and 100% specific. And these results took twice as long to obtain.
While continuing to validate the new testing method in CJD patients, the scientists are looking to expand their research to diagnose forms of prion diseases in sheep, cattle, and wildlife. The team also hopes to replace the nasal brush with an even simpler swabbing approach.
Urine test for variant CJD
In another NEJM article, Fabio Moda, PhD, of the University of Texas Medical School at Houston, and his colleagues described results observed with their urine test.
The team noted that the infectious agent in transmissible spongiform encephalopathies appears to be composed exclusively of the misfolded form of the prion protein, PrPSc. So they set out to determine if they could detect PrPSc in the urine of patients with CJD.
The researchers analyzed urine samples from healthy individuals (n=52) and patients with variant CJD (n=68), sporadic CJD (n=14), genetic forms of prion disease (n=4), other neurodegenerative disorders (n=50), and nondegenerative neurologic disorders (n=50).
The group found they could only detect PrPSc in samples from patients with variant CJD. They found “minute quantities” of PrPSc in 13 of the 14 urine samples from variant CJD patients, but PrPSc was not present in any of the samples from the other patients or the healthy individuals.
This suggests the test has a sensitivity of 92.9% and a specificity of 100%.
Gene plays crucial role in cancer development, team says
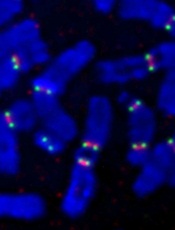
Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded.

Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded.

Credit: Beth A. Sullivan
New research suggests DNA ligase 3 is crucial for the evolutionary processes that drive cancer.
“We have identified a gene that, as cells age, seems to regulate whether the cells become cancerous or not,” said Eric A. Hendrickson, PhD, of the University of Minnesota in Minneapolis.
“This gene has never been identified before in this role, so this makes it a potentially very important therapeutic target.”
Dr Hendrickson and his colleagues recounted this discovery in Cell Reports.
The researchers noted that short, dysfunctional telomeres can fuse, thereby generating dicentric chromosomes and initiating breakage-fusion-bridge cycles. The cells that manage to escape the subsequent crisis have genomic rearrangements that drive clonal evolution and malignant progression.
The team wanted to determine exactly what allows these malignant cells to escape telomere-driven crisis and avoid death.
To find out, the group disabled certain genes in human cells and then studied the impact this had on telomere fusion.
They found that cells escaped death when ligase 3 was active but not when its action, which appears to promote fusion within like chromosomes rather than between different chromosomes, was inhibited.
“Telomere dysfunction has been identified in many human cancers,” said study author Duncan Baird, PhD, of Cardiff University in the UK.
“And, as we have shown previously, short telomeres can predict the outcome of patients with [chronic lymphocytic leukemia] and probably many other tumor types. Thus, the discovery that ligase 3 is required for this process is fundamentally important.”
This research was made possible by a chance meeting between Dr Baird and Dr Hendrickson at an international conference. The pair discovered they were both looking at the role of ligase 3 in cancer and decided to collaborate.
“The collaboration paid off, as we were able to uncover something that neither one of us could have done on our own,” Dr Hendrickson said.
Additional studies are already underway. The researchers are investigating the discovery that the reliance on ligase 3 appears to be dependent upon the activity of another key DNA repair gene, p53.
“Since p53 is the most commonly mutated gene in human cancer, it now behooves us to discover how these two genes are interacting and to see if we can’t use that information to develop synergistic treatment modalities,” Dr Hendrickson concluded.
Malaria treatment in pregnancy often not up to snuff

Credit: Nina Matthews
Pregnant women are at risk of being undertreated or inappropriately treated for malaria, a new study suggests.
The research showed that some pregnant women with symptoms of malaria did not seek care from their formal healthcare system.
Those who did seek care sometimes received inappropriate treatment because healthcare providers did not to adhere to standard diagnostic and treatment guidelines recommended by the World Health Organization (WHO).
Jenny Hill, of the Liverpool School of Tropical Medicine in the UK, and her colleagues reported these discoveries in PLOS Medicine.
The team reviewed 37 studies investigating pregnant women’s access to malaria treatment and healthcare provider practices for managing malaria during pregnancy. The studies were conducted in Africa (30), Asia (4), Brazil (2), and Yemen (1).
Twenty-five percent to 75% of the women studied reported malaria episodes during pregnancy. More than 85% of women who reported a malaria episode during pregnancy sought some form of treatment, though this included self-treatment, herbal remedies, and other options not recommended by WHO.
Barriers to WHO-approved treatment included poor knowledge of drug safety, prohibitive costs, and self-treatment practices. Factors that determined whether a woman sought professional treatment included education, previous miscarriage, and prior use of antenatal care.
Other barriers to appropriate malaria treatment included healthcare providers’ reliance on a clinical diagnosis of malaria and poor adherence to treatment policy.
Seventy-two percent of healthcare providers followed standard treatment guidelines for malaria during a patient’s second or third trimester, but only 28% of providers followed the guidelines for patients in their first trimester (P=0.02).
Healthcare providers’ prescribing practices were driven by concerns about drug safety, patient preference, drug availability, and cost. Other factors that determined provider practices included access to training and the type of healthcare facility (public vs private).
Hill and her colleagues noted that this research is limited by the sparseness of data and the inconsistencies in study methodologies. Nevertheless, these findings highlight the need to develop interventions to improve access to and delivery of appropriate malaria treatment in pregnant women.

Credit: Nina Matthews
Pregnant women are at risk of being undertreated or inappropriately treated for malaria, a new study suggests.
The research showed that some pregnant women with symptoms of malaria did not seek care from their formal healthcare system.
Those who did seek care sometimes received inappropriate treatment because healthcare providers did not to adhere to standard diagnostic and treatment guidelines recommended by the World Health Organization (WHO).
Jenny Hill, of the Liverpool School of Tropical Medicine in the UK, and her colleagues reported these discoveries in PLOS Medicine.
The team reviewed 37 studies investigating pregnant women’s access to malaria treatment and healthcare provider practices for managing malaria during pregnancy. The studies were conducted in Africa (30), Asia (4), Brazil (2), and Yemen (1).
Twenty-five percent to 75% of the women studied reported malaria episodes during pregnancy. More than 85% of women who reported a malaria episode during pregnancy sought some form of treatment, though this included self-treatment, herbal remedies, and other options not recommended by WHO.
Barriers to WHO-approved treatment included poor knowledge of drug safety, prohibitive costs, and self-treatment practices. Factors that determined whether a woman sought professional treatment included education, previous miscarriage, and prior use of antenatal care.
Other barriers to appropriate malaria treatment included healthcare providers’ reliance on a clinical diagnosis of malaria and poor adherence to treatment policy.
Seventy-two percent of healthcare providers followed standard treatment guidelines for malaria during a patient’s second or third trimester, but only 28% of providers followed the guidelines for patients in their first trimester (P=0.02).
Healthcare providers’ prescribing practices were driven by concerns about drug safety, patient preference, drug availability, and cost. Other factors that determined provider practices included access to training and the type of healthcare facility (public vs private).
Hill and her colleagues noted that this research is limited by the sparseness of data and the inconsistencies in study methodologies. Nevertheless, these findings highlight the need to develop interventions to improve access to and delivery of appropriate malaria treatment in pregnant women.

Credit: Nina Matthews
Pregnant women are at risk of being undertreated or inappropriately treated for malaria, a new study suggests.
The research showed that some pregnant women with symptoms of malaria did not seek care from their formal healthcare system.
Those who did seek care sometimes received inappropriate treatment because healthcare providers did not to adhere to standard diagnostic and treatment guidelines recommended by the World Health Organization (WHO).
Jenny Hill, of the Liverpool School of Tropical Medicine in the UK, and her colleagues reported these discoveries in PLOS Medicine.
The team reviewed 37 studies investigating pregnant women’s access to malaria treatment and healthcare provider practices for managing malaria during pregnancy. The studies were conducted in Africa (30), Asia (4), Brazil (2), and Yemen (1).
Twenty-five percent to 75% of the women studied reported malaria episodes during pregnancy. More than 85% of women who reported a malaria episode during pregnancy sought some form of treatment, though this included self-treatment, herbal remedies, and other options not recommended by WHO.
Barriers to WHO-approved treatment included poor knowledge of drug safety, prohibitive costs, and self-treatment practices. Factors that determined whether a woman sought professional treatment included education, previous miscarriage, and prior use of antenatal care.
Other barriers to appropriate malaria treatment included healthcare providers’ reliance on a clinical diagnosis of malaria and poor adherence to treatment policy.
Seventy-two percent of healthcare providers followed standard treatment guidelines for malaria during a patient’s second or third trimester, but only 28% of providers followed the guidelines for patients in their first trimester (P=0.02).
Healthcare providers’ prescribing practices were driven by concerns about drug safety, patient preference, drug availability, and cost. Other factors that determined provider practices included access to training and the type of healthcare facility (public vs private).
Hill and her colleagues noted that this research is limited by the sparseness of data and the inconsistencies in study methodologies. Nevertheless, these findings highlight the need to develop interventions to improve access to and delivery of appropriate malaria treatment in pregnant women.
Malaria prophylaxis appears safe, effective in kids

Credit: Malayaka house
Year-round prophylaxis with a newer antimalaria treatment can reduce the risk of malaria in young children without posing a risk of serious adverse events, according to a study published in PLOS Medicine.
The researchers found that dihydroartemisinin-piperaquine (DP), an artemisinin-based combination therapy, was the most effective of 3 treatments at reducing malaria risk in children aged 6 months to 24 months.
The other 2 treatments, the antifolates sulfadoxine-pyrimethamine (SP) and trimethoprim-sulfamethoxazole (TS), have been in use longer than DP. And, in many locations, the malaria parasite has developed a resistance to them.
The researchers conducted this study to determine if the benefits of malaria prophylaxis outweighed the potential risk of anemia and other side effects from the drugs.
And they found the benefits did outweigh the risks. There was no significant increase in grade 3/4 adverse events with any of the treatments when compared to a control group.
The researchers also wanted to look specifically at the effects of year-round treatment. They noted that most previous studies of malaria prophylaxis have been limited to areas where there is only a seasonal risk of the disease. But this study took place in Uganda, where the risk persists throughout the year.
“Our study showed that preventive drug treatment can greatly reduce malaria in young children in areas where there are year-round high rates of transmission,” said study author Grant Dorsey, MD, of the University of California, San Francisco.
To make this discovery, the researchers studied 393 children from Tororo, Uganda. Beginning at 6 months of age, the children were randomized to 1 of 4 groups: monthly DP, monthly SP, daily TS, or a group that didn’t receive any prophylaxis, which is the standard medical practice in the area.
Treatments were given at home without supervision. Piperaquine levels were used as a measure of compliance in the DP arm.
All of the families involved in the study received insecticide-treated bed nets to put over the children when they slept. By 24 months of age, 352 children were still taking part in the study.
There were 3.02 malaria episodes per person-year in the DP group, 5.21 in the TS group, 6.73 in the SP group, and 6.95 in the control group. Protective efficacy measured 58% for the DP group, 28% for the TS group, and 7% for the SP group.
Piperaquine levels were below the detection limit 52% of the time when malaria was diagnosed in the DP group, which suggests non-adherence to treatment.
Between the groups, there was no significant difference in the rate of grade 3/4 adverse events related to treatment. There were 8 such events in the SP group, 8 in the TS group, and 3 in the DP group. Events included elevated temperature, anemia, neutropenia, thrombocytopenia, and elevated ALT/AST.
Considering all grade 3/4 adverse events regardless of their relationship to treatment, the researchers found the overall incidence was significantly lower in the DP group, but not the SP or TS groups, compared to the control group. The same was true for the incidence of elevated temperature, anemia, and thrombocytopenia.
After discontinuing the children’s treatment at 24 months of age, the researchers followed the children until age 3 and found no difference in malaria rates between the groups.
The team said these results suggest monthly administration of DP is a safe and effective option for reducing malaria among infants in regions with year-round transmission and high resistance to antifolates.
The findings also help to allay any concerns that continuous treatment might interfere with the children’s ability to develop an immune response against malaria, thereby making them more likely to contract the disease after treatment stops.
The researchers noted, however, that additional research is needed to evaluate the preventive efficacy of DP in other areas, maintain surveillance for potential selection of drug-resistant parasites, and evaluate the role of preventative treatment in the context of other malaria control interventions.

Credit: Malayaka house
Year-round prophylaxis with a newer antimalaria treatment can reduce the risk of malaria in young children without posing a risk of serious adverse events, according to a study published in PLOS Medicine.
The researchers found that dihydroartemisinin-piperaquine (DP), an artemisinin-based combination therapy, was the most effective of 3 treatments at reducing malaria risk in children aged 6 months to 24 months.
The other 2 treatments, the antifolates sulfadoxine-pyrimethamine (SP) and trimethoprim-sulfamethoxazole (TS), have been in use longer than DP. And, in many locations, the malaria parasite has developed a resistance to them.
The researchers conducted this study to determine if the benefits of malaria prophylaxis outweighed the potential risk of anemia and other side effects from the drugs.
And they found the benefits did outweigh the risks. There was no significant increase in grade 3/4 adverse events with any of the treatments when compared to a control group.
The researchers also wanted to look specifically at the effects of year-round treatment. They noted that most previous studies of malaria prophylaxis have been limited to areas where there is only a seasonal risk of the disease. But this study took place in Uganda, where the risk persists throughout the year.
“Our study showed that preventive drug treatment can greatly reduce malaria in young children in areas where there are year-round high rates of transmission,” said study author Grant Dorsey, MD, of the University of California, San Francisco.
To make this discovery, the researchers studied 393 children from Tororo, Uganda. Beginning at 6 months of age, the children were randomized to 1 of 4 groups: monthly DP, monthly SP, daily TS, or a group that didn’t receive any prophylaxis, which is the standard medical practice in the area.
Treatments were given at home without supervision. Piperaquine levels were used as a measure of compliance in the DP arm.
All of the families involved in the study received insecticide-treated bed nets to put over the children when they slept. By 24 months of age, 352 children were still taking part in the study.
There were 3.02 malaria episodes per person-year in the DP group, 5.21 in the TS group, 6.73 in the SP group, and 6.95 in the control group. Protective efficacy measured 58% for the DP group, 28% for the TS group, and 7% for the SP group.
Piperaquine levels were below the detection limit 52% of the time when malaria was diagnosed in the DP group, which suggests non-adherence to treatment.
Between the groups, there was no significant difference in the rate of grade 3/4 adverse events related to treatment. There were 8 such events in the SP group, 8 in the TS group, and 3 in the DP group. Events included elevated temperature, anemia, neutropenia, thrombocytopenia, and elevated ALT/AST.
Considering all grade 3/4 adverse events regardless of their relationship to treatment, the researchers found the overall incidence was significantly lower in the DP group, but not the SP or TS groups, compared to the control group. The same was true for the incidence of elevated temperature, anemia, and thrombocytopenia.
After discontinuing the children’s treatment at 24 months of age, the researchers followed the children until age 3 and found no difference in malaria rates between the groups.
The team said these results suggest monthly administration of DP is a safe and effective option for reducing malaria among infants in regions with year-round transmission and high resistance to antifolates.
The findings also help to allay any concerns that continuous treatment might interfere with the children’s ability to develop an immune response against malaria, thereby making them more likely to contract the disease after treatment stops.
The researchers noted, however, that additional research is needed to evaluate the preventive efficacy of DP in other areas, maintain surveillance for potential selection of drug-resistant parasites, and evaluate the role of preventative treatment in the context of other malaria control interventions.

Credit: Malayaka house
Year-round prophylaxis with a newer antimalaria treatment can reduce the risk of malaria in young children without posing a risk of serious adverse events, according to a study published in PLOS Medicine.
The researchers found that dihydroartemisinin-piperaquine (DP), an artemisinin-based combination therapy, was the most effective of 3 treatments at reducing malaria risk in children aged 6 months to 24 months.
The other 2 treatments, the antifolates sulfadoxine-pyrimethamine (SP) and trimethoprim-sulfamethoxazole (TS), have been in use longer than DP. And, in many locations, the malaria parasite has developed a resistance to them.
The researchers conducted this study to determine if the benefits of malaria prophylaxis outweighed the potential risk of anemia and other side effects from the drugs.
And they found the benefits did outweigh the risks. There was no significant increase in grade 3/4 adverse events with any of the treatments when compared to a control group.
The researchers also wanted to look specifically at the effects of year-round treatment. They noted that most previous studies of malaria prophylaxis have been limited to areas where there is only a seasonal risk of the disease. But this study took place in Uganda, where the risk persists throughout the year.
“Our study showed that preventive drug treatment can greatly reduce malaria in young children in areas where there are year-round high rates of transmission,” said study author Grant Dorsey, MD, of the University of California, San Francisco.
To make this discovery, the researchers studied 393 children from Tororo, Uganda. Beginning at 6 months of age, the children were randomized to 1 of 4 groups: monthly DP, monthly SP, daily TS, or a group that didn’t receive any prophylaxis, which is the standard medical practice in the area.
Treatments were given at home without supervision. Piperaquine levels were used as a measure of compliance in the DP arm.
All of the families involved in the study received insecticide-treated bed nets to put over the children when they slept. By 24 months of age, 352 children were still taking part in the study.
There were 3.02 malaria episodes per person-year in the DP group, 5.21 in the TS group, 6.73 in the SP group, and 6.95 in the control group. Protective efficacy measured 58% for the DP group, 28% for the TS group, and 7% for the SP group.
Piperaquine levels were below the detection limit 52% of the time when malaria was diagnosed in the DP group, which suggests non-adherence to treatment.
Between the groups, there was no significant difference in the rate of grade 3/4 adverse events related to treatment. There were 8 such events in the SP group, 8 in the TS group, and 3 in the DP group. Events included elevated temperature, anemia, neutropenia, thrombocytopenia, and elevated ALT/AST.
Considering all grade 3/4 adverse events regardless of their relationship to treatment, the researchers found the overall incidence was significantly lower in the DP group, but not the SP or TS groups, compared to the control group. The same was true for the incidence of elevated temperature, anemia, and thrombocytopenia.
After discontinuing the children’s treatment at 24 months of age, the researchers followed the children until age 3 and found no difference in malaria rates between the groups.
The team said these results suggest monthly administration of DP is a safe and effective option for reducing malaria among infants in regions with year-round transmission and high resistance to antifolates.
The findings also help to allay any concerns that continuous treatment might interfere with the children’s ability to develop an immune response against malaria, thereby making them more likely to contract the disease after treatment stops.
The researchers noted, however, that additional research is needed to evaluate the preventive efficacy of DP in other areas, maintain surveillance for potential selection of drug-resistant parasites, and evaluate the role of preventative treatment in the context of other malaria control interventions.