User login
Wealth appears to affect distribution of cancer types

Credit: Rhoda Baer
Results of a large study suggest certain malignanices are more concentrated in areas with high levels of poverty, while other cancer types arise more often in wealthy regions.
The research, conducted using data from nearly 3 million US cancer cases, also indicated that areas with higher poverty levels tended to have a lower cancer incidence but higher mortality rate than areas with lower poverty levels.
Researchers reported these findings in the journal Cancer.
Francis Boscoe, PhD, of the New York State Cancer Registry in Albany, and his colleagues conducted this research to evaluate the role of socioeconomics in cancer incidence and mortality.
So the team compared individuals living in areas with the highest poverty levels to those living in areas with the lowest poverty levels.
The researchers collected information on nearly 3 million cancer cases diagnosed between 2005 and 2009 in16 states, plus Los Angeles (an area covering 42% of the US population). Cases were divided into 1 of 4 groupings based on the poverty rate of the residential census tract at the time of diagnosis.
For all malignancies combined, there was a negligible association between cancer incidence and poverty. However, 32 of 39 cancer types showed a significant association with poverty.
Fourteen cancers were positively associated with poverty, and 18 were negatively associated. Myeloma, Hodgkin lymphoma, and non-Hodgkin lymphoma (NHL) were among the negatively associated malignancies.
The cancers most significantly associated with higher poverty levels were Kaposi sarcoma and cancers of the larynx, cervix, penis, and liver. The cancers most significantly associated with lower poverty levels were melanoma, thyroid cancer, other non-epithelial skin cancers, and testis cancer.
Overall, male malignancy rates were more sensitive to poverty level than female rates. And, in general, the race-specific cancer incidence rates differed, but the poverty gradients were similar.
For example, there was a roughly 20-fold difference in rates of melanoma between white and black individuals, but the relationship between poverty and incidence remained regardless of race.
This was not true for NHL, however. Among black individuals, the incidence of NHL increased as the poverty level increased. But among white individuals, the incidence of NHL decreased as the poverty level increased.
The researchers pointed out that—for all cancer sites, races, and sexes combined—the difference in risk between the greatest and lowest poverty category was less than 2%.
“At first glance, the effects seem to cancel one another out,” Dr Boscoe said. “But the cancers more associated with poverty have lower incidence and higher mortality, and those associated with wealth have higher incidence and lower mortality. When it comes to cancer, the poor are more likely to die of the disease, while the affluent are more likely to die with the disease.”
Dr Boscoe noted that recent gains in technology have made it much easier to link patient addresses with neighborhood characteristics, therefore making it possible to incorporate socioeconomic status into cancer surveillance.
“Our hope is that our paper will illustrate the value and necessity of doing this routinely in the future,” he concluded. ![]()

Credit: Rhoda Baer
Results of a large study suggest certain malignanices are more concentrated in areas with high levels of poverty, while other cancer types arise more often in wealthy regions.
The research, conducted using data from nearly 3 million US cancer cases, also indicated that areas with higher poverty levels tended to have a lower cancer incidence but higher mortality rate than areas with lower poverty levels.
Researchers reported these findings in the journal Cancer.
Francis Boscoe, PhD, of the New York State Cancer Registry in Albany, and his colleagues conducted this research to evaluate the role of socioeconomics in cancer incidence and mortality.
So the team compared individuals living in areas with the highest poverty levels to those living in areas with the lowest poverty levels.
The researchers collected information on nearly 3 million cancer cases diagnosed between 2005 and 2009 in16 states, plus Los Angeles (an area covering 42% of the US population). Cases were divided into 1 of 4 groupings based on the poverty rate of the residential census tract at the time of diagnosis.
For all malignancies combined, there was a negligible association between cancer incidence and poverty. However, 32 of 39 cancer types showed a significant association with poverty.
Fourteen cancers were positively associated with poverty, and 18 were negatively associated. Myeloma, Hodgkin lymphoma, and non-Hodgkin lymphoma (NHL) were among the negatively associated malignancies.
The cancers most significantly associated with higher poverty levels were Kaposi sarcoma and cancers of the larynx, cervix, penis, and liver. The cancers most significantly associated with lower poverty levels were melanoma, thyroid cancer, other non-epithelial skin cancers, and testis cancer.
Overall, male malignancy rates were more sensitive to poverty level than female rates. And, in general, the race-specific cancer incidence rates differed, but the poverty gradients were similar.
For example, there was a roughly 20-fold difference in rates of melanoma between white and black individuals, but the relationship between poverty and incidence remained regardless of race.
This was not true for NHL, however. Among black individuals, the incidence of NHL increased as the poverty level increased. But among white individuals, the incidence of NHL decreased as the poverty level increased.
The researchers pointed out that—for all cancer sites, races, and sexes combined—the difference in risk between the greatest and lowest poverty category was less than 2%.
“At first glance, the effects seem to cancel one another out,” Dr Boscoe said. “But the cancers more associated with poverty have lower incidence and higher mortality, and those associated with wealth have higher incidence and lower mortality. When it comes to cancer, the poor are more likely to die of the disease, while the affluent are more likely to die with the disease.”
Dr Boscoe noted that recent gains in technology have made it much easier to link patient addresses with neighborhood characteristics, therefore making it possible to incorporate socioeconomic status into cancer surveillance.
“Our hope is that our paper will illustrate the value and necessity of doing this routinely in the future,” he concluded. ![]()

Credit: Rhoda Baer
Results of a large study suggest certain malignanices are more concentrated in areas with high levels of poverty, while other cancer types arise more often in wealthy regions.
The research, conducted using data from nearly 3 million US cancer cases, also indicated that areas with higher poverty levels tended to have a lower cancer incidence but higher mortality rate than areas with lower poverty levels.
Researchers reported these findings in the journal Cancer.
Francis Boscoe, PhD, of the New York State Cancer Registry in Albany, and his colleagues conducted this research to evaluate the role of socioeconomics in cancer incidence and mortality.
So the team compared individuals living in areas with the highest poverty levels to those living in areas with the lowest poverty levels.
The researchers collected information on nearly 3 million cancer cases diagnosed between 2005 and 2009 in16 states, plus Los Angeles (an area covering 42% of the US population). Cases were divided into 1 of 4 groupings based on the poverty rate of the residential census tract at the time of diagnosis.
For all malignancies combined, there was a negligible association between cancer incidence and poverty. However, 32 of 39 cancer types showed a significant association with poverty.
Fourteen cancers were positively associated with poverty, and 18 were negatively associated. Myeloma, Hodgkin lymphoma, and non-Hodgkin lymphoma (NHL) were among the negatively associated malignancies.
The cancers most significantly associated with higher poverty levels were Kaposi sarcoma and cancers of the larynx, cervix, penis, and liver. The cancers most significantly associated with lower poverty levels were melanoma, thyroid cancer, other non-epithelial skin cancers, and testis cancer.
Overall, male malignancy rates were more sensitive to poverty level than female rates. And, in general, the race-specific cancer incidence rates differed, but the poverty gradients were similar.
For example, there was a roughly 20-fold difference in rates of melanoma between white and black individuals, but the relationship between poverty and incidence remained regardless of race.
This was not true for NHL, however. Among black individuals, the incidence of NHL increased as the poverty level increased. But among white individuals, the incidence of NHL decreased as the poverty level increased.
The researchers pointed out that—for all cancer sites, races, and sexes combined—the difference in risk between the greatest and lowest poverty category was less than 2%.
“At first glance, the effects seem to cancel one another out,” Dr Boscoe said. “But the cancers more associated with poverty have lower incidence and higher mortality, and those associated with wealth have higher incidence and lower mortality. When it comes to cancer, the poor are more likely to die of the disease, while the affluent are more likely to die with the disease.”
Dr Boscoe noted that recent gains in technology have made it much easier to link patient addresses with neighborhood characteristics, therefore making it possible to incorporate socioeconomic status into cancer surveillance.
“Our hope is that our paper will illustrate the value and necessity of doing this routinely in the future,” he concluded. ![]()
Refugees struggle to access cancer treatment
Credit: UK Department
for International Development
A new study published in The Lancet Oncology reveals a high demand for costly cancer treatment among refugees from the recent conflicts in Iraq and Syria, with host countries struggling to find the money and medicine to treat their new patients.
The findings prompted a call from study author Paul Spiegel, MD, the United Nations High Commissioner for Refugees (UNHCR) Chief Medical Expert, for schemes to improve access to affordable cancer care for refugees.
In the first study of its kind, Dr Spiegel and his colleagues examined data from funding applications made to the UNHCR Exceptional Care Committee (ECC) from refugees in Jordan and Syria whose cancer treatment costs were likely to exceed US$2000 a year.
The results indicate that cancer is an important public health problem in refugee settings, the researchers said. The study also highlights the challenges and costs national health systems and humanitarian organizations face when overwhelmed by massive influxes of refugees.
For example, in Jordan, the ECC assessed 1989 applications for treatment between 2010 and 2012. Roughly a quarter of these (511) were for cancer, with breast cancer and colorectal cancer being the most common. Around half (48%) of these cases were approved and funded.
Funding was often denied because the patient had a poor prognosis (43% of cases in 2011 and 31% in 2012) or the treatment was too costly (25% in 2011). The average amount requested from the ECC for cancer treatment was $11,540 in 2011 and $5151 in 2012. However, the amounts approved were substantially lower—$4626 in 2011 and $3501 in 2012.
“The countries in the Middle East have welcomed millions of refugees, first from Iraq and then Syria,” Dr Spiegel said. “This massive influx has strained health systems at all levels. Despite help from international organizations and donors to expand health facilities and pay for additional personnel and drugs, it has been insufficient.”
“The burden has fallen disproportionately on the host countries to absorb the costs. For example, the Jordanian Ministry of Health footed an estimated $53 million bill for medical care for refugees in the first 4 months of 2013.”
Dr Spiegel and his colleagues are therefore calling for improved cancer prevention and treatment in refugee settings through the use of innovative financing schemes; better primary care, including screening for common cancers (eg, colonoscopies and mammograms); and the development of web-based cancer registries to prevent the interruption of treatment.
“Until now, the responses to humanitarian crises have been primarily based on experiences from refugee camps in sub-Saharan Africa where infectious diseases and malnutrition have been the priority,” Dr Spiegel said. “In the 21st century, refugee situations are substantially longer and increasingly occur in middle-income countries where the levels of chronic diseases, including cancer, are higher.”
“Cancer diagnosis and care in humanitarian emergencies typifies a growing trend towards more costly chronic disease care, something that seems to have been overlooked but is of increasing importance because the number of refugees is growing.” ![]()

Credit: UK Department
for International Development
A new study published in The Lancet Oncology reveals a high demand for costly cancer treatment among refugees from the recent conflicts in Iraq and Syria, with host countries struggling to find the money and medicine to treat their new patients.
The findings prompted a call from study author Paul Spiegel, MD, the United Nations High Commissioner for Refugees (UNHCR) Chief Medical Expert, for schemes to improve access to affordable cancer care for refugees.
In the first study of its kind, Dr Spiegel and his colleagues examined data from funding applications made to the UNHCR Exceptional Care Committee (ECC) from refugees in Jordan and Syria whose cancer treatment costs were likely to exceed US$2000 a year.
The results indicate that cancer is an important public health problem in refugee settings, the researchers said. The study also highlights the challenges and costs national health systems and humanitarian organizations face when overwhelmed by massive influxes of refugees.
For example, in Jordan, the ECC assessed 1989 applications for treatment between 2010 and 2012. Roughly a quarter of these (511) were for cancer, with breast cancer and colorectal cancer being the most common. Around half (48%) of these cases were approved and funded.
Funding was often denied because the patient had a poor prognosis (43% of cases in 2011 and 31% in 2012) or the treatment was too costly (25% in 2011). The average amount requested from the ECC for cancer treatment was $11,540 in 2011 and $5151 in 2012. However, the amounts approved were substantially lower—$4626 in 2011 and $3501 in 2012.
“The countries in the Middle East have welcomed millions of refugees, first from Iraq and then Syria,” Dr Spiegel said. “This massive influx has strained health systems at all levels. Despite help from international organizations and donors to expand health facilities and pay for additional personnel and drugs, it has been insufficient.”
“The burden has fallen disproportionately on the host countries to absorb the costs. For example, the Jordanian Ministry of Health footed an estimated $53 million bill for medical care for refugees in the first 4 months of 2013.”
Dr Spiegel and his colleagues are therefore calling for improved cancer prevention and treatment in refugee settings through the use of innovative financing schemes; better primary care, including screening for common cancers (eg, colonoscopies and mammograms); and the development of web-based cancer registries to prevent the interruption of treatment.
“Until now, the responses to humanitarian crises have been primarily based on experiences from refugee camps in sub-Saharan Africa where infectious diseases and malnutrition have been the priority,” Dr Spiegel said. “In the 21st century, refugee situations are substantially longer and increasingly occur in middle-income countries where the levels of chronic diseases, including cancer, are higher.”
“Cancer diagnosis and care in humanitarian emergencies typifies a growing trend towards more costly chronic disease care, something that seems to have been overlooked but is of increasing importance because the number of refugees is growing.” ![]()

Credit: UK Department
for International Development
A new study published in The Lancet Oncology reveals a high demand for costly cancer treatment among refugees from the recent conflicts in Iraq and Syria, with host countries struggling to find the money and medicine to treat their new patients.
The findings prompted a call from study author Paul Spiegel, MD, the United Nations High Commissioner for Refugees (UNHCR) Chief Medical Expert, for schemes to improve access to affordable cancer care for refugees.
In the first study of its kind, Dr Spiegel and his colleagues examined data from funding applications made to the UNHCR Exceptional Care Committee (ECC) from refugees in Jordan and Syria whose cancer treatment costs were likely to exceed US$2000 a year.
The results indicate that cancer is an important public health problem in refugee settings, the researchers said. The study also highlights the challenges and costs national health systems and humanitarian organizations face when overwhelmed by massive influxes of refugees.
For example, in Jordan, the ECC assessed 1989 applications for treatment between 2010 and 2012. Roughly a quarter of these (511) were for cancer, with breast cancer and colorectal cancer being the most common. Around half (48%) of these cases were approved and funded.
Funding was often denied because the patient had a poor prognosis (43% of cases in 2011 and 31% in 2012) or the treatment was too costly (25% in 2011). The average amount requested from the ECC for cancer treatment was $11,540 in 2011 and $5151 in 2012. However, the amounts approved were substantially lower—$4626 in 2011 and $3501 in 2012.
“The countries in the Middle East have welcomed millions of refugees, first from Iraq and then Syria,” Dr Spiegel said. “This massive influx has strained health systems at all levels. Despite help from international organizations and donors to expand health facilities and pay for additional personnel and drugs, it has been insufficient.”
“The burden has fallen disproportionately on the host countries to absorb the costs. For example, the Jordanian Ministry of Health footed an estimated $53 million bill for medical care for refugees in the first 4 months of 2013.”
Dr Spiegel and his colleagues are therefore calling for improved cancer prevention and treatment in refugee settings through the use of innovative financing schemes; better primary care, including screening for common cancers (eg, colonoscopies and mammograms); and the development of web-based cancer registries to prevent the interruption of treatment.
“Until now, the responses to humanitarian crises have been primarily based on experiences from refugee camps in sub-Saharan Africa where infectious diseases and malnutrition have been the priority,” Dr Spiegel said. “In the 21st century, refugee situations are substantially longer and increasingly occur in middle-income countries where the levels of chronic diseases, including cancer, are higher.”
“Cancer diagnosis and care in humanitarian emergencies typifies a growing trend towards more costly chronic disease care, something that seems to have been overlooked but is of increasing importance because the number of refugees is growing.” ![]()
Trapping parasites to fight malaria

Credit: St Jude Children’s
Research Hospital
Investigators have identified antibodies that prevent malaria-causing parasites at the schizont stage from rupturing and spilling into the bloodstream.
These antibodies reduced loads of the parasite significantly in mice and humans, and they might one day be exploited to create a malaria vaccine, according to the researchers.
Jonathan Kurtis, MD, PhD, of Rhode Island Hospital in Providence, and his colleagues described the antibodies and their effects in Science.
The investigators studied the plasma of malaria-resistant 2-year-olds in Tanzania, where the disease is endemic. The team thought the naturally acquired immunity in these chronically exposed individuals provided a good model through which to identify vaccine antigens.
The analysis revealed that a particular Plasmodium falciparum antigen, known as P falciparum schizont egress antigen-1 (PfSEA-1), triggered antibodies in the children that, in turn, blocked replication of the parasite.
When the researchers vaccinated malaria-infected mice with the antigen or passively transferred PfSEA-1 antibodies to the rodents, they observed a 4-fold reduction of malaria parasites in the animals’ blood.
“When my post-doctoral fellow, Dipak Raj, discovered that antibodies to this protein, PfSEA-1, effectively trapped the malaria-causing parasite within the red blood cells, it was truly a moment of discovery,” Dr Kurtis said.
“Many researchers are trying to find ways to develop a malaria vaccine by preventing the parasite from entering the red blood cell, and, here, we found a way to block it from leaving the cell once it has entered. If it’s trapped in the red blood cell, it can’t go anywhere. It can’t do any further damage.”
The presence of PfSEA-1 antibodies also appeared to protect the Tanzanian study participants from severe cases of malaria. The investigators measured antibodies to PfSEA-1 in the entire cohort of 785 children and found that, among those with antibodies to PfSEA-1, there were no cases of severe malaria.
To generalize their results, the researchers then went back to serum samples they had collected from 140 children in Kenya in 1997. Analyses revealed that individuals with antibodies to PfSEA-1 had 50% lower parasitemia than individuals without these antibodies during a high-transmission season.
The investigators believe these findings could bring researchers a step closer to an effective malaria vaccine that targets parasites at multiple life stages.
“We still have additional trials ahead of us, first in another animal model, but we hope to begin phase 1 trials in humans very soon,” Dr Kurtis said.
“Our findings support PfSEA-1 as a potential vaccine candidate. And we are confident that, by partnering with our colleagues at the National Institutes of Health and other researchers focused on vaccines to prevent the parasites from entering red blood cells, we can approach the parasite from all angles, which could help us develop a truly effective vaccine to prevent this infectious disease that kills millions of children every year.” ![]()

Credit: St Jude Children’s
Research Hospital
Investigators have identified antibodies that prevent malaria-causing parasites at the schizont stage from rupturing and spilling into the bloodstream.
These antibodies reduced loads of the parasite significantly in mice and humans, and they might one day be exploited to create a malaria vaccine, according to the researchers.
Jonathan Kurtis, MD, PhD, of Rhode Island Hospital in Providence, and his colleagues described the antibodies and their effects in Science.
The investigators studied the plasma of malaria-resistant 2-year-olds in Tanzania, where the disease is endemic. The team thought the naturally acquired immunity in these chronically exposed individuals provided a good model through which to identify vaccine antigens.
The analysis revealed that a particular Plasmodium falciparum antigen, known as P falciparum schizont egress antigen-1 (PfSEA-1), triggered antibodies in the children that, in turn, blocked replication of the parasite.
When the researchers vaccinated malaria-infected mice with the antigen or passively transferred PfSEA-1 antibodies to the rodents, they observed a 4-fold reduction of malaria parasites in the animals’ blood.
“When my post-doctoral fellow, Dipak Raj, discovered that antibodies to this protein, PfSEA-1, effectively trapped the malaria-causing parasite within the red blood cells, it was truly a moment of discovery,” Dr Kurtis said.
“Many researchers are trying to find ways to develop a malaria vaccine by preventing the parasite from entering the red blood cell, and, here, we found a way to block it from leaving the cell once it has entered. If it’s trapped in the red blood cell, it can’t go anywhere. It can’t do any further damage.”
The presence of PfSEA-1 antibodies also appeared to protect the Tanzanian study participants from severe cases of malaria. The investigators measured antibodies to PfSEA-1 in the entire cohort of 785 children and found that, among those with antibodies to PfSEA-1, there were no cases of severe malaria.
To generalize their results, the researchers then went back to serum samples they had collected from 140 children in Kenya in 1997. Analyses revealed that individuals with antibodies to PfSEA-1 had 50% lower parasitemia than individuals without these antibodies during a high-transmission season.
The investigators believe these findings could bring researchers a step closer to an effective malaria vaccine that targets parasites at multiple life stages.
“We still have additional trials ahead of us, first in another animal model, but we hope to begin phase 1 trials in humans very soon,” Dr Kurtis said.
“Our findings support PfSEA-1 as a potential vaccine candidate. And we are confident that, by partnering with our colleagues at the National Institutes of Health and other researchers focused on vaccines to prevent the parasites from entering red blood cells, we can approach the parasite from all angles, which could help us develop a truly effective vaccine to prevent this infectious disease that kills millions of children every year.” ![]()

Credit: St Jude Children’s
Research Hospital
Investigators have identified antibodies that prevent malaria-causing parasites at the schizont stage from rupturing and spilling into the bloodstream.
These antibodies reduced loads of the parasite significantly in mice and humans, and they might one day be exploited to create a malaria vaccine, according to the researchers.
Jonathan Kurtis, MD, PhD, of Rhode Island Hospital in Providence, and his colleagues described the antibodies and their effects in Science.
The investigators studied the plasma of malaria-resistant 2-year-olds in Tanzania, where the disease is endemic. The team thought the naturally acquired immunity in these chronically exposed individuals provided a good model through which to identify vaccine antigens.
The analysis revealed that a particular Plasmodium falciparum antigen, known as P falciparum schizont egress antigen-1 (PfSEA-1), triggered antibodies in the children that, in turn, blocked replication of the parasite.
When the researchers vaccinated malaria-infected mice with the antigen or passively transferred PfSEA-1 antibodies to the rodents, they observed a 4-fold reduction of malaria parasites in the animals’ blood.
“When my post-doctoral fellow, Dipak Raj, discovered that antibodies to this protein, PfSEA-1, effectively trapped the malaria-causing parasite within the red blood cells, it was truly a moment of discovery,” Dr Kurtis said.
“Many researchers are trying to find ways to develop a malaria vaccine by preventing the parasite from entering the red blood cell, and, here, we found a way to block it from leaving the cell once it has entered. If it’s trapped in the red blood cell, it can’t go anywhere. It can’t do any further damage.”
The presence of PfSEA-1 antibodies also appeared to protect the Tanzanian study participants from severe cases of malaria. The investigators measured antibodies to PfSEA-1 in the entire cohort of 785 children and found that, among those with antibodies to PfSEA-1, there were no cases of severe malaria.
To generalize their results, the researchers then went back to serum samples they had collected from 140 children in Kenya in 1997. Analyses revealed that individuals with antibodies to PfSEA-1 had 50% lower parasitemia than individuals without these antibodies during a high-transmission season.
The investigators believe these findings could bring researchers a step closer to an effective malaria vaccine that targets parasites at multiple life stages.
“We still have additional trials ahead of us, first in another animal model, but we hope to begin phase 1 trials in humans very soon,” Dr Kurtis said.
“Our findings support PfSEA-1 as a potential vaccine candidate. And we are confident that, by partnering with our colleagues at the National Institutes of Health and other researchers focused on vaccines to prevent the parasites from entering red blood cells, we can approach the parasite from all angles, which could help us develop a truly effective vaccine to prevent this infectious disease that kills millions of children every year.” ![]()
Large-volume infusion pump recalled

Credit: CDC
The medical technology company CareFusion has announced a Class I recall of its Alaris Pump model 8100, software version 9.1.18.
This large-volume infusion pump is used for the delivery of fluids, medicines, blood, and blood products.
Version 9.1.18 of the Alaris Pump model 8100 is being recalled due to the possibility of a software failure in which the pump module will not properly delay an infusion when the “Delay Until” option or “Multidose” feature is used.
There have been no reports of adverse events or deaths related to this malfunction, but it does pose risks. CareFusion has received 1 report where the device malfunctioned when the “Delay Until” option was selected.
The software failure also prevents the pump from properly delivering a multidose infusion under the following conditions:
- When the first dose is programmed to infuse when the system time is earlier than 7 pm and a subsequent dose is intended to infuse between 7 pm and 11:59 pm
- When the first dose is programmed to infuse when the system time is between 7 pm and 11:59 pm and a subsequent dose is intended to infuse between 12 am and 6:59 pm the next day.
If the infusion starts earlier or later than intended and is not immediately detected and stopped, serious injury or death could result. Therefore, healthcare professionals should not use the Alaris Pump module “Delay Until” option or the “Multidose” option.
However, CareFusion said it has identified the root cause of the issue and recommends that the previous Alaris Pump module software version 9.1.17 be installed to address this recall. The company said it will contact all affected customers to schedule the installation of software version 9.1.17.
As an interim guidance, customers may update their dataset to disable both “Delay” options (“Delay Until” and “Delay For”) and/or the “Multidose” option across all profiles to prevent the use of these features. These are shared configurations with the Alaris Syringe module and, if disabled, would prevent use of these features with the Alaris Syringe module as well.
For more information on this recall, see CareFusion’s recall notice, or contact the CareFusion Support Center at 888-562-6018 or SupportCenter@carefusion.com.
To report adverse reactions or quality problems associated with this product, visit the Food and Drug Administration’s MedWatch website. ![]()

Credit: CDC
The medical technology company CareFusion has announced a Class I recall of its Alaris Pump model 8100, software version 9.1.18.
This large-volume infusion pump is used for the delivery of fluids, medicines, blood, and blood products.
Version 9.1.18 of the Alaris Pump model 8100 is being recalled due to the possibility of a software failure in which the pump module will not properly delay an infusion when the “Delay Until” option or “Multidose” feature is used.
There have been no reports of adverse events or deaths related to this malfunction, but it does pose risks. CareFusion has received 1 report where the device malfunctioned when the “Delay Until” option was selected.
The software failure also prevents the pump from properly delivering a multidose infusion under the following conditions:
- When the first dose is programmed to infuse when the system time is earlier than 7 pm and a subsequent dose is intended to infuse between 7 pm and 11:59 pm
- When the first dose is programmed to infuse when the system time is between 7 pm and 11:59 pm and a subsequent dose is intended to infuse between 12 am and 6:59 pm the next day.
If the infusion starts earlier or later than intended and is not immediately detected and stopped, serious injury or death could result. Therefore, healthcare professionals should not use the Alaris Pump module “Delay Until” option or the “Multidose” option.
However, CareFusion said it has identified the root cause of the issue and recommends that the previous Alaris Pump module software version 9.1.17 be installed to address this recall. The company said it will contact all affected customers to schedule the installation of software version 9.1.17.
As an interim guidance, customers may update their dataset to disable both “Delay” options (“Delay Until” and “Delay For”) and/or the “Multidose” option across all profiles to prevent the use of these features. These are shared configurations with the Alaris Syringe module and, if disabled, would prevent use of these features with the Alaris Syringe module as well.
For more information on this recall, see CareFusion’s recall notice, or contact the CareFusion Support Center at 888-562-6018 or SupportCenter@carefusion.com.
To report adverse reactions or quality problems associated with this product, visit the Food and Drug Administration’s MedWatch website. ![]()

Credit: CDC
The medical technology company CareFusion has announced a Class I recall of its Alaris Pump model 8100, software version 9.1.18.
This large-volume infusion pump is used for the delivery of fluids, medicines, blood, and blood products.
Version 9.1.18 of the Alaris Pump model 8100 is being recalled due to the possibility of a software failure in which the pump module will not properly delay an infusion when the “Delay Until” option or “Multidose” feature is used.
There have been no reports of adverse events or deaths related to this malfunction, but it does pose risks. CareFusion has received 1 report where the device malfunctioned when the “Delay Until” option was selected.
The software failure also prevents the pump from properly delivering a multidose infusion under the following conditions:
- When the first dose is programmed to infuse when the system time is earlier than 7 pm and a subsequent dose is intended to infuse between 7 pm and 11:59 pm
- When the first dose is programmed to infuse when the system time is between 7 pm and 11:59 pm and a subsequent dose is intended to infuse between 12 am and 6:59 pm the next day.
If the infusion starts earlier or later than intended and is not immediately detected and stopped, serious injury or death could result. Therefore, healthcare professionals should not use the Alaris Pump module “Delay Until” option or the “Multidose” option.
However, CareFusion said it has identified the root cause of the issue and recommends that the previous Alaris Pump module software version 9.1.17 be installed to address this recall. The company said it will contact all affected customers to schedule the installation of software version 9.1.17.
As an interim guidance, customers may update their dataset to disable both “Delay” options (“Delay Until” and “Delay For”) and/or the “Multidose” option across all profiles to prevent the use of these features. These are shared configurations with the Alaris Syringe module and, if disabled, would prevent use of these features with the Alaris Syringe module as well.
For more information on this recall, see CareFusion’s recall notice, or contact the CareFusion Support Center at 888-562-6018 or SupportCenter@carefusion.com.
To report adverse reactions or quality problems associated with this product, visit the Food and Drug Administration’s MedWatch website. ![]()
TPN calculation software recalled
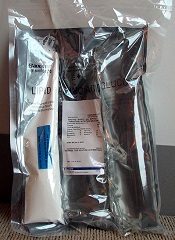
The US Food and Drug Administration (FDA) has announced a Class I recall of Baxter Corporation Englewood’s ABACUS Total Parenteral Nutrition (TPN) Calculation Software, versions 3.1, 3.0, 2.1, and 2.0.
Baxter has received 2 reports of malfunctioning software and said errors with this software may cause adverse effects.
ABACUS TPN Calculation Software is a Windows-based software application used by pharmacists to calculate or order TPN formulas.
The errors explained
Due to software failures, the following errors may occur:
- ABACUS v3.1 may calculate quantities of electrolytes that are double the expected values during the creation of TPN orders.
- ABACUS v3.1 may automatically add additional sterile water to a formula equal to the volume of a premix, resulting in an over-dilution.
- All software versions of ABACUS software display the calcium phosphate curve points for Premasol incorrectly.
- All software versions of ABACUS may display an inaccurate estimation for calcium and phosphate precipitation in certain circumstances where multiple ingredients provide calcium.
If any of these failures occur, patients may be at risk of developing overdose symptoms. The symptoms are varied and depend on the type of software failure and composition of the fluid being compounded.
Symptoms may be non-specific and include nausea, vomiting, dizziness, or fatigue. Some more severe symptoms include cardiac arrhythmia, pulmonary edema, congestive heart failure, and seizures. A fatal outcome is possible, especially in the high-risk population.
Actions to take
Baxter is recommending that customers contact the company to ensure the ABACUS software is configured correctly.
Customers with a software version earlier than 3.1 will have software version 3.1 installed, which addresses the issues that prompted the recall. In addition, Baxter Support Services will schedule upgrades and assist customers with establishing the proper ABACUS configuration in the customers’ facilities.
Baxter has also requested that customers follow safe compounding practices. Namely, use the “Summary” button to verify the order against the calculated amounts prior to completing the order.
In addition, verify that the ordered ingredients and quantities displayed in the software and printed on the Bag label and the Solution Formula label match the PN prescription prior to preparation. And use a filter for administration of a PN bag.
For more information on the recall, see the FDA’s recall notice, or contact Baxter at 303-617-2242. For technical support, call 1-800-678-2292 or email COtechsupport@baxter.com.
To report adverse reactions or quality problems related to this product, visit the FDA’s MedWatch website. ![]()

The US Food and Drug Administration (FDA) has announced a Class I recall of Baxter Corporation Englewood’s ABACUS Total Parenteral Nutrition (TPN) Calculation Software, versions 3.1, 3.0, 2.1, and 2.0.
Baxter has received 2 reports of malfunctioning software and said errors with this software may cause adverse effects.
ABACUS TPN Calculation Software is a Windows-based software application used by pharmacists to calculate or order TPN formulas.
The errors explained
Due to software failures, the following errors may occur:
- ABACUS v3.1 may calculate quantities of electrolytes that are double the expected values during the creation of TPN orders.
- ABACUS v3.1 may automatically add additional sterile water to a formula equal to the volume of a premix, resulting in an over-dilution.
- All software versions of ABACUS software display the calcium phosphate curve points for Premasol incorrectly.
- All software versions of ABACUS may display an inaccurate estimation for calcium and phosphate precipitation in certain circumstances where multiple ingredients provide calcium.
If any of these failures occur, patients may be at risk of developing overdose symptoms. The symptoms are varied and depend on the type of software failure and composition of the fluid being compounded.
Symptoms may be non-specific and include nausea, vomiting, dizziness, or fatigue. Some more severe symptoms include cardiac arrhythmia, pulmonary edema, congestive heart failure, and seizures. A fatal outcome is possible, especially in the high-risk population.
Actions to take
Baxter is recommending that customers contact the company to ensure the ABACUS software is configured correctly.
Customers with a software version earlier than 3.1 will have software version 3.1 installed, which addresses the issues that prompted the recall. In addition, Baxter Support Services will schedule upgrades and assist customers with establishing the proper ABACUS configuration in the customers’ facilities.
Baxter has also requested that customers follow safe compounding practices. Namely, use the “Summary” button to verify the order against the calculated amounts prior to completing the order.
In addition, verify that the ordered ingredients and quantities displayed in the software and printed on the Bag label and the Solution Formula label match the PN prescription prior to preparation. And use a filter for administration of a PN bag.
For more information on the recall, see the FDA’s recall notice, or contact Baxter at 303-617-2242. For technical support, call 1-800-678-2292 or email COtechsupport@baxter.com.
To report adverse reactions or quality problems related to this product, visit the FDA’s MedWatch website. ![]()

The US Food and Drug Administration (FDA) has announced a Class I recall of Baxter Corporation Englewood’s ABACUS Total Parenteral Nutrition (TPN) Calculation Software, versions 3.1, 3.0, 2.1, and 2.0.
Baxter has received 2 reports of malfunctioning software and said errors with this software may cause adverse effects.
ABACUS TPN Calculation Software is a Windows-based software application used by pharmacists to calculate or order TPN formulas.
The errors explained
Due to software failures, the following errors may occur:
- ABACUS v3.1 may calculate quantities of electrolytes that are double the expected values during the creation of TPN orders.
- ABACUS v3.1 may automatically add additional sterile water to a formula equal to the volume of a premix, resulting in an over-dilution.
- All software versions of ABACUS software display the calcium phosphate curve points for Premasol incorrectly.
- All software versions of ABACUS may display an inaccurate estimation for calcium and phosphate precipitation in certain circumstances where multiple ingredients provide calcium.
If any of these failures occur, patients may be at risk of developing overdose symptoms. The symptoms are varied and depend on the type of software failure and composition of the fluid being compounded.
Symptoms may be non-specific and include nausea, vomiting, dizziness, or fatigue. Some more severe symptoms include cardiac arrhythmia, pulmonary edema, congestive heart failure, and seizures. A fatal outcome is possible, especially in the high-risk population.
Actions to take
Baxter is recommending that customers contact the company to ensure the ABACUS software is configured correctly.
Customers with a software version earlier than 3.1 will have software version 3.1 installed, which addresses the issues that prompted the recall. In addition, Baxter Support Services will schedule upgrades and assist customers with establishing the proper ABACUS configuration in the customers’ facilities.
Baxter has also requested that customers follow safe compounding practices. Namely, use the “Summary” button to verify the order against the calculated amounts prior to completing the order.
In addition, verify that the ordered ingredients and quantities displayed in the software and printed on the Bag label and the Solution Formula label match the PN prescription prior to preparation. And use a filter for administration of a PN bag.
For more information on the recall, see the FDA’s recall notice, or contact Baxter at 303-617-2242. For technical support, call 1-800-678-2292 or email COtechsupport@baxter.com.
To report adverse reactions or quality problems related to this product, visit the FDA’s MedWatch website. ![]()
Chemists discover true structure of anticancer agent

Credit: NIH
Chemists say they have determined the correct structure of a compound that has shown activity against lymphoma and a range of other cancers.
Their research, published in Angewandte Chemie, focused on a compound called TIC10.
The team showed that TIC10’s structure differs subtly from a version described by another group last year, and the previous structure associated with TIC10 actually describes a molecule that lacks TIC10’s anticancer activity.
The newly identified structure describes a molecule with potent anticancer effects in animals, representing a new family of biologically active structures that can now be explored for possible therapeutic uses.
“This new structure should generate much interest in the cancer research community,” said study author Kim D. Janda, PhD, of The Scripps Research Institute in La Jolla, California.
Antitumor potential
TIC10 was first described in Science Translational Medicine in early 2013. The authors identified the compound, within a library of thousands of molecules maintained by the National Cancer Institute (NCI), for its ability to boost cells’ production of the natural antitumor protein TRAIL. (TIC10 stands for TRAIL-inducing compound #10.)
As a small molecule, TIC10 would be easier to deliver in a therapy than the TRAIL protein itself. The paper’s authors reported that TIC10 was orally active and dramatically shrank a variety of tumors in mice.
Tumors can develop resistance to TRAIL, but Dr Janda had been studying compounds that defeat this resistance. The news about TIC10 therefore got his attention.
“I thought, ‘They have this molecule for upregulating TRAIL, and we have these molecules that can overcome tumor-cell TRAIL resistance—the combination could be important,’” he said.
The original publication on TIC10 included a figure showing its predicted structure. So Dr Janda asked one of his postdoctoral researchers, Jonathan Lockner, to make TIC10 using that information.
Although the original TIC10 research team had seemingly confirmed the predicted structure with mass spectrometry, no one had published a thorough characterization of the TIC10 molecule.
“There were no nuclear magnetic resonance data or X-ray crystallography data, and there was definitely no procedure for the synthesis,” Dr Lockner said. “My background was chemistry, though, so I was able to find a way to synthesize it starting from simple compounds.”
Surprising inactivity
There was just one problem with Dr Lockner’s newly synthesized “TIC10.” When tested, it failed to induce TRAIL expression in cells, even at high doses.
“Of course, I was nervous,” Dr Lockner said. “As a chemist, you never want to make a mistake and give biologists the wrong material.”
To try and verify they had the right material, Dr Janda’s team obtained a sample of TIC10 directly from the NCI.
“When we got that sample and tested it, we saw that it had the expected TRAIL-upregulating effect,” said Nicholas Jacob, a graduate student in the Janda Lab and coauthor of the new paper.
“That prompted us to look more closely at the structures of these 2 compounds.”
The researchers spent months characterizing their own synthesized material and the NCI material, using an array of sophisticated structural analysis tools. They also tested the 2 compounds’ biological effects.
The team eventually concluded that the TIC10 compound from the NCI library does boost TRAIL production in cells and remains promising as the basis for anticancer therapies, but it does not have the structure that was originally published.
The right structure
The originally published structure has a core made of 3 carbon-nitrogen rings in a straight line and does not induce TRAIL activity. The correct, TRAIL-inducing structure differs subtly, with an end ring that sticks out at an angle.
In chemists’ parlance, the 2 compounds are constitutional isomers: a linear imidazolinopyrimidinone and an angular imidazolinopyrimidinone.
And Dr Lockner found that the angular, TRAIL-inducing structure was easier to synthesize than the one originally described.
Now, with the correct molecule in hand and a solid understanding of its structure and synthesis, Dr Janda and his team are moving forward with their original plan to study TIC10 in combination with TRAIL-resistance-thwarting molecules as an anticancer therapy.
The therapeutic implications of TIC10 may even go beyond cancer, according to the researchers. The angular core of the TRAIL-inducing molecule Dr Janda’s team discovered is a novel type of a biologically active structure, or pharmacophore, from which chemists may now be able to build a new class of candidate drugs, possibly for a variety of ailments. ![]()

Credit: NIH
Chemists say they have determined the correct structure of a compound that has shown activity against lymphoma and a range of other cancers.
Their research, published in Angewandte Chemie, focused on a compound called TIC10.
The team showed that TIC10’s structure differs subtly from a version described by another group last year, and the previous structure associated with TIC10 actually describes a molecule that lacks TIC10’s anticancer activity.
The newly identified structure describes a molecule with potent anticancer effects in animals, representing a new family of biologically active structures that can now be explored for possible therapeutic uses.
“This new structure should generate much interest in the cancer research community,” said study author Kim D. Janda, PhD, of The Scripps Research Institute in La Jolla, California.
Antitumor potential
TIC10 was first described in Science Translational Medicine in early 2013. The authors identified the compound, within a library of thousands of molecules maintained by the National Cancer Institute (NCI), for its ability to boost cells’ production of the natural antitumor protein TRAIL. (TIC10 stands for TRAIL-inducing compound #10.)
As a small molecule, TIC10 would be easier to deliver in a therapy than the TRAIL protein itself. The paper’s authors reported that TIC10 was orally active and dramatically shrank a variety of tumors in mice.
Tumors can develop resistance to TRAIL, but Dr Janda had been studying compounds that defeat this resistance. The news about TIC10 therefore got his attention.
“I thought, ‘They have this molecule for upregulating TRAIL, and we have these molecules that can overcome tumor-cell TRAIL resistance—the combination could be important,’” he said.
The original publication on TIC10 included a figure showing its predicted structure. So Dr Janda asked one of his postdoctoral researchers, Jonathan Lockner, to make TIC10 using that information.
Although the original TIC10 research team had seemingly confirmed the predicted structure with mass spectrometry, no one had published a thorough characterization of the TIC10 molecule.
“There were no nuclear magnetic resonance data or X-ray crystallography data, and there was definitely no procedure for the synthesis,” Dr Lockner said. “My background was chemistry, though, so I was able to find a way to synthesize it starting from simple compounds.”
Surprising inactivity
There was just one problem with Dr Lockner’s newly synthesized “TIC10.” When tested, it failed to induce TRAIL expression in cells, even at high doses.
“Of course, I was nervous,” Dr Lockner said. “As a chemist, you never want to make a mistake and give biologists the wrong material.”
To try and verify they had the right material, Dr Janda’s team obtained a sample of TIC10 directly from the NCI.
“When we got that sample and tested it, we saw that it had the expected TRAIL-upregulating effect,” said Nicholas Jacob, a graduate student in the Janda Lab and coauthor of the new paper.
“That prompted us to look more closely at the structures of these 2 compounds.”
The researchers spent months characterizing their own synthesized material and the NCI material, using an array of sophisticated structural analysis tools. They also tested the 2 compounds’ biological effects.
The team eventually concluded that the TIC10 compound from the NCI library does boost TRAIL production in cells and remains promising as the basis for anticancer therapies, but it does not have the structure that was originally published.
The right structure
The originally published structure has a core made of 3 carbon-nitrogen rings in a straight line and does not induce TRAIL activity. The correct, TRAIL-inducing structure differs subtly, with an end ring that sticks out at an angle.
In chemists’ parlance, the 2 compounds are constitutional isomers: a linear imidazolinopyrimidinone and an angular imidazolinopyrimidinone.
And Dr Lockner found that the angular, TRAIL-inducing structure was easier to synthesize than the one originally described.
Now, with the correct molecule in hand and a solid understanding of its structure and synthesis, Dr Janda and his team are moving forward with their original plan to study TIC10 in combination with TRAIL-resistance-thwarting molecules as an anticancer therapy.
The therapeutic implications of TIC10 may even go beyond cancer, according to the researchers. The angular core of the TRAIL-inducing molecule Dr Janda’s team discovered is a novel type of a biologically active structure, or pharmacophore, from which chemists may now be able to build a new class of candidate drugs, possibly for a variety of ailments. ![]()

Credit: NIH
Chemists say they have determined the correct structure of a compound that has shown activity against lymphoma and a range of other cancers.
Their research, published in Angewandte Chemie, focused on a compound called TIC10.
The team showed that TIC10’s structure differs subtly from a version described by another group last year, and the previous structure associated with TIC10 actually describes a molecule that lacks TIC10’s anticancer activity.
The newly identified structure describes a molecule with potent anticancer effects in animals, representing a new family of biologically active structures that can now be explored for possible therapeutic uses.
“This new structure should generate much interest in the cancer research community,” said study author Kim D. Janda, PhD, of The Scripps Research Institute in La Jolla, California.
Antitumor potential
TIC10 was first described in Science Translational Medicine in early 2013. The authors identified the compound, within a library of thousands of molecules maintained by the National Cancer Institute (NCI), for its ability to boost cells’ production of the natural antitumor protein TRAIL. (TIC10 stands for TRAIL-inducing compound #10.)
As a small molecule, TIC10 would be easier to deliver in a therapy than the TRAIL protein itself. The paper’s authors reported that TIC10 was orally active and dramatically shrank a variety of tumors in mice.
Tumors can develop resistance to TRAIL, but Dr Janda had been studying compounds that defeat this resistance. The news about TIC10 therefore got his attention.
“I thought, ‘They have this molecule for upregulating TRAIL, and we have these molecules that can overcome tumor-cell TRAIL resistance—the combination could be important,’” he said.
The original publication on TIC10 included a figure showing its predicted structure. So Dr Janda asked one of his postdoctoral researchers, Jonathan Lockner, to make TIC10 using that information.
Although the original TIC10 research team had seemingly confirmed the predicted structure with mass spectrometry, no one had published a thorough characterization of the TIC10 molecule.
“There were no nuclear magnetic resonance data or X-ray crystallography data, and there was definitely no procedure for the synthesis,” Dr Lockner said. “My background was chemistry, though, so I was able to find a way to synthesize it starting from simple compounds.”
Surprising inactivity
There was just one problem with Dr Lockner’s newly synthesized “TIC10.” When tested, it failed to induce TRAIL expression in cells, even at high doses.
“Of course, I was nervous,” Dr Lockner said. “As a chemist, you never want to make a mistake and give biologists the wrong material.”
To try and verify they had the right material, Dr Janda’s team obtained a sample of TIC10 directly from the NCI.
“When we got that sample and tested it, we saw that it had the expected TRAIL-upregulating effect,” said Nicholas Jacob, a graduate student in the Janda Lab and coauthor of the new paper.
“That prompted us to look more closely at the structures of these 2 compounds.”
The researchers spent months characterizing their own synthesized material and the NCI material, using an array of sophisticated structural analysis tools. They also tested the 2 compounds’ biological effects.
The team eventually concluded that the TIC10 compound from the NCI library does boost TRAIL production in cells and remains promising as the basis for anticancer therapies, but it does not have the structure that was originally published.
The right structure
The originally published structure has a core made of 3 carbon-nitrogen rings in a straight line and does not induce TRAIL activity. The correct, TRAIL-inducing structure differs subtly, with an end ring that sticks out at an angle.
In chemists’ parlance, the 2 compounds are constitutional isomers: a linear imidazolinopyrimidinone and an angular imidazolinopyrimidinone.
And Dr Lockner found that the angular, TRAIL-inducing structure was easier to synthesize than the one originally described.
Now, with the correct molecule in hand and a solid understanding of its structure and synthesis, Dr Janda and his team are moving forward with their original plan to study TIC10 in combination with TRAIL-resistance-thwarting molecules as an anticancer therapy.
The therapeutic implications of TIC10 may even go beyond cancer, according to the researchers. The angular core of the TRAIL-inducing molecule Dr Janda’s team discovered is a novel type of a biologically active structure, or pharmacophore, from which chemists may now be able to build a new class of candidate drugs, possibly for a variety of ailments. ![]()
Cancer trial publications often omit minority accrual rates

Credit: Rhoda Baer
A review of clinical trial data from 2012 suggests Hispanic patients are underrepresented in US cancer studies, and many trial publications fail to provide information on patients’ racial/ethnic backgrounds.
Researchers analyzed 159 reports of phase 2 and 3 trials and found that roughly 21% included information on the number of minority patients enrolled.
About 8% of the publications included data on the number of Hispanic patients enrolled.
And from this data, the investigators found the accrual rate for Hispanic patients was about 4%.
According to the researchers, this lack of information and low representation inhibits physicians’ ability to provide optimal treatment to Hispanic cancer patients and patients belonging to other minority groups.
“We have a major responsibility to ensure adequate representation,” said study author Ian M. Thompson Jr, MD, of The University of Texas Health Science Center at San Antonio.
“How else will we know how best to treat our patients, and how else are we going to reduce the health disparities in [the Hispanic] population?”
Dr Thompson and his colleagues have a particular interest in the Hispanic population because 58% of San Antonio residents are Hispanic, as are 68% of residents in South Texas as a whole.
So the investigators wanted to determine Hispanic accrual rates in randomized trials of cancer patients. The team evaluated data from phase 2 and 3 cancer trials published in 2012. They focused on studies that were considered likely to change the standard of care and were published in “high-impact” journals.
The researchers identified 159 trials—68 phase 2 studies and 91 phase 3 studies. They discovered that 33 of the trial publications—about 21%—disclosed data on minority accrual. And 13 publications—about 8%—included data on the accrual of Hispanic cancer patients.
Of the 4154 patients enrolled on those 13 trials, 162 were Hispanic, which translates to an overall accrual rate of 3.9%. The enrollment of Hispanic patients ranged from 1 patient (0.5%) in a phase 2 trial of lung cancer to 17 patients (26%) in a phase 2 study of acute lymphoblastic leukemia.
“Fundamentally, in the most recent published cancer clinical trials, either the number and proportion of Hispanics are not reported or are far below their actual representation in the national population,” Dr Thompson summarized.
“For institutions like ours that serve a ‘minority-majority’ population, it’s a major responsibility for us to ensure adequate representation so that we can tell our patients how they can best be treated and how we can reduce the disparities of this rapidly growing population.”
Dr Thompson and his colleagues described this research in the Journal of Clinical Oncology. ![]()

Credit: Rhoda Baer
A review of clinical trial data from 2012 suggests Hispanic patients are underrepresented in US cancer studies, and many trial publications fail to provide information on patients’ racial/ethnic backgrounds.
Researchers analyzed 159 reports of phase 2 and 3 trials and found that roughly 21% included information on the number of minority patients enrolled.
About 8% of the publications included data on the number of Hispanic patients enrolled.
And from this data, the investigators found the accrual rate for Hispanic patients was about 4%.
According to the researchers, this lack of information and low representation inhibits physicians’ ability to provide optimal treatment to Hispanic cancer patients and patients belonging to other minority groups.
“We have a major responsibility to ensure adequate representation,” said study author Ian M. Thompson Jr, MD, of The University of Texas Health Science Center at San Antonio.
“How else will we know how best to treat our patients, and how else are we going to reduce the health disparities in [the Hispanic] population?”
Dr Thompson and his colleagues have a particular interest in the Hispanic population because 58% of San Antonio residents are Hispanic, as are 68% of residents in South Texas as a whole.
So the investigators wanted to determine Hispanic accrual rates in randomized trials of cancer patients. The team evaluated data from phase 2 and 3 cancer trials published in 2012. They focused on studies that were considered likely to change the standard of care and were published in “high-impact” journals.
The researchers identified 159 trials—68 phase 2 studies and 91 phase 3 studies. They discovered that 33 of the trial publications—about 21%—disclosed data on minority accrual. And 13 publications—about 8%—included data on the accrual of Hispanic cancer patients.
Of the 4154 patients enrolled on those 13 trials, 162 were Hispanic, which translates to an overall accrual rate of 3.9%. The enrollment of Hispanic patients ranged from 1 patient (0.5%) in a phase 2 trial of lung cancer to 17 patients (26%) in a phase 2 study of acute lymphoblastic leukemia.
“Fundamentally, in the most recent published cancer clinical trials, either the number and proportion of Hispanics are not reported or are far below their actual representation in the national population,” Dr Thompson summarized.
“For institutions like ours that serve a ‘minority-majority’ population, it’s a major responsibility for us to ensure adequate representation so that we can tell our patients how they can best be treated and how we can reduce the disparities of this rapidly growing population.”
Dr Thompson and his colleagues described this research in the Journal of Clinical Oncology. ![]()

Credit: Rhoda Baer
A review of clinical trial data from 2012 suggests Hispanic patients are underrepresented in US cancer studies, and many trial publications fail to provide information on patients’ racial/ethnic backgrounds.
Researchers analyzed 159 reports of phase 2 and 3 trials and found that roughly 21% included information on the number of minority patients enrolled.
About 8% of the publications included data on the number of Hispanic patients enrolled.
And from this data, the investigators found the accrual rate for Hispanic patients was about 4%.
According to the researchers, this lack of information and low representation inhibits physicians’ ability to provide optimal treatment to Hispanic cancer patients and patients belonging to other minority groups.
“We have a major responsibility to ensure adequate representation,” said study author Ian M. Thompson Jr, MD, of The University of Texas Health Science Center at San Antonio.
“How else will we know how best to treat our patients, and how else are we going to reduce the health disparities in [the Hispanic] population?”
Dr Thompson and his colleagues have a particular interest in the Hispanic population because 58% of San Antonio residents are Hispanic, as are 68% of residents in South Texas as a whole.
So the investigators wanted to determine Hispanic accrual rates in randomized trials of cancer patients. The team evaluated data from phase 2 and 3 cancer trials published in 2012. They focused on studies that were considered likely to change the standard of care and were published in “high-impact” journals.
The researchers identified 159 trials—68 phase 2 studies and 91 phase 3 studies. They discovered that 33 of the trial publications—about 21%—disclosed data on minority accrual. And 13 publications—about 8%—included data on the accrual of Hispanic cancer patients.
Of the 4154 patients enrolled on those 13 trials, 162 were Hispanic, which translates to an overall accrual rate of 3.9%. The enrollment of Hispanic patients ranged from 1 patient (0.5%) in a phase 2 trial of lung cancer to 17 patients (26%) in a phase 2 study of acute lymphoblastic leukemia.
“Fundamentally, in the most recent published cancer clinical trials, either the number and proportion of Hispanics are not reported or are far below their actual representation in the national population,” Dr Thompson summarized.
“For institutions like ours that serve a ‘minority-majority’ population, it’s a major responsibility for us to ensure adequate representation so that we can tell our patients how they can best be treated and how we can reduce the disparities of this rapidly growing population.”
Dr Thompson and his colleagues described this research in the Journal of Clinical Oncology.
C difficile vaccine generates immune response
on a blood agar plate
Credit: CDC
BOSTON—Results of a phase 2 study suggest an investigational vaccine may be able to prevent Clostridium difficile infection (CDI).
The vaccine generated an immune response against C difficile toxins A and B. And adverse reactions were generally mild and of short duration.
Researchers presented these results at the 114th General Meeting of the American Society for Microbiology. The study was sponsored by Sanofi, the company developing the vaccine.
“C diff infection threatens the many people who frequently use antibiotics, as well as older hospitalized patients and residents in long-term healthcare facilities,” said study investigator Jamshid Saleh, MD of the Northern California Clinical Research Center in Redding, California.
“It would be great if we could offer patients a way to help prevent this contagious and debilitating disease versus just treating it after it happens.”
To find out if Sanofi’s vaccine would do the job, Dr Saleh and his colleagues conducted a randomized, multicenter trial split into 2 stages.
The first stage, conducted with 455 volunteers, was placebo-controlled, double-blind, and designed for dose and formulation selection. The second stage, which included 206 additional volunteers, was designed to compare the dose and formulation chosen in the first stage against 2 alternate dosing schedules.
Volunteers ranged in age from 40 to 75 years and were at risk of CDI due to impending hospitalization or residence in a long-term healthcare facility.
In stage 1, volunteers were randomized into 1 of 5 study groups: high-dose or low-dose vaccine, either with or without adjuvant, or placebo. Each formulation was administered on days 0, 7, and 30.
Researchers measured immune responses using both enzyme linked immunosorbent assay (ELISA), which assesses antitoxin A and B immunoglobulin G (IgG) concentrations, and toxin neutralization activity (TNA), which measures antitoxin A and B neutralizing activity.
Composite ELISA ranking analysis determined that the high-dose plus adjuvant vaccine formulation (group 3) generated the greatest immune response over a 60-day period. ELISA results also showed a 4-fold increase in the development of detectable antibodies for both toxins A and B.
So the researchers selected the high-dose plus adjuvant vaccine formulation for further study in stage 2 of the trial. They compared its use across 3 schedules: days 0, 7, and 30 (group 3, n=101); days 0, 7, and 180 (group 6, n=103); and days 0, 30, and 180 (group 7, n=103). The team analyzed subjects on days 0, 7, 14, 30, 60, 180, and 210.
There were increased immune responses in all vaccine groups and with each dose, according to ELISA and TNA. Overall, group 3 demonstrated the most favorable immune profile over the 30-, 60- and 180-day periods, particularly in volunteers aged 65-75 years.
The safety profile of all vaccine doses was deemed acceptable throughout the study. Reactions were monitored until day 210 and were generally grade 1, of short duration, did not lead to study discontinuation, and were not considered clinically significant.
“Sanofi Pasteur’s investigational vaccine stimulates a person’s immune system to fight C diff toxins upon exposure and, ultimately, may help prevent a future CDI from occurring,” Dr Saleh said.
“Like other toxoid vaccines—such as tetanus, diphtheria, and whooping cough—this investigational vaccine targets the symptom-causing toxins generated by C diff bacteria and could be an important public health measure to help protect individuals from CDI.”
Based on the phase 2 results, researchers started a phase 3 trial in August 2013.

on a blood agar plate
Credit: CDC
BOSTON—Results of a phase 2 study suggest an investigational vaccine may be able to prevent Clostridium difficile infection (CDI).
The vaccine generated an immune response against C difficile toxins A and B. And adverse reactions were generally mild and of short duration.
Researchers presented these results at the 114th General Meeting of the American Society for Microbiology. The study was sponsored by Sanofi, the company developing the vaccine.
“C diff infection threatens the many people who frequently use antibiotics, as well as older hospitalized patients and residents in long-term healthcare facilities,” said study investigator Jamshid Saleh, MD of the Northern California Clinical Research Center in Redding, California.
“It would be great if we could offer patients a way to help prevent this contagious and debilitating disease versus just treating it after it happens.”
To find out if Sanofi’s vaccine would do the job, Dr Saleh and his colleagues conducted a randomized, multicenter trial split into 2 stages.
The first stage, conducted with 455 volunteers, was placebo-controlled, double-blind, and designed for dose and formulation selection. The second stage, which included 206 additional volunteers, was designed to compare the dose and formulation chosen in the first stage against 2 alternate dosing schedules.
Volunteers ranged in age from 40 to 75 years and were at risk of CDI due to impending hospitalization or residence in a long-term healthcare facility.
In stage 1, volunteers were randomized into 1 of 5 study groups: high-dose or low-dose vaccine, either with or without adjuvant, or placebo. Each formulation was administered on days 0, 7, and 30.
Researchers measured immune responses using both enzyme linked immunosorbent assay (ELISA), which assesses antitoxin A and B immunoglobulin G (IgG) concentrations, and toxin neutralization activity (TNA), which measures antitoxin A and B neutralizing activity.
Composite ELISA ranking analysis determined that the high-dose plus adjuvant vaccine formulation (group 3) generated the greatest immune response over a 60-day period. ELISA results also showed a 4-fold increase in the development of detectable antibodies for both toxins A and B.
So the researchers selected the high-dose plus adjuvant vaccine formulation for further study in stage 2 of the trial. They compared its use across 3 schedules: days 0, 7, and 30 (group 3, n=101); days 0, 7, and 180 (group 6, n=103); and days 0, 30, and 180 (group 7, n=103). The team analyzed subjects on days 0, 7, 14, 30, 60, 180, and 210.
There were increased immune responses in all vaccine groups and with each dose, according to ELISA and TNA. Overall, group 3 demonstrated the most favorable immune profile over the 30-, 60- and 180-day periods, particularly in volunteers aged 65-75 years.
The safety profile of all vaccine doses was deemed acceptable throughout the study. Reactions were monitored until day 210 and were generally grade 1, of short duration, did not lead to study discontinuation, and were not considered clinically significant.
“Sanofi Pasteur’s investigational vaccine stimulates a person’s immune system to fight C diff toxins upon exposure and, ultimately, may help prevent a future CDI from occurring,” Dr Saleh said.
“Like other toxoid vaccines—such as tetanus, diphtheria, and whooping cough—this investigational vaccine targets the symptom-causing toxins generated by C diff bacteria and could be an important public health measure to help protect individuals from CDI.”
Based on the phase 2 results, researchers started a phase 3 trial in August 2013.

on a blood agar plate
Credit: CDC
BOSTON—Results of a phase 2 study suggest an investigational vaccine may be able to prevent Clostridium difficile infection (CDI).
The vaccine generated an immune response against C difficile toxins A and B. And adverse reactions were generally mild and of short duration.
Researchers presented these results at the 114th General Meeting of the American Society for Microbiology. The study was sponsored by Sanofi, the company developing the vaccine.
“C diff infection threatens the many people who frequently use antibiotics, as well as older hospitalized patients and residents in long-term healthcare facilities,” said study investigator Jamshid Saleh, MD of the Northern California Clinical Research Center in Redding, California.
“It would be great if we could offer patients a way to help prevent this contagious and debilitating disease versus just treating it after it happens.”
To find out if Sanofi’s vaccine would do the job, Dr Saleh and his colleagues conducted a randomized, multicenter trial split into 2 stages.
The first stage, conducted with 455 volunteers, was placebo-controlled, double-blind, and designed for dose and formulation selection. The second stage, which included 206 additional volunteers, was designed to compare the dose and formulation chosen in the first stage against 2 alternate dosing schedules.
Volunteers ranged in age from 40 to 75 years and were at risk of CDI due to impending hospitalization or residence in a long-term healthcare facility.
In stage 1, volunteers were randomized into 1 of 5 study groups: high-dose or low-dose vaccine, either with or without adjuvant, or placebo. Each formulation was administered on days 0, 7, and 30.
Researchers measured immune responses using both enzyme linked immunosorbent assay (ELISA), which assesses antitoxin A and B immunoglobulin G (IgG) concentrations, and toxin neutralization activity (TNA), which measures antitoxin A and B neutralizing activity.
Composite ELISA ranking analysis determined that the high-dose plus adjuvant vaccine formulation (group 3) generated the greatest immune response over a 60-day period. ELISA results also showed a 4-fold increase in the development of detectable antibodies for both toxins A and B.
So the researchers selected the high-dose plus adjuvant vaccine formulation for further study in stage 2 of the trial. They compared its use across 3 schedules: days 0, 7, and 30 (group 3, n=101); days 0, 7, and 180 (group 6, n=103); and days 0, 30, and 180 (group 7, n=103). The team analyzed subjects on days 0, 7, 14, 30, 60, 180, and 210.
There were increased immune responses in all vaccine groups and with each dose, according to ELISA and TNA. Overall, group 3 demonstrated the most favorable immune profile over the 30-, 60- and 180-day periods, particularly in volunteers aged 65-75 years.
The safety profile of all vaccine doses was deemed acceptable throughout the study. Reactions were monitored until day 210 and were generally grade 1, of short duration, did not lead to study discontinuation, and were not considered clinically significant.
“Sanofi Pasteur’s investigational vaccine stimulates a person’s immune system to fight C diff toxins upon exposure and, ultimately, may help prevent a future CDI from occurring,” Dr Saleh said.
“Like other toxoid vaccines—such as tetanus, diphtheria, and whooping cough—this investigational vaccine targets the symptom-causing toxins generated by C diff bacteria and could be an important public health measure to help protect individuals from CDI.”
Based on the phase 2 results, researchers started a phase 3 trial in August 2013.
Olive oil may protect against adverse vascular effects
SAN DIEGO—Taking olive oil supplements may counteract some of the adverse cardiovascular effects of air pollution, according to a new study.
“Exposure to airborne particulate matter can lead to endothelial dysfunction, a condition in which the endothelium of blood vessels does not function normally, which is a risk factor for clinical cardiovascular events and progression of atherosclerosis,” said Haiyan Tong,
MD, PhD, of the US Environmental Protection Agency.
“As olive oil and fish oil are known to have beneficial effects on endothelial dysfunction, we examined whether use of these supplements would counteract the adverse cardiovascular effects of exposure to concentrated ambient particulate matter in a controlled setting.”
Dr Tong and his colleagues presented their findings at the American Thoracic Society’s 2014 International Conference (abstract 55100).
Their study involved 42 healthy adults who were randomized to receive 3 g/day of olive oil, fish oil, or no supplements for 4 weeks. Subjects then underwent controlled, 2-hour exposures to filtered air, followed on the next day by exposure to fine/ultrafine concentrated ambient particulate matter (CAP, mean mass concentration 253±16 µg/m3) in a controlled-exposure chamber.
The researchers assessed endothelial function by sonographic measurement of flow-mediated dilation of the brachial artery before, immediately after, and 20 hours after exposure to air and CAP. They also measured blood markers of vasoconstriction and fibrinolysis.
Immediately after exposure to CAP, there were significant particulate matter mass-dependent reductions in flow-mediated dilation in the control group (-19.4±8.4% per 100 µg/m3 increase in CAP concentration relative to pre-filtered air levels) and the fish oil group (-13.7±5.3%), but the decrease in the olive oil group was not significant (-7.6±6.8%).
Tissue plasminogen activator increased immediately after CAP exposure in the olive oil group (11.6±5%), and this effect persisted for up to 20 hours.
Olive oil supplementation also ameliorated changes in blood markers associated with vasoconstriction and fibrinolysis, while fish oil supplementation had no effect on endothelial function or fibrinolysis after CAP exposure.
“Our study suggests that use of olive oil supplements may protect against the adverse vascular effects of exposure to air pollution particles,” Dr Tong said. “If these results are replicated in further studies, use of these supplements might offer a safe, low-cost, and effective means of counteracting some of the health consequences of exposure to air pollution.”

SAN DIEGO—Taking olive oil supplements may counteract some of the adverse cardiovascular effects of air pollution, according to a new study.
“Exposure to airborne particulate matter can lead to endothelial dysfunction, a condition in which the endothelium of blood vessels does not function normally, which is a risk factor for clinical cardiovascular events and progression of atherosclerosis,” said Haiyan Tong,
MD, PhD, of the US Environmental Protection Agency.
“As olive oil and fish oil are known to have beneficial effects on endothelial dysfunction, we examined whether use of these supplements would counteract the adverse cardiovascular effects of exposure to concentrated ambient particulate matter in a controlled setting.”
Dr Tong and his colleagues presented their findings at the American Thoracic Society’s 2014 International Conference (abstract 55100).
Their study involved 42 healthy adults who were randomized to receive 3 g/day of olive oil, fish oil, or no supplements for 4 weeks. Subjects then underwent controlled, 2-hour exposures to filtered air, followed on the next day by exposure to fine/ultrafine concentrated ambient particulate matter (CAP, mean mass concentration 253±16 µg/m3) in a controlled-exposure chamber.
The researchers assessed endothelial function by sonographic measurement of flow-mediated dilation of the brachial artery before, immediately after, and 20 hours after exposure to air and CAP. They also measured blood markers of vasoconstriction and fibrinolysis.
Immediately after exposure to CAP, there were significant particulate matter mass-dependent reductions in flow-mediated dilation in the control group (-19.4±8.4% per 100 µg/m3 increase in CAP concentration relative to pre-filtered air levels) and the fish oil group (-13.7±5.3%), but the decrease in the olive oil group was not significant (-7.6±6.8%).
Tissue plasminogen activator increased immediately after CAP exposure in the olive oil group (11.6±5%), and this effect persisted for up to 20 hours.
Olive oil supplementation also ameliorated changes in blood markers associated with vasoconstriction and fibrinolysis, while fish oil supplementation had no effect on endothelial function or fibrinolysis after CAP exposure.
“Our study suggests that use of olive oil supplements may protect against the adverse vascular effects of exposure to air pollution particles,” Dr Tong said. “If these results are replicated in further studies, use of these supplements might offer a safe, low-cost, and effective means of counteracting some of the health consequences of exposure to air pollution.”

SAN DIEGO—Taking olive oil supplements may counteract some of the adverse cardiovascular effects of air pollution, according to a new study.
“Exposure to airborne particulate matter can lead to endothelial dysfunction, a condition in which the endothelium of blood vessels does not function normally, which is a risk factor for clinical cardiovascular events and progression of atherosclerosis,” said Haiyan Tong,
MD, PhD, of the US Environmental Protection Agency.
“As olive oil and fish oil are known to have beneficial effects on endothelial dysfunction, we examined whether use of these supplements would counteract the adverse cardiovascular effects of exposure to concentrated ambient particulate matter in a controlled setting.”
Dr Tong and his colleagues presented their findings at the American Thoracic Society’s 2014 International Conference (abstract 55100).
Their study involved 42 healthy adults who were randomized to receive 3 g/day of olive oil, fish oil, or no supplements for 4 weeks. Subjects then underwent controlled, 2-hour exposures to filtered air, followed on the next day by exposure to fine/ultrafine concentrated ambient particulate matter (CAP, mean mass concentration 253±16 µg/m3) in a controlled-exposure chamber.
The researchers assessed endothelial function by sonographic measurement of flow-mediated dilation of the brachial artery before, immediately after, and 20 hours after exposure to air and CAP. They also measured blood markers of vasoconstriction and fibrinolysis.
Immediately after exposure to CAP, there were significant particulate matter mass-dependent reductions in flow-mediated dilation in the control group (-19.4±8.4% per 100 µg/m3 increase in CAP concentration relative to pre-filtered air levels) and the fish oil group (-13.7±5.3%), but the decrease in the olive oil group was not significant (-7.6±6.8%).
Tissue plasminogen activator increased immediately after CAP exposure in the olive oil group (11.6±5%), and this effect persisted for up to 20 hours.
Olive oil supplementation also ameliorated changes in blood markers associated with vasoconstriction and fibrinolysis, while fish oil supplementation had no effect on endothelial function or fibrinolysis after CAP exposure.
“Our study suggests that use of olive oil supplements may protect against the adverse vascular effects of exposure to air pollution particles,” Dr Tong said. “If these results are replicated in further studies, use of these supplements might offer a safe, low-cost, and effective means of counteracting some of the health consequences of exposure to air pollution.”
Chip may allow for early cancer detection

Institute of Photonic Sciences
Scientists say they’ve developed a lab-on-a-chip device capable of detecting protein markers for cancer.
The device can detect very low concentrations of protein markers in the blood, enabling cancer diagnosis in its earliest stages, the team says.
Romain Quidant, PhD, of The Institute of Photonic Sciences in Barcelona, Spain, and his colleagues described the device in Nano Letters.
The lab on a chip hosts 32 sensing sites distributed across a network of 8 fluidic microchannels that enables it to conduct multiple analyses.
Gold nanoparticles lie on the surface of the chip and are chemically programed with an antibody receptor in such a way that they are capable of specifically attracting the protein markers circulating in blood.
When a drop of blood is injected into the chip, it circulates through the microchannels, and, if cancer markers are present in the blood, they will stick to the nanoparticles located on the microchannels as they pass by, setting off changes in what is known as the plasmonic resonance.
The device monitors these changes, the magnitude of which is directly related to the concentration/number of markers in the patient’s blood. In this way, it provides a direct assessment of the patient’s risk of developing cancer.
“The most fascinating finding is that we are capable of detecting extremely low concentrations of this protein in a matter of minutes, making this device an ultra-high-sensitivity, state-of-the-art, powerful instrument that will benefit early detection and treatment monitoring of cancer,” Dr Quidant said.

Institute of Photonic Sciences
Scientists say they’ve developed a lab-on-a-chip device capable of detecting protein markers for cancer.
The device can detect very low concentrations of protein markers in the blood, enabling cancer diagnosis in its earliest stages, the team says.
Romain Quidant, PhD, of The Institute of Photonic Sciences in Barcelona, Spain, and his colleagues described the device in Nano Letters.
The lab on a chip hosts 32 sensing sites distributed across a network of 8 fluidic microchannels that enables it to conduct multiple analyses.
Gold nanoparticles lie on the surface of the chip and are chemically programed with an antibody receptor in such a way that they are capable of specifically attracting the protein markers circulating in blood.
When a drop of blood is injected into the chip, it circulates through the microchannels, and, if cancer markers are present in the blood, they will stick to the nanoparticles located on the microchannels as they pass by, setting off changes in what is known as the plasmonic resonance.
The device monitors these changes, the magnitude of which is directly related to the concentration/number of markers in the patient’s blood. In this way, it provides a direct assessment of the patient’s risk of developing cancer.
“The most fascinating finding is that we are capable of detecting extremely low concentrations of this protein in a matter of minutes, making this device an ultra-high-sensitivity, state-of-the-art, powerful instrument that will benefit early detection and treatment monitoring of cancer,” Dr Quidant said.

Institute of Photonic Sciences
Scientists say they’ve developed a lab-on-a-chip device capable of detecting protein markers for cancer.
The device can detect very low concentrations of protein markers in the blood, enabling cancer diagnosis in its earliest stages, the team says.
Romain Quidant, PhD, of The Institute of Photonic Sciences in Barcelona, Spain, and his colleagues described the device in Nano Letters.
The lab on a chip hosts 32 sensing sites distributed across a network of 8 fluidic microchannels that enables it to conduct multiple analyses.
Gold nanoparticles lie on the surface of the chip and are chemically programed with an antibody receptor in such a way that they are capable of specifically attracting the protein markers circulating in blood.
When a drop of blood is injected into the chip, it circulates through the microchannels, and, if cancer markers are present in the blood, they will stick to the nanoparticles located on the microchannels as they pass by, setting off changes in what is known as the plasmonic resonance.
The device monitors these changes, the magnitude of which is directly related to the concentration/number of markers in the patient’s blood. In this way, it provides a direct assessment of the patient’s risk of developing cancer.
“The most fascinating finding is that we are capable of detecting extremely low concentrations of this protein in a matter of minutes, making this device an ultra-high-sensitivity, state-of-the-art, powerful instrument that will benefit early detection and treatment monitoring of cancer,” Dr Quidant said.