User login
Denosumab boosts bone strength in glucocorticoid users
Bone strength and microarchitecture remained stronger at 24 months after treatment with denosumab compared to risedronate, in a study of 110 adults using glucocorticoids.
Patients using glucocorticoids are at increased risk for vertebral and nonvertebral fractures at both the start of treatment or as treatment continues, wrote Piet Geusens, MD, of Maastricht University, the Netherlands, and colleagues.
Imaging data collected via high-resolution peripheral quantitative computed tomography (HR-pQCT) allow for the assessment of bone microarchitecture and strength, but specific data comparing the impact of bone treatment in patients using glucocorticoids are lacking, they said.
In a study published in the Journal of Bone and Mineral Research, the researchers identified a subset of 56 patients randomized to denosumab and 54 to risedronate patients out of a total of 590 patients who were enrolled in a phase 3 randomized, controlled trial of denosumab vs. risedronate for bone mineral density. The main results of the larger trial – presented at EULAR 2018 – showed greater increases in bone strength with denosumab over risedronate in patients receiving glucocorticoids.
In the current study, the researchers reviewed HR-pQCT scans of the distal radius and tibia at baseline, 12 months, and 24 months. Bone strength and microarchitecture were defined in terms of failure load (FL) as a primary outcome. Patients also were divided into subpopulations of those initiating glucocorticoid treatment (GC-I) and continuing treatment (GC-C).
Baseline characteristics were mainly balanced among the treatment groups within the GC-I and GC-C categories.
Among the GC-I patients, in the denosumab group, FL increased significantly from baseline to 12 months at the radius at tibia (1.8% and 1.7%, respectively) but did not change significantly in the risedronate group, which translated to a significant treatment difference between the drugs of 3.3% for radius and 2.5% for tibia.
At 24 months, the radius measure of FL was unchanged from baseline in denosumab patients but significantly decreased in risedronate patients, with a difference of –4.1%, which translated to a significant between-treatment difference at the radius of 5.6% (P < .001). Changes at the tibia were not significantly different between the groups at 24 months.
Among the GC-C patients, FL was unchanged from baseline to 12 months for both the denosumab and risedronate groups. However, FL significantly increased with denosumab (4.3%) and remained unchanged in the risedronate group.
The researchers also found significant differences between denosumab and risedronate in percentage changes in cortical bone mineral density, and less prominent changes and differences in trabecular bone mineral density.
The study findings were limited by several factors including the use of the HR-pQCT scanner, which limits the measurement of trabecular microarchitecture, and the use of only standard HR-pQCT parameters, which do not allow insight into endosteal changes, and the inability to correct for multiplicity of data, the researchers noted.
However, the results support the superiority of denosumab over risedronate for preventing FL and total bone mineral density loss at the radius and tibia in new glucocorticoid users, and for increasing FL and total bone mineral density at the radius in long-term glucocorticoid users, they said.
Denosumab therefore could be a useful therapeutic option and could inform decision-making in patients initiating GC-therapy or on long-term GC-therapy, they concluded.
The study was supported by Amgen. Dr. Geusens disclosed grants from Amgen, Celgene, Lilly, Merck, Pfizer, Roche, UCB, Fresenius, Mylan, and Sandoz, and grants and other funding from AbbVie, outside the current study.
Bone strength and microarchitecture remained stronger at 24 months after treatment with denosumab compared to risedronate, in a study of 110 adults using glucocorticoids.
Patients using glucocorticoids are at increased risk for vertebral and nonvertebral fractures at both the start of treatment or as treatment continues, wrote Piet Geusens, MD, of Maastricht University, the Netherlands, and colleagues.
Imaging data collected via high-resolution peripheral quantitative computed tomography (HR-pQCT) allow for the assessment of bone microarchitecture and strength, but specific data comparing the impact of bone treatment in patients using glucocorticoids are lacking, they said.
In a study published in the Journal of Bone and Mineral Research, the researchers identified a subset of 56 patients randomized to denosumab and 54 to risedronate patients out of a total of 590 patients who were enrolled in a phase 3 randomized, controlled trial of denosumab vs. risedronate for bone mineral density. The main results of the larger trial – presented at EULAR 2018 – showed greater increases in bone strength with denosumab over risedronate in patients receiving glucocorticoids.
In the current study, the researchers reviewed HR-pQCT scans of the distal radius and tibia at baseline, 12 months, and 24 months. Bone strength and microarchitecture were defined in terms of failure load (FL) as a primary outcome. Patients also were divided into subpopulations of those initiating glucocorticoid treatment (GC-I) and continuing treatment (GC-C).
Baseline characteristics were mainly balanced among the treatment groups within the GC-I and GC-C categories.
Among the GC-I patients, in the denosumab group, FL increased significantly from baseline to 12 months at the radius at tibia (1.8% and 1.7%, respectively) but did not change significantly in the risedronate group, which translated to a significant treatment difference between the drugs of 3.3% for radius and 2.5% for tibia.
At 24 months, the radius measure of FL was unchanged from baseline in denosumab patients but significantly decreased in risedronate patients, with a difference of –4.1%, which translated to a significant between-treatment difference at the radius of 5.6% (P < .001). Changes at the tibia were not significantly different between the groups at 24 months.
Among the GC-C patients, FL was unchanged from baseline to 12 months for both the denosumab and risedronate groups. However, FL significantly increased with denosumab (4.3%) and remained unchanged in the risedronate group.
The researchers also found significant differences between denosumab and risedronate in percentage changes in cortical bone mineral density, and less prominent changes and differences in trabecular bone mineral density.
The study findings were limited by several factors including the use of the HR-pQCT scanner, which limits the measurement of trabecular microarchitecture, and the use of only standard HR-pQCT parameters, which do not allow insight into endosteal changes, and the inability to correct for multiplicity of data, the researchers noted.
However, the results support the superiority of denosumab over risedronate for preventing FL and total bone mineral density loss at the radius and tibia in new glucocorticoid users, and for increasing FL and total bone mineral density at the radius in long-term glucocorticoid users, they said.
Denosumab therefore could be a useful therapeutic option and could inform decision-making in patients initiating GC-therapy or on long-term GC-therapy, they concluded.
The study was supported by Amgen. Dr. Geusens disclosed grants from Amgen, Celgene, Lilly, Merck, Pfizer, Roche, UCB, Fresenius, Mylan, and Sandoz, and grants and other funding from AbbVie, outside the current study.
Bone strength and microarchitecture remained stronger at 24 months after treatment with denosumab compared to risedronate, in a study of 110 adults using glucocorticoids.
Patients using glucocorticoids are at increased risk for vertebral and nonvertebral fractures at both the start of treatment or as treatment continues, wrote Piet Geusens, MD, of Maastricht University, the Netherlands, and colleagues.
Imaging data collected via high-resolution peripheral quantitative computed tomography (HR-pQCT) allow for the assessment of bone microarchitecture and strength, but specific data comparing the impact of bone treatment in patients using glucocorticoids are lacking, they said.
In a study published in the Journal of Bone and Mineral Research, the researchers identified a subset of 56 patients randomized to denosumab and 54 to risedronate patients out of a total of 590 patients who were enrolled in a phase 3 randomized, controlled trial of denosumab vs. risedronate for bone mineral density. The main results of the larger trial – presented at EULAR 2018 – showed greater increases in bone strength with denosumab over risedronate in patients receiving glucocorticoids.
In the current study, the researchers reviewed HR-pQCT scans of the distal radius and tibia at baseline, 12 months, and 24 months. Bone strength and microarchitecture were defined in terms of failure load (FL) as a primary outcome. Patients also were divided into subpopulations of those initiating glucocorticoid treatment (GC-I) and continuing treatment (GC-C).
Baseline characteristics were mainly balanced among the treatment groups within the GC-I and GC-C categories.
Among the GC-I patients, in the denosumab group, FL increased significantly from baseline to 12 months at the radius at tibia (1.8% and 1.7%, respectively) but did not change significantly in the risedronate group, which translated to a significant treatment difference between the drugs of 3.3% for radius and 2.5% for tibia.
At 24 months, the radius measure of FL was unchanged from baseline in denosumab patients but significantly decreased in risedronate patients, with a difference of –4.1%, which translated to a significant between-treatment difference at the radius of 5.6% (P < .001). Changes at the tibia were not significantly different between the groups at 24 months.
Among the GC-C patients, FL was unchanged from baseline to 12 months for both the denosumab and risedronate groups. However, FL significantly increased with denosumab (4.3%) and remained unchanged in the risedronate group.
The researchers also found significant differences between denosumab and risedronate in percentage changes in cortical bone mineral density, and less prominent changes and differences in trabecular bone mineral density.
The study findings were limited by several factors including the use of the HR-pQCT scanner, which limits the measurement of trabecular microarchitecture, and the use of only standard HR-pQCT parameters, which do not allow insight into endosteal changes, and the inability to correct for multiplicity of data, the researchers noted.
However, the results support the superiority of denosumab over risedronate for preventing FL and total bone mineral density loss at the radius and tibia in new glucocorticoid users, and for increasing FL and total bone mineral density at the radius in long-term glucocorticoid users, they said.
Denosumab therefore could be a useful therapeutic option and could inform decision-making in patients initiating GC-therapy or on long-term GC-therapy, they concluded.
The study was supported by Amgen. Dr. Geusens disclosed grants from Amgen, Celgene, Lilly, Merck, Pfizer, Roche, UCB, Fresenius, Mylan, and Sandoz, and grants and other funding from AbbVie, outside the current study.
FROM THE JOURNAL OF BONE AND MINERAL RESEARCH
Medical assistants identify strategies and barriers to clinic efficiency
ABSTRACT
Background: Medical assistant (MA) roles have expanded rapidly as primary care has evolved and MAs take on new patient care duties. Research that looks at the MA experience and factors that enhance or reduce efficiency among MAs is limited.
Methods: We surveyed all MAs working in 6 clinics run by a large academic family medicine department in Ann Arbor, Michigan. MAs deemed by peers as “most efficient” were selected for follow-up interviews. We evaluated personal strategies for efficiency, barriers to efficient care, impact of physician actions on efficiency, and satisfaction.
Results: A total of 75/86 MAs (87%) responded to at least some survey questions and 61/86 (71%) completed the full survey. We interviewed 18 MAs face to face. Most saw their role as essential to clinic functioning and viewed health care as a personal calling. MAs identified common strategies to improve efficiency and described the MA role to orchestrate the flow of the clinic day. Staff recognized differing priorities of patients, staff, and physicians and articulated frustrations with hierarchy and competing priorities as well as behaviors that impeded clinic efficiency. Respondents emphasized the importance of feeling valued by others on their team.
Conclusions: With the evolving demands made on MAs’ time, it is critical to understand how the most effective staff members manage their role and highlight the strategies they employ to provide efficient clinical care. Understanding factors that increase or decrease MA job satisfaction can help identify high-efficiency practices and promote a clinic culture that values and supports all staff.
As primary care continues to evolve into more team-based practice, the role of the medical assistant (MA) has rapidly transformed.1 Staff may assist with patient management, documentation in the electronic medical record, order entry, pre-visit planning, and fulfillment of quality metrics, particularly in a Primary Care Medical Home (PCMH).2 From 2012 through 2014, MA job postings per graduate increased from 1.3 to 2.3, suggesting twice as many job postings as graduates.3 As the demand for experienced MAs increases, the ability to recruit and retain high-performing staff members will be critical.
MAs are referenced in medical literature as early as the 1800s.4 The American Association of Medical Assistants was founded in 1956, which led to educational standardization and certifications.5 Despite the important role that MAs have long played in the proper functioning of a medical clinic—and the knowledge that team configurations impact a clinic’s efficiency and quality6,7—few investigations have sought out the MA’s perspective.8,9 Given the increasing clinical demands placed on all members of the primary care team (and the burnout that often results), it seems that MA insights into clinic efficiency could be valuable.
METHODS
This cross-sectional study was conducted from February to April 2019 at a large academic institution with 6 regional ambulatory care family medicine clinics, each one with 11,000 to 18,000 patient visits annually. Faculty work at all 6 clinics and residents at 2 of them. All MAs are hired, paid, and managed by a central administrative department rather than by the family medicine department. The family medicine clinics are currently PCMH certified, with a mix of fee-for-service and capitated reimbursement.
Continue to: We developed and piloted...
We developed and piloted a voluntary, anonymous 39-question (29 closed-ended and 10 brief open-ended) online Qualtrics survey, which we distributed via an email link to all the MAs in the department. The survey included clinic site, years as an MA, perceptions of the clinic environment, perception of teamwork at their site, identification of efficient practices, and feedback for physicians to improve efficiency and flow. Most questions were Likert-style with 5 choices ranging from “strongly agree” to “strongly disagree” or short answer. Age and gender were omitted to protect confidentiality, as most MAs in the department are female. Participants could opt to enter in a drawing for three $25 gift cards. The survey was reviewed by the University of Michigan Institutional Review Board and deemed exempt.
We asked MAs to nominate peers in their clinic who were “especially efficient and do their jobs well—people that others can learn from.” The staff members who were nominated most frequently by their peers were invited to share additional perspectives via a 10- to 30-minute semi-structured interview with the first author. Interviews covered highly efficient practices, barriers and facilitators to efficient care, and physician behaviors that impaired efficiency. We interviewed a minimum of 2 MAs per clinic and increased the number of interviews through snowball sampling, as needed, to reach data saturation (eg, the point at which we were no longer hearing new content). MAs were assured that all comments would be anonymized. There was no monetary incentive for the interviews. The interviewer had previously met only 3 of the 18 MAs interviewed.
Analysis. Summary statistics were calculated for quantitative data. To compare subgroups (such as individual clinics), a chi-square test was used. In cases when there were small cell sizes (< 5 subjects), we used the Fisher’s Exact test. Qualitative data was collected with real-time typewritten notes during the interviews to capture ideas and verbatim quotes when possible. We also included open-ended comments shared on the Qualtrics survey. Data were organized by theme using a deductive coding approach. Both authors reviewed and discussed observations, and coding was conducted by the first author. Reporting followed the STROBE Statement checklist for cross-sectional studies.10 Results were shared with MAs, supervisory staff, and physicians, which allowed for feedback and comments and served as “member-checking.” MAs reported that the data reflected their lived experiences.
RESULTS
Surveys were distributed to all 86 MAs working in family medicine clinics. A total of 75 (87%) responded to at least some questions (typically just demographics). We used those who completed the full survey (n = 61; 71%) for data analysis. Eighteen MAs participated in face-to-face interviews. Among respondents, 35 (47%) had worked at least 10 years as an MA and 21 (28%) had worked at least a decade in the family medicine department.
Perception of role
All respondents (n = 61; 100%) somewhat or strongly agreed that the MA role was “very important to keep the clinic functioning” and 58 (95%) reported that working in health care was “a calling” for them. Only 7 (11%) agreed that family medicine was an easier environment for MAs compared to a specialty clinic; 30 (49%) disagreed with this. Among respondents, 32 (53%) strongly or somewhat agreed that their work was very stressful and just half (n = 28; 46%) agreed there were adequate MA staff at their clinic.
Continue to: Efficiency and competing priorities
Efficiency and competing priorities
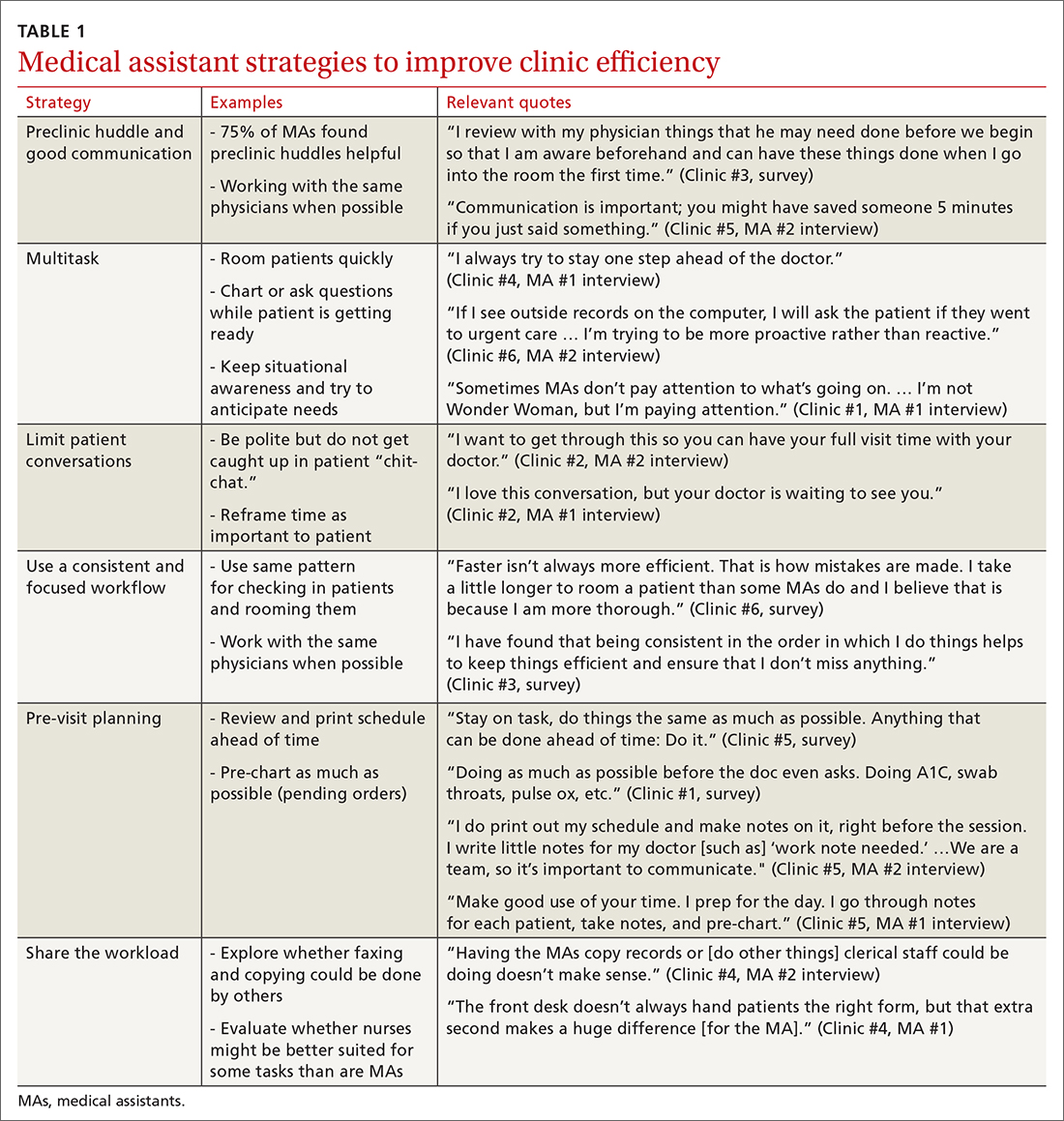
MAs described important work values that increased their efficiency. These included clinic culture (good communication and strong teamwork), as well as individual strategies such as multitasking, limiting patient conversations, and doing tasks in a consistent way to improve accuracy. (See TABLE 1.) They identified ways physicians bolster or hurt efficiency and ways in which the relationship between the physician and the MA shapes the MA’s perception of their value in clinic.
Communication was emphasized as critical for efficient care, and MAs encouraged the use of preclinic huddles and communication as priorities. Seventy-five percent of MAs reported preclinic huddles to plan for patient care were helpful, but only half said huddles took place “always” or “most of the time.” Many described reviewing the schedule and completing tasks ahead of patient arrival as critical to efficiency.
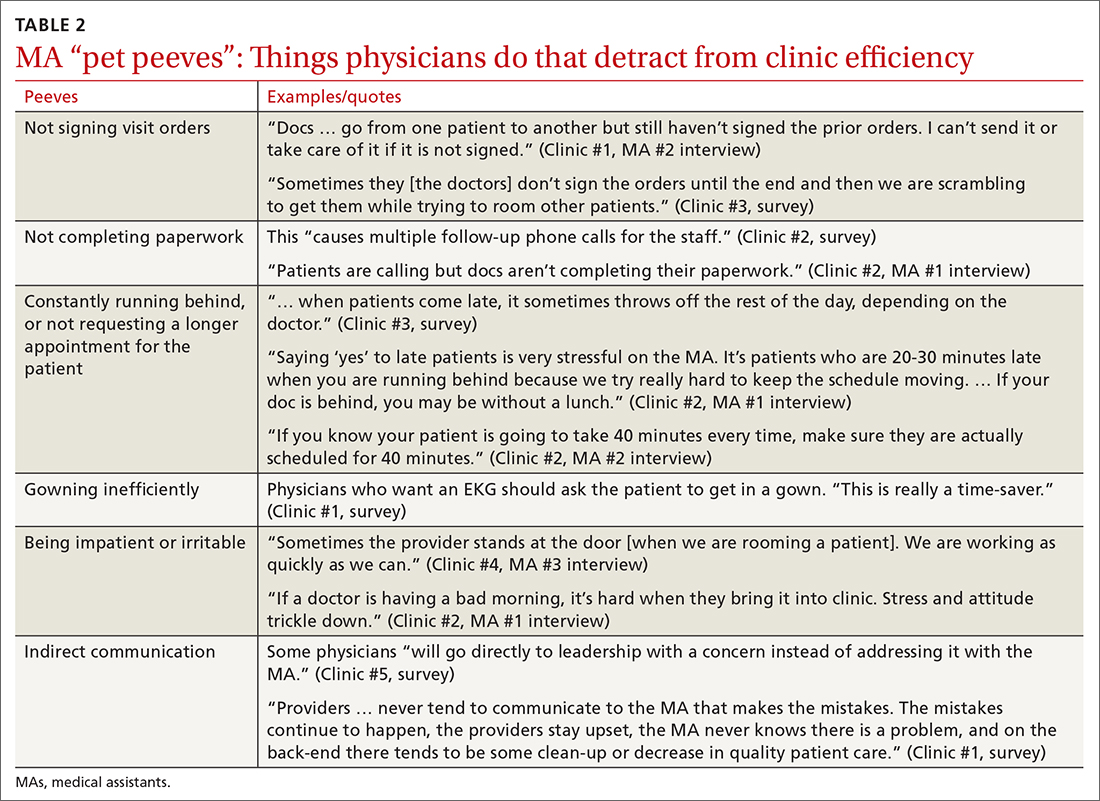
Participants described the tension between their identified role of orchestrating clinic flow and responding to directives by others that disrupted the flow. Several MAs found it challenging when physicians agreed to see very late patients and felt frustrated when decisions that changed the flow were made by the physician or front desk staff without including the MA. MAs were also able to articulate how they managed competing priorities within the clinic, such as when a patient- or physician-driven need to extend appointments was at odds with maintaining a timely schedule. They were eager to share personal tips for time management and prided themselves on careful and accurate performance and skills they had learned on the job. MAs also described how efficiency could be adversely affected by the behaviors or attitudes of physicians. (See TABLE 2.)
Clinic environment
Thirty-six MAs (59%) reported that other MAs on their team were willing to help them out in clinic “a great deal” or “a lot” of the time, by helping to room a patient, acting as a chaperone for an exam, or doing a point-of-care lab. This sense of support varied across clinics (38% to 91% reported good support), suggesting that cultures vary by site. Some MAs expressed frustration at peers they saw as resistant to helping, exemplified by this verbatim quote from an interview:
“ Some don’t want to help out. They may sigh. It’s how they react—you just know.” (Clinic #1, MA #2 interview)
Efficient MAs stressed the need for situational awareness to recognize when co-workers need help:
“ [Peers often] are not aware that another MA is drowning. There’s 5 people who could have done that, and here I am running around and nobody budged.” (Clinic #5, MA #2 interview)
Continue to: A minority of staff...
A minority of staff used the open-ended survey sections to describe clinic hierarchy. When asked about “pet peeves,” a few advised that physicians should not “talk down” to staff and should try to teach rather than criticize. Another asked that physicians not “bark orders” or have “low gratitude” for staff work. MAs found micromanaging stressful—particularly when the physician prompted the MA about patient arrivals:
“[I don’t like] when providers will make a comment about a patient arriving when you already know this information. You then rush to put [the] patient in [a] room, then [the] provider ends up making [the] patient wait an extensive amount of time. I’m perfectly capable of knowing when a patient arrives.” (Clinic #6, survey)
MAs did not like physicians “talking bad about us” or blaming the MA if the clinic is running behind.
Despite these concerns, most MAs reported feeling appreciated for the job they do. Only 10 (16%) reported that the people they work with rarely say “thank you,” and 2 (3%) stated they were not well supported by the physicians in clinic. Most (n = 38; 62%) strongly agreed or agreed that they felt part of the team and that their opinions matter. In the interviews, many expanded on this idea:
“I really feel like I’m valued, so I want to do everything I can to make [my doctor’s] day go better. If you want a good clinic, the best thing a doc can do is make the MA feel valued.” (Clinic #1, MA #1 interview)
DISCUSSION
Participants described their role much as an orchestra director, with MAs as the key to clinic flow and timeliness.9 Respondents articulated multiple common strategies used to increase their own efficiency and clinic flow; these may be considered best practices and incorporated as part of the basic training. Most MAs reported their day-to-day jobs were stressful and believed this was underrecognized, so efficiency strategies are critical. With staff completing multiple time-sensitive tasks during clinic, consistent co-worker support is crucial and may impact efficiency.8 Proper training of managers to provide that support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial.
Several comments reflected the power differential within medical offices. One study reported that MAs and physicians “occupy roles at opposite ends of social and occupational hierarchies.”11 It’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.9 Prior research has found that MAs have higher perceptions of their own competence than do the physicians working with them.12 If there is a fundamental lack of trust between the 2 groups, this will undoubtedly hinder team-building. Attention to this issue is key to a more favorable work environment.
Continue to: Almost all respondents...
Almost all respondents reported health care was a “calling,” which mirrors physician research that suggests seeing work as a “calling” is protective against burnout.13,14 Open-ended comments indicated great pride in contributions, and most staff members felt appreciated by their teams. Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier. Staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients.15 We also uncovered some MA “pet peeves” that hinder efficiency and could be shared with physicians to emphasize the importance of patience and civility.
One barrier to expansion of MA roles within PCMH practices is the limited pay and career ladder for MAs who adopt new job responsibilities that require advanced skills or training.1,2 The mean MA salary at our institution ($37,372) is higher than in our state overall ($33,760), which may impact satisfaction.16 In addition, 93% of MAs are women; thus, they may continue to struggle more with lower pay than do workers in male-dominated professions.17,18 Expected job growth from 2018-2028 is predicted at 23%, which may help to boost salaries.19 Prior studies describe the lack of a job ladder or promotion opportunities as a challenge1,20; this was not formally assessed in our study.
MAs see work in family medicine as much harder than it is in other specialty clinics. Being trusted with more responsibility, greater autonomy,21-23 and expanded patient care roles can boost MA self-efficacy, which can reduce burnout for both physicians and MAs.8,24 However, new responsibilities should include appropriate training, support, and compensation, and match staff interests.7
Study limitations. The study was limited to 6 clinics in 1 department at a large academic medical center. Interviewed participants were selected by convenience and snowball sampling and thus, the results cannot be generalized to the population of MAs as a whole. As the initial interview goal was simply to gather efficiency tips, the project was not designed to be formal qualitative research. However, the discussions built on open-ended comments from the written survey helped contextualize our quantitative findings about efficiency. Notes were documented in real time by a single interviewer with rapid typing skills, which allowed capture of quotes verbatim. Subsequent studies would benefit from more formal qualitative research methods (recording and transcribing interviews, multiple coders to reduce risk of bias, and more complex thematic analysis).
Our research demonstrated how MAs perceive their roles in primary care and the facilitators and barriers to high efficiency in the workplace, which begins to fill an important knowledge gap in primary care. Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.
CORRESPONDENCE
Katherine J. Gold, MD, MSW, MS, Department of Family Medicine and Department of Obstetrics and Gynecology, University of Michigan, 1018 Fuller Street, Ann Arbor, MI 48104-1213; ktgold@umich.edu
1. Chapman SA, Blash LK. New roles for medical assistants in innovative primary care practices. Health Serv Res. 2017;52(suppl 1):383-406.
2. Ferrante JM, Shaw EK, Bayly JE, et al. Barriers and facilitators to expanding roles of medical assistants in patient-centered medical homes (PCMHs). J Am Board Fam Med. 2018;31:226-235.
3. Atkins B. The outlook for medical assisting in 2016 and beyond. Accessed January 27, 2022. www.medicalassistantdegrees.net/articles/medical-assisting-trends/
4. Unqualified medical “assistants.” Hospital (Lond 1886). 1897;23:163-164.
5. Ameritech College of Healthcare. The origins of the AAMA. Accessed January 27, 2022. www.ameritech.edu/blog/medical-assisting-history/
6. Dai M, Willard-Grace R, Knox M, et al. Team configurations, efficiency, and family physician burnout. J Am Board Fam Med. 2020;33:368-377.
7. Harper PG, Van Riper K, Ramer T, et al. Team-based care: an expanded medical assistant role—enhanced rooming and visit assistance. J Interprof Care. 2018:1-7.
8. Sheridan B, Chien AT, Peters AS, et al. Team-based primary care: the medical assistant perspective. Health Care Manage Rev. 2018;43:115-125.
9. Tache S, Hill-Sakurai L. Medical assistants: the invisible “glue” of primary health care practices in the United States? J Health Organ Manag. 2010;24:288-305.
10. STROBE checklist for cohort, case-control, and cross-sectional studies. Accessed January 27, 2022. www.strobe-statement.org/fileadmin/Strobe/uploads/checklists/STROBE_checklist_v4_combined.pdf
11. Gray CP, Harrison MI, Hung D. Medical assistants as flow managers in primary care: challenges and recommendations. J Healthc Manag. 2016;61:181-191.
12. Elder NC, Jacobson CJ, Bolon SK, et al. Patterns of relating between physicians and medical assistants in small family medicine offices. Ann Fam Med. 2014;12:150-157.
13. Jager AJ, Tutty MA, Kao AC. Association between physician burnout and identification with medicine as a calling. Mayo Clinic Proc. 2017;92:415-422.
14. Yoon JD, Daley BM, Curlin FA. The association between a sense of calling and physician well-being: a national study of primary care physicians and psychiatrists. Acad Psychiatry. 2017;41:167-173.
15. Mohr DC, Young GJ, Meterko M, et al. Job satisfaction of primary care team members and quality of care. Am J Med Qual. 2011;26:18-25.
16. US Bureau of Labor Statistics. Occupational employment and wage statistics. Accessed January 27, 2022. https://www.bls.gov/oes/current/oes319092.htm
17. Chapman SA, Marks A, Dower C. Positioning medical assistants for a greater role in the era of health reform. Acad Med. 2015;90:1347-1352.
18. Mandel H. The role of occupational attributes in gender earnings inequality, 1970-2010. Soc Sci Res. 2016;55:122-138.
19. US Bureau of Labor Statistics. Occupational outlook handbook: medical assistants. Accessed January 27, 2022. www.bls.gov/ooh/healthcare/medical-assistants.htm
20. Skillman SM, Dahal A, Frogner BK, et al. Frontline workers’ career pathways: a detailed look at Washington state’s medical assistant workforce. Med Care Res Rev. 2018:1077558718812950.
21. Morse G, Salyers MP, Rollins AL, et al. Burnout in mental health services: a review of the problem and its remediation. Adm Policy Ment Health. 2012;39:341-352.
22. Dubois CA, Bentein K, Ben Mansour JB, et al. Why some employees adopt or resist reorganization of work practices in health care: associations between perceived loss of resources, burnout, and attitudes to change. Int J Environ Res Pub Health. 2014;11:187-201.
23. Aronsson G, Theorell T, Grape T, et al. A systematic review including meta-analysis of work environment and burnout symptoms. BMC Public Health. 2017;17:264.
24. O’Malley AS, Gourevitch R, Draper K, et al. Overcoming challenges to teamwork in patient-centered medical homes: a qualitative study. J Gen Intern Med. 2015;30:183-192.
ABSTRACT
Background: Medical assistant (MA) roles have expanded rapidly as primary care has evolved and MAs take on new patient care duties. Research that looks at the MA experience and factors that enhance or reduce efficiency among MAs is limited.
Methods: We surveyed all MAs working in 6 clinics run by a large academic family medicine department in Ann Arbor, Michigan. MAs deemed by peers as “most efficient” were selected for follow-up interviews. We evaluated personal strategies for efficiency, barriers to efficient care, impact of physician actions on efficiency, and satisfaction.
Results: A total of 75/86 MAs (87%) responded to at least some survey questions and 61/86 (71%) completed the full survey. We interviewed 18 MAs face to face. Most saw their role as essential to clinic functioning and viewed health care as a personal calling. MAs identified common strategies to improve efficiency and described the MA role to orchestrate the flow of the clinic day. Staff recognized differing priorities of patients, staff, and physicians and articulated frustrations with hierarchy and competing priorities as well as behaviors that impeded clinic efficiency. Respondents emphasized the importance of feeling valued by others on their team.
Conclusions: With the evolving demands made on MAs’ time, it is critical to understand how the most effective staff members manage their role and highlight the strategies they employ to provide efficient clinical care. Understanding factors that increase or decrease MA job satisfaction can help identify high-efficiency practices and promote a clinic culture that values and supports all staff.
As primary care continues to evolve into more team-based practice, the role of the medical assistant (MA) has rapidly transformed.1 Staff may assist with patient management, documentation in the electronic medical record, order entry, pre-visit planning, and fulfillment of quality metrics, particularly in a Primary Care Medical Home (PCMH).2 From 2012 through 2014, MA job postings per graduate increased from 1.3 to 2.3, suggesting twice as many job postings as graduates.3 As the demand for experienced MAs increases, the ability to recruit and retain high-performing staff members will be critical.
MAs are referenced in medical literature as early as the 1800s.4 The American Association of Medical Assistants was founded in 1956, which led to educational standardization and certifications.5 Despite the important role that MAs have long played in the proper functioning of a medical clinic—and the knowledge that team configurations impact a clinic’s efficiency and quality6,7—few investigations have sought out the MA’s perspective.8,9 Given the increasing clinical demands placed on all members of the primary care team (and the burnout that often results), it seems that MA insights into clinic efficiency could be valuable.
METHODS
This cross-sectional study was conducted from February to April 2019 at a large academic institution with 6 regional ambulatory care family medicine clinics, each one with 11,000 to 18,000 patient visits annually. Faculty work at all 6 clinics and residents at 2 of them. All MAs are hired, paid, and managed by a central administrative department rather than by the family medicine department. The family medicine clinics are currently PCMH certified, with a mix of fee-for-service and capitated reimbursement.
Continue to: We developed and piloted...
We developed and piloted a voluntary, anonymous 39-question (29 closed-ended and 10 brief open-ended) online Qualtrics survey, which we distributed via an email link to all the MAs in the department. The survey included clinic site, years as an MA, perceptions of the clinic environment, perception of teamwork at their site, identification of efficient practices, and feedback for physicians to improve efficiency and flow. Most questions were Likert-style with 5 choices ranging from “strongly agree” to “strongly disagree” or short answer. Age and gender were omitted to protect confidentiality, as most MAs in the department are female. Participants could opt to enter in a drawing for three $25 gift cards. The survey was reviewed by the University of Michigan Institutional Review Board and deemed exempt.
We asked MAs to nominate peers in their clinic who were “especially efficient and do their jobs well—people that others can learn from.” The staff members who were nominated most frequently by their peers were invited to share additional perspectives via a 10- to 30-minute semi-structured interview with the first author. Interviews covered highly efficient practices, barriers and facilitators to efficient care, and physician behaviors that impaired efficiency. We interviewed a minimum of 2 MAs per clinic and increased the number of interviews through snowball sampling, as needed, to reach data saturation (eg, the point at which we were no longer hearing new content). MAs were assured that all comments would be anonymized. There was no monetary incentive for the interviews. The interviewer had previously met only 3 of the 18 MAs interviewed.
Analysis. Summary statistics were calculated for quantitative data. To compare subgroups (such as individual clinics), a chi-square test was used. In cases when there were small cell sizes (< 5 subjects), we used the Fisher’s Exact test. Qualitative data was collected with real-time typewritten notes during the interviews to capture ideas and verbatim quotes when possible. We also included open-ended comments shared on the Qualtrics survey. Data were organized by theme using a deductive coding approach. Both authors reviewed and discussed observations, and coding was conducted by the first author. Reporting followed the STROBE Statement checklist for cross-sectional studies.10 Results were shared with MAs, supervisory staff, and physicians, which allowed for feedback and comments and served as “member-checking.” MAs reported that the data reflected their lived experiences.
RESULTS
Surveys were distributed to all 86 MAs working in family medicine clinics. A total of 75 (87%) responded to at least some questions (typically just demographics). We used those who completed the full survey (n = 61; 71%) for data analysis. Eighteen MAs participated in face-to-face interviews. Among respondents, 35 (47%) had worked at least 10 years as an MA and 21 (28%) had worked at least a decade in the family medicine department.
Perception of role
All respondents (n = 61; 100%) somewhat or strongly agreed that the MA role was “very important to keep the clinic functioning” and 58 (95%) reported that working in health care was “a calling” for them. Only 7 (11%) agreed that family medicine was an easier environment for MAs compared to a specialty clinic; 30 (49%) disagreed with this. Among respondents, 32 (53%) strongly or somewhat agreed that their work was very stressful and just half (n = 28; 46%) agreed there were adequate MA staff at their clinic.
Continue to: Efficiency and competing priorities
Efficiency and competing priorities
MAs described important work values that increased their efficiency. These included clinic culture (good communication and strong teamwork), as well as individual strategies such as multitasking, limiting patient conversations, and doing tasks in a consistent way to improve accuracy. (See TABLE 1.) They identified ways physicians bolster or hurt efficiency and ways in which the relationship between the physician and the MA shapes the MA’s perception of their value in clinic.
Communication was emphasized as critical for efficient care, and MAs encouraged the use of preclinic huddles and communication as priorities. Seventy-five percent of MAs reported preclinic huddles to plan for patient care were helpful, but only half said huddles took place “always” or “most of the time.” Many described reviewing the schedule and completing tasks ahead of patient arrival as critical to efficiency.
Participants described the tension between their identified role of orchestrating clinic flow and responding to directives by others that disrupted the flow. Several MAs found it challenging when physicians agreed to see very late patients and felt frustrated when decisions that changed the flow were made by the physician or front desk staff without including the MA. MAs were also able to articulate how they managed competing priorities within the clinic, such as when a patient- or physician-driven need to extend appointments was at odds with maintaining a timely schedule. They were eager to share personal tips for time management and prided themselves on careful and accurate performance and skills they had learned on the job. MAs also described how efficiency could be adversely affected by the behaviors or attitudes of physicians. (See TABLE 2.)
Clinic environment
Thirty-six MAs (59%) reported that other MAs on their team were willing to help them out in clinic “a great deal” or “a lot” of the time, by helping to room a patient, acting as a chaperone for an exam, or doing a point-of-care lab. This sense of support varied across clinics (38% to 91% reported good support), suggesting that cultures vary by site. Some MAs expressed frustration at peers they saw as resistant to helping, exemplified by this verbatim quote from an interview:
“ Some don’t want to help out. They may sigh. It’s how they react—you just know.” (Clinic #1, MA #2 interview)
Efficient MAs stressed the need for situational awareness to recognize when co-workers need help:
“ [Peers often] are not aware that another MA is drowning. There’s 5 people who could have done that, and here I am running around and nobody budged.” (Clinic #5, MA #2 interview)
Continue to: A minority of staff...
A minority of staff used the open-ended survey sections to describe clinic hierarchy. When asked about “pet peeves,” a few advised that physicians should not “talk down” to staff and should try to teach rather than criticize. Another asked that physicians not “bark orders” or have “low gratitude” for staff work. MAs found micromanaging stressful—particularly when the physician prompted the MA about patient arrivals:
“[I don’t like] when providers will make a comment about a patient arriving when you already know this information. You then rush to put [the] patient in [a] room, then [the] provider ends up making [the] patient wait an extensive amount of time. I’m perfectly capable of knowing when a patient arrives.” (Clinic #6, survey)
MAs did not like physicians “talking bad about us” or blaming the MA if the clinic is running behind.
Despite these concerns, most MAs reported feeling appreciated for the job they do. Only 10 (16%) reported that the people they work with rarely say “thank you,” and 2 (3%) stated they were not well supported by the physicians in clinic. Most (n = 38; 62%) strongly agreed or agreed that they felt part of the team and that their opinions matter. In the interviews, many expanded on this idea:
“I really feel like I’m valued, so I want to do everything I can to make [my doctor’s] day go better. If you want a good clinic, the best thing a doc can do is make the MA feel valued.” (Clinic #1, MA #1 interview)
DISCUSSION
Participants described their role much as an orchestra director, with MAs as the key to clinic flow and timeliness.9 Respondents articulated multiple common strategies used to increase their own efficiency and clinic flow; these may be considered best practices and incorporated as part of the basic training. Most MAs reported their day-to-day jobs were stressful and believed this was underrecognized, so efficiency strategies are critical. With staff completing multiple time-sensitive tasks during clinic, consistent co-worker support is crucial and may impact efficiency.8 Proper training of managers to provide that support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial.
Several comments reflected the power differential within medical offices. One study reported that MAs and physicians “occupy roles at opposite ends of social and occupational hierarchies.”11 It’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.9 Prior research has found that MAs have higher perceptions of their own competence than do the physicians working with them.12 If there is a fundamental lack of trust between the 2 groups, this will undoubtedly hinder team-building. Attention to this issue is key to a more favorable work environment.
Continue to: Almost all respondents...
Almost all respondents reported health care was a “calling,” which mirrors physician research that suggests seeing work as a “calling” is protective against burnout.13,14 Open-ended comments indicated great pride in contributions, and most staff members felt appreciated by their teams. Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier. Staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients.15 We also uncovered some MA “pet peeves” that hinder efficiency and could be shared with physicians to emphasize the importance of patience and civility.
One barrier to expansion of MA roles within PCMH practices is the limited pay and career ladder for MAs who adopt new job responsibilities that require advanced skills or training.1,2 The mean MA salary at our institution ($37,372) is higher than in our state overall ($33,760), which may impact satisfaction.16 In addition, 93% of MAs are women; thus, they may continue to struggle more with lower pay than do workers in male-dominated professions.17,18 Expected job growth from 2018-2028 is predicted at 23%, which may help to boost salaries.19 Prior studies describe the lack of a job ladder or promotion opportunities as a challenge1,20; this was not formally assessed in our study.
MAs see work in family medicine as much harder than it is in other specialty clinics. Being trusted with more responsibility, greater autonomy,21-23 and expanded patient care roles can boost MA self-efficacy, which can reduce burnout for both physicians and MAs.8,24 However, new responsibilities should include appropriate training, support, and compensation, and match staff interests.7
Study limitations. The study was limited to 6 clinics in 1 department at a large academic medical center. Interviewed participants were selected by convenience and snowball sampling and thus, the results cannot be generalized to the population of MAs as a whole. As the initial interview goal was simply to gather efficiency tips, the project was not designed to be formal qualitative research. However, the discussions built on open-ended comments from the written survey helped contextualize our quantitative findings about efficiency. Notes were documented in real time by a single interviewer with rapid typing skills, which allowed capture of quotes verbatim. Subsequent studies would benefit from more formal qualitative research methods (recording and transcribing interviews, multiple coders to reduce risk of bias, and more complex thematic analysis).
Our research demonstrated how MAs perceive their roles in primary care and the facilitators and barriers to high efficiency in the workplace, which begins to fill an important knowledge gap in primary care. Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.
CORRESPONDENCE
Katherine J. Gold, MD, MSW, MS, Department of Family Medicine and Department of Obstetrics and Gynecology, University of Michigan, 1018 Fuller Street, Ann Arbor, MI 48104-1213; ktgold@umich.edu
ABSTRACT
Background: Medical assistant (MA) roles have expanded rapidly as primary care has evolved and MAs take on new patient care duties. Research that looks at the MA experience and factors that enhance or reduce efficiency among MAs is limited.
Methods: We surveyed all MAs working in 6 clinics run by a large academic family medicine department in Ann Arbor, Michigan. MAs deemed by peers as “most efficient” were selected for follow-up interviews. We evaluated personal strategies for efficiency, barriers to efficient care, impact of physician actions on efficiency, and satisfaction.
Results: A total of 75/86 MAs (87%) responded to at least some survey questions and 61/86 (71%) completed the full survey. We interviewed 18 MAs face to face. Most saw their role as essential to clinic functioning and viewed health care as a personal calling. MAs identified common strategies to improve efficiency and described the MA role to orchestrate the flow of the clinic day. Staff recognized differing priorities of patients, staff, and physicians and articulated frustrations with hierarchy and competing priorities as well as behaviors that impeded clinic efficiency. Respondents emphasized the importance of feeling valued by others on their team.
Conclusions: With the evolving demands made on MAs’ time, it is critical to understand how the most effective staff members manage their role and highlight the strategies they employ to provide efficient clinical care. Understanding factors that increase or decrease MA job satisfaction can help identify high-efficiency practices and promote a clinic culture that values and supports all staff.
As primary care continues to evolve into more team-based practice, the role of the medical assistant (MA) has rapidly transformed.1 Staff may assist with patient management, documentation in the electronic medical record, order entry, pre-visit planning, and fulfillment of quality metrics, particularly in a Primary Care Medical Home (PCMH).2 From 2012 through 2014, MA job postings per graduate increased from 1.3 to 2.3, suggesting twice as many job postings as graduates.3 As the demand for experienced MAs increases, the ability to recruit and retain high-performing staff members will be critical.
MAs are referenced in medical literature as early as the 1800s.4 The American Association of Medical Assistants was founded in 1956, which led to educational standardization and certifications.5 Despite the important role that MAs have long played in the proper functioning of a medical clinic—and the knowledge that team configurations impact a clinic’s efficiency and quality6,7—few investigations have sought out the MA’s perspective.8,9 Given the increasing clinical demands placed on all members of the primary care team (and the burnout that often results), it seems that MA insights into clinic efficiency could be valuable.
METHODS
This cross-sectional study was conducted from February to April 2019 at a large academic institution with 6 regional ambulatory care family medicine clinics, each one with 11,000 to 18,000 patient visits annually. Faculty work at all 6 clinics and residents at 2 of them. All MAs are hired, paid, and managed by a central administrative department rather than by the family medicine department. The family medicine clinics are currently PCMH certified, with a mix of fee-for-service and capitated reimbursement.
Continue to: We developed and piloted...
We developed and piloted a voluntary, anonymous 39-question (29 closed-ended and 10 brief open-ended) online Qualtrics survey, which we distributed via an email link to all the MAs in the department. The survey included clinic site, years as an MA, perceptions of the clinic environment, perception of teamwork at their site, identification of efficient practices, and feedback for physicians to improve efficiency and flow. Most questions were Likert-style with 5 choices ranging from “strongly agree” to “strongly disagree” or short answer. Age and gender were omitted to protect confidentiality, as most MAs in the department are female. Participants could opt to enter in a drawing for three $25 gift cards. The survey was reviewed by the University of Michigan Institutional Review Board and deemed exempt.
We asked MAs to nominate peers in their clinic who were “especially efficient and do their jobs well—people that others can learn from.” The staff members who were nominated most frequently by their peers were invited to share additional perspectives via a 10- to 30-minute semi-structured interview with the first author. Interviews covered highly efficient practices, barriers and facilitators to efficient care, and physician behaviors that impaired efficiency. We interviewed a minimum of 2 MAs per clinic and increased the number of interviews through snowball sampling, as needed, to reach data saturation (eg, the point at which we were no longer hearing new content). MAs were assured that all comments would be anonymized. There was no monetary incentive for the interviews. The interviewer had previously met only 3 of the 18 MAs interviewed.
Analysis. Summary statistics were calculated for quantitative data. To compare subgroups (such as individual clinics), a chi-square test was used. In cases when there were small cell sizes (< 5 subjects), we used the Fisher’s Exact test. Qualitative data was collected with real-time typewritten notes during the interviews to capture ideas and verbatim quotes when possible. We also included open-ended comments shared on the Qualtrics survey. Data were organized by theme using a deductive coding approach. Both authors reviewed and discussed observations, and coding was conducted by the first author. Reporting followed the STROBE Statement checklist for cross-sectional studies.10 Results were shared with MAs, supervisory staff, and physicians, which allowed for feedback and comments and served as “member-checking.” MAs reported that the data reflected their lived experiences.
RESULTS
Surveys were distributed to all 86 MAs working in family medicine clinics. A total of 75 (87%) responded to at least some questions (typically just demographics). We used those who completed the full survey (n = 61; 71%) for data analysis. Eighteen MAs participated in face-to-face interviews. Among respondents, 35 (47%) had worked at least 10 years as an MA and 21 (28%) had worked at least a decade in the family medicine department.
Perception of role
All respondents (n = 61; 100%) somewhat or strongly agreed that the MA role was “very important to keep the clinic functioning” and 58 (95%) reported that working in health care was “a calling” for them. Only 7 (11%) agreed that family medicine was an easier environment for MAs compared to a specialty clinic; 30 (49%) disagreed with this. Among respondents, 32 (53%) strongly or somewhat agreed that their work was very stressful and just half (n = 28; 46%) agreed there were adequate MA staff at their clinic.
Continue to: Efficiency and competing priorities
Efficiency and competing priorities
MAs described important work values that increased their efficiency. These included clinic culture (good communication and strong teamwork), as well as individual strategies such as multitasking, limiting patient conversations, and doing tasks in a consistent way to improve accuracy. (See TABLE 1.) They identified ways physicians bolster or hurt efficiency and ways in which the relationship between the physician and the MA shapes the MA’s perception of their value in clinic.
Communication was emphasized as critical for efficient care, and MAs encouraged the use of preclinic huddles and communication as priorities. Seventy-five percent of MAs reported preclinic huddles to plan for patient care were helpful, but only half said huddles took place “always” or “most of the time.” Many described reviewing the schedule and completing tasks ahead of patient arrival as critical to efficiency.
Participants described the tension between their identified role of orchestrating clinic flow and responding to directives by others that disrupted the flow. Several MAs found it challenging when physicians agreed to see very late patients and felt frustrated when decisions that changed the flow were made by the physician or front desk staff without including the MA. MAs were also able to articulate how they managed competing priorities within the clinic, such as when a patient- or physician-driven need to extend appointments was at odds with maintaining a timely schedule. They were eager to share personal tips for time management and prided themselves on careful and accurate performance and skills they had learned on the job. MAs also described how efficiency could be adversely affected by the behaviors or attitudes of physicians. (See TABLE 2.)
Clinic environment
Thirty-six MAs (59%) reported that other MAs on their team were willing to help them out in clinic “a great deal” or “a lot” of the time, by helping to room a patient, acting as a chaperone for an exam, or doing a point-of-care lab. This sense of support varied across clinics (38% to 91% reported good support), suggesting that cultures vary by site. Some MAs expressed frustration at peers they saw as resistant to helping, exemplified by this verbatim quote from an interview:
“ Some don’t want to help out. They may sigh. It’s how they react—you just know.” (Clinic #1, MA #2 interview)
Efficient MAs stressed the need for situational awareness to recognize when co-workers need help:
“ [Peers often] are not aware that another MA is drowning. There’s 5 people who could have done that, and here I am running around and nobody budged.” (Clinic #5, MA #2 interview)
Continue to: A minority of staff...
A minority of staff used the open-ended survey sections to describe clinic hierarchy. When asked about “pet peeves,” a few advised that physicians should not “talk down” to staff and should try to teach rather than criticize. Another asked that physicians not “bark orders” or have “low gratitude” for staff work. MAs found micromanaging stressful—particularly when the physician prompted the MA about patient arrivals:
“[I don’t like] when providers will make a comment about a patient arriving when you already know this information. You then rush to put [the] patient in [a] room, then [the] provider ends up making [the] patient wait an extensive amount of time. I’m perfectly capable of knowing when a patient arrives.” (Clinic #6, survey)
MAs did not like physicians “talking bad about us” or blaming the MA if the clinic is running behind.
Despite these concerns, most MAs reported feeling appreciated for the job they do. Only 10 (16%) reported that the people they work with rarely say “thank you,” and 2 (3%) stated they were not well supported by the physicians in clinic. Most (n = 38; 62%) strongly agreed or agreed that they felt part of the team and that their opinions matter. In the interviews, many expanded on this idea:
“I really feel like I’m valued, so I want to do everything I can to make [my doctor’s] day go better. If you want a good clinic, the best thing a doc can do is make the MA feel valued.” (Clinic #1, MA #1 interview)
DISCUSSION
Participants described their role much as an orchestra director, with MAs as the key to clinic flow and timeliness.9 Respondents articulated multiple common strategies used to increase their own efficiency and clinic flow; these may be considered best practices and incorporated as part of the basic training. Most MAs reported their day-to-day jobs were stressful and believed this was underrecognized, so efficiency strategies are critical. With staff completing multiple time-sensitive tasks during clinic, consistent co-worker support is crucial and may impact efficiency.8 Proper training of managers to provide that support and ensure equitable workloads may be one strategy to ensure that staff members feel the workplace is fair and collegial.
Several comments reflected the power differential within medical offices. One study reported that MAs and physicians “occupy roles at opposite ends of social and occupational hierarchies.”11 It’s important for physicians to be cognizant of these patterns and clinic culture, as reducing a hierarchy-based environment will be appreciated by MAs.9 Prior research has found that MAs have higher perceptions of their own competence than do the physicians working with them.12 If there is a fundamental lack of trust between the 2 groups, this will undoubtedly hinder team-building. Attention to this issue is key to a more favorable work environment.
Continue to: Almost all respondents...
Almost all respondents reported health care was a “calling,” which mirrors physician research that suggests seeing work as a “calling” is protective against burnout.13,14 Open-ended comments indicated great pride in contributions, and most staff members felt appreciated by their teams. Many described the working relationships with physicians as critical to their satisfaction at work and indicated that strong partnerships motivated them to do their best to make the physician’s day easier. Staff job satisfaction is linked to improved quality of care, so treating staff well contributes to high-value care for patients.15 We also uncovered some MA “pet peeves” that hinder efficiency and could be shared with physicians to emphasize the importance of patience and civility.
One barrier to expansion of MA roles within PCMH practices is the limited pay and career ladder for MAs who adopt new job responsibilities that require advanced skills or training.1,2 The mean MA salary at our institution ($37,372) is higher than in our state overall ($33,760), which may impact satisfaction.16 In addition, 93% of MAs are women; thus, they may continue to struggle more with lower pay than do workers in male-dominated professions.17,18 Expected job growth from 2018-2028 is predicted at 23%, which may help to boost salaries.19 Prior studies describe the lack of a job ladder or promotion opportunities as a challenge1,20; this was not formally assessed in our study.
MAs see work in family medicine as much harder than it is in other specialty clinics. Being trusted with more responsibility, greater autonomy,21-23 and expanded patient care roles can boost MA self-efficacy, which can reduce burnout for both physicians and MAs.8,24 However, new responsibilities should include appropriate training, support, and compensation, and match staff interests.7
Study limitations. The study was limited to 6 clinics in 1 department at a large academic medical center. Interviewed participants were selected by convenience and snowball sampling and thus, the results cannot be generalized to the population of MAs as a whole. As the initial interview goal was simply to gather efficiency tips, the project was not designed to be formal qualitative research. However, the discussions built on open-ended comments from the written survey helped contextualize our quantitative findings about efficiency. Notes were documented in real time by a single interviewer with rapid typing skills, which allowed capture of quotes verbatim. Subsequent studies would benefit from more formal qualitative research methods (recording and transcribing interviews, multiple coders to reduce risk of bias, and more complex thematic analysis).
Our research demonstrated how MAs perceive their roles in primary care and the facilitators and barriers to high efficiency in the workplace, which begins to fill an important knowledge gap in primary care. Disseminating practices that staff members themselves have identified as effective, and being attentive to how staff members are treated, may increase individual efficiency while improving staff retention and satisfaction.
CORRESPONDENCE
Katherine J. Gold, MD, MSW, MS, Department of Family Medicine and Department of Obstetrics and Gynecology, University of Michigan, 1018 Fuller Street, Ann Arbor, MI 48104-1213; ktgold@umich.edu
1. Chapman SA, Blash LK. New roles for medical assistants in innovative primary care practices. Health Serv Res. 2017;52(suppl 1):383-406.
2. Ferrante JM, Shaw EK, Bayly JE, et al. Barriers and facilitators to expanding roles of medical assistants in patient-centered medical homes (PCMHs). J Am Board Fam Med. 2018;31:226-235.
3. Atkins B. The outlook for medical assisting in 2016 and beyond. Accessed January 27, 2022. www.medicalassistantdegrees.net/articles/medical-assisting-trends/
4. Unqualified medical “assistants.” Hospital (Lond 1886). 1897;23:163-164.
5. Ameritech College of Healthcare. The origins of the AAMA. Accessed January 27, 2022. www.ameritech.edu/blog/medical-assisting-history/
6. Dai M, Willard-Grace R, Knox M, et al. Team configurations, efficiency, and family physician burnout. J Am Board Fam Med. 2020;33:368-377.
7. Harper PG, Van Riper K, Ramer T, et al. Team-based care: an expanded medical assistant role—enhanced rooming and visit assistance. J Interprof Care. 2018:1-7.
8. Sheridan B, Chien AT, Peters AS, et al. Team-based primary care: the medical assistant perspective. Health Care Manage Rev. 2018;43:115-125.
9. Tache S, Hill-Sakurai L. Medical assistants: the invisible “glue” of primary health care practices in the United States? J Health Organ Manag. 2010;24:288-305.
10. STROBE checklist for cohort, case-control, and cross-sectional studies. Accessed January 27, 2022. www.strobe-statement.org/fileadmin/Strobe/uploads/checklists/STROBE_checklist_v4_combined.pdf
11. Gray CP, Harrison MI, Hung D. Medical assistants as flow managers in primary care: challenges and recommendations. J Healthc Manag. 2016;61:181-191.
12. Elder NC, Jacobson CJ, Bolon SK, et al. Patterns of relating between physicians and medical assistants in small family medicine offices. Ann Fam Med. 2014;12:150-157.
13. Jager AJ, Tutty MA, Kao AC. Association between physician burnout and identification with medicine as a calling. Mayo Clinic Proc. 2017;92:415-422.
14. Yoon JD, Daley BM, Curlin FA. The association between a sense of calling and physician well-being: a national study of primary care physicians and psychiatrists. Acad Psychiatry. 2017;41:167-173.
15. Mohr DC, Young GJ, Meterko M, et al. Job satisfaction of primary care team members and quality of care. Am J Med Qual. 2011;26:18-25.
16. US Bureau of Labor Statistics. Occupational employment and wage statistics. Accessed January 27, 2022. https://www.bls.gov/oes/current/oes319092.htm
17. Chapman SA, Marks A, Dower C. Positioning medical assistants for a greater role in the era of health reform. Acad Med. 2015;90:1347-1352.
18. Mandel H. The role of occupational attributes in gender earnings inequality, 1970-2010. Soc Sci Res. 2016;55:122-138.
19. US Bureau of Labor Statistics. Occupational outlook handbook: medical assistants. Accessed January 27, 2022. www.bls.gov/ooh/healthcare/medical-assistants.htm
20. Skillman SM, Dahal A, Frogner BK, et al. Frontline workers’ career pathways: a detailed look at Washington state’s medical assistant workforce. Med Care Res Rev. 2018:1077558718812950.
21. Morse G, Salyers MP, Rollins AL, et al. Burnout in mental health services: a review of the problem and its remediation. Adm Policy Ment Health. 2012;39:341-352.
22. Dubois CA, Bentein K, Ben Mansour JB, et al. Why some employees adopt or resist reorganization of work practices in health care: associations between perceived loss of resources, burnout, and attitudes to change. Int J Environ Res Pub Health. 2014;11:187-201.
23. Aronsson G, Theorell T, Grape T, et al. A systematic review including meta-analysis of work environment and burnout symptoms. BMC Public Health. 2017;17:264.
24. O’Malley AS, Gourevitch R, Draper K, et al. Overcoming challenges to teamwork in patient-centered medical homes: a qualitative study. J Gen Intern Med. 2015;30:183-192.
1. Chapman SA, Blash LK. New roles for medical assistants in innovative primary care practices. Health Serv Res. 2017;52(suppl 1):383-406.
2. Ferrante JM, Shaw EK, Bayly JE, et al. Barriers and facilitators to expanding roles of medical assistants in patient-centered medical homes (PCMHs). J Am Board Fam Med. 2018;31:226-235.
3. Atkins B. The outlook for medical assisting in 2016 and beyond. Accessed January 27, 2022. www.medicalassistantdegrees.net/articles/medical-assisting-trends/
4. Unqualified medical “assistants.” Hospital (Lond 1886). 1897;23:163-164.
5. Ameritech College of Healthcare. The origins of the AAMA. Accessed January 27, 2022. www.ameritech.edu/blog/medical-assisting-history/
6. Dai M, Willard-Grace R, Knox M, et al. Team configurations, efficiency, and family physician burnout. J Am Board Fam Med. 2020;33:368-377.
7. Harper PG, Van Riper K, Ramer T, et al. Team-based care: an expanded medical assistant role—enhanced rooming and visit assistance. J Interprof Care. 2018:1-7.
8. Sheridan B, Chien AT, Peters AS, et al. Team-based primary care: the medical assistant perspective. Health Care Manage Rev. 2018;43:115-125.
9. Tache S, Hill-Sakurai L. Medical assistants: the invisible “glue” of primary health care practices in the United States? J Health Organ Manag. 2010;24:288-305.
10. STROBE checklist for cohort, case-control, and cross-sectional studies. Accessed January 27, 2022. www.strobe-statement.org/fileadmin/Strobe/uploads/checklists/STROBE_checklist_v4_combined.pdf
11. Gray CP, Harrison MI, Hung D. Medical assistants as flow managers in primary care: challenges and recommendations. J Healthc Manag. 2016;61:181-191.
12. Elder NC, Jacobson CJ, Bolon SK, et al. Patterns of relating between physicians and medical assistants in small family medicine offices. Ann Fam Med. 2014;12:150-157.
13. Jager AJ, Tutty MA, Kao AC. Association between physician burnout and identification with medicine as a calling. Mayo Clinic Proc. 2017;92:415-422.
14. Yoon JD, Daley BM, Curlin FA. The association between a sense of calling and physician well-being: a national study of primary care physicians and psychiatrists. Acad Psychiatry. 2017;41:167-173.
15. Mohr DC, Young GJ, Meterko M, et al. Job satisfaction of primary care team members and quality of care. Am J Med Qual. 2011;26:18-25.
16. US Bureau of Labor Statistics. Occupational employment and wage statistics. Accessed January 27, 2022. https://www.bls.gov/oes/current/oes319092.htm
17. Chapman SA, Marks A, Dower C. Positioning medical assistants for a greater role in the era of health reform. Acad Med. 2015;90:1347-1352.
18. Mandel H. The role of occupational attributes in gender earnings inequality, 1970-2010. Soc Sci Res. 2016;55:122-138.
19. US Bureau of Labor Statistics. Occupational outlook handbook: medical assistants. Accessed January 27, 2022. www.bls.gov/ooh/healthcare/medical-assistants.htm
20. Skillman SM, Dahal A, Frogner BK, et al. Frontline workers’ career pathways: a detailed look at Washington state’s medical assistant workforce. Med Care Res Rev. 2018:1077558718812950.
21. Morse G, Salyers MP, Rollins AL, et al. Burnout in mental health services: a review of the problem and its remediation. Adm Policy Ment Health. 2012;39:341-352.
22. Dubois CA, Bentein K, Ben Mansour JB, et al. Why some employees adopt or resist reorganization of work practices in health care: associations between perceived loss of resources, burnout, and attitudes to change. Int J Environ Res Pub Health. 2014;11:187-201.
23. Aronsson G, Theorell T, Grape T, et al. A systematic review including meta-analysis of work environment and burnout symptoms. BMC Public Health. 2017;17:264.
24. O’Malley AS, Gourevitch R, Draper K, et al. Overcoming challenges to teamwork in patient-centered medical homes: a qualitative study. J Gen Intern Med. 2015;30:183-192.
Diagnostic challenges in primary care: Identifying and avoiding cognitive bias
Medical errors in all settings contributed to as many as 250,000 deaths per year in the United States between 2000 and 2008, according to a 2016 study.1 Diagnostic error, in particular, remains a leading cause of morbidity and mortality in the United States and worldwide. In 2017, 12 million patients (roughly 5% of all US adults) who sought outpatient care experienced missed, delayed, or incorrect diagnosis at least once.2
In his classic work, How Doctors Think, Jerome Groopman, MD, explored the diagnostic process with a focus on the role of cognitive bias in clinical decision-making. Groopman examined how physicians can become sidetracked in their thinking and “blinded” to potential alternative diagnoses.3 Medical error is not necessarily because of a deficiency in medical knowledge; rather, physicians become susceptible to medical error when defective and faulty reasoning distort their diagnostic ability.4
Cognitive bias in the diagnostic process has been extensively studied, and a full review is beyond the scope of this article.5 However, here we will examine pathways leading to diagnostic errors in the primary care setting, specifically the role of cognitive bias in the work-up of polymyalgia rheumatica (PMR), ovarian cancer (OC), Lewy body dementia (LBD), and fibromyalgia (FM). As these 4 disease states are seen with low-to-moderate frequency in primary care, cognitive bias can complicate accurate diagnosis. But first, a word about how to understand clinical reasoning.
There are 2 types of reasoning (and 1 is more prone to error)
Physician clinical reasoning can be divided into 2 different cognitive approaches.
Type 1 reasoning employs intuition and heuristics; this type is automatic, reflexive, and quick.5 While the use of mental shortcuts in type 1 increases the speed with which decisions are made, it also makes this form of reasoning more prone to error.
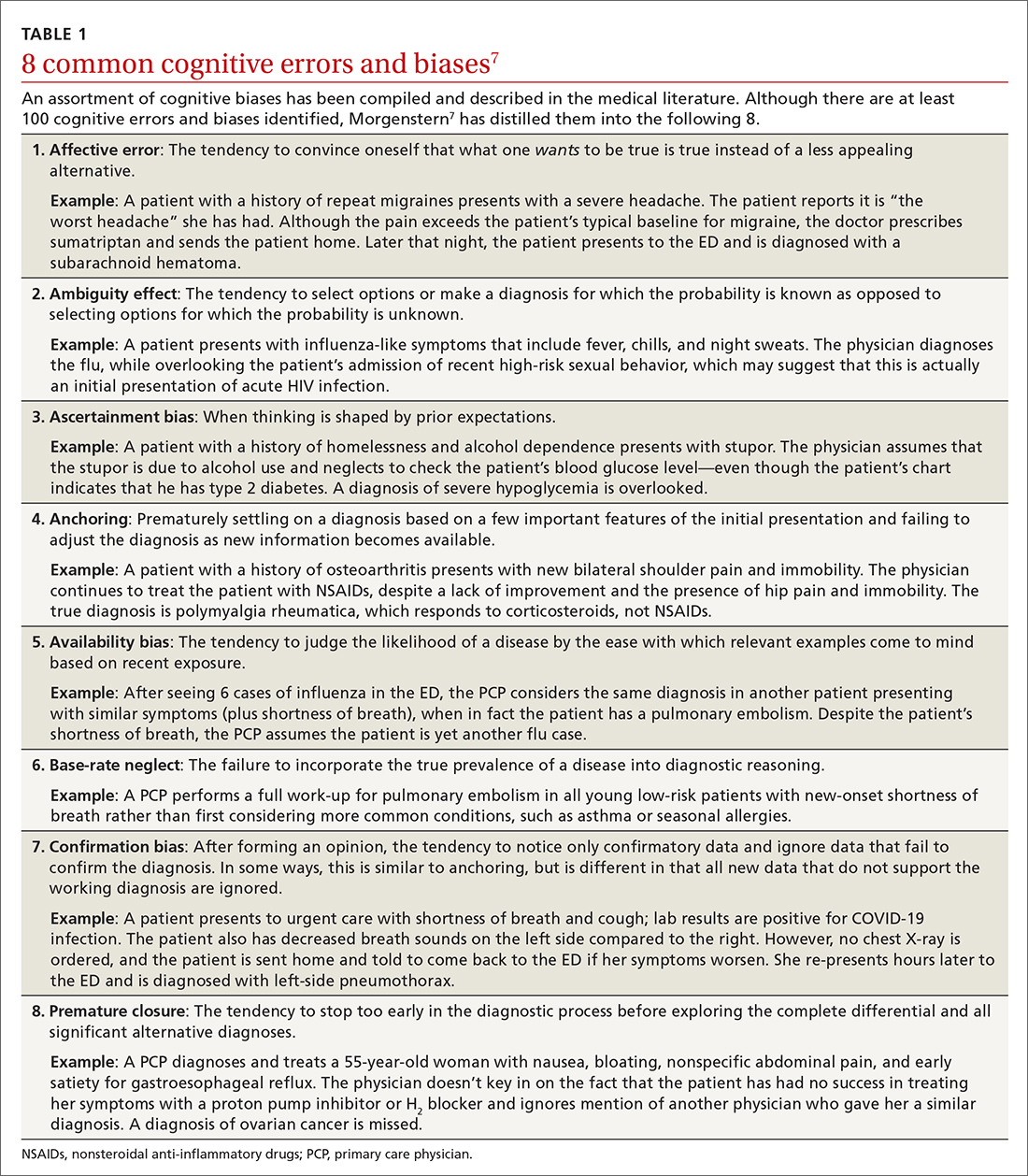
Type 2 reasoning requires conscious effort. It is goal directed and rigorous and therefore slower than type 1 reasoning. Extrapolated to the clinical context, clinicians transition from type 2 to type 1 reasoning as they gain experience and training throughout their careers and develop their own conscious and subconscious heuristics. Deviations from accurate decision-making occur in a systematic manner due to cognitive biases and result in medical error.6table 17 lists common types of cognitive bias.
An important question to ask. Physicians tend to fall into a pattern of quick, type 1 reasoning. However, it’s important to strive to maintain a broad differential diagnosis and avoid premature closure of the diagnostic process. It’s critical that we consider alternative diagnoses (ie, consciously move from type 1 to type 2 thinking) and continue to ask ourselves, “What else?” while working through differential diagnoses. This can be a powerful debiasing technique.
Continue to: The discussion...
The discussion of the following 4 disease states demonstrates how cognitive bias can lead to diagnostic error.
The patient is barely able to ambulate and appears to be in considerable pain. She is relying heavily on her walker and is assisted by her granddaughter. The primary care physician (PCP) obtains a detailed history that includes chronic shoulder and hip pain. Given that the patient has not responded to NSAID treatment over the previous 6 months, the PCP takes a moment to reconsider the diagnosis of OA and considers other options.
In light of the high prevalence of PMR in older women, the physician pursues a more specific physical examination tailored to ferret out PMR. He had learned this diagnostic shortcut as a resident, remembered it, and adeptly applied it whenever circumstances warranted. He asks the patient to raise her arms above her head (goalpost sign). She is unable to perform this task and experiences severe bilateral shoulder pain on trial. The PCP then places the patient on the examining table and attempts to assist her in rolling toward him. The patient is also unable to perform this maneuver and experiences significant bilateral hip pain on trial.
Based primarily on the patient’s history and physical exam findings, the PCP makes a presumptive diagnosis of PMR vs OA vs combined PMR with OA, orders an erythrocyte sedimentation rate (ESR) and basic rheumatologic
PMR can be mistaken for OA
PMR is the most common inflammatory rheumatic disease in older patients.8 It is a debilitating illness with simple, effective treatment but has devastating consequences if missed or left untreated.9 PMR typically manifests in patients older than age 50, with a peak incidence at 80 years of age. It is also far more common in women.10
Approximately 80% of patients with PMR initially present to their PCP, often posing a diagnostic challenge to many clinicians.11 Due to overlap in symptoms, the condition is often misdiagnosed as OA, a more common condition seen by PCPs. Also, there are no specific diagnostic tests for PMR. An elevated ESR can help confirm the diagnosis, but one-third of patients with PMR have a normal ESR.12 Therefore, the diagnostic conundrum the physician faces is OA vs rheumatoid arthritis (RA), PMR, or another condition.
Continue to: The consequences...
The consequences of a missed and delayed PMR diagnosis range from seriously impaired quality of life to significantly increased risk of vascular events (eg, blindness, stroke) due to temporal arteritis.13 Early diagnosis is even more critical as the risk of a vascular event and death is highest during initial phases of the disease course.14
FPs often miss this Dx. A timely diagnosis relies almost exclusively on an accurate, thorough history and physical exam. However, PCPs often struggle to correctly diagnose PMR. According to a study by Bahlas and colleagues,15 the accuracy rate for correctly diagnosing PMR was 24% among a cohort of family physicians.
The differential diagnosis for PMR is broad and includes seronegative spondyloarthropathies, malignancy, Lyme disease, hypothyroidism, and both RA and OA.16
PCPs are extremely adept at correctly diagnosing RA, but not PMR. A study by Blaauw and colleagues17 comparing PCPs and rheumatologists found PCPs correctly identified 92% of RA cases but only 55% of PMR cases. When rheumatologists reviewed these same cases, they correctly identified PMR and RA almost 100% of the time.17 The difference in diagnostic accuracy between rheumatologists and PCPs suggests limited experience and gaps in fund of knowledge.
Making the diagnosis. The diagnosis of PMR is often made on empiric response to corticosteroid treatment, but doing so based solely on a patient’s response is controversial.18 There are rare instances in which patients with PMR fail to respond to treatment. On the other hand, some inflammatory conditions that mimic or share symptoms with PMR also respond to corticosteroids, potentially resulting in erroneous confirmation bias.
Some classification criteria use rapid response to low-dose prednisone/prednisolone (≤ 20 mg) to confirm the diagnosis,19 while other more recent guidelines no longer include this approach.20 If PMR continues to be suspected after a trial of steroids is unsuccessful, the PCP can try another course of higher dose steroids or consult with Rheumatology.
Continue to: A full history...
A full history and physical exam revealed a myriad of gastrointestinal (GI) complaints, such as diarrhea. But the PCP recalled a recent roundtable discussion on debiasing techniques specifically related to gynecologic disorders, including OC. Therefore, he decided to include OC in the differential diagnosis—something he would not routinely have done given the preponderance of GI symptoms. Despite the patient’s reluctance and time constraints, the PCP ordered a transvaginal ultrasound. Findings from the ultrasound study revealed stage II OC, which carries a good prognosis. The patient is currently undergoing treatment and was last reported as doing well.
Early signs of ovarian cancer can be chalked up to a “GI issue”
OC is the second most common gynecologic cancer21 and the fifth leading cause of cancer-related death22 in US women. Compared to other cancers, the prognosis for localized early-stage OC is surprisingly good, with a 5-year survival rate approaching 93%.23 However, most disease is detected in later stages, and the 5-year survival rate drops to a low of 29%.24
There remains no established screening protocol for OC. Fewer than a quarter of all cases are diagnosed in stage I, and detection of OC relies heavily on the physician’s ability to decipher vague symptomatology that overlaps with other, more common maladies. This poses an obvious diagnostic challenge and, not surprisingly, a high level of susceptibility to cognitive bias.
More than 90% of patients with OC present with some combination of the following symptoms prior to diagnosis: abdominal (77%), GI (70%), pain (58%), constitutional (50%), urinary (34%), and pelvic (26%).25 The 3 most common isolated symptoms in patients with OC are abdominal bloating, decrease in appetite, and frank abdominal pain.26 Patients with biopsy-confirmed OC experience these symptoms an average of 6 months prior to actual diagnosis.27
Knowledge gaps play a role. Studies assessing the ability of health care providers to identify presenting symptoms of OC reveal specific knowledge gaps. For instance, in a survey by Gajjar and colleagues,28 most PCPs correctly identified bloating as a key symptom of OC; however, they weren’t as good at identifying less common symptoms, such as inability to finish a meal and early satiety. Moreover, survey participants misinterpreted or missed GI symptoms as an important manifestation of early OC disease.28 These specific knowledge gaps combine with physician errors in thinking, further obscuring and extending the diagnostic process. The point prevalence for OC is relatively low, and many PCPs only encounter a few cases during their entire career.29 This low pre-test probability may also fuel the delay in diagnosis.
Watch for these forms of bias. Since nonspecific symptoms of early-stage OC resemble those of other more benign conditions, a form of anchoring error known as multiple alternatives bias can arise. In this scenario, clinicians investigate only 1 potential plausible diagnosis and remain focused on that single, often faulty, conclusion. This persists despite other equally plausible alternatives that arise as the investigation proceeds.28
Affective error may also play a role in missed or delayed diagnosis. For example, a physician would prefer to diagnose and treat a common GI illness than consider OC. Another distortion involves outcome bias wherein the physician gives more significance to benign conditions such as irritable bowel syndrome because they have a more favorable outcome and clear treatment path. Physicians also favor these benign conditions because they encounter them more frequently than OC in the clinic setting. (This is known as availability bias.) Outcome bias and multiple alternatives bias can result in noninvestigation of symptoms and inefficient or improper management, leading to a delay in arriving at the correct diagnosis or anchoring on a plausible but incorrect diagnosis.
Continue to: An incorrect initial diagnostic...
An incorrect initial diagnostic path often triggers a cascade of subsequent errors. The physician orders additional unhelpful and expensive tests in an effort to characterize the suspected GI pathology. This then leads the physician to prematurely terminate the work-up and accept the most favored diagnosis. Lastly, sunk-cost fallacy comes into play: The physician has “invested” time and energy investigating a particular diagnosis and rather than abandon the presumed diagnosis, continues to put more time and effort in going down an incorrect diagnostic path.
A series of failures. These biases and miscues have been observed in several studies. For example, a survey of 1725 women by Goff and colleagues30 sought to identify factors related to delayed OC diagnosis. The authors found that the following factors were significantly associated with a delayed diagnosis: omission of a pelvic exam at initial presentation, a separate exploration of a multitude of collateral symptoms, a failure to order ultrasound/computed tomography/CA-125 test, and a failure to consider age as a factor (especially if the patient was outside the norm).
Responses from the survey also revealed that physicians initially ordered work-ups related to GI etiology and only later considered a pelvic work-up. This suggests that well-known presenting signs and symptoms or a constellation of typical and atypical symptoms of OC often failed to trigger physician recognition. Understandably, patients presenting with menorrhagia or gynecologic complaints are more likely to have OC detected at an earlier stage than patients who present with GI or abdominal signs alone.31 table 27 summarizes some of the cognitive biases seen in the diagnostic path of OC.
While in the hospital, he becomes acutely upset by the hallucinations and is given haloperidol and lorazepam by house staff. In the morning, the patient exhibits severe signs of Parkinson disease that include rigidity and masked facies.
Given the patient’s poor response to haloperidol and continued confusion, the team consulted Neurology and Psychiatry. Gathering a more detailed history from the patient and family, the patient is given a diagnosis of classic LBD. The antipsychotic medications are stopped. The patient and his family receive education about LBD treatment and management, and the patient is discharged to outpatient care.
Psychiatric symptoms can be an early “misdirect” in cases of Lewy body disease
LBD, the second leading neurodegenerative dementia after Alzheimer disease (AD), affects 1.5 million Americans,32 representing about 10% of all dementia cases. LBD and AD overlap in 25% of dementia cases.33 In patients older than 85 years, the prevalence jumps to 5% of the general population and 22% of all cases of dementia.33 Despite its prevalence, a recent study showed that only 6% of PCPs correctly identified LBD as the primary diagnosis when presented with typical case examples.32
Continue to: 3 stages of presentation
3 stages of presentation. Unlike other forms of dementia, LBD typically presents first with psychiatric symptoms, then with cognitive impairment, and last with parkinsonian symptoms. Additionally, rapid eye movement sleep behavior disorder and often subtle elements of nonmemory cognitive impairment distinguish LBD from both AD and vascular dementia.32 The primary cognitive deficit in LBD is not in memory but in attention, executive functioning, and visuospatial ability.34 Only in the later stages of the disease do patients exhibit gradual and progressive memory loss.
Mistaken for many things. When evaluating patients exhibiting signs of dementia, it’s important to include LBD in the differential, with increased suspicion for patients experiencing episodes of psychosis or delirium. The uniqueness of LBD lies in its psychotic symptomatology, particularly during earlier stages of the disease. This feature helps distinguish LBD from both AD and vascular dementia. As seen in the case, LBD can also be confused with acute delirium.
Older adult patients presenting to the ED or clinic with visual hallucinations, delirium, and mental confusion may receive a false diagnosis of schizophrenia, medication- or substance-induced psychosis, Parkinson disease, or delirium of unknown etiology.35 Unfortunately, LBD is often overlooked and not considered in the differential diagnosis. Due to underrecognition, patients may receive treatment with typical antipsychotics. The addition of a neuroleptic to help control the psychotic symptoms causes patients with LBD to develop severe extrapyramidal symptoms and worsening mental status,36 leading to severe parkinsonian signs, which further muddies the diagnostic process. In addition, treatment for suspected Parkinson disease, including carbidopa-levodopa, has no benefit for patients with LBD and may increase psychotic symptoms.37
First-line treatment for LBD includes psychoeducation for the patient and family, cholinesterase inhibitors (eg, rivastigmine), and avoidance of high-potency antipsychotics, such as haloperidol. Although persistent hallucinations and psychosis remain difficult to treat in LBD, low-dose quetiapine is 1 option. Incorrectly diagnosing and prescribing treatment for another condition exacerbates symptoms in this patient population.
The patient has been experiencing chronic pain for the past few years after a motor vehicle accident. She has seen a physiatrist and another provider, both of whom found no “objective” causes of her chronic pain. They started the patient on sertraline for depression and an analgesic, both of which were ineffective.
The patient likes to exercise at a gym twice a week by doing light cardio (treadmill) exercise and light weightlifting. Lately, however, she has been unable to exercise due to the pain. At this visit, she mentions having low energy, poor sleep, frequent fatigue, and generalized soreness and pain in multiple areas of her body. The PCP recognizes the patient’s presenting symptoms as significant for FM and starts her on pregabalin and hydrotherapy, with positive results.
Continue to: Fibromyalgia skepticism may lead to a Dx of depression
Fibromyalgia skepticism may lead to a Dx of depression
FM, the second most common disorder seen in rheumatologic practice after OA, is estimated to affect approximately 1 in 20 patients (approximately 5 million Americans) in the primary care setting.38,39 The condition has a high female-to-male preponderance (3.4% vs 0.5%).40 While the primary symptom of FM is chronic pain, patients commonly present with fatigue and sleep disturbance.41 Comorbid conditions include headaches, irritable bowel syndrome, and mood disturbances (most commonly anxiety and depression).
Several studies have explored reasons for the misdiagnosis and underdiagnosis of FM. One important factor is ongoing skepticism among some physicians and the public, in general, as to whether FM is a real disease. This issue was addressed by a study by White and colleagues,42 who estimated and compared the point prevalence of FM and related disorders in Amish vs non-Amish adults. The authors hypothesized that if litigation and/or compensation availability have a major impact on FM prevalence, then there would be a near zero prevalence of FM in the Amish community. And yet, researchers found an overall age- and sex-adjusted FM prevalence of 7.3% (95% CI; 5.3%-9.7%); this was both statistically greater than zero (P < .0001) and greater than 2 control populations of non-Amish adults (both P < .05).
Many physicians consider FM fundamentally an emotional disturbance, and the high preponderance of FM in female patients may contribute to this misconception as reports of pain and emotional distress by women are often dismissed as hysteria.43 Physicians often explore the emotional aspects of FM, incorrectly diagnosing patients with depression and subsequently treating them with a psychotropic drug.39 Alternatively, they may focus on the musculoskeletal presentations of FM and prescribe analgesics or physical therapy, both of which do little to alleviate FM.
To make the correct diagnosis of FM, the American College of Rheumatology created a specific set of criteria in 1990, which was updated in 2010.44 For a diagnosis of FM, a patient must have at least a 3-month history of bilateral pain above and below the waist and along the axial skeletal spine. Although not included in the updated 2010 criteria, many clinicians continue to check for tender points, following the 1990 criteria requiring the presence of 11 of 18 points to make the diagnosis.
At least 3 cognitive biases relating to FM apply: anchoring, availability, and fundamental attribution error (see table 3).7 Anchoring occurs when the PCP settles on a psychiatric diagnosis of exaggerated pain syndrome, muscle overuse, or OA and fails to explore alternative etiology. Availability bias may obscure the true diagnosis of FM. Since PCPs see many patients with RA or OA, they may overlook or dismiss the possibility of FM. Attribution error happens when physicians dismiss the complaints of patients with FM as merely due to psychological distress, hysteria, or acting out.43
Patients with FM, who are often otherwise healthy, often present multiple times to the same PCP with a chief complaint of chronic pain. These repeat presentations can result in compassion fatigue and impact care. As Aloush and colleagues40 noted in their study, “FM patients were perceived as more difficult than RA patients, with a high level of concern and emotional response. A high proportion of physicians were reluctant to accept them because they feel emotional/psychological difficulties meeting and coping with these patients.”In response, patients with undiagnosed FM or inadequately treated FM may visit other PCPs, which may or may not result in a correct diagnosis and treatment.
We can do better
Primary care physicians face the daunting task of diagnosing and treating a wide range of common conditions while also trying to recognize less-common conditions with atypical presentations—all during a busy clinic workday. Nonetheless, we should strive to overcome internal (eg, cognitive bias and fund-of-knowledge deficits) and external (eg, time constraints, limited resources) pressures to improve diagnostic accuracy and care.
Each of the 4 disease states we’ve discussed have high rates of missed and/or delayed diagnosis. Each presents a unique set of confounders: PMR with its overlapping symptoms of many other rheumatologic diseases; OC with its often vague and misleading GI symptomatology; LBD with overlapping features of AD and Parkinson disease; and FM with skepticism. As gatekeepers to health care, it falls on PCPs to sort out these diagnostic dilemmas to avoid medical errors. Fundamental knowledge of each disease, its unique pathophysiology and symptoms, and varying presentations can be learned, internalized, and subsequently put into clinical practice to improve patient outcomes.
CORRESPONDENCE
Paul D. Rosen MD, Brooklyn Hospital Center, Department of Family Medicine, 121 Dekalb Avenue, Brooklyn, New York 11201; drpaulie2000@hotmail.com
1. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139. doi: 10.1136/bmj.i2139
2. Van Such M, Lohr R, Beckman T, et al. Extent of diagnostic agreement among medical referrals. J Eval Clin Pract. 2017;23:870-874. doi: 10.1111/jep.12747
3. Groopman JE. How Doctors Think. Houghton Mifflin; 2007.
4. Tversky A, Kahneman D. Judgment under uncertainty: heuristics and biases. Science. 1974;185:1124-1131. doi: 10.1126/science.185.4157.1124
5. Norman GR, Monteiro SD, Sherbino J, et al. The causes of errors in clinical reasoning: Cognitive biases, knowledge deficits, and dual process thinking. Acad Med. 2017;92:23-30. doi: 10.1097/ACM.0000000000001421
6. Croskerry P. The importance of cognitive errors in diagnosis and strategies to minimize them. Acad Med. 2003;78:775-780. doi: 10.1097/00001888-200308000-00003
7. Morgenstern J. Cognitive errors in medicine: The common errors. First10EM blog. September 15, 2015. Updated September 22, 2019. Accessed February 8, 2022. https://first10em.com/cognitive-errors/
8. Gazitt T, Zisman D, Gardner G. Polymyalgia rheumatica: a common disease in seniors. Curr Rheumatol Rep. 2020;22:40. doi: 10.1007/s11926-020-00919-2
9. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35. doi: 10.1002/art.23176
10. Doran MF, Crowson CS, O’Fallon WM, et al. Trends in the incidence of polymyalgia rheumatica over a 30 year period in Olmsted County, Minnesota, USA. J Rheumatol. 2002;29:1694-1697.
11. Barraclough K, Liddell WG, du Toit J, et al. Polymyalgia rheumatica in primary care: a cohort study of the diagnostic criteria and outcome. Fam Pract. 2008;25:328-33. doi: 10.1093/fampra/cmn044
12. Manzo C. Polymyalgia rheumatica (PMR) with normal values of both erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) concentration at the time of diagnosis in a centenarian man: a case report. Diseases. 2018;6:84. doi: 10.3390/diseases6040084
13. Crowson CS, Matteson EL. Contemporary prevalence estimates for giant cell arteritis and polymyalgia rheumatica, 2015. Semin Arthritis Rheum. 2017;47:253-256. doi: 10.1016/j.semarthrit.2017.04.001
14. Nordborg E, Bengtsson BA. Death rates and causes of death in 284 consecutive patients with giant cell arteritis confirmed by biopsy. BMJ. 1989;299:549-550. doi: 10.1136/bmj.299.6698.549
15. Bahlas S, Ramos-Remus C, Davis P. Utilisation and costs of investigations, and accuracy of diagnosis of polymyalgia rheumatica by family physicians. Clin Rheumatol. 2000;19:278-280. doi: 10.1007/s100670070045
16. Brooks RC, McGee SR. Diagnostic dilemmas in polymyalgia rheumatica. Arch Intern Med. 1997;157:162-168.
17. Blaauw AA, Schuwirth LW, van der Vleuten CP, et al. Assessing clinical competence: recognition of case descriptions of rheumatic diseases by general practitioners. Br J Rheumatol. 1995;34:375-379. doi:10.1093/rheumatology/34.4.375
18. Mager DR. Polymylagia rheumatica: common disease, elusive diagnosis. Home Healthc Now. 2015;33:132-138. doi:10.1097/NHH.0000000000000199
19. Kermani TA, Warrington KJ. Polymyalgia rheumatica. Lancet. 381;63-72. doi: 10.1016/S0140-6736(12)60680-1. Published correction appears in Lancet. 20135;381:28.
20. Liew DF, Owen CE, Buchanan RR. Prescribing for polymyalgia rheumatica. Aust Prescr. 2018;41:14-19. doi: 10.18773/austprescr.2018.001
21. Ovarian cancer statistics. Centers for Disease Control and Prevention. Reviewed June 8, 2021. Accessed February 22, 2022. www.cdc.gov/cancer/ovarian/statistics/index.htm
22. Key statistics for ovarian cancer. American Cancer Society. Revised January 12, 2022. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/about/key-statistics.html
23. Survival rates for ovarian cancer. American Cancer Society. Revised January 25, 2021. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/detection-diagnosis-staging/survival-rates.html
24. Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med. 2017;14:9-32. doi: 10.20892/j.issn.2095-3941.2016.0084
25. Goff BA, Mandel LS, Melancon CH, et al. Frequency of symptoms of ovarian cancer in women presenting to primary care clinics. JAMA. 2004;291:2705-2712. doi: 10.1001/jama.291.22.2705
26. Olson SH, Mignone L, Nakraseive C, et al. Symptoms of ovarian cancer. Obstet Gynecol. 2001;98:212-217. doi: 10.1016/s0029-7844(01)01457-0
27. Allgar VL, Neal RD. Delays in the diagnosis of six cancers: analysis of data from the National Survey of NHS patients: Cancer. Br J Cancer. 2005;92:1959-1970. doi: 10.1038/sj.bjc.6602587
28. Gajjar K, Ogden G, Mujahid MI, et al. Symptoms and risk factors of ovarian cancer: a survey in primary care. Obstet Gynecol. 2012:754197. doi: 10.5402/2012/754197
29. Austoker J. Diagnosis of ovarian cancer in primary care. BMJ. 2009;339:b3286. doi: 10.1136/bmj.b3286
30. Goff BA, Mandel L, Muntz HG, et al. Ovarian carcinoma diagnosis: results of a national ovarian cancer survey. Cancer. 2000;89:2068-2075. doi: 10.1002/1097-0142(20001115)89:10<2068::aid-cncr6>3.0.co;2-z
31. Smith EM, Anderson B. The effects of symptoms and delay in seeking diagnosis on stage of disease at diagnosis among women with cancers of the ovary. Cancer. 1985;56:2727-2732. doi: 10.1002/1097-0142(19851201)56:11<2727::aid-cncr2820561138>3.0.co;2-8
32. Galvin JE, Duda JE, Kaufer DI, et al. Lewy body dementia: the caregiver experience of clinical care. Parkinsonism Relat Disord. 2010;16:388-392. doi: 10.1016/j.parkreldis.2010.03.007
33. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6:333-341. doi: 10.31887/DCNS.2004.6.3/imckeith
34. Mrak RE, Griffin WS. Dementia with Lewy bodies: definitions, diagnosis, and pathogenic relationship to Alzheimer’s disease. Neuropsychiatr Dis Treat. 2007;3:619-625.
35. Khotianov N, Singh R, Singh S. Lewy body dementia: case report and discussion. J Am Board Fam Pract. 2002;15:50-54.
36. Stinton C, McKeith I, Taylor JP, et al. Pharmacological management of Lewy body dementia: a systematic review and meta-analysis. Am J Psychiatry. 2015;172:731-742. doi: 10.1176/appi.ajp.2015.14121582
37. Velayudhan L, Ffytche D, Ballard C, et al. New therapeutic strategies of Lewy body dementias. Curr Neurol Neurosci Rep. 2017;17:68. doi: 10.1007/s11910-017-0778-2
38. Arnold LM, Clauw DJ, McCarberg BH; FibroCollaborative. Improving the recognition and diagnosis of fibromyalgia. Mayo Clin Proc. 2011;86:457-464. doi: 10.4065/mcp.2010.0738
39. Arnold LM, Gebke KB, Choy EHS. Fibromyalgia: management strategies for primary care providers. Int J Clin Pract. 2016;70:99-112. doi: 10.1111/ijcp.12757
40. Aloush V, Niv D, Ablin JN, et al. Good pain, bad pain: illness perception and physician attitudes towards rheumatoid arthritis and fibromyalgia patients. Clin Exp Rheumatol. 2021;39(suppl 130):54-60.
41. Vincent A, Lahr BD, Wolfe F, et al. Prevalence of fibromyalgia: a population-based study in Olmsted County, Minnesota, utilizing the Rochester Epidemiology Project. Arthritis Care Res (Hoboken). 2013;65:786-792. doi: 10.1002/acr.21896
42. White KP, Thompson J. Fibromyalgia syndrome in an Amish community: a controlled study to determine disease and symptom prevalence. J Rheumatol. 2003;30:1835-1840.
43. Lobo CP, Pfalzgraf AR, Giannetti V, et al. Impact of invalidation and trust in physicians on health outcomes in fibromyalgia patients. Prim Care Companion CNS Disord. 2014;16:10.4088/PCC.14m01664. doi: 10.4088/PCC.14m01664
44. Wolfe F, Clauw DJ, Fitzcharles MA, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken). 2010;62:600-610. doi:10.1002/acr.20140
Medical errors in all settings contributed to as many as 250,000 deaths per year in the United States between 2000 and 2008, according to a 2016 study.1 Diagnostic error, in particular, remains a leading cause of morbidity and mortality in the United States and worldwide. In 2017, 12 million patients (roughly 5% of all US adults) who sought outpatient care experienced missed, delayed, or incorrect diagnosis at least once.2
In his classic work, How Doctors Think, Jerome Groopman, MD, explored the diagnostic process with a focus on the role of cognitive bias in clinical decision-making. Groopman examined how physicians can become sidetracked in their thinking and “blinded” to potential alternative diagnoses.3 Medical error is not necessarily because of a deficiency in medical knowledge; rather, physicians become susceptible to medical error when defective and faulty reasoning distort their diagnostic ability.4
Cognitive bias in the diagnostic process has been extensively studied, and a full review is beyond the scope of this article.5 However, here we will examine pathways leading to diagnostic errors in the primary care setting, specifically the role of cognitive bias in the work-up of polymyalgia rheumatica (PMR), ovarian cancer (OC), Lewy body dementia (LBD), and fibromyalgia (FM). As these 4 disease states are seen with low-to-moderate frequency in primary care, cognitive bias can complicate accurate diagnosis. But first, a word about how to understand clinical reasoning.
There are 2 types of reasoning (and 1 is more prone to error)
Physician clinical reasoning can be divided into 2 different cognitive approaches.
Type 1 reasoning employs intuition and heuristics; this type is automatic, reflexive, and quick.5 While the use of mental shortcuts in type 1 increases the speed with which decisions are made, it also makes this form of reasoning more prone to error.
Type 2 reasoning requires conscious effort. It is goal directed and rigorous and therefore slower than type 1 reasoning. Extrapolated to the clinical context, clinicians transition from type 2 to type 1 reasoning as they gain experience and training throughout their careers and develop their own conscious and subconscious heuristics. Deviations from accurate decision-making occur in a systematic manner due to cognitive biases and result in medical error.6table 17 lists common types of cognitive bias.
An important question to ask. Physicians tend to fall into a pattern of quick, type 1 reasoning. However, it’s important to strive to maintain a broad differential diagnosis and avoid premature closure of the diagnostic process. It’s critical that we consider alternative diagnoses (ie, consciously move from type 1 to type 2 thinking) and continue to ask ourselves, “What else?” while working through differential diagnoses. This can be a powerful debiasing technique.
Continue to: The discussion...
The discussion of the following 4 disease states demonstrates how cognitive bias can lead to diagnostic error.
The patient is barely able to ambulate and appears to be in considerable pain. She is relying heavily on her walker and is assisted by her granddaughter. The primary care physician (PCP) obtains a detailed history that includes chronic shoulder and hip pain. Given that the patient has not responded to NSAID treatment over the previous 6 months, the PCP takes a moment to reconsider the diagnosis of OA and considers other options.
In light of the high prevalence of PMR in older women, the physician pursues a more specific physical examination tailored to ferret out PMR. He had learned this diagnostic shortcut as a resident, remembered it, and adeptly applied it whenever circumstances warranted. He asks the patient to raise her arms above her head (goalpost sign). She is unable to perform this task and experiences severe bilateral shoulder pain on trial. The PCP then places the patient on the examining table and attempts to assist her in rolling toward him. The patient is also unable to perform this maneuver and experiences significant bilateral hip pain on trial.
Based primarily on the patient’s history and physical exam findings, the PCP makes a presumptive diagnosis of PMR vs OA vs combined PMR with OA, orders an erythrocyte sedimentation rate (ESR) and basic rheumatologic
PMR can be mistaken for OA
PMR is the most common inflammatory rheumatic disease in older patients.8 It is a debilitating illness with simple, effective treatment but has devastating consequences if missed or left untreated.9 PMR typically manifests in patients older than age 50, with a peak incidence at 80 years of age. It is also far more common in women.10
Approximately 80% of patients with PMR initially present to their PCP, often posing a diagnostic challenge to many clinicians.11 Due to overlap in symptoms, the condition is often misdiagnosed as OA, a more common condition seen by PCPs. Also, there are no specific diagnostic tests for PMR. An elevated ESR can help confirm the diagnosis, but one-third of patients with PMR have a normal ESR.12 Therefore, the diagnostic conundrum the physician faces is OA vs rheumatoid arthritis (RA), PMR, or another condition.
Continue to: The consequences...
The consequences of a missed and delayed PMR diagnosis range from seriously impaired quality of life to significantly increased risk of vascular events (eg, blindness, stroke) due to temporal arteritis.13 Early diagnosis is even more critical as the risk of a vascular event and death is highest during initial phases of the disease course.14
FPs often miss this Dx. A timely diagnosis relies almost exclusively on an accurate, thorough history and physical exam. However, PCPs often struggle to correctly diagnose PMR. According to a study by Bahlas and colleagues,15 the accuracy rate for correctly diagnosing PMR was 24% among a cohort of family physicians.
The differential diagnosis for PMR is broad and includes seronegative spondyloarthropathies, malignancy, Lyme disease, hypothyroidism, and both RA and OA.16
PCPs are extremely adept at correctly diagnosing RA, but not PMR. A study by Blaauw and colleagues17 comparing PCPs and rheumatologists found PCPs correctly identified 92% of RA cases but only 55% of PMR cases. When rheumatologists reviewed these same cases, they correctly identified PMR and RA almost 100% of the time.17 The difference in diagnostic accuracy between rheumatologists and PCPs suggests limited experience and gaps in fund of knowledge.
Making the diagnosis. The diagnosis of PMR is often made on empiric response to corticosteroid treatment, but doing so based solely on a patient’s response is controversial.18 There are rare instances in which patients with PMR fail to respond to treatment. On the other hand, some inflammatory conditions that mimic or share symptoms with PMR also respond to corticosteroids, potentially resulting in erroneous confirmation bias.
Some classification criteria use rapid response to low-dose prednisone/prednisolone (≤ 20 mg) to confirm the diagnosis,19 while other more recent guidelines no longer include this approach.20 If PMR continues to be suspected after a trial of steroids is unsuccessful, the PCP can try another course of higher dose steroids or consult with Rheumatology.
Continue to: A full history...
A full history and physical exam revealed a myriad of gastrointestinal (GI) complaints, such as diarrhea. But the PCP recalled a recent roundtable discussion on debiasing techniques specifically related to gynecologic disorders, including OC. Therefore, he decided to include OC in the differential diagnosis—something he would not routinely have done given the preponderance of GI symptoms. Despite the patient’s reluctance and time constraints, the PCP ordered a transvaginal ultrasound. Findings from the ultrasound study revealed stage II OC, which carries a good prognosis. The patient is currently undergoing treatment and was last reported as doing well.
Early signs of ovarian cancer can be chalked up to a “GI issue”
OC is the second most common gynecologic cancer21 and the fifth leading cause of cancer-related death22 in US women. Compared to other cancers, the prognosis for localized early-stage OC is surprisingly good, with a 5-year survival rate approaching 93%.23 However, most disease is detected in later stages, and the 5-year survival rate drops to a low of 29%.24
There remains no established screening protocol for OC. Fewer than a quarter of all cases are diagnosed in stage I, and detection of OC relies heavily on the physician’s ability to decipher vague symptomatology that overlaps with other, more common maladies. This poses an obvious diagnostic challenge and, not surprisingly, a high level of susceptibility to cognitive bias.
More than 90% of patients with OC present with some combination of the following symptoms prior to diagnosis: abdominal (77%), GI (70%), pain (58%), constitutional (50%), urinary (34%), and pelvic (26%).25 The 3 most common isolated symptoms in patients with OC are abdominal bloating, decrease in appetite, and frank abdominal pain.26 Patients with biopsy-confirmed OC experience these symptoms an average of 6 months prior to actual diagnosis.27
Knowledge gaps play a role. Studies assessing the ability of health care providers to identify presenting symptoms of OC reveal specific knowledge gaps. For instance, in a survey by Gajjar and colleagues,28 most PCPs correctly identified bloating as a key symptom of OC; however, they weren’t as good at identifying less common symptoms, such as inability to finish a meal and early satiety. Moreover, survey participants misinterpreted or missed GI symptoms as an important manifestation of early OC disease.28 These specific knowledge gaps combine with physician errors in thinking, further obscuring and extending the diagnostic process. The point prevalence for OC is relatively low, and many PCPs only encounter a few cases during their entire career.29 This low pre-test probability may also fuel the delay in diagnosis.
Watch for these forms of bias. Since nonspecific symptoms of early-stage OC resemble those of other more benign conditions, a form of anchoring error known as multiple alternatives bias can arise. In this scenario, clinicians investigate only 1 potential plausible diagnosis and remain focused on that single, often faulty, conclusion. This persists despite other equally plausible alternatives that arise as the investigation proceeds.28
Affective error may also play a role in missed or delayed diagnosis. For example, a physician would prefer to diagnose and treat a common GI illness than consider OC. Another distortion involves outcome bias wherein the physician gives more significance to benign conditions such as irritable bowel syndrome because they have a more favorable outcome and clear treatment path. Physicians also favor these benign conditions because they encounter them more frequently than OC in the clinic setting. (This is known as availability bias.) Outcome bias and multiple alternatives bias can result in noninvestigation of symptoms and inefficient or improper management, leading to a delay in arriving at the correct diagnosis or anchoring on a plausible but incorrect diagnosis.
Continue to: An incorrect initial diagnostic...
An incorrect initial diagnostic path often triggers a cascade of subsequent errors. The physician orders additional unhelpful and expensive tests in an effort to characterize the suspected GI pathology. This then leads the physician to prematurely terminate the work-up and accept the most favored diagnosis. Lastly, sunk-cost fallacy comes into play: The physician has “invested” time and energy investigating a particular diagnosis and rather than abandon the presumed diagnosis, continues to put more time and effort in going down an incorrect diagnostic path.
A series of failures. These biases and miscues have been observed in several studies. For example, a survey of 1725 women by Goff and colleagues30 sought to identify factors related to delayed OC diagnosis. The authors found that the following factors were significantly associated with a delayed diagnosis: omission of a pelvic exam at initial presentation, a separate exploration of a multitude of collateral symptoms, a failure to order ultrasound/computed tomography/CA-125 test, and a failure to consider age as a factor (especially if the patient was outside the norm).
Responses from the survey also revealed that physicians initially ordered work-ups related to GI etiology and only later considered a pelvic work-up. This suggests that well-known presenting signs and symptoms or a constellation of typical and atypical symptoms of OC often failed to trigger physician recognition. Understandably, patients presenting with menorrhagia or gynecologic complaints are more likely to have OC detected at an earlier stage than patients who present with GI or abdominal signs alone.31 table 27 summarizes some of the cognitive biases seen in the diagnostic path of OC.
While in the hospital, he becomes acutely upset by the hallucinations and is given haloperidol and lorazepam by house staff. In the morning, the patient exhibits severe signs of Parkinson disease that include rigidity and masked facies.
Given the patient’s poor response to haloperidol and continued confusion, the team consulted Neurology and Psychiatry. Gathering a more detailed history from the patient and family, the patient is given a diagnosis of classic LBD. The antipsychotic medications are stopped. The patient and his family receive education about LBD treatment and management, and the patient is discharged to outpatient care.
Psychiatric symptoms can be an early “misdirect” in cases of Lewy body disease
LBD, the second leading neurodegenerative dementia after Alzheimer disease (AD), affects 1.5 million Americans,32 representing about 10% of all dementia cases. LBD and AD overlap in 25% of dementia cases.33 In patients older than 85 years, the prevalence jumps to 5% of the general population and 22% of all cases of dementia.33 Despite its prevalence, a recent study showed that only 6% of PCPs correctly identified LBD as the primary diagnosis when presented with typical case examples.32
Continue to: 3 stages of presentation
3 stages of presentation. Unlike other forms of dementia, LBD typically presents first with psychiatric symptoms, then with cognitive impairment, and last with parkinsonian symptoms. Additionally, rapid eye movement sleep behavior disorder and often subtle elements of nonmemory cognitive impairment distinguish LBD from both AD and vascular dementia.32 The primary cognitive deficit in LBD is not in memory but in attention, executive functioning, and visuospatial ability.34 Only in the later stages of the disease do patients exhibit gradual and progressive memory loss.
Mistaken for many things. When evaluating patients exhibiting signs of dementia, it’s important to include LBD in the differential, with increased suspicion for patients experiencing episodes of psychosis or delirium. The uniqueness of LBD lies in its psychotic symptomatology, particularly during earlier stages of the disease. This feature helps distinguish LBD from both AD and vascular dementia. As seen in the case, LBD can also be confused with acute delirium.
Older adult patients presenting to the ED or clinic with visual hallucinations, delirium, and mental confusion may receive a false diagnosis of schizophrenia, medication- or substance-induced psychosis, Parkinson disease, or delirium of unknown etiology.35 Unfortunately, LBD is often overlooked and not considered in the differential diagnosis. Due to underrecognition, patients may receive treatment with typical antipsychotics. The addition of a neuroleptic to help control the psychotic symptoms causes patients with LBD to develop severe extrapyramidal symptoms and worsening mental status,36 leading to severe parkinsonian signs, which further muddies the diagnostic process. In addition, treatment for suspected Parkinson disease, including carbidopa-levodopa, has no benefit for patients with LBD and may increase psychotic symptoms.37
First-line treatment for LBD includes psychoeducation for the patient and family, cholinesterase inhibitors (eg, rivastigmine), and avoidance of high-potency antipsychotics, such as haloperidol. Although persistent hallucinations and psychosis remain difficult to treat in LBD, low-dose quetiapine is 1 option. Incorrectly diagnosing and prescribing treatment for another condition exacerbates symptoms in this patient population.
The patient has been experiencing chronic pain for the past few years after a motor vehicle accident. She has seen a physiatrist and another provider, both of whom found no “objective” causes of her chronic pain. They started the patient on sertraline for depression and an analgesic, both of which were ineffective.
The patient likes to exercise at a gym twice a week by doing light cardio (treadmill) exercise and light weightlifting. Lately, however, she has been unable to exercise due to the pain. At this visit, she mentions having low energy, poor sleep, frequent fatigue, and generalized soreness and pain in multiple areas of her body. The PCP recognizes the patient’s presenting symptoms as significant for FM and starts her on pregabalin and hydrotherapy, with positive results.
Continue to: Fibromyalgia skepticism may lead to a Dx of depression
Fibromyalgia skepticism may lead to a Dx of depression
FM, the second most common disorder seen in rheumatologic practice after OA, is estimated to affect approximately 1 in 20 patients (approximately 5 million Americans) in the primary care setting.38,39 The condition has a high female-to-male preponderance (3.4% vs 0.5%).40 While the primary symptom of FM is chronic pain, patients commonly present with fatigue and sleep disturbance.41 Comorbid conditions include headaches, irritable bowel syndrome, and mood disturbances (most commonly anxiety and depression).
Several studies have explored reasons for the misdiagnosis and underdiagnosis of FM. One important factor is ongoing skepticism among some physicians and the public, in general, as to whether FM is a real disease. This issue was addressed by a study by White and colleagues,42 who estimated and compared the point prevalence of FM and related disorders in Amish vs non-Amish adults. The authors hypothesized that if litigation and/or compensation availability have a major impact on FM prevalence, then there would be a near zero prevalence of FM in the Amish community. And yet, researchers found an overall age- and sex-adjusted FM prevalence of 7.3% (95% CI; 5.3%-9.7%); this was both statistically greater than zero (P < .0001) and greater than 2 control populations of non-Amish adults (both P < .05).
Many physicians consider FM fundamentally an emotional disturbance, and the high preponderance of FM in female patients may contribute to this misconception as reports of pain and emotional distress by women are often dismissed as hysteria.43 Physicians often explore the emotional aspects of FM, incorrectly diagnosing patients with depression and subsequently treating them with a psychotropic drug.39 Alternatively, they may focus on the musculoskeletal presentations of FM and prescribe analgesics or physical therapy, both of which do little to alleviate FM.
To make the correct diagnosis of FM, the American College of Rheumatology created a specific set of criteria in 1990, which was updated in 2010.44 For a diagnosis of FM, a patient must have at least a 3-month history of bilateral pain above and below the waist and along the axial skeletal spine. Although not included in the updated 2010 criteria, many clinicians continue to check for tender points, following the 1990 criteria requiring the presence of 11 of 18 points to make the diagnosis.
At least 3 cognitive biases relating to FM apply: anchoring, availability, and fundamental attribution error (see table 3).7 Anchoring occurs when the PCP settles on a psychiatric diagnosis of exaggerated pain syndrome, muscle overuse, or OA and fails to explore alternative etiology. Availability bias may obscure the true diagnosis of FM. Since PCPs see many patients with RA or OA, they may overlook or dismiss the possibility of FM. Attribution error happens when physicians dismiss the complaints of patients with FM as merely due to psychological distress, hysteria, or acting out.43
Patients with FM, who are often otherwise healthy, often present multiple times to the same PCP with a chief complaint of chronic pain. These repeat presentations can result in compassion fatigue and impact care. As Aloush and colleagues40 noted in their study, “FM patients were perceived as more difficult than RA patients, with a high level of concern and emotional response. A high proportion of physicians were reluctant to accept them because they feel emotional/psychological difficulties meeting and coping with these patients.”In response, patients with undiagnosed FM or inadequately treated FM may visit other PCPs, which may or may not result in a correct diagnosis and treatment.
We can do better
Primary care physicians face the daunting task of diagnosing and treating a wide range of common conditions while also trying to recognize less-common conditions with atypical presentations—all during a busy clinic workday. Nonetheless, we should strive to overcome internal (eg, cognitive bias and fund-of-knowledge deficits) and external (eg, time constraints, limited resources) pressures to improve diagnostic accuracy and care.
Each of the 4 disease states we’ve discussed have high rates of missed and/or delayed diagnosis. Each presents a unique set of confounders: PMR with its overlapping symptoms of many other rheumatologic diseases; OC with its often vague and misleading GI symptomatology; LBD with overlapping features of AD and Parkinson disease; and FM with skepticism. As gatekeepers to health care, it falls on PCPs to sort out these diagnostic dilemmas to avoid medical errors. Fundamental knowledge of each disease, its unique pathophysiology and symptoms, and varying presentations can be learned, internalized, and subsequently put into clinical practice to improve patient outcomes.
CORRESPONDENCE
Paul D. Rosen MD, Brooklyn Hospital Center, Department of Family Medicine, 121 Dekalb Avenue, Brooklyn, New York 11201; drpaulie2000@hotmail.com
Medical errors in all settings contributed to as many as 250,000 deaths per year in the United States between 2000 and 2008, according to a 2016 study.1 Diagnostic error, in particular, remains a leading cause of morbidity and mortality in the United States and worldwide. In 2017, 12 million patients (roughly 5% of all US adults) who sought outpatient care experienced missed, delayed, or incorrect diagnosis at least once.2
In his classic work, How Doctors Think, Jerome Groopman, MD, explored the diagnostic process with a focus on the role of cognitive bias in clinical decision-making. Groopman examined how physicians can become sidetracked in their thinking and “blinded” to potential alternative diagnoses.3 Medical error is not necessarily because of a deficiency in medical knowledge; rather, physicians become susceptible to medical error when defective and faulty reasoning distort their diagnostic ability.4
Cognitive bias in the diagnostic process has been extensively studied, and a full review is beyond the scope of this article.5 However, here we will examine pathways leading to diagnostic errors in the primary care setting, specifically the role of cognitive bias in the work-up of polymyalgia rheumatica (PMR), ovarian cancer (OC), Lewy body dementia (LBD), and fibromyalgia (FM). As these 4 disease states are seen with low-to-moderate frequency in primary care, cognitive bias can complicate accurate diagnosis. But first, a word about how to understand clinical reasoning.
There are 2 types of reasoning (and 1 is more prone to error)
Physician clinical reasoning can be divided into 2 different cognitive approaches.
Type 1 reasoning employs intuition and heuristics; this type is automatic, reflexive, and quick.5 While the use of mental shortcuts in type 1 increases the speed with which decisions are made, it also makes this form of reasoning more prone to error.
Type 2 reasoning requires conscious effort. It is goal directed and rigorous and therefore slower than type 1 reasoning. Extrapolated to the clinical context, clinicians transition from type 2 to type 1 reasoning as they gain experience and training throughout their careers and develop their own conscious and subconscious heuristics. Deviations from accurate decision-making occur in a systematic manner due to cognitive biases and result in medical error.6table 17 lists common types of cognitive bias.
An important question to ask. Physicians tend to fall into a pattern of quick, type 1 reasoning. However, it’s important to strive to maintain a broad differential diagnosis and avoid premature closure of the diagnostic process. It’s critical that we consider alternative diagnoses (ie, consciously move from type 1 to type 2 thinking) and continue to ask ourselves, “What else?” while working through differential diagnoses. This can be a powerful debiasing technique.
Continue to: The discussion...
The discussion of the following 4 disease states demonstrates how cognitive bias can lead to diagnostic error.
The patient is barely able to ambulate and appears to be in considerable pain. She is relying heavily on her walker and is assisted by her granddaughter. The primary care physician (PCP) obtains a detailed history that includes chronic shoulder and hip pain. Given that the patient has not responded to NSAID treatment over the previous 6 months, the PCP takes a moment to reconsider the diagnosis of OA and considers other options.
In light of the high prevalence of PMR in older women, the physician pursues a more specific physical examination tailored to ferret out PMR. He had learned this diagnostic shortcut as a resident, remembered it, and adeptly applied it whenever circumstances warranted. He asks the patient to raise her arms above her head (goalpost sign). She is unable to perform this task and experiences severe bilateral shoulder pain on trial. The PCP then places the patient on the examining table and attempts to assist her in rolling toward him. The patient is also unable to perform this maneuver and experiences significant bilateral hip pain on trial.
Based primarily on the patient’s history and physical exam findings, the PCP makes a presumptive diagnosis of PMR vs OA vs combined PMR with OA, orders an erythrocyte sedimentation rate (ESR) and basic rheumatologic
PMR can be mistaken for OA
PMR is the most common inflammatory rheumatic disease in older patients.8 It is a debilitating illness with simple, effective treatment but has devastating consequences if missed or left untreated.9 PMR typically manifests in patients older than age 50, with a peak incidence at 80 years of age. It is also far more common in women.10
Approximately 80% of patients with PMR initially present to their PCP, often posing a diagnostic challenge to many clinicians.11 Due to overlap in symptoms, the condition is often misdiagnosed as OA, a more common condition seen by PCPs. Also, there are no specific diagnostic tests for PMR. An elevated ESR can help confirm the diagnosis, but one-third of patients with PMR have a normal ESR.12 Therefore, the diagnostic conundrum the physician faces is OA vs rheumatoid arthritis (RA), PMR, or another condition.
Continue to: The consequences...
The consequences of a missed and delayed PMR diagnosis range from seriously impaired quality of life to significantly increased risk of vascular events (eg, blindness, stroke) due to temporal arteritis.13 Early diagnosis is even more critical as the risk of a vascular event and death is highest during initial phases of the disease course.14
FPs often miss this Dx. A timely diagnosis relies almost exclusively on an accurate, thorough history and physical exam. However, PCPs often struggle to correctly diagnose PMR. According to a study by Bahlas and colleagues,15 the accuracy rate for correctly diagnosing PMR was 24% among a cohort of family physicians.
The differential diagnosis for PMR is broad and includes seronegative spondyloarthropathies, malignancy, Lyme disease, hypothyroidism, and both RA and OA.16
PCPs are extremely adept at correctly diagnosing RA, but not PMR. A study by Blaauw and colleagues17 comparing PCPs and rheumatologists found PCPs correctly identified 92% of RA cases but only 55% of PMR cases. When rheumatologists reviewed these same cases, they correctly identified PMR and RA almost 100% of the time.17 The difference in diagnostic accuracy between rheumatologists and PCPs suggests limited experience and gaps in fund of knowledge.
Making the diagnosis. The diagnosis of PMR is often made on empiric response to corticosteroid treatment, but doing so based solely on a patient’s response is controversial.18 There are rare instances in which patients with PMR fail to respond to treatment. On the other hand, some inflammatory conditions that mimic or share symptoms with PMR also respond to corticosteroids, potentially resulting in erroneous confirmation bias.
Some classification criteria use rapid response to low-dose prednisone/prednisolone (≤ 20 mg) to confirm the diagnosis,19 while other more recent guidelines no longer include this approach.20 If PMR continues to be suspected after a trial of steroids is unsuccessful, the PCP can try another course of higher dose steroids or consult with Rheumatology.
Continue to: A full history...
A full history and physical exam revealed a myriad of gastrointestinal (GI) complaints, such as diarrhea. But the PCP recalled a recent roundtable discussion on debiasing techniques specifically related to gynecologic disorders, including OC. Therefore, he decided to include OC in the differential diagnosis—something he would not routinely have done given the preponderance of GI symptoms. Despite the patient’s reluctance and time constraints, the PCP ordered a transvaginal ultrasound. Findings from the ultrasound study revealed stage II OC, which carries a good prognosis. The patient is currently undergoing treatment and was last reported as doing well.
Early signs of ovarian cancer can be chalked up to a “GI issue”
OC is the second most common gynecologic cancer21 and the fifth leading cause of cancer-related death22 in US women. Compared to other cancers, the prognosis for localized early-stage OC is surprisingly good, with a 5-year survival rate approaching 93%.23 However, most disease is detected in later stages, and the 5-year survival rate drops to a low of 29%.24
There remains no established screening protocol for OC. Fewer than a quarter of all cases are diagnosed in stage I, and detection of OC relies heavily on the physician’s ability to decipher vague symptomatology that overlaps with other, more common maladies. This poses an obvious diagnostic challenge and, not surprisingly, a high level of susceptibility to cognitive bias.
More than 90% of patients with OC present with some combination of the following symptoms prior to diagnosis: abdominal (77%), GI (70%), pain (58%), constitutional (50%), urinary (34%), and pelvic (26%).25 The 3 most common isolated symptoms in patients with OC are abdominal bloating, decrease in appetite, and frank abdominal pain.26 Patients with biopsy-confirmed OC experience these symptoms an average of 6 months prior to actual diagnosis.27
Knowledge gaps play a role. Studies assessing the ability of health care providers to identify presenting symptoms of OC reveal specific knowledge gaps. For instance, in a survey by Gajjar and colleagues,28 most PCPs correctly identified bloating as a key symptom of OC; however, they weren’t as good at identifying less common symptoms, such as inability to finish a meal and early satiety. Moreover, survey participants misinterpreted or missed GI symptoms as an important manifestation of early OC disease.28 These specific knowledge gaps combine with physician errors in thinking, further obscuring and extending the diagnostic process. The point prevalence for OC is relatively low, and many PCPs only encounter a few cases during their entire career.29 This low pre-test probability may also fuel the delay in diagnosis.
Watch for these forms of bias. Since nonspecific symptoms of early-stage OC resemble those of other more benign conditions, a form of anchoring error known as multiple alternatives bias can arise. In this scenario, clinicians investigate only 1 potential plausible diagnosis and remain focused on that single, often faulty, conclusion. This persists despite other equally plausible alternatives that arise as the investigation proceeds.28
Affective error may also play a role in missed or delayed diagnosis. For example, a physician would prefer to diagnose and treat a common GI illness than consider OC. Another distortion involves outcome bias wherein the physician gives more significance to benign conditions such as irritable bowel syndrome because they have a more favorable outcome and clear treatment path. Physicians also favor these benign conditions because they encounter them more frequently than OC in the clinic setting. (This is known as availability bias.) Outcome bias and multiple alternatives bias can result in noninvestigation of symptoms and inefficient or improper management, leading to a delay in arriving at the correct diagnosis or anchoring on a plausible but incorrect diagnosis.
Continue to: An incorrect initial diagnostic...
An incorrect initial diagnostic path often triggers a cascade of subsequent errors. The physician orders additional unhelpful and expensive tests in an effort to characterize the suspected GI pathology. This then leads the physician to prematurely terminate the work-up and accept the most favored diagnosis. Lastly, sunk-cost fallacy comes into play: The physician has “invested” time and energy investigating a particular diagnosis and rather than abandon the presumed diagnosis, continues to put more time and effort in going down an incorrect diagnostic path.
A series of failures. These biases and miscues have been observed in several studies. For example, a survey of 1725 women by Goff and colleagues30 sought to identify factors related to delayed OC diagnosis. The authors found that the following factors were significantly associated with a delayed diagnosis: omission of a pelvic exam at initial presentation, a separate exploration of a multitude of collateral symptoms, a failure to order ultrasound/computed tomography/CA-125 test, and a failure to consider age as a factor (especially if the patient was outside the norm).
Responses from the survey also revealed that physicians initially ordered work-ups related to GI etiology and only later considered a pelvic work-up. This suggests that well-known presenting signs and symptoms or a constellation of typical and atypical symptoms of OC often failed to trigger physician recognition. Understandably, patients presenting with menorrhagia or gynecologic complaints are more likely to have OC detected at an earlier stage than patients who present with GI or abdominal signs alone.31 table 27 summarizes some of the cognitive biases seen in the diagnostic path of OC.
While in the hospital, he becomes acutely upset by the hallucinations and is given haloperidol and lorazepam by house staff. In the morning, the patient exhibits severe signs of Parkinson disease that include rigidity and masked facies.
Given the patient’s poor response to haloperidol and continued confusion, the team consulted Neurology and Psychiatry. Gathering a more detailed history from the patient and family, the patient is given a diagnosis of classic LBD. The antipsychotic medications are stopped. The patient and his family receive education about LBD treatment and management, and the patient is discharged to outpatient care.
Psychiatric symptoms can be an early “misdirect” in cases of Lewy body disease
LBD, the second leading neurodegenerative dementia after Alzheimer disease (AD), affects 1.5 million Americans,32 representing about 10% of all dementia cases. LBD and AD overlap in 25% of dementia cases.33 In patients older than 85 years, the prevalence jumps to 5% of the general population and 22% of all cases of dementia.33 Despite its prevalence, a recent study showed that only 6% of PCPs correctly identified LBD as the primary diagnosis when presented with typical case examples.32
Continue to: 3 stages of presentation
3 stages of presentation. Unlike other forms of dementia, LBD typically presents first with psychiatric symptoms, then with cognitive impairment, and last with parkinsonian symptoms. Additionally, rapid eye movement sleep behavior disorder and often subtle elements of nonmemory cognitive impairment distinguish LBD from both AD and vascular dementia.32 The primary cognitive deficit in LBD is not in memory but in attention, executive functioning, and visuospatial ability.34 Only in the later stages of the disease do patients exhibit gradual and progressive memory loss.
Mistaken for many things. When evaluating patients exhibiting signs of dementia, it’s important to include LBD in the differential, with increased suspicion for patients experiencing episodes of psychosis or delirium. The uniqueness of LBD lies in its psychotic symptomatology, particularly during earlier stages of the disease. This feature helps distinguish LBD from both AD and vascular dementia. As seen in the case, LBD can also be confused with acute delirium.
Older adult patients presenting to the ED or clinic with visual hallucinations, delirium, and mental confusion may receive a false diagnosis of schizophrenia, medication- or substance-induced psychosis, Parkinson disease, or delirium of unknown etiology.35 Unfortunately, LBD is often overlooked and not considered in the differential diagnosis. Due to underrecognition, patients may receive treatment with typical antipsychotics. The addition of a neuroleptic to help control the psychotic symptoms causes patients with LBD to develop severe extrapyramidal symptoms and worsening mental status,36 leading to severe parkinsonian signs, which further muddies the diagnostic process. In addition, treatment for suspected Parkinson disease, including carbidopa-levodopa, has no benefit for patients with LBD and may increase psychotic symptoms.37
First-line treatment for LBD includes psychoeducation for the patient and family, cholinesterase inhibitors (eg, rivastigmine), and avoidance of high-potency antipsychotics, such as haloperidol. Although persistent hallucinations and psychosis remain difficult to treat in LBD, low-dose quetiapine is 1 option. Incorrectly diagnosing and prescribing treatment for another condition exacerbates symptoms in this patient population.
The patient has been experiencing chronic pain for the past few years after a motor vehicle accident. She has seen a physiatrist and another provider, both of whom found no “objective” causes of her chronic pain. They started the patient on sertraline for depression and an analgesic, both of which were ineffective.
The patient likes to exercise at a gym twice a week by doing light cardio (treadmill) exercise and light weightlifting. Lately, however, she has been unable to exercise due to the pain. At this visit, she mentions having low energy, poor sleep, frequent fatigue, and generalized soreness and pain in multiple areas of her body. The PCP recognizes the patient’s presenting symptoms as significant for FM and starts her on pregabalin and hydrotherapy, with positive results.
Continue to: Fibromyalgia skepticism may lead to a Dx of depression
Fibromyalgia skepticism may lead to a Dx of depression
FM, the second most common disorder seen in rheumatologic practice after OA, is estimated to affect approximately 1 in 20 patients (approximately 5 million Americans) in the primary care setting.38,39 The condition has a high female-to-male preponderance (3.4% vs 0.5%).40 While the primary symptom of FM is chronic pain, patients commonly present with fatigue and sleep disturbance.41 Comorbid conditions include headaches, irritable bowel syndrome, and mood disturbances (most commonly anxiety and depression).
Several studies have explored reasons for the misdiagnosis and underdiagnosis of FM. One important factor is ongoing skepticism among some physicians and the public, in general, as to whether FM is a real disease. This issue was addressed by a study by White and colleagues,42 who estimated and compared the point prevalence of FM and related disorders in Amish vs non-Amish adults. The authors hypothesized that if litigation and/or compensation availability have a major impact on FM prevalence, then there would be a near zero prevalence of FM in the Amish community. And yet, researchers found an overall age- and sex-adjusted FM prevalence of 7.3% (95% CI; 5.3%-9.7%); this was both statistically greater than zero (P < .0001) and greater than 2 control populations of non-Amish adults (both P < .05).
Many physicians consider FM fundamentally an emotional disturbance, and the high preponderance of FM in female patients may contribute to this misconception as reports of pain and emotional distress by women are often dismissed as hysteria.43 Physicians often explore the emotional aspects of FM, incorrectly diagnosing patients with depression and subsequently treating them with a psychotropic drug.39 Alternatively, they may focus on the musculoskeletal presentations of FM and prescribe analgesics or physical therapy, both of which do little to alleviate FM.
To make the correct diagnosis of FM, the American College of Rheumatology created a specific set of criteria in 1990, which was updated in 2010.44 For a diagnosis of FM, a patient must have at least a 3-month history of bilateral pain above and below the waist and along the axial skeletal spine. Although not included in the updated 2010 criteria, many clinicians continue to check for tender points, following the 1990 criteria requiring the presence of 11 of 18 points to make the diagnosis.
At least 3 cognitive biases relating to FM apply: anchoring, availability, and fundamental attribution error (see table 3).7 Anchoring occurs when the PCP settles on a psychiatric diagnosis of exaggerated pain syndrome, muscle overuse, or OA and fails to explore alternative etiology. Availability bias may obscure the true diagnosis of FM. Since PCPs see many patients with RA or OA, they may overlook or dismiss the possibility of FM. Attribution error happens when physicians dismiss the complaints of patients with FM as merely due to psychological distress, hysteria, or acting out.43
Patients with FM, who are often otherwise healthy, often present multiple times to the same PCP with a chief complaint of chronic pain. These repeat presentations can result in compassion fatigue and impact care. As Aloush and colleagues40 noted in their study, “FM patients were perceived as more difficult than RA patients, with a high level of concern and emotional response. A high proportion of physicians were reluctant to accept them because they feel emotional/psychological difficulties meeting and coping with these patients.”In response, patients with undiagnosed FM or inadequately treated FM may visit other PCPs, which may or may not result in a correct diagnosis and treatment.
We can do better
Primary care physicians face the daunting task of diagnosing and treating a wide range of common conditions while also trying to recognize less-common conditions with atypical presentations—all during a busy clinic workday. Nonetheless, we should strive to overcome internal (eg, cognitive bias and fund-of-knowledge deficits) and external (eg, time constraints, limited resources) pressures to improve diagnostic accuracy and care.
Each of the 4 disease states we’ve discussed have high rates of missed and/or delayed diagnosis. Each presents a unique set of confounders: PMR with its overlapping symptoms of many other rheumatologic diseases; OC with its often vague and misleading GI symptomatology; LBD with overlapping features of AD and Parkinson disease; and FM with skepticism. As gatekeepers to health care, it falls on PCPs to sort out these diagnostic dilemmas to avoid medical errors. Fundamental knowledge of each disease, its unique pathophysiology and symptoms, and varying presentations can be learned, internalized, and subsequently put into clinical practice to improve patient outcomes.
CORRESPONDENCE
Paul D. Rosen MD, Brooklyn Hospital Center, Department of Family Medicine, 121 Dekalb Avenue, Brooklyn, New York 11201; drpaulie2000@hotmail.com
1. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139. doi: 10.1136/bmj.i2139
2. Van Such M, Lohr R, Beckman T, et al. Extent of diagnostic agreement among medical referrals. J Eval Clin Pract. 2017;23:870-874. doi: 10.1111/jep.12747
3. Groopman JE. How Doctors Think. Houghton Mifflin; 2007.
4. Tversky A, Kahneman D. Judgment under uncertainty: heuristics and biases. Science. 1974;185:1124-1131. doi: 10.1126/science.185.4157.1124
5. Norman GR, Monteiro SD, Sherbino J, et al. The causes of errors in clinical reasoning: Cognitive biases, knowledge deficits, and dual process thinking. Acad Med. 2017;92:23-30. doi: 10.1097/ACM.0000000000001421
6. Croskerry P. The importance of cognitive errors in diagnosis and strategies to minimize them. Acad Med. 2003;78:775-780. doi: 10.1097/00001888-200308000-00003
7. Morgenstern J. Cognitive errors in medicine: The common errors. First10EM blog. September 15, 2015. Updated September 22, 2019. Accessed February 8, 2022. https://first10em.com/cognitive-errors/
8. Gazitt T, Zisman D, Gardner G. Polymyalgia rheumatica: a common disease in seniors. Curr Rheumatol Rep. 2020;22:40. doi: 10.1007/s11926-020-00919-2
9. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35. doi: 10.1002/art.23176
10. Doran MF, Crowson CS, O’Fallon WM, et al. Trends in the incidence of polymyalgia rheumatica over a 30 year period in Olmsted County, Minnesota, USA. J Rheumatol. 2002;29:1694-1697.
11. Barraclough K, Liddell WG, du Toit J, et al. Polymyalgia rheumatica in primary care: a cohort study of the diagnostic criteria and outcome. Fam Pract. 2008;25:328-33. doi: 10.1093/fampra/cmn044
12. Manzo C. Polymyalgia rheumatica (PMR) with normal values of both erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) concentration at the time of diagnosis in a centenarian man: a case report. Diseases. 2018;6:84. doi: 10.3390/diseases6040084
13. Crowson CS, Matteson EL. Contemporary prevalence estimates for giant cell arteritis and polymyalgia rheumatica, 2015. Semin Arthritis Rheum. 2017;47:253-256. doi: 10.1016/j.semarthrit.2017.04.001
14. Nordborg E, Bengtsson BA. Death rates and causes of death in 284 consecutive patients with giant cell arteritis confirmed by biopsy. BMJ. 1989;299:549-550. doi: 10.1136/bmj.299.6698.549
15. Bahlas S, Ramos-Remus C, Davis P. Utilisation and costs of investigations, and accuracy of diagnosis of polymyalgia rheumatica by family physicians. Clin Rheumatol. 2000;19:278-280. doi: 10.1007/s100670070045
16. Brooks RC, McGee SR. Diagnostic dilemmas in polymyalgia rheumatica. Arch Intern Med. 1997;157:162-168.
17. Blaauw AA, Schuwirth LW, van der Vleuten CP, et al. Assessing clinical competence: recognition of case descriptions of rheumatic diseases by general practitioners. Br J Rheumatol. 1995;34:375-379. doi:10.1093/rheumatology/34.4.375
18. Mager DR. Polymylagia rheumatica: common disease, elusive diagnosis. Home Healthc Now. 2015;33:132-138. doi:10.1097/NHH.0000000000000199
19. Kermani TA, Warrington KJ. Polymyalgia rheumatica. Lancet. 381;63-72. doi: 10.1016/S0140-6736(12)60680-1. Published correction appears in Lancet. 20135;381:28.
20. Liew DF, Owen CE, Buchanan RR. Prescribing for polymyalgia rheumatica. Aust Prescr. 2018;41:14-19. doi: 10.18773/austprescr.2018.001
21. Ovarian cancer statistics. Centers for Disease Control and Prevention. Reviewed June 8, 2021. Accessed February 22, 2022. www.cdc.gov/cancer/ovarian/statistics/index.htm
22. Key statistics for ovarian cancer. American Cancer Society. Revised January 12, 2022. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/about/key-statistics.html
23. Survival rates for ovarian cancer. American Cancer Society. Revised January 25, 2021. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/detection-diagnosis-staging/survival-rates.html
24. Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med. 2017;14:9-32. doi: 10.20892/j.issn.2095-3941.2016.0084
25. Goff BA, Mandel LS, Melancon CH, et al. Frequency of symptoms of ovarian cancer in women presenting to primary care clinics. JAMA. 2004;291:2705-2712. doi: 10.1001/jama.291.22.2705
26. Olson SH, Mignone L, Nakraseive C, et al. Symptoms of ovarian cancer. Obstet Gynecol. 2001;98:212-217. doi: 10.1016/s0029-7844(01)01457-0
27. Allgar VL, Neal RD. Delays in the diagnosis of six cancers: analysis of data from the National Survey of NHS patients: Cancer. Br J Cancer. 2005;92:1959-1970. doi: 10.1038/sj.bjc.6602587
28. Gajjar K, Ogden G, Mujahid MI, et al. Symptoms and risk factors of ovarian cancer: a survey in primary care. Obstet Gynecol. 2012:754197. doi: 10.5402/2012/754197
29. Austoker J. Diagnosis of ovarian cancer in primary care. BMJ. 2009;339:b3286. doi: 10.1136/bmj.b3286
30. Goff BA, Mandel L, Muntz HG, et al. Ovarian carcinoma diagnosis: results of a national ovarian cancer survey. Cancer. 2000;89:2068-2075. doi: 10.1002/1097-0142(20001115)89:10<2068::aid-cncr6>3.0.co;2-z
31. Smith EM, Anderson B. The effects of symptoms and delay in seeking diagnosis on stage of disease at diagnosis among women with cancers of the ovary. Cancer. 1985;56:2727-2732. doi: 10.1002/1097-0142(19851201)56:11<2727::aid-cncr2820561138>3.0.co;2-8
32. Galvin JE, Duda JE, Kaufer DI, et al. Lewy body dementia: the caregiver experience of clinical care. Parkinsonism Relat Disord. 2010;16:388-392. doi: 10.1016/j.parkreldis.2010.03.007
33. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6:333-341. doi: 10.31887/DCNS.2004.6.3/imckeith
34. Mrak RE, Griffin WS. Dementia with Lewy bodies: definitions, diagnosis, and pathogenic relationship to Alzheimer’s disease. Neuropsychiatr Dis Treat. 2007;3:619-625.
35. Khotianov N, Singh R, Singh S. Lewy body dementia: case report and discussion. J Am Board Fam Pract. 2002;15:50-54.
36. Stinton C, McKeith I, Taylor JP, et al. Pharmacological management of Lewy body dementia: a systematic review and meta-analysis. Am J Psychiatry. 2015;172:731-742. doi: 10.1176/appi.ajp.2015.14121582
37. Velayudhan L, Ffytche D, Ballard C, et al. New therapeutic strategies of Lewy body dementias. Curr Neurol Neurosci Rep. 2017;17:68. doi: 10.1007/s11910-017-0778-2
38. Arnold LM, Clauw DJ, McCarberg BH; FibroCollaborative. Improving the recognition and diagnosis of fibromyalgia. Mayo Clin Proc. 2011;86:457-464. doi: 10.4065/mcp.2010.0738
39. Arnold LM, Gebke KB, Choy EHS. Fibromyalgia: management strategies for primary care providers. Int J Clin Pract. 2016;70:99-112. doi: 10.1111/ijcp.12757
40. Aloush V, Niv D, Ablin JN, et al. Good pain, bad pain: illness perception and physician attitudes towards rheumatoid arthritis and fibromyalgia patients. Clin Exp Rheumatol. 2021;39(suppl 130):54-60.
41. Vincent A, Lahr BD, Wolfe F, et al. Prevalence of fibromyalgia: a population-based study in Olmsted County, Minnesota, utilizing the Rochester Epidemiology Project. Arthritis Care Res (Hoboken). 2013;65:786-792. doi: 10.1002/acr.21896
42. White KP, Thompson J. Fibromyalgia syndrome in an Amish community: a controlled study to determine disease and symptom prevalence. J Rheumatol. 2003;30:1835-1840.
43. Lobo CP, Pfalzgraf AR, Giannetti V, et al. Impact of invalidation and trust in physicians on health outcomes in fibromyalgia patients. Prim Care Companion CNS Disord. 2014;16:10.4088/PCC.14m01664. doi: 10.4088/PCC.14m01664
44. Wolfe F, Clauw DJ, Fitzcharles MA, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken). 2010;62:600-610. doi:10.1002/acr.20140
1. Makary MA, Daniel M. Medical error—the third leading cause of death in the US. BMJ. 2016;353:i2139. doi: 10.1136/bmj.i2139
2. Van Such M, Lohr R, Beckman T, et al. Extent of diagnostic agreement among medical referrals. J Eval Clin Pract. 2017;23:870-874. doi: 10.1111/jep.12747
3. Groopman JE. How Doctors Think. Houghton Mifflin; 2007.
4. Tversky A, Kahneman D. Judgment under uncertainty: heuristics and biases. Science. 1974;185:1124-1131. doi: 10.1126/science.185.4157.1124
5. Norman GR, Monteiro SD, Sherbino J, et al. The causes of errors in clinical reasoning: Cognitive biases, knowledge deficits, and dual process thinking. Acad Med. 2017;92:23-30. doi: 10.1097/ACM.0000000000001421
6. Croskerry P. The importance of cognitive errors in diagnosis and strategies to minimize them. Acad Med. 2003;78:775-780. doi: 10.1097/00001888-200308000-00003
7. Morgenstern J. Cognitive errors in medicine: The common errors. First10EM blog. September 15, 2015. Updated September 22, 2019. Accessed February 8, 2022. https://first10em.com/cognitive-errors/
8. Gazitt T, Zisman D, Gardner G. Polymyalgia rheumatica: a common disease in seniors. Curr Rheumatol Rep. 2020;22:40. doi: 10.1007/s11926-020-00919-2
9. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35. doi: 10.1002/art.23176
10. Doran MF, Crowson CS, O’Fallon WM, et al. Trends in the incidence of polymyalgia rheumatica over a 30 year period in Olmsted County, Minnesota, USA. J Rheumatol. 2002;29:1694-1697.
11. Barraclough K, Liddell WG, du Toit J, et al. Polymyalgia rheumatica in primary care: a cohort study of the diagnostic criteria and outcome. Fam Pract. 2008;25:328-33. doi: 10.1093/fampra/cmn044
12. Manzo C. Polymyalgia rheumatica (PMR) with normal values of both erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) concentration at the time of diagnosis in a centenarian man: a case report. Diseases. 2018;6:84. doi: 10.3390/diseases6040084
13. Crowson CS, Matteson EL. Contemporary prevalence estimates for giant cell arteritis and polymyalgia rheumatica, 2015. Semin Arthritis Rheum. 2017;47:253-256. doi: 10.1016/j.semarthrit.2017.04.001
14. Nordborg E, Bengtsson BA. Death rates and causes of death in 284 consecutive patients with giant cell arteritis confirmed by biopsy. BMJ. 1989;299:549-550. doi: 10.1136/bmj.299.6698.549
15. Bahlas S, Ramos-Remus C, Davis P. Utilisation and costs of investigations, and accuracy of diagnosis of polymyalgia rheumatica by family physicians. Clin Rheumatol. 2000;19:278-280. doi: 10.1007/s100670070045
16. Brooks RC, McGee SR. Diagnostic dilemmas in polymyalgia rheumatica. Arch Intern Med. 1997;157:162-168.
17. Blaauw AA, Schuwirth LW, van der Vleuten CP, et al. Assessing clinical competence: recognition of case descriptions of rheumatic diseases by general practitioners. Br J Rheumatol. 1995;34:375-379. doi:10.1093/rheumatology/34.4.375
18. Mager DR. Polymylagia rheumatica: common disease, elusive diagnosis. Home Healthc Now. 2015;33:132-138. doi:10.1097/NHH.0000000000000199
19. Kermani TA, Warrington KJ. Polymyalgia rheumatica. Lancet. 381;63-72. doi: 10.1016/S0140-6736(12)60680-1. Published correction appears in Lancet. 20135;381:28.
20. Liew DF, Owen CE, Buchanan RR. Prescribing for polymyalgia rheumatica. Aust Prescr. 2018;41:14-19. doi: 10.18773/austprescr.2018.001
21. Ovarian cancer statistics. Centers for Disease Control and Prevention. Reviewed June 8, 2021. Accessed February 22, 2022. www.cdc.gov/cancer/ovarian/statistics/index.htm
22. Key statistics for ovarian cancer. American Cancer Society. Revised January 12, 2022. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/about/key-statistics.html
23. Survival rates for ovarian cancer. American Cancer Society. Revised January 25, 2021. Accessed February 22, 2022. www.cancer.org/cancer/ovarian-cancer/detection-diagnosis-staging/survival-rates.html
24. Reid BM, Permuth JB, Sellers TA. Epidemiology of ovarian cancer: a review. Cancer Biol Med. 2017;14:9-32. doi: 10.20892/j.issn.2095-3941.2016.0084
25. Goff BA, Mandel LS, Melancon CH, et al. Frequency of symptoms of ovarian cancer in women presenting to primary care clinics. JAMA. 2004;291:2705-2712. doi: 10.1001/jama.291.22.2705
26. Olson SH, Mignone L, Nakraseive C, et al. Symptoms of ovarian cancer. Obstet Gynecol. 2001;98:212-217. doi: 10.1016/s0029-7844(01)01457-0
27. Allgar VL, Neal RD. Delays in the diagnosis of six cancers: analysis of data from the National Survey of NHS patients: Cancer. Br J Cancer. 2005;92:1959-1970. doi: 10.1038/sj.bjc.6602587
28. Gajjar K, Ogden G, Mujahid MI, et al. Symptoms and risk factors of ovarian cancer: a survey in primary care. Obstet Gynecol. 2012:754197. doi: 10.5402/2012/754197
29. Austoker J. Diagnosis of ovarian cancer in primary care. BMJ. 2009;339:b3286. doi: 10.1136/bmj.b3286
30. Goff BA, Mandel L, Muntz HG, et al. Ovarian carcinoma diagnosis: results of a national ovarian cancer survey. Cancer. 2000;89:2068-2075. doi: 10.1002/1097-0142(20001115)89:10<2068::aid-cncr6>3.0.co;2-z
31. Smith EM, Anderson B. The effects of symptoms and delay in seeking diagnosis on stage of disease at diagnosis among women with cancers of the ovary. Cancer. 1985;56:2727-2732. doi: 10.1002/1097-0142(19851201)56:11<2727::aid-cncr2820561138>3.0.co;2-8
32. Galvin JE, Duda JE, Kaufer DI, et al. Lewy body dementia: the caregiver experience of clinical care. Parkinsonism Relat Disord. 2010;16:388-392. doi: 10.1016/j.parkreldis.2010.03.007
33. McKeith I. Dementia with Lewy bodies. Dialogues Clin Neurosci. 2004;6:333-341. doi: 10.31887/DCNS.2004.6.3/imckeith
34. Mrak RE, Griffin WS. Dementia with Lewy bodies: definitions, diagnosis, and pathogenic relationship to Alzheimer’s disease. Neuropsychiatr Dis Treat. 2007;3:619-625.
35. Khotianov N, Singh R, Singh S. Lewy body dementia: case report and discussion. J Am Board Fam Pract. 2002;15:50-54.
36. Stinton C, McKeith I, Taylor JP, et al. Pharmacological management of Lewy body dementia: a systematic review and meta-analysis. Am J Psychiatry. 2015;172:731-742. doi: 10.1176/appi.ajp.2015.14121582
37. Velayudhan L, Ffytche D, Ballard C, et al. New therapeutic strategies of Lewy body dementias. Curr Neurol Neurosci Rep. 2017;17:68. doi: 10.1007/s11910-017-0778-2
38. Arnold LM, Clauw DJ, McCarberg BH; FibroCollaborative. Improving the recognition and diagnosis of fibromyalgia. Mayo Clin Proc. 2011;86:457-464. doi: 10.4065/mcp.2010.0738
39. Arnold LM, Gebke KB, Choy EHS. Fibromyalgia: management strategies for primary care providers. Int J Clin Pract. 2016;70:99-112. doi: 10.1111/ijcp.12757
40. Aloush V, Niv D, Ablin JN, et al. Good pain, bad pain: illness perception and physician attitudes towards rheumatoid arthritis and fibromyalgia patients. Clin Exp Rheumatol. 2021;39(suppl 130):54-60.
41. Vincent A, Lahr BD, Wolfe F, et al. Prevalence of fibromyalgia: a population-based study in Olmsted County, Minnesota, utilizing the Rochester Epidemiology Project. Arthritis Care Res (Hoboken). 2013;65:786-792. doi: 10.1002/acr.21896
42. White KP, Thompson J. Fibromyalgia syndrome in an Amish community: a controlled study to determine disease and symptom prevalence. J Rheumatol. 2003;30:1835-1840.
43. Lobo CP, Pfalzgraf AR, Giannetti V, et al. Impact of invalidation and trust in physicians on health outcomes in fibromyalgia patients. Prim Care Companion CNS Disord. 2014;16:10.4088/PCC.14m01664. doi: 10.4088/PCC.14m01664
44. Wolfe F, Clauw DJ, Fitzcharles MA, et al. The American College of Rheumatology preliminary diagnostic criteria for fibromyalgia and measurement of symptom severity. Arthritis Care Res (Hoboken). 2010;62:600-610. doi:10.1002/acr.20140
Strategies for improved management of hypothyroidism
The hormones thyroxine (T4) and triiodothyronine (T3), produced by the thyroid gland, are crucial for maintaining metabolism. A deficit of thyroid hormone production—hypothyroidism—is a common endocrine disorder seen in primary care.
Although the diagnosis and management of hypothyroidism are considered straightforward, many patients with hypothyroidism do not achieve optimal treatment goals or see an improvement in their quality of life. In this article, we address the questionable utility of screening; outline the diagnostic approach, including the central role of laboratory testing; and explain why treatment requires a precise approach to address the range of patient types.
Epidemiology and classification
Estimates are that almost 5% of Americans 12 years or older have hypothyroidism; older people and women are more likely to develop the condition. 1 In the US National Health and Nutrition Examination Survey (NHANES III) of 13,344 people without known thyroid disease or a family history, hypothyroidism was found in 4.6% (overt [clinical] in 0.3% and subclinical in 4.3%); 11% had a high serum thyroid peroxidase antibody level, which increases their risk of hypothyroidism, and is treated the same as hypothyroidism of other causes; and, overall, lower serum thyroid-stimulating hormone (TSH) levels were seen in Blacks, compared to Whites and Mexican Americans.1
Primary hypothyroidism accounts for > 95% of cases of hypothyroidism, representing a failure of the thyroid gland to produce sufficient hormone. It has been shown that, in iodine-replete countries such as the United States, the prevalence of spontaneous hypothyroidism is 1% to 2%, and it is 10 times more common in women.2,3
Central hypothyroidism is caused by insufficient stimulation of the thyroid gland by TSH, due to pituitary (secondary hypothyroidism) or hypothalamic (tertiary hypothyroidism) disease and is estimated to occur in 1 in every 20,000 to 80,000 people in the general population.4
How does hypothyroidism manifest?
Signs and symptoms. Manifestations of hypothyroidism range from life-threatening to minimal or no clinical signs and symptoms (TABLE W1). Signs and symptoms of low thyroid function vary by the degree of hypothyroidism at presentation.
Common signs and symptoms of low thyroid function include fatigue, weight gain, dry skin, brittle hair, hair loss, morning stiffness, muscle aches, joint pain, cold intolerance, diffuse headache, constipation, difficulty concentrating, low libido, depression, and menstrual irregularities. On physical examination, a patient might present with bradycardia, hypotension, hypothermia with slow speech or movement, coarse facial appearance, goiter, diffuse hair loss, cold hands and feet, and a prolonged Achilles tendon reflex.5 Skin findings, such as keratosis pilaris, palmoplantar keratoderma (thickening of the skin), and pityriasis rubra pilar, can be associated with autoimmune hypothyroidism.6,7
Continue to: Carpal tunnel syndrome...
Carpal tunnel syndrome, plantar fasciitis, infertility or miscarriage, dyspepsia, and small intestinal bacterial overgrowth can be associated with hypothyroidism; thyroid function should therefore be assessed in patients who have any of these conditions, along with other signs and symptoms of low thyroid function.8,9 A patient with severe hypothyroidism might present with hemodynamic instability, pericardial or pleural effusion, and myxedema coma.10
Clues in the history and from the lab. A history of radiation to the head, neck, or chest area and a history or family history of autoimmune disorders are risk factors for autoimmune thyroid disease.11,12 Laboratory findings can include markers of oxidative stress, such as elevated levels of low-density lipoprotein cholesterol and serum malondialdehyde.13-15
Screening and diagnosis
Screening. The US Preventive Services Task Force has asserted that evidence is insufficient by which to evaluate the benefits and risks of routine screening for thyroid dysfunction in nonpregnant, asymptomatic adults.16 According to the American Thyroid Association and the American Association of Clinical Endocrinologists, screening should be considered in high-risk patients, including those who take medication that affects thyroid function or the results of thyroid hormone assays (TABLE W2, available at mdedge.com/familymedicine).17-20
Screening inpatients is challenging and usually not recommended unless thyroid disease is strongly suspected. This is because changes in the levels of thyroid hormones, binding proteins, and the TSH concentration can occur in severe nonthyroidal illness; in addition, assay interference by antibodies and other substances can affect thyroid hormone measurement.21
Testing strategy. Generally, screening and diagnosis of hypothyroidism are based primarily on laboratory testing, because signs and symptoms are nonspecific (FIGURE 15). A serum TSH level is usually the initial test when screening for thyroid dysfunction. A normal serum TSH value ranges from 0.5-5.0 mIU/L.
When an abnormal serum TSH level is found, further tests can be performed to investigate, including a serum free thyroxine (FT4) test. (Our preference is to order TSH and FT4 assays simultaneously to facilitate and confirm the diagnosis.) An FT4 test measures the amount of unattached, or free, thyroxine in blood by immunoassay. A normal FT4 value usually ranges from 0.7-1.9 ng/dL.
The combination of a high TSH level and a low FT4 level could be an indication of an underactive thyroid gland (ie, clinical or overt hypothyroidism). Milder, subclinical hypothyroidism is characterized by a higher-than-normal TSH level but a normal FT4 level.22 Central (secondary) hypothyroidism is characterized by a low serum FT4 level and a serum TSH level that can be below the reference range, low normal, or even slightly high.4
Continue to: These measurements...
These measurements must be interpreted within the context of the laboratory-specific normal range for each test. Third-generation serum TSH assays are more sensitive and specific than serum FT4 measurements for hypothyroidism. FT4 is usually measured by automated analogue immunoassay, which generally provides reliable results; abnormal binding proteins or other interferences occur in some patients, however, resulting in reporting of a falsely high, or falsely low, FT4 level. In such cases, FT4 by direct dialysis, or total T4, can be measured for further evaluation. In primary care, you are most likely to encounter primary hypothyroidism; secondary (central) hypothyroidism is much rarer (< 5% of cases).4
The ins and outs of treatment
For most patients, hypothyroidism is a permanent disorder requiring lifelong thyroid hormone replacement therapy—unless the disease is transient (ie, painless or subacute thyroiditis); reversible, because it is caused by medication; or responsive to medical intervention that addresses the underlying autoimmune condition.19 Goals of treatment (Figure 25,23) are to:
- normalize the TSH level to 0.5-5.0 mIU/L (the main goal), with an age-related shift toward a higher TSH goal in older patients (and an upper limit of normal of 7.5 mIU/L in patients who are ≥ 80 years of age)20
- restore the euthyroid state
- relieve symptoms
- reduce any goiter
- avoid overtreatment (iatrogenic thyrotoxicosis).
Desiccated thyroid extract (DTE), developed in the late 1880s and made from the dried thyroid gland of pigs, sheep, or cows, was the earliest treatment for hypothyroidism. The use of DTE has declined since the introduction of synthetic thyroxine (T4, or levothyroxine [here, referred to as LT4]), which is now the standard treatment.20-22 LT4 is deiodinated in peripheral tissues to form T3, the active thyroid hormone; this process accounts for 80% of total T3 production daily.24
LT4 formulations. LT4 is commercially available in tablet, soft-gel, and liquid preparations. Most patients are treated with the tablet; the soft-gel capsule or liquid is an option for patients who absorb the tablet poorly (because of atrophic gastritis, celiac disease, or gluten sensitivity or because they are post bariatric surgery). Increasing the dosage of the tablet form of LT4, with ongoing TSH monitoring, is more cost effective than moving to an alternative preparation.
If a switch of LT4 formulation is made (ie, from one manufacturer to another), test the serum TSH level to ensure that the therapeutic goal is being reached. Also, in our experience, it is best to prescribe a brand-name preparation of levothyroxine, not a generic, whenever possible, due to the variability in generic formulations and the potential presence of other (inert) ingredients.25
Dosing (TABLE 320,23). The average full replacement dosage of LT4 for a young, healthy adult is approximately 1.6 mcg/kg/d. Older patients (> 65 years) or those with coronary artery disease (CAD) should be started on a lower dosage (25-50 mcg/d) and titrated to goal accordingly.
LT4 (tablets, soft-gel capsules, or liquid) should be administered on an empty stomach, with water only, 30 to 60 minutes before breakfast. Medications that interfere with LT4 absorption (eg, bile acid resins, calcium carbonate, ferrous sulfate) should be taken several hours after LT4. For patients who cannot take LT4 in the morning, taking it at bedtime (≥ 2-3 hours after the last meal) is acceptable.
Continue to: Monitoring and titrating
Monitoring and titrating. Hypothyroid symptoms usually improve after 2 or 3 weeks of LT4 treatment; in severe hypothyroidism, complete recovery might take months. Approximately 6 weeks after LT4 therapy is initiated, serum TSH should be measured. After assessing whether administration of LT4 at the starting dosage is appropriate, that dosage can be increased, or decreased, every 4 to 6 weeks until the TSH goal is reached. Once the patient is maintained at a given dosage, measure serum TSH once a year—more often if there is an abnormal result or a change in the patient’s health status.23
Adverse effects of LT4 therapy are rare, unless over-replacement occurs. Rarely, patients have an allergy to the dye or an excipient (filler) in the tablet.26-28 The white, 50-mcg tablets can be given safely to patients with dye sensitivity. For those who have an allergy to an excipient (except gelatin) or gluten intolerance, the LT4 soft-gel capsule or liquid preparation (Tirosint) can be prescribed.
Pure LT4, in a capsule made from vegetable sources, can be ordered through a compounding pharmacy for patients who are allergic to animal products.
Anemia, especially iron-deficiency anemia, can cause intolerance to LT4 therapy; in such patients, lowering the starting dosage and treating anemia are indicated.29
Persistent symptoms (despite a normal TSH level). Because many hypothyroid symptoms are nonspecific, patients might come to think that their LT4 dosage is inadequate if they feel tired or gain weight. Persistent hypothyroid symptoms despite a normal serum TSH level might be due to (1) the inability of LT4 therapy to restore tissue thyroid hormone levels to normal or (2) other variables unrelated to hypothyroidism, including disorders associated with inflammation or autoimmune disease, certain medications, diet, lifestyle, and environmental toxins.
These patients might benefit from a detailed history to identify other causes and a switch to either LT4 + liothyronine (LT3; synthetic T3) combination therapy or DTE26,30-33 (TABLE 434), although a beneficial effect of LT4 + LT3 therapy was not seen in several studies.35,36 Over-replacement with LT4 should be discouraged, due to concerns about thyrotoxicosis and its complications (eg, atrial fibrillation, accelerated bone loss).
DTE and LT4 + LT3. Use of DTE has decreased since the 1970s, when LT4 became the therapy of choice. Subsequently, anecdotal evidence emerged that some patients did not feel well on LT4 and preferred to return to DTE.32,33
Continue to: Several clinical trials...
Several clinical trials addressed the question of whether residual symptoms could be resolved through LT4 + LT3 combination therapy31-39 (TABLE 434), but evidence of any consistent superiority of combination therapy was not demonstrated.35-39 In selected cases, patients might prefer the combination approach.31,33,39 The quality of life of hypothyroid patients was found to be similarly improved with LT4 or DTE, but the latter was associated with modest weight loss (approximately 4 lbs); nearly 50% of study patients preferred treatment with DTE over LT4.33 A follow-up study did not confirm weight loss with DTE, however.34
When LT4 monotherapy and LT4 + LT3 combination therapy were compared, results were mixed31-39; responsiveness to therapy containing LT3 might therefore depend on multiple variables, including genetic background, nutritional and lifestyle factors, stress, presence of comorbidities and autoimmune disorders, and other unidentified or poorly defined variables.40-48
Although combination therapy and DTE are not generally recommended over LT4 monotherapy, they might offer better options for patients who are still symptomatic when being treated with LT4 only: In a randomized, double-blind, crossover study that compared LT4 with DTE and with LT4 + LT3, one-third of the most highly symptomatic patients who had low scores on mood, cognitive, and quality-of-life assessments improved significantly after they were switched to combination therapy or DTE.34
The 2014 American Thyroid Association guidelines24 do not support routine use of LT4 + LT3 in hypothyroid patients who have residual symptoms after LT4 monotherapy; however, a therapeutic trial of LT4 + LT3, while maintaining a normal serum TSH, is reasonable in selected patients. Candidates for DTE or LT4 + LT3 might include patients who do not feel well on LT4 monotherapy, are post thyroidectomy or post radioiodine therapy, or have a low serum T3 level. DTE and combination therapy are discouraged in older patients, patients who have underlying CAD, and pregnant patients.
Special treatment circumstances
A number of patient variables have the potential to alter management strategies for hypothyroidism.18,20,23,40,49-53
Age, comorbidity. Older patients (> 65 years) and patients with cardiopulmonary disease or CAD should be treated with LT4, 25 to 50 mcg/d, initially; that dosage can be titrated upward by 12.5 to 25 mcg/d every 4 to 6 weeks until the TSH goal is reached—preferably, in the range of 4 to 8 mIU/L. An increase in the dosage of LT4 might be required in the presence of malabsorption (eg, gastrointestinal disorders, celiac disease) and in nephrotic syndrome.18,20,23
Body weight. A decrease in the dosage of LT4 might be indicated in the setting of significant weight loss (> 10% body weight).23
Continue to: Co-pharmacy
Co-pharmacy. An increase in the dosage of LT4 might be required when other drugs (eg, phenytoin, phenobarbital, rifampin, and carbamazepine) have led to an increased rate of thyroid hormone metabolism. A decrease in the dosage of LT4 might be necessary after initiation of androgen therapy.23
Pregnancy. Women with pre-existing hypothyroidism require an increase of 25% to 50% in their LT4 dosage during pregnancy to maintain a TSH level in the recommended pregnancy reference range. Thyroid function should be monitored every 4 to 6 weeks to ensure that the TSH target for each trimester is reached (first trimester, 0.1-4 mIU/L; second trimester, 0.2-4 mIU/L; third trimester, 0.3-4 mIU/L). Postpartum, LT4 can be reduced to the prepartum dosage; TSH should be checked every 4 to 6 weeks to maintain the TSH goal.23
Estrogen therapy. Hypothyroid women who are receiving estrogen therapy might require an increase in their LT4 dosage because serum thyroxine-binding globulin levels are increased by estrogens or through other mechanisms that have not been identified.23
Surgical candidacy. Observational studies show few adverse outcomes in surgical patients with mild (subclinical) hypothyroidism or moderate hypothyroidism; however, the risk of adverse surgical outcome might be increased in patients with severe hypothyroidism. For patients in whom surgery is planned and who have:
- subclinical hypothyroidism (elevated TSH and normal FT4), we recommend that surgery—urgent or elective—not be posptoned but proceed.
- moderate (overt) hypothyroidism who require urgent surgery, we recommend not postponing surgery, even though minor perioperative complications might develop. Such patients should be treated with LT4 as soon as the diagnosis for which surgery is required has been made. Alternatively, when moderate hypothyroidism is discovered in a patient who is being evaluated for elective surgery, we recommend postponing surgery until the euthyroid state is restored.
- severe hypothyroidism (myxedema coma [discussed in a bit]; severe clinical symptoms of chronic hypothyroidism, such as altered mental status, pericardial effusion, or heart failure; or a very low level of T4), surgery should be delayed until hypothyroidism has been treated. When emergency surgery is required for a severely hypothyroid patient, they should be treated with LT4 as soon as the diagnosis for which surgery is indicated has been made. When emergency surgery must be performed in a patient with myxedema coma, we recommend treatment with LT4 + LT3, rather than LT4 alone, often administered intravenously because LT4 is poorly absorbed in these patients.
Nonadherence. For patients who do not take LT4 regularly or do not respond to efforts to improve adherence, LT4 can be given weekly, instead of daily, although this interval is not ideal. Weekly dosing should not be used in older patients with CAD.23
Thyroid cancer. Patients who are post total thyroidectomy for thyroid cancer need to take LT4 to treat hypothyroidism and to prevent recurrence of thyroid cancer. The goal TSH level should be based on the cancer stage and risk of recurrence and should be monitored by an endocrinologist.
Myxedema coma. This medical emergency has high mortality. Myxedema coma occurs when severe hypothyroidism leads to any, or a combination, of the following: diminished mentation; hypothermia; bradycardia; hyponatremia; hypotension; cardiovascular, respiratory, and gastrointestinal dysfunction; and renal insufficiency. LT4, LT3, and glucocorticoids should be administered intravenously and the patient monitored closely—preferably in consultation with an endocrinologist.
Continue to: When to seek consultation
When to seek consultation
A patient with hypothyroidism should be referred to Endocrinology if they are < 18 years of age, pregnant, unresponsive to therapy, or have cardiac disease, coexisting endocrine disease, suspected myxedema coma, goiter or thyroid nodules, or a structural thyroid abnormality.
What we know about nutrition and hypothyroidism
Although it is commonly recognized that iodine is essential for production of thyroid hormone, other nutritional factors might contribute to proper production of thyroid hormones, including:
- adequate intake of iron, tyrosine, selenium, zinc, and vitamins E, B2, B3, B6, C, and D44,45
- selenium and zinc, which increase conversion of T4 to T3 and might be important in the management of hypothyroid patients40,46
- vitamin A, zinc, and regular exercise, which have been shown to improve cellular sensitivity to thyroid hormones.
Low iron stores can contribute to persistent symptoms and poor quality of life in patients with hypothyroidism, despite their being treated according to guidelines.29,47
Despite what is known about these nutritional connections, there is insufficient evidence that improving nutrition can reverse hypothyroidism.
Prevention
Prevention of hypothyroidism should take into account variables that affect or inhibit thyroid function, such as stress, infection (eg, Epstein-Barr virus), excessive fluoride intake, toxins (eg, pesticides, solvents, mercury, cadmium, and lead), autoimmune disease (eg, celiac disease), and food sensitivity.54,55 Oxidative stress can also cause thyroid impairment.40-48,54-58
Otherwise, there are, at present, no effective strategies for preventing thyroid disorders.
Subclinical hypothyroidism: Elusive management target
Subclinical hypothyroidism is defined as a normal serum FT4 level in the presence of an elevated serum TSH level. The prevalence of subclinical hypothyroidism varies from 3% to 15%, depending on the population studied; a higher incidence has been noted in women and older people.59 In the NHANES III,1 which excluded people with previously diagnosed thyroid disease, the incidence of subclinical hypothyroidism was 4.3%.
Continue to: Causes of subclinical...
Causes of subclinical hypothyroidism are the same as those of overt hypothyroidism, and include Hashimoto disease. The combination of an elevated TSH level and a normal FT4 level is associated with disorders characterized by protein-binding variations (eg, pregnancy, genetic disorders, drugs), TSH-secreting pituitary adenoma, class II and III obesity (respectively, body mass index, ≥ 35 but < 40 and ≥ 40), and assay variability.49,51
Lab diagnosis: Fraught with difficulty
The serum TSH level and either the total T4 level or the FT4 level should be measured to make a diagnosis of subclinical hypothyroidism. Most laboratories use a 1-step analogue immunoassay to determine free thyroid hormones; protein-binding variations can thus affect measurement of FT4.
Several scenarios that can result in inaccurate measurement of FT4 by radioimmunoassay include genetic disorders that affect binding proteins; pregnancy; use of certain drugs, including heparin, furosemide, antiepileptic agents, salicylate, ferrous sulfate, and cholesterol-binding resins; and some medical conditions, including cardiac surgery, critical illness, and renal failure. Variables that inhibit proper production of thyroid hormones—stress, infection, fluoride (an iodine antagonist), toxins (pesticides, mercury, cadmium, lead) and autoimmune conditions, such as celiac disease—should be considered when attempting to determine the cause of subclinical hypothyroidism.
Liquid chromatography–mass spectrometry measurement of thyroid hormones might be more accurate than immunoassay.53 Measuring serum total T4 and FT4 by dialysis, free from interfering proteins, might also be useful when measurement of FT4 by immunoassay is affected by binding-protein variations.
Features of subclinical hypothyroidism
Most patients who have subclinical hypothyroidism and a serum TSH level < 10 mIU/L are asymptomatic. Some might have nonspecific symptoms of hypothyroidism, however, such as reduced quality of life, poor cognitive function, and poor memory—symptoms that do not typically correlate with the serum TSH level.
It has been suggested that some elderly people normally have a higher level of serum TSH, and that they might have even a prolonged lifespan.51 Additionally, it has been shown that, in nonpregnant adult patients with subclinical hypothyroidism and a serum TSH level of 4.5 to 10 mIU/L, treatment with LT4 was not associated with improvement in thyroid-related symptoms or general quality of life.52
Treat, or don't treat, subclinical hypothyroidism?
It is well accepted that the goal of therapy in hypothyroid patients is to normalize the serum TSH level; however, the American Thyroid Association and the American Association of Clinical Endocrinology recommend starting LT4 in patients with a serum TSH level ≥ 10 mIU/L (TABLE 5).59-62 The principal reason for not treating subclinical hypothyroidism is the lack of benefit in reducing the risk of cardiovascular morbidity and mortality when the TSH level is between 7.5 and 10 mIU/L.62
Continue to: Routine treatment
Routine treatment of patients with a serum TSH level of 4.5 to 10 mIU/L remains controversial. When TSH is 7.0 to 9.9 mIU/L, treatment is recommended for (1) patients < 65 years and (2) for older patients (> 65 years) only when there are convincing hypothyroid symptoms because of concern about unintended overtreatment.
When the TSH level is anywhere above the upper limit of normal to 6.9 mIU/L, treatment is recommended for patients < 65 years old, patients who have a high titer of thyroid peroxidase antibodies, and patients with goiter—but not for patients > 65 years (and, especially, not for octogenarians) because their upper limit of normal could be as high as 6 to 8 mIU/L, especially if they are otherwise healthy.
Treatment should be considered for women with subclinical hypothyroidism who are trying to conceive or experiencing an infertility problem.
For patients with subclinical hypothyroidism who are not being treated, monitor thyroid function every 6 to 12 months by testing TSH and FT4.
CORRESPONDENCE
Thanh D. Hoang, DO, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; thanh.d.hoang.mil@mail.mil
1. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499. doi: 10.1210/jcem.87.2.8182
2. Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99:39-51. doi: 10.1093/bmb/ldr030
3. Canaris GJ, Manowitz NR, Mayor G, et al. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160:526-534. doi: 10.1001/archinte.160.4.526
4. Persani L. Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab. 2012;97:3068-3078. doi: 10.1210/jc.2012-1616
5. Almandoz JP, Gharib H. Hypothyroidism: etiology, diagnosis, and management. Med Clin North Am. 2012;96:203-221. doi: 10.1016/j.mcna.2012.01.005
6. Ai J, Leonhardt JM, Heymann WR. Autoimmune thyroid diseases: etiology, pathogenesis and dermatologic manifestations. J Am Acad Dermatol. 2003;48:641-659. doi: 10.1067/mjd.2003.257
7. Franzotti AM, Avelar JCD, Cardoso TA, et al. Pityriasis rubra pilar and hypothyroidism. An Bras Dermatol. 2014;89:497-500. doi: 10.1590/abd1806-4841.20142994
8. Yaylali O, Kirac S, Yilmaz M, et al. Does hypothyroidism affect gastrointestinal motility? Gastroenterol Res Pract. 2009;2009:529802. doi: 10.1155/2009/529802
9. Patil AD. Link between hypothyroidism and small intestinal bacterial overgrowth. Indian J Endocrinol Metab. 2014;18:307-309.
10. Ono Y, Ono S, Yasunaga H, et al. Clinical characteristics and outcomes of myxedema coma: analysis of a national inpatient database in Japan. J Epidemiol. 2017;27:117-122. doi: 10.1016/j.je.2016.04.002
11. Boomsma MJ, Bijl HP, Langendijk JA. Radiation-induced hypothyroidism in head and neck cancer patients: a systematic review. Radiother Oncol. 2011;99:1-5. doi: 10.1016/j.radonc.2011.03.002
12. Boelaert K, Newby PR, Simmonds MJ, et al. Prevalence and relative risk of other autoimmune diseases in subjects with autoimmune thyroid disease. Am J Med. 2010;123:183.e1-e9. doi: 10.1016/j.amjmed.2009.06.030
13. Cheserek MJ, Wu G-R, Ntazinda A, et al. Association between thyroid hormones, lipids and oxidative stress markers in subclinical hypothyroidism. J Med Biochem. 2015;34:323-331. doi: 10.2478/jomb-2014-0044
14. Zha K, Zuo C, Wang A, et al. LDL in patients with subclinical hypothyroidism shows increased lipid peroxidation. Lipids Health Dis. 2015;14:95. doi: 10.1186/s12944-015-0092-4
15. Tejovathi B, Suchitra MM, Suresh V, et al. Association of lipid oxidation with endothelial dysfunction in patients with overt hypothyroidism. Exp Clin Endocrinol Diabetes. 2013;121:306-309. doi: 10.1055/s-0032-1333298
16. LeFevre ML; U.S. Preventive Services Task Force. Screening for thyroid dysfunction: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015;162:641-650. doi: 10.7326/M15-0483
17. Chaker L, Bianco AC, Jonklaas J, et al. Hypothyroidism. Lancet. 2017;390:1550-1562. doi: 10.1016/S0140-6736(17)30703-1
18. Vaidya B, Pearce SHS. Management of hypothyroidism in adults. BMJ. 2008;337:a801. doi: 10.1136/bmj.a801
19. Iyer PC, Cabanillas ME, Waguespack SG, et al. Immune-related thyroiditis with immune checkpoint inhibitors. Thyroid. 2018;28:1243-1251. doi: 10.1089/thy.2018.0116
20. Garber JR, Cobin RH, Gharib H, et al; American Association Of Clinical Endocrinologists And American Thyroid Association Taskforce On Hypothyroidism In Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid. 2012;22:1200-1235. doi: 10.1089/thy.2012.0205
21. Maiden MJ, Torpy DJ. Thyroid hormones in critical illness. Crit Care Clin. 2019;35:375-388. doi: 10.1016/j.ccc.2018.11.012
22. Peeters RP. Subclinical hypothyroidism. N Engl J Med. 2017;376:2556-2565. doi: 10.1056/NEJMcp1611144
23. Benvenga S, Carlé A. Levothyroxine formulations: pharmacological and clinical implications of generic substitution. Adv Ther. 2019;36(suppl 2):59-71. doi: 10.1007/s12325-019-01079-1
24. Jonklaas J, Bianco AC, Bauer AJ, et al; . Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association Task Force on Thyroid Hormone Replacement. Thyroid. 2014;24:1670-1751. doi: 10.1089/thy.2014.0028
25. Engler D, Burger AG. The deiodination of the iodothyronines and of their derivatives in man. Endocr Rev. 1984;5:151-184. doi: 10.1210/edrv-5-2-151
26. Ettleson MD, Bianco AC. Individualized therapy for hypothyroidism: is T4 enough for everyone? J Clin Endocrinol Metab. 2020;105:e3090-e3104. doi: 10.1210/clinem/dgaa430
27. Slayden TA, Shakir MKM, Hoang TD. A bull in a pill shop: alpha-gal allergy complicating treatment options for postprocedural hypothyroidism. AACE Clin Case Rep. 2020;6:e101-e104. doi: 10.4158/ACCR-2019-0495
28. Chamorro-Pareja N, Carrillo-Martin I, Haehn DA, et al. Self-reported allergy to thyroid replacement therapy: a multicenter retrospective chart review. Endocr Pract. 2020;26:761-767. doi: 10.4158/EP-2019-0488
29. Shakir MKM, Turton D, Aprill BS, et al. Anemia: a cause of intolerance to thyroxine sodium. Mayo Clin Proc. 2000;75:189-192.
30. Jonklaas J, Bianco AC, Cappola AR, et al. Evidence-based use of levothyroxine/liothyronine combinations in treating hypothyroidism: a consensus document. Thyroid. 2021;31:156-182. doi: 10.1089/thy.2020.0720
31. Appelhof BC, Fliers E, Wekking EM, et al. Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J Clin Endocrinol Metab. 2005;90:2666-2674. doi: 10.1210/jc.2004-2111
32. Escobar-Morreale HF, Botella-Carretero JI, M, et al. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann Intern Med. 2005;142:412-424. doi: 10.7326/0003-4819-142-6-200503150-00007
33. Hoang TD, Olsen CH, Mai VQ, et al. Desiccated thyroid extract compared with levothyroxine in the treatment of hypothyroidism: a randomized, double-blind, crossover study. J Clin Endocrinol Metab. 2013;98:1982-1990. doi: 10.1210/jc.2012-4107
34. Shakir MKM, Brooks DI, McAninch EA, et al. Comparative effectiveness of levothyroxine, desiccated thyroid extract, and levothyroxine+liothyronine in hypothyroidism. J Clin Endocrinol Metab. 2021;106:e4400-e4413. doi: 10.1210/clinem/dgab478
35. Valizadeh M, Seyyed-Majidi MR, Hajibeigloo H, et al. Efficacy of combined levothyroxine and liothyronine as compared with levothyroxine monotherapy in primary hypothyroidism: a randomized controlled trial. Endocr Res. 2009;34:80-89. doi: 10.1080/07435800903156340
36. Walsh JP, Shiels L, Lim EM, et al. Combined thyroxine/liothyronine treatment does not improve well-being, quality of life, or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J Clin Endocrinol Metab. 2003;88:4543-4550. doi: 10.1210/jc.2003-030249
37. Rodriguez T, Lavis VR, Meininger JC, et al. Substitution of liothyronine at a 1:5 ratio for a portion of levothyroxine: effect on fatigue, symptoms of depression, and working memory versus treatment with levothyroxine alone. Endocr Pract. 2005;11:223-233. doi: 10.4158/EP.11.4.223
38. Sawka AM, Gerstein HC, Marriott MJ, et al. Does a combination regimen of thyroxine (T4) and 3,5,3’-triiodothyronine improve depressive symptoms better than T4 alone in patients with hypothyroidism? Results of a double-blind, randomized, controlled trial. J Clin Endocrinol Metab. 2003;88:4551-4555. doi: 10.1210/jc.2003-030139
39. Clyde PW, Harari AE, Getka EJ, et al. Combined levothyroxine plus liothyronine compared with levothyroxine alone in primary hypothyroidism: a randomized controlled trial. JAMA. 2003;290:2952-2958. doi: 10.1001/jama.290.22.2952
40. Duntas LH. Selenium and the thyroid: a close-knit connection. J Clin Endocrinol Metab. 2010;95:5180-5188. doi: 10.1210/jc.2010-0191
41. Winther KH, Wichman JEM, Bonnema SJ, et al. Insufficient documentation for clinical efficacy of selenium supplementation in chronic autoimmune thyroiditis, based on a systematic review and meta-analysis. Endocrine. 2017;55:376-385. doi: 10.1007/s12020-016-1098-z
42. Parva NR, Tadepalli S, Singh P, et al. Prevalence of vitamin D deficiency and associated risk factors in the US population (2011-2012). Cureus. 2018;10:e2741. doi: 10.7759/cureus.2741
43. Wang J, Lv S, Chen G, et al. Meta-analysis of the association between vitamin D and autoimmune thyroid disease. Nutrients. 2015,7:2485-2498. doi: 10.3390/nu7042485
44. Wilson MM, Reedy J, Krebs-Smith SM. American diet quality: where it is, where it is heading, and what it could be. J Acad Nutr Diet. 2016;116:302-310.e1. doi: 10.1016/j.jand.2015.09.020
45. Babiker A, Alawi A, Al Atawi M, et al. The role of micronutrients in thyroid dysfunction. Sudan J Paediatr. 2020;20:13-19. doi: 10.24911/SJP.106-1587138942
46. Knezevic J, Starchl C, Tmava Berisha A, et al. Thyroid-gut-axis: How does the microbiota influence thyroid function? Nutrients. 2020;12:1769. doi: 10.3390/nu12061769
47. Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78:34-44. doi: 10.1017/S0029665118001192
48. Chakrabarti SK, Ghosh S, Banerjee S, et al. Oxidative stress in hypothyroid patients and the role of antioxidant supplementation. Indian J Endocrinol Metab. 2016;20:674-678. doi: 10.4103/2230-8210.190555
49. Tseng F-Y, Lin W-Y, Lin C-C, et al. Subclinical hypothyroidism is associated with increased risk for all-cause and cardiovascular mortality in adults. J Am Coll Cardiol. 2012;60:730-737. doi: 10.1016/j.jacc.2012.03.047
50. Roberts LM, Pattison H, Roalfe A, et al. Is subclinical thyroid dysfunction in the elderly associated with depression or cognitive dysfunction? Ann Intern Med. 2006;145:573-581. doi: 10.7326/0003-4819-145-8-200610170-00006
51. Gussekloo J, van Exel E, de Craen AJM, et al. Thyroid status, disability and cognitive function, and survival in old age. JAMA. 2004;292:2591-2599. doi: 10.1001/jama.292.21.2591
52. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism: a systematic review and meta-analysis. JAMA. 2018;320:1349-1359. doi: 10.1001/jama.2018.13770
53. Monzani F, Dardano A, Caraccio N. Does treating subclinical hypothyroidism improve markers of cardiovascular risk? Treat Endocrinol. 2006;5:65-81. doi: 10.2165/00024677-200605020-00001
54. Duntas LH. Does celiac disease trigger autoimmune thyroiditis? Nat Rev Endocrinol. 2009;5:190-191. doi: 10.1038/nrendo.2009.46
55. Lerner A, Jeremias P, Matthias T. Gut-thyroid axis and celiac disease. Endocr Connect. 2017;6:R52-R58. doi: 10.1530/EC-17-0021
56. Janegova A, Janega P, Rychly B, et al. The role of Epstein-Barr virus infection in the development of autoimmune thyroid diseases. Endokrynol Pol. 2015;66:132-136. doi: 10.5603/EP.2015.0020
57. Brent GA. Environmental exposures and autoimmune thyroid disease. Thyroid. 2010;20:755-761. doi: 10.1089/thy.2010.1636
58. Valko M, Leibfritz D, Moncol J, et al. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44-84. doi: 10.1016/j.biocel.2006.07.001
59. Garber JR, Cobin RH, Gharib H, et al; American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028. doi: 10.4158/EP12280.GL
60. Welsh KJ, Soldin SJ. Diagnosis of endocrine disease: How reliable are free thyroid and total T3 hormone assays? Eur J Endocrinol. 2016;175:R255-R263. doi: 10.1530/EJE-16-0193
61. Grossman A, Feldhamer I, Meyerovitch J. Treatment with levothyroxin in subclinical hypothyroidism is associated with increased mortality in the elderly. Eur J Intern Med. 2018;50:65-68. doi: 10.1016/j.ejim.2017.11.010
62. Pearce SHS, Brabant G, Duntas LH, et al. 2013 ETA Guideline: management of subclinical hypothyroidism. Eur Thyroid J. 2013;2:215-228. doi: 10.1159/000356507
The hormones thyroxine (T4) and triiodothyronine (T3), produced by the thyroid gland, are crucial for maintaining metabolism. A deficit of thyroid hormone production—hypothyroidism—is a common endocrine disorder seen in primary care.
Although the diagnosis and management of hypothyroidism are considered straightforward, many patients with hypothyroidism do not achieve optimal treatment goals or see an improvement in their quality of life. In this article, we address the questionable utility of screening; outline the diagnostic approach, including the central role of laboratory testing; and explain why treatment requires a precise approach to address the range of patient types.
Epidemiology and classification
Estimates are that almost 5% of Americans 12 years or older have hypothyroidism; older people and women are more likely to develop the condition. 1 In the US National Health and Nutrition Examination Survey (NHANES III) of 13,344 people without known thyroid disease or a family history, hypothyroidism was found in 4.6% (overt [clinical] in 0.3% and subclinical in 4.3%); 11% had a high serum thyroid peroxidase antibody level, which increases their risk of hypothyroidism, and is treated the same as hypothyroidism of other causes; and, overall, lower serum thyroid-stimulating hormone (TSH) levels were seen in Blacks, compared to Whites and Mexican Americans.1
Primary hypothyroidism accounts for > 95% of cases of hypothyroidism, representing a failure of the thyroid gland to produce sufficient hormone. It has been shown that, in iodine-replete countries such as the United States, the prevalence of spontaneous hypothyroidism is 1% to 2%, and it is 10 times more common in women.2,3
Central hypothyroidism is caused by insufficient stimulation of the thyroid gland by TSH, due to pituitary (secondary hypothyroidism) or hypothalamic (tertiary hypothyroidism) disease and is estimated to occur in 1 in every 20,000 to 80,000 people in the general population.4
How does hypothyroidism manifest?
Signs and symptoms. Manifestations of hypothyroidism range from life-threatening to minimal or no clinical signs and symptoms (TABLE W1). Signs and symptoms of low thyroid function vary by the degree of hypothyroidism at presentation.
Common signs and symptoms of low thyroid function include fatigue, weight gain, dry skin, brittle hair, hair loss, morning stiffness, muscle aches, joint pain, cold intolerance, diffuse headache, constipation, difficulty concentrating, low libido, depression, and menstrual irregularities. On physical examination, a patient might present with bradycardia, hypotension, hypothermia with slow speech or movement, coarse facial appearance, goiter, diffuse hair loss, cold hands and feet, and a prolonged Achilles tendon reflex.5 Skin findings, such as keratosis pilaris, palmoplantar keratoderma (thickening of the skin), and pityriasis rubra pilar, can be associated with autoimmune hypothyroidism.6,7
Continue to: Carpal tunnel syndrome...
Carpal tunnel syndrome, plantar fasciitis, infertility or miscarriage, dyspepsia, and small intestinal bacterial overgrowth can be associated with hypothyroidism; thyroid function should therefore be assessed in patients who have any of these conditions, along with other signs and symptoms of low thyroid function.8,9 A patient with severe hypothyroidism might present with hemodynamic instability, pericardial or pleural effusion, and myxedema coma.10
Clues in the history and from the lab. A history of radiation to the head, neck, or chest area and a history or family history of autoimmune disorders are risk factors for autoimmune thyroid disease.11,12 Laboratory findings can include markers of oxidative stress, such as elevated levels of low-density lipoprotein cholesterol and serum malondialdehyde.13-15
Screening and diagnosis
Screening. The US Preventive Services Task Force has asserted that evidence is insufficient by which to evaluate the benefits and risks of routine screening for thyroid dysfunction in nonpregnant, asymptomatic adults.16 According to the American Thyroid Association and the American Association of Clinical Endocrinologists, screening should be considered in high-risk patients, including those who take medication that affects thyroid function or the results of thyroid hormone assays (TABLE W2, available at mdedge.com/familymedicine).17-20
Screening inpatients is challenging and usually not recommended unless thyroid disease is strongly suspected. This is because changes in the levels of thyroid hormones, binding proteins, and the TSH concentration can occur in severe nonthyroidal illness; in addition, assay interference by antibodies and other substances can affect thyroid hormone measurement.21
Testing strategy. Generally, screening and diagnosis of hypothyroidism are based primarily on laboratory testing, because signs and symptoms are nonspecific (FIGURE 15). A serum TSH level is usually the initial test when screening for thyroid dysfunction. A normal serum TSH value ranges from 0.5-5.0 mIU/L.
When an abnormal serum TSH level is found, further tests can be performed to investigate, including a serum free thyroxine (FT4) test. (Our preference is to order TSH and FT4 assays simultaneously to facilitate and confirm the diagnosis.) An FT4 test measures the amount of unattached, or free, thyroxine in blood by immunoassay. A normal FT4 value usually ranges from 0.7-1.9 ng/dL.
The combination of a high TSH level and a low FT4 level could be an indication of an underactive thyroid gland (ie, clinical or overt hypothyroidism). Milder, subclinical hypothyroidism is characterized by a higher-than-normal TSH level but a normal FT4 level.22 Central (secondary) hypothyroidism is characterized by a low serum FT4 level and a serum TSH level that can be below the reference range, low normal, or even slightly high.4
Continue to: These measurements...
These measurements must be interpreted within the context of the laboratory-specific normal range for each test. Third-generation serum TSH assays are more sensitive and specific than serum FT4 measurements for hypothyroidism. FT4 is usually measured by automated analogue immunoassay, which generally provides reliable results; abnormal binding proteins or other interferences occur in some patients, however, resulting in reporting of a falsely high, or falsely low, FT4 level. In such cases, FT4 by direct dialysis, or total T4, can be measured for further evaluation. In primary care, you are most likely to encounter primary hypothyroidism; secondary (central) hypothyroidism is much rarer (< 5% of cases).4
The ins and outs of treatment
For most patients, hypothyroidism is a permanent disorder requiring lifelong thyroid hormone replacement therapy—unless the disease is transient (ie, painless or subacute thyroiditis); reversible, because it is caused by medication; or responsive to medical intervention that addresses the underlying autoimmune condition.19 Goals of treatment (Figure 25,23) are to:
- normalize the TSH level to 0.5-5.0 mIU/L (the main goal), with an age-related shift toward a higher TSH goal in older patients (and an upper limit of normal of 7.5 mIU/L in patients who are ≥ 80 years of age)20
- restore the euthyroid state
- relieve symptoms
- reduce any goiter
- avoid overtreatment (iatrogenic thyrotoxicosis).
Desiccated thyroid extract (DTE), developed in the late 1880s and made from the dried thyroid gland of pigs, sheep, or cows, was the earliest treatment for hypothyroidism. The use of DTE has declined since the introduction of synthetic thyroxine (T4, or levothyroxine [here, referred to as LT4]), which is now the standard treatment.20-22 LT4 is deiodinated in peripheral tissues to form T3, the active thyroid hormone; this process accounts for 80% of total T3 production daily.24
LT4 formulations. LT4 is commercially available in tablet, soft-gel, and liquid preparations. Most patients are treated with the tablet; the soft-gel capsule or liquid is an option for patients who absorb the tablet poorly (because of atrophic gastritis, celiac disease, or gluten sensitivity or because they are post bariatric surgery). Increasing the dosage of the tablet form of LT4, with ongoing TSH monitoring, is more cost effective than moving to an alternative preparation.
If a switch of LT4 formulation is made (ie, from one manufacturer to another), test the serum TSH level to ensure that the therapeutic goal is being reached. Also, in our experience, it is best to prescribe a brand-name preparation of levothyroxine, not a generic, whenever possible, due to the variability in generic formulations and the potential presence of other (inert) ingredients.25
Dosing (TABLE 320,23). The average full replacement dosage of LT4 for a young, healthy adult is approximately 1.6 mcg/kg/d. Older patients (> 65 years) or those with coronary artery disease (CAD) should be started on a lower dosage (25-50 mcg/d) and titrated to goal accordingly.
LT4 (tablets, soft-gel capsules, or liquid) should be administered on an empty stomach, with water only, 30 to 60 minutes before breakfast. Medications that interfere with LT4 absorption (eg, bile acid resins, calcium carbonate, ferrous sulfate) should be taken several hours after LT4. For patients who cannot take LT4 in the morning, taking it at bedtime (≥ 2-3 hours after the last meal) is acceptable.
Continue to: Monitoring and titrating
Monitoring and titrating. Hypothyroid symptoms usually improve after 2 or 3 weeks of LT4 treatment; in severe hypothyroidism, complete recovery might take months. Approximately 6 weeks after LT4 therapy is initiated, serum TSH should be measured. After assessing whether administration of LT4 at the starting dosage is appropriate, that dosage can be increased, or decreased, every 4 to 6 weeks until the TSH goal is reached. Once the patient is maintained at a given dosage, measure serum TSH once a year—more often if there is an abnormal result or a change in the patient’s health status.23
Adverse effects of LT4 therapy are rare, unless over-replacement occurs. Rarely, patients have an allergy to the dye or an excipient (filler) in the tablet.26-28 The white, 50-mcg tablets can be given safely to patients with dye sensitivity. For those who have an allergy to an excipient (except gelatin) or gluten intolerance, the LT4 soft-gel capsule or liquid preparation (Tirosint) can be prescribed.
Pure LT4, in a capsule made from vegetable sources, can be ordered through a compounding pharmacy for patients who are allergic to animal products.
Anemia, especially iron-deficiency anemia, can cause intolerance to LT4 therapy; in such patients, lowering the starting dosage and treating anemia are indicated.29
Persistent symptoms (despite a normal TSH level). Because many hypothyroid symptoms are nonspecific, patients might come to think that their LT4 dosage is inadequate if they feel tired or gain weight. Persistent hypothyroid symptoms despite a normal serum TSH level might be due to (1) the inability of LT4 therapy to restore tissue thyroid hormone levels to normal or (2) other variables unrelated to hypothyroidism, including disorders associated with inflammation or autoimmune disease, certain medications, diet, lifestyle, and environmental toxins.
These patients might benefit from a detailed history to identify other causes and a switch to either LT4 + liothyronine (LT3; synthetic T3) combination therapy or DTE26,30-33 (TABLE 434), although a beneficial effect of LT4 + LT3 therapy was not seen in several studies.35,36 Over-replacement with LT4 should be discouraged, due to concerns about thyrotoxicosis and its complications (eg, atrial fibrillation, accelerated bone loss).
DTE and LT4 + LT3. Use of DTE has decreased since the 1970s, when LT4 became the therapy of choice. Subsequently, anecdotal evidence emerged that some patients did not feel well on LT4 and preferred to return to DTE.32,33
Continue to: Several clinical trials...
Several clinical trials addressed the question of whether residual symptoms could be resolved through LT4 + LT3 combination therapy31-39 (TABLE 434), but evidence of any consistent superiority of combination therapy was not demonstrated.35-39 In selected cases, patients might prefer the combination approach.31,33,39 The quality of life of hypothyroid patients was found to be similarly improved with LT4 or DTE, but the latter was associated with modest weight loss (approximately 4 lbs); nearly 50% of study patients preferred treatment with DTE over LT4.33 A follow-up study did not confirm weight loss with DTE, however.34
When LT4 monotherapy and LT4 + LT3 combination therapy were compared, results were mixed31-39; responsiveness to therapy containing LT3 might therefore depend on multiple variables, including genetic background, nutritional and lifestyle factors, stress, presence of comorbidities and autoimmune disorders, and other unidentified or poorly defined variables.40-48
Although combination therapy and DTE are not generally recommended over LT4 monotherapy, they might offer better options for patients who are still symptomatic when being treated with LT4 only: In a randomized, double-blind, crossover study that compared LT4 with DTE and with LT4 + LT3, one-third of the most highly symptomatic patients who had low scores on mood, cognitive, and quality-of-life assessments improved significantly after they were switched to combination therapy or DTE.34
The 2014 American Thyroid Association guidelines24 do not support routine use of LT4 + LT3 in hypothyroid patients who have residual symptoms after LT4 monotherapy; however, a therapeutic trial of LT4 + LT3, while maintaining a normal serum TSH, is reasonable in selected patients. Candidates for DTE or LT4 + LT3 might include patients who do not feel well on LT4 monotherapy, are post thyroidectomy or post radioiodine therapy, or have a low serum T3 level. DTE and combination therapy are discouraged in older patients, patients who have underlying CAD, and pregnant patients.
Special treatment circumstances
A number of patient variables have the potential to alter management strategies for hypothyroidism.18,20,23,40,49-53
Age, comorbidity. Older patients (> 65 years) and patients with cardiopulmonary disease or CAD should be treated with LT4, 25 to 50 mcg/d, initially; that dosage can be titrated upward by 12.5 to 25 mcg/d every 4 to 6 weeks until the TSH goal is reached—preferably, in the range of 4 to 8 mIU/L. An increase in the dosage of LT4 might be required in the presence of malabsorption (eg, gastrointestinal disorders, celiac disease) and in nephrotic syndrome.18,20,23
Body weight. A decrease in the dosage of LT4 might be indicated in the setting of significant weight loss (> 10% body weight).23
Continue to: Co-pharmacy
Co-pharmacy. An increase in the dosage of LT4 might be required when other drugs (eg, phenytoin, phenobarbital, rifampin, and carbamazepine) have led to an increased rate of thyroid hormone metabolism. A decrease in the dosage of LT4 might be necessary after initiation of androgen therapy.23
Pregnancy. Women with pre-existing hypothyroidism require an increase of 25% to 50% in their LT4 dosage during pregnancy to maintain a TSH level in the recommended pregnancy reference range. Thyroid function should be monitored every 4 to 6 weeks to ensure that the TSH target for each trimester is reached (first trimester, 0.1-4 mIU/L; second trimester, 0.2-4 mIU/L; third trimester, 0.3-4 mIU/L). Postpartum, LT4 can be reduced to the prepartum dosage; TSH should be checked every 4 to 6 weeks to maintain the TSH goal.23
Estrogen therapy. Hypothyroid women who are receiving estrogen therapy might require an increase in their LT4 dosage because serum thyroxine-binding globulin levels are increased by estrogens or through other mechanisms that have not been identified.23
Surgical candidacy. Observational studies show few adverse outcomes in surgical patients with mild (subclinical) hypothyroidism or moderate hypothyroidism; however, the risk of adverse surgical outcome might be increased in patients with severe hypothyroidism. For patients in whom surgery is planned and who have:
- subclinical hypothyroidism (elevated TSH and normal FT4), we recommend that surgery—urgent or elective—not be posptoned but proceed.
- moderate (overt) hypothyroidism who require urgent surgery, we recommend not postponing surgery, even though minor perioperative complications might develop. Such patients should be treated with LT4 as soon as the diagnosis for which surgery is required has been made. Alternatively, when moderate hypothyroidism is discovered in a patient who is being evaluated for elective surgery, we recommend postponing surgery until the euthyroid state is restored.
- severe hypothyroidism (myxedema coma [discussed in a bit]; severe clinical symptoms of chronic hypothyroidism, such as altered mental status, pericardial effusion, or heart failure; or a very low level of T4), surgery should be delayed until hypothyroidism has been treated. When emergency surgery is required for a severely hypothyroid patient, they should be treated with LT4 as soon as the diagnosis for which surgery is indicated has been made. When emergency surgery must be performed in a patient with myxedema coma, we recommend treatment with LT4 + LT3, rather than LT4 alone, often administered intravenously because LT4 is poorly absorbed in these patients.
Nonadherence. For patients who do not take LT4 regularly or do not respond to efforts to improve adherence, LT4 can be given weekly, instead of daily, although this interval is not ideal. Weekly dosing should not be used in older patients with CAD.23
Thyroid cancer. Patients who are post total thyroidectomy for thyroid cancer need to take LT4 to treat hypothyroidism and to prevent recurrence of thyroid cancer. The goal TSH level should be based on the cancer stage and risk of recurrence and should be monitored by an endocrinologist.
Myxedema coma. This medical emergency has high mortality. Myxedema coma occurs when severe hypothyroidism leads to any, or a combination, of the following: diminished mentation; hypothermia; bradycardia; hyponatremia; hypotension; cardiovascular, respiratory, and gastrointestinal dysfunction; and renal insufficiency. LT4, LT3, and glucocorticoids should be administered intravenously and the patient monitored closely—preferably in consultation with an endocrinologist.
Continue to: When to seek consultation
When to seek consultation
A patient with hypothyroidism should be referred to Endocrinology if they are < 18 years of age, pregnant, unresponsive to therapy, or have cardiac disease, coexisting endocrine disease, suspected myxedema coma, goiter or thyroid nodules, or a structural thyroid abnormality.
What we know about nutrition and hypothyroidism
Although it is commonly recognized that iodine is essential for production of thyroid hormone, other nutritional factors might contribute to proper production of thyroid hormones, including:
- adequate intake of iron, tyrosine, selenium, zinc, and vitamins E, B2, B3, B6, C, and D44,45
- selenium and zinc, which increase conversion of T4 to T3 and might be important in the management of hypothyroid patients40,46
- vitamin A, zinc, and regular exercise, which have been shown to improve cellular sensitivity to thyroid hormones.
Low iron stores can contribute to persistent symptoms and poor quality of life in patients with hypothyroidism, despite their being treated according to guidelines.29,47
Despite what is known about these nutritional connections, there is insufficient evidence that improving nutrition can reverse hypothyroidism.
Prevention
Prevention of hypothyroidism should take into account variables that affect or inhibit thyroid function, such as stress, infection (eg, Epstein-Barr virus), excessive fluoride intake, toxins (eg, pesticides, solvents, mercury, cadmium, and lead), autoimmune disease (eg, celiac disease), and food sensitivity.54,55 Oxidative stress can also cause thyroid impairment.40-48,54-58
Otherwise, there are, at present, no effective strategies for preventing thyroid disorders.
Subclinical hypothyroidism: Elusive management target
Subclinical hypothyroidism is defined as a normal serum FT4 level in the presence of an elevated serum TSH level. The prevalence of subclinical hypothyroidism varies from 3% to 15%, depending on the population studied; a higher incidence has been noted in women and older people.59 In the NHANES III,1 which excluded people with previously diagnosed thyroid disease, the incidence of subclinical hypothyroidism was 4.3%.
Continue to: Causes of subclinical...
Causes of subclinical hypothyroidism are the same as those of overt hypothyroidism, and include Hashimoto disease. The combination of an elevated TSH level and a normal FT4 level is associated with disorders characterized by protein-binding variations (eg, pregnancy, genetic disorders, drugs), TSH-secreting pituitary adenoma, class II and III obesity (respectively, body mass index, ≥ 35 but < 40 and ≥ 40), and assay variability.49,51
Lab diagnosis: Fraught with difficulty
The serum TSH level and either the total T4 level or the FT4 level should be measured to make a diagnosis of subclinical hypothyroidism. Most laboratories use a 1-step analogue immunoassay to determine free thyroid hormones; protein-binding variations can thus affect measurement of FT4.
Several scenarios that can result in inaccurate measurement of FT4 by radioimmunoassay include genetic disorders that affect binding proteins; pregnancy; use of certain drugs, including heparin, furosemide, antiepileptic agents, salicylate, ferrous sulfate, and cholesterol-binding resins; and some medical conditions, including cardiac surgery, critical illness, and renal failure. Variables that inhibit proper production of thyroid hormones—stress, infection, fluoride (an iodine antagonist), toxins (pesticides, mercury, cadmium, lead) and autoimmune conditions, such as celiac disease—should be considered when attempting to determine the cause of subclinical hypothyroidism.
Liquid chromatography–mass spectrometry measurement of thyroid hormones might be more accurate than immunoassay.53 Measuring serum total T4 and FT4 by dialysis, free from interfering proteins, might also be useful when measurement of FT4 by immunoassay is affected by binding-protein variations.
Features of subclinical hypothyroidism
Most patients who have subclinical hypothyroidism and a serum TSH level < 10 mIU/L are asymptomatic. Some might have nonspecific symptoms of hypothyroidism, however, such as reduced quality of life, poor cognitive function, and poor memory—symptoms that do not typically correlate with the serum TSH level.
It has been suggested that some elderly people normally have a higher level of serum TSH, and that they might have even a prolonged lifespan.51 Additionally, it has been shown that, in nonpregnant adult patients with subclinical hypothyroidism and a serum TSH level of 4.5 to 10 mIU/L, treatment with LT4 was not associated with improvement in thyroid-related symptoms or general quality of life.52
Treat, or don't treat, subclinical hypothyroidism?
It is well accepted that the goal of therapy in hypothyroid patients is to normalize the serum TSH level; however, the American Thyroid Association and the American Association of Clinical Endocrinology recommend starting LT4 in patients with a serum TSH level ≥ 10 mIU/L (TABLE 5).59-62 The principal reason for not treating subclinical hypothyroidism is the lack of benefit in reducing the risk of cardiovascular morbidity and mortality when the TSH level is between 7.5 and 10 mIU/L.62
Continue to: Routine treatment
Routine treatment of patients with a serum TSH level of 4.5 to 10 mIU/L remains controversial. When TSH is 7.0 to 9.9 mIU/L, treatment is recommended for (1) patients < 65 years and (2) for older patients (> 65 years) only when there are convincing hypothyroid symptoms because of concern about unintended overtreatment.
When the TSH level is anywhere above the upper limit of normal to 6.9 mIU/L, treatment is recommended for patients < 65 years old, patients who have a high titer of thyroid peroxidase antibodies, and patients with goiter—but not for patients > 65 years (and, especially, not for octogenarians) because their upper limit of normal could be as high as 6 to 8 mIU/L, especially if they are otherwise healthy.
Treatment should be considered for women with subclinical hypothyroidism who are trying to conceive or experiencing an infertility problem.
For patients with subclinical hypothyroidism who are not being treated, monitor thyroid function every 6 to 12 months by testing TSH and FT4.
CORRESPONDENCE
Thanh D. Hoang, DO, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; thanh.d.hoang.mil@mail.mil
The hormones thyroxine (T4) and triiodothyronine (T3), produced by the thyroid gland, are crucial for maintaining metabolism. A deficit of thyroid hormone production—hypothyroidism—is a common endocrine disorder seen in primary care.
Although the diagnosis and management of hypothyroidism are considered straightforward, many patients with hypothyroidism do not achieve optimal treatment goals or see an improvement in their quality of life. In this article, we address the questionable utility of screening; outline the diagnostic approach, including the central role of laboratory testing; and explain why treatment requires a precise approach to address the range of patient types.
Epidemiology and classification
Estimates are that almost 5% of Americans 12 years or older have hypothyroidism; older people and women are more likely to develop the condition. 1 In the US National Health and Nutrition Examination Survey (NHANES III) of 13,344 people without known thyroid disease or a family history, hypothyroidism was found in 4.6% (overt [clinical] in 0.3% and subclinical in 4.3%); 11% had a high serum thyroid peroxidase antibody level, which increases their risk of hypothyroidism, and is treated the same as hypothyroidism of other causes; and, overall, lower serum thyroid-stimulating hormone (TSH) levels were seen in Blacks, compared to Whites and Mexican Americans.1
Primary hypothyroidism accounts for > 95% of cases of hypothyroidism, representing a failure of the thyroid gland to produce sufficient hormone. It has been shown that, in iodine-replete countries such as the United States, the prevalence of spontaneous hypothyroidism is 1% to 2%, and it is 10 times more common in women.2,3
Central hypothyroidism is caused by insufficient stimulation of the thyroid gland by TSH, due to pituitary (secondary hypothyroidism) or hypothalamic (tertiary hypothyroidism) disease and is estimated to occur in 1 in every 20,000 to 80,000 people in the general population.4
How does hypothyroidism manifest?
Signs and symptoms. Manifestations of hypothyroidism range from life-threatening to minimal or no clinical signs and symptoms (TABLE W1). Signs and symptoms of low thyroid function vary by the degree of hypothyroidism at presentation.
Common signs and symptoms of low thyroid function include fatigue, weight gain, dry skin, brittle hair, hair loss, morning stiffness, muscle aches, joint pain, cold intolerance, diffuse headache, constipation, difficulty concentrating, low libido, depression, and menstrual irregularities. On physical examination, a patient might present with bradycardia, hypotension, hypothermia with slow speech or movement, coarse facial appearance, goiter, diffuse hair loss, cold hands and feet, and a prolonged Achilles tendon reflex.5 Skin findings, such as keratosis pilaris, palmoplantar keratoderma (thickening of the skin), and pityriasis rubra pilar, can be associated with autoimmune hypothyroidism.6,7
Continue to: Carpal tunnel syndrome...
Carpal tunnel syndrome, plantar fasciitis, infertility or miscarriage, dyspepsia, and small intestinal bacterial overgrowth can be associated with hypothyroidism; thyroid function should therefore be assessed in patients who have any of these conditions, along with other signs and symptoms of low thyroid function.8,9 A patient with severe hypothyroidism might present with hemodynamic instability, pericardial or pleural effusion, and myxedema coma.10
Clues in the history and from the lab. A history of radiation to the head, neck, or chest area and a history or family history of autoimmune disorders are risk factors for autoimmune thyroid disease.11,12 Laboratory findings can include markers of oxidative stress, such as elevated levels of low-density lipoprotein cholesterol and serum malondialdehyde.13-15
Screening and diagnosis
Screening. The US Preventive Services Task Force has asserted that evidence is insufficient by which to evaluate the benefits and risks of routine screening for thyroid dysfunction in nonpregnant, asymptomatic adults.16 According to the American Thyroid Association and the American Association of Clinical Endocrinologists, screening should be considered in high-risk patients, including those who take medication that affects thyroid function or the results of thyroid hormone assays (TABLE W2, available at mdedge.com/familymedicine).17-20
Screening inpatients is challenging and usually not recommended unless thyroid disease is strongly suspected. This is because changes in the levels of thyroid hormones, binding proteins, and the TSH concentration can occur in severe nonthyroidal illness; in addition, assay interference by antibodies and other substances can affect thyroid hormone measurement.21
Testing strategy. Generally, screening and diagnosis of hypothyroidism are based primarily on laboratory testing, because signs and symptoms are nonspecific (FIGURE 15). A serum TSH level is usually the initial test when screening for thyroid dysfunction. A normal serum TSH value ranges from 0.5-5.0 mIU/L.
When an abnormal serum TSH level is found, further tests can be performed to investigate, including a serum free thyroxine (FT4) test. (Our preference is to order TSH and FT4 assays simultaneously to facilitate and confirm the diagnosis.) An FT4 test measures the amount of unattached, or free, thyroxine in blood by immunoassay. A normal FT4 value usually ranges from 0.7-1.9 ng/dL.
The combination of a high TSH level and a low FT4 level could be an indication of an underactive thyroid gland (ie, clinical or overt hypothyroidism). Milder, subclinical hypothyroidism is characterized by a higher-than-normal TSH level but a normal FT4 level.22 Central (secondary) hypothyroidism is characterized by a low serum FT4 level and a serum TSH level that can be below the reference range, low normal, or even slightly high.4
Continue to: These measurements...
These measurements must be interpreted within the context of the laboratory-specific normal range for each test. Third-generation serum TSH assays are more sensitive and specific than serum FT4 measurements for hypothyroidism. FT4 is usually measured by automated analogue immunoassay, which generally provides reliable results; abnormal binding proteins or other interferences occur in some patients, however, resulting in reporting of a falsely high, or falsely low, FT4 level. In such cases, FT4 by direct dialysis, or total T4, can be measured for further evaluation. In primary care, you are most likely to encounter primary hypothyroidism; secondary (central) hypothyroidism is much rarer (< 5% of cases).4
The ins and outs of treatment
For most patients, hypothyroidism is a permanent disorder requiring lifelong thyroid hormone replacement therapy—unless the disease is transient (ie, painless or subacute thyroiditis); reversible, because it is caused by medication; or responsive to medical intervention that addresses the underlying autoimmune condition.19 Goals of treatment (Figure 25,23) are to:
- normalize the TSH level to 0.5-5.0 mIU/L (the main goal), with an age-related shift toward a higher TSH goal in older patients (and an upper limit of normal of 7.5 mIU/L in patients who are ≥ 80 years of age)20
- restore the euthyroid state
- relieve symptoms
- reduce any goiter
- avoid overtreatment (iatrogenic thyrotoxicosis).
Desiccated thyroid extract (DTE), developed in the late 1880s and made from the dried thyroid gland of pigs, sheep, or cows, was the earliest treatment for hypothyroidism. The use of DTE has declined since the introduction of synthetic thyroxine (T4, or levothyroxine [here, referred to as LT4]), which is now the standard treatment.20-22 LT4 is deiodinated in peripheral tissues to form T3, the active thyroid hormone; this process accounts for 80% of total T3 production daily.24
LT4 formulations. LT4 is commercially available in tablet, soft-gel, and liquid preparations. Most patients are treated with the tablet; the soft-gel capsule or liquid is an option for patients who absorb the tablet poorly (because of atrophic gastritis, celiac disease, or gluten sensitivity or because they are post bariatric surgery). Increasing the dosage of the tablet form of LT4, with ongoing TSH monitoring, is more cost effective than moving to an alternative preparation.
If a switch of LT4 formulation is made (ie, from one manufacturer to another), test the serum TSH level to ensure that the therapeutic goal is being reached. Also, in our experience, it is best to prescribe a brand-name preparation of levothyroxine, not a generic, whenever possible, due to the variability in generic formulations and the potential presence of other (inert) ingredients.25
Dosing (TABLE 320,23). The average full replacement dosage of LT4 for a young, healthy adult is approximately 1.6 mcg/kg/d. Older patients (> 65 years) or those with coronary artery disease (CAD) should be started on a lower dosage (25-50 mcg/d) and titrated to goal accordingly.
LT4 (tablets, soft-gel capsules, or liquid) should be administered on an empty stomach, with water only, 30 to 60 minutes before breakfast. Medications that interfere with LT4 absorption (eg, bile acid resins, calcium carbonate, ferrous sulfate) should be taken several hours after LT4. For patients who cannot take LT4 in the morning, taking it at bedtime (≥ 2-3 hours after the last meal) is acceptable.
Continue to: Monitoring and titrating
Monitoring and titrating. Hypothyroid symptoms usually improve after 2 or 3 weeks of LT4 treatment; in severe hypothyroidism, complete recovery might take months. Approximately 6 weeks after LT4 therapy is initiated, serum TSH should be measured. After assessing whether administration of LT4 at the starting dosage is appropriate, that dosage can be increased, or decreased, every 4 to 6 weeks until the TSH goal is reached. Once the patient is maintained at a given dosage, measure serum TSH once a year—more often if there is an abnormal result or a change in the patient’s health status.23
Adverse effects of LT4 therapy are rare, unless over-replacement occurs. Rarely, patients have an allergy to the dye or an excipient (filler) in the tablet.26-28 The white, 50-mcg tablets can be given safely to patients with dye sensitivity. For those who have an allergy to an excipient (except gelatin) or gluten intolerance, the LT4 soft-gel capsule or liquid preparation (Tirosint) can be prescribed.
Pure LT4, in a capsule made from vegetable sources, can be ordered through a compounding pharmacy for patients who are allergic to animal products.
Anemia, especially iron-deficiency anemia, can cause intolerance to LT4 therapy; in such patients, lowering the starting dosage and treating anemia are indicated.29
Persistent symptoms (despite a normal TSH level). Because many hypothyroid symptoms are nonspecific, patients might come to think that their LT4 dosage is inadequate if they feel tired or gain weight. Persistent hypothyroid symptoms despite a normal serum TSH level might be due to (1) the inability of LT4 therapy to restore tissue thyroid hormone levels to normal or (2) other variables unrelated to hypothyroidism, including disorders associated with inflammation or autoimmune disease, certain medications, diet, lifestyle, and environmental toxins.
These patients might benefit from a detailed history to identify other causes and a switch to either LT4 + liothyronine (LT3; synthetic T3) combination therapy or DTE26,30-33 (TABLE 434), although a beneficial effect of LT4 + LT3 therapy was not seen in several studies.35,36 Over-replacement with LT4 should be discouraged, due to concerns about thyrotoxicosis and its complications (eg, atrial fibrillation, accelerated bone loss).
DTE and LT4 + LT3. Use of DTE has decreased since the 1970s, when LT4 became the therapy of choice. Subsequently, anecdotal evidence emerged that some patients did not feel well on LT4 and preferred to return to DTE.32,33
Continue to: Several clinical trials...
Several clinical trials addressed the question of whether residual symptoms could be resolved through LT4 + LT3 combination therapy31-39 (TABLE 434), but evidence of any consistent superiority of combination therapy was not demonstrated.35-39 In selected cases, patients might prefer the combination approach.31,33,39 The quality of life of hypothyroid patients was found to be similarly improved with LT4 or DTE, but the latter was associated with modest weight loss (approximately 4 lbs); nearly 50% of study patients preferred treatment with DTE over LT4.33 A follow-up study did not confirm weight loss with DTE, however.34
When LT4 monotherapy and LT4 + LT3 combination therapy were compared, results were mixed31-39; responsiveness to therapy containing LT3 might therefore depend on multiple variables, including genetic background, nutritional and lifestyle factors, stress, presence of comorbidities and autoimmune disorders, and other unidentified or poorly defined variables.40-48
Although combination therapy and DTE are not generally recommended over LT4 monotherapy, they might offer better options for patients who are still symptomatic when being treated with LT4 only: In a randomized, double-blind, crossover study that compared LT4 with DTE and with LT4 + LT3, one-third of the most highly symptomatic patients who had low scores on mood, cognitive, and quality-of-life assessments improved significantly after they were switched to combination therapy or DTE.34
The 2014 American Thyroid Association guidelines24 do not support routine use of LT4 + LT3 in hypothyroid patients who have residual symptoms after LT4 monotherapy; however, a therapeutic trial of LT4 + LT3, while maintaining a normal serum TSH, is reasonable in selected patients. Candidates for DTE or LT4 + LT3 might include patients who do not feel well on LT4 monotherapy, are post thyroidectomy or post radioiodine therapy, or have a low serum T3 level. DTE and combination therapy are discouraged in older patients, patients who have underlying CAD, and pregnant patients.
Special treatment circumstances
A number of patient variables have the potential to alter management strategies for hypothyroidism.18,20,23,40,49-53
Age, comorbidity. Older patients (> 65 years) and patients with cardiopulmonary disease or CAD should be treated with LT4, 25 to 50 mcg/d, initially; that dosage can be titrated upward by 12.5 to 25 mcg/d every 4 to 6 weeks until the TSH goal is reached—preferably, in the range of 4 to 8 mIU/L. An increase in the dosage of LT4 might be required in the presence of malabsorption (eg, gastrointestinal disorders, celiac disease) and in nephrotic syndrome.18,20,23
Body weight. A decrease in the dosage of LT4 might be indicated in the setting of significant weight loss (> 10% body weight).23
Continue to: Co-pharmacy
Co-pharmacy. An increase in the dosage of LT4 might be required when other drugs (eg, phenytoin, phenobarbital, rifampin, and carbamazepine) have led to an increased rate of thyroid hormone metabolism. A decrease in the dosage of LT4 might be necessary after initiation of androgen therapy.23
Pregnancy. Women with pre-existing hypothyroidism require an increase of 25% to 50% in their LT4 dosage during pregnancy to maintain a TSH level in the recommended pregnancy reference range. Thyroid function should be monitored every 4 to 6 weeks to ensure that the TSH target for each trimester is reached (first trimester, 0.1-4 mIU/L; second trimester, 0.2-4 mIU/L; third trimester, 0.3-4 mIU/L). Postpartum, LT4 can be reduced to the prepartum dosage; TSH should be checked every 4 to 6 weeks to maintain the TSH goal.23
Estrogen therapy. Hypothyroid women who are receiving estrogen therapy might require an increase in their LT4 dosage because serum thyroxine-binding globulin levels are increased by estrogens or through other mechanisms that have not been identified.23
Surgical candidacy. Observational studies show few adverse outcomes in surgical patients with mild (subclinical) hypothyroidism or moderate hypothyroidism; however, the risk of adverse surgical outcome might be increased in patients with severe hypothyroidism. For patients in whom surgery is planned and who have:
- subclinical hypothyroidism (elevated TSH and normal FT4), we recommend that surgery—urgent or elective—not be posptoned but proceed.
- moderate (overt) hypothyroidism who require urgent surgery, we recommend not postponing surgery, even though minor perioperative complications might develop. Such patients should be treated with LT4 as soon as the diagnosis for which surgery is required has been made. Alternatively, when moderate hypothyroidism is discovered in a patient who is being evaluated for elective surgery, we recommend postponing surgery until the euthyroid state is restored.
- severe hypothyroidism (myxedema coma [discussed in a bit]; severe clinical symptoms of chronic hypothyroidism, such as altered mental status, pericardial effusion, or heart failure; or a very low level of T4), surgery should be delayed until hypothyroidism has been treated. When emergency surgery is required for a severely hypothyroid patient, they should be treated with LT4 as soon as the diagnosis for which surgery is indicated has been made. When emergency surgery must be performed in a patient with myxedema coma, we recommend treatment with LT4 + LT3, rather than LT4 alone, often administered intravenously because LT4 is poorly absorbed in these patients.
Nonadherence. For patients who do not take LT4 regularly or do not respond to efforts to improve adherence, LT4 can be given weekly, instead of daily, although this interval is not ideal. Weekly dosing should not be used in older patients with CAD.23
Thyroid cancer. Patients who are post total thyroidectomy for thyroid cancer need to take LT4 to treat hypothyroidism and to prevent recurrence of thyroid cancer. The goal TSH level should be based on the cancer stage and risk of recurrence and should be monitored by an endocrinologist.
Myxedema coma. This medical emergency has high mortality. Myxedema coma occurs when severe hypothyroidism leads to any, or a combination, of the following: diminished mentation; hypothermia; bradycardia; hyponatremia; hypotension; cardiovascular, respiratory, and gastrointestinal dysfunction; and renal insufficiency. LT4, LT3, and glucocorticoids should be administered intravenously and the patient monitored closely—preferably in consultation with an endocrinologist.
Continue to: When to seek consultation
When to seek consultation
A patient with hypothyroidism should be referred to Endocrinology if they are < 18 years of age, pregnant, unresponsive to therapy, or have cardiac disease, coexisting endocrine disease, suspected myxedema coma, goiter or thyroid nodules, or a structural thyroid abnormality.
What we know about nutrition and hypothyroidism
Although it is commonly recognized that iodine is essential for production of thyroid hormone, other nutritional factors might contribute to proper production of thyroid hormones, including:
- adequate intake of iron, tyrosine, selenium, zinc, and vitamins E, B2, B3, B6, C, and D44,45
- selenium and zinc, which increase conversion of T4 to T3 and might be important in the management of hypothyroid patients40,46
- vitamin A, zinc, and regular exercise, which have been shown to improve cellular sensitivity to thyroid hormones.
Low iron stores can contribute to persistent symptoms and poor quality of life in patients with hypothyroidism, despite their being treated according to guidelines.29,47
Despite what is known about these nutritional connections, there is insufficient evidence that improving nutrition can reverse hypothyroidism.
Prevention
Prevention of hypothyroidism should take into account variables that affect or inhibit thyroid function, such as stress, infection (eg, Epstein-Barr virus), excessive fluoride intake, toxins (eg, pesticides, solvents, mercury, cadmium, and lead), autoimmune disease (eg, celiac disease), and food sensitivity.54,55 Oxidative stress can also cause thyroid impairment.40-48,54-58
Otherwise, there are, at present, no effective strategies for preventing thyroid disorders.
Subclinical hypothyroidism: Elusive management target
Subclinical hypothyroidism is defined as a normal serum FT4 level in the presence of an elevated serum TSH level. The prevalence of subclinical hypothyroidism varies from 3% to 15%, depending on the population studied; a higher incidence has been noted in women and older people.59 In the NHANES III,1 which excluded people with previously diagnosed thyroid disease, the incidence of subclinical hypothyroidism was 4.3%.
Continue to: Causes of subclinical...
Causes of subclinical hypothyroidism are the same as those of overt hypothyroidism, and include Hashimoto disease. The combination of an elevated TSH level and a normal FT4 level is associated with disorders characterized by protein-binding variations (eg, pregnancy, genetic disorders, drugs), TSH-secreting pituitary adenoma, class II and III obesity (respectively, body mass index, ≥ 35 but < 40 and ≥ 40), and assay variability.49,51
Lab diagnosis: Fraught with difficulty
The serum TSH level and either the total T4 level or the FT4 level should be measured to make a diagnosis of subclinical hypothyroidism. Most laboratories use a 1-step analogue immunoassay to determine free thyroid hormones; protein-binding variations can thus affect measurement of FT4.
Several scenarios that can result in inaccurate measurement of FT4 by radioimmunoassay include genetic disorders that affect binding proteins; pregnancy; use of certain drugs, including heparin, furosemide, antiepileptic agents, salicylate, ferrous sulfate, and cholesterol-binding resins; and some medical conditions, including cardiac surgery, critical illness, and renal failure. Variables that inhibit proper production of thyroid hormones—stress, infection, fluoride (an iodine antagonist), toxins (pesticides, mercury, cadmium, lead) and autoimmune conditions, such as celiac disease—should be considered when attempting to determine the cause of subclinical hypothyroidism.
Liquid chromatography–mass spectrometry measurement of thyroid hormones might be more accurate than immunoassay.53 Measuring serum total T4 and FT4 by dialysis, free from interfering proteins, might also be useful when measurement of FT4 by immunoassay is affected by binding-protein variations.
Features of subclinical hypothyroidism
Most patients who have subclinical hypothyroidism and a serum TSH level < 10 mIU/L are asymptomatic. Some might have nonspecific symptoms of hypothyroidism, however, such as reduced quality of life, poor cognitive function, and poor memory—symptoms that do not typically correlate with the serum TSH level.
It has been suggested that some elderly people normally have a higher level of serum TSH, and that they might have even a prolonged lifespan.51 Additionally, it has been shown that, in nonpregnant adult patients with subclinical hypothyroidism and a serum TSH level of 4.5 to 10 mIU/L, treatment with LT4 was not associated with improvement in thyroid-related symptoms or general quality of life.52
Treat, or don't treat, subclinical hypothyroidism?
It is well accepted that the goal of therapy in hypothyroid patients is to normalize the serum TSH level; however, the American Thyroid Association and the American Association of Clinical Endocrinology recommend starting LT4 in patients with a serum TSH level ≥ 10 mIU/L (TABLE 5).59-62 The principal reason for not treating subclinical hypothyroidism is the lack of benefit in reducing the risk of cardiovascular morbidity and mortality when the TSH level is between 7.5 and 10 mIU/L.62
Continue to: Routine treatment
Routine treatment of patients with a serum TSH level of 4.5 to 10 mIU/L remains controversial. When TSH is 7.0 to 9.9 mIU/L, treatment is recommended for (1) patients < 65 years and (2) for older patients (> 65 years) only when there are convincing hypothyroid symptoms because of concern about unintended overtreatment.
When the TSH level is anywhere above the upper limit of normal to 6.9 mIU/L, treatment is recommended for patients < 65 years old, patients who have a high titer of thyroid peroxidase antibodies, and patients with goiter—but not for patients > 65 years (and, especially, not for octogenarians) because their upper limit of normal could be as high as 6 to 8 mIU/L, especially if they are otherwise healthy.
Treatment should be considered for women with subclinical hypothyroidism who are trying to conceive or experiencing an infertility problem.
For patients with subclinical hypothyroidism who are not being treated, monitor thyroid function every 6 to 12 months by testing TSH and FT4.
CORRESPONDENCE
Thanh D. Hoang, DO, Division of Endocrinology, Walter Reed National Military Medical Center, 8901 Wisconsin Avenue, Bethesda, MD 20889; thanh.d.hoang.mil@mail.mil
1. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499. doi: 10.1210/jcem.87.2.8182
2. Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99:39-51. doi: 10.1093/bmb/ldr030
3. Canaris GJ, Manowitz NR, Mayor G, et al. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160:526-534. doi: 10.1001/archinte.160.4.526
4. Persani L. Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab. 2012;97:3068-3078. doi: 10.1210/jc.2012-1616
5. Almandoz JP, Gharib H. Hypothyroidism: etiology, diagnosis, and management. Med Clin North Am. 2012;96:203-221. doi: 10.1016/j.mcna.2012.01.005
6. Ai J, Leonhardt JM, Heymann WR. Autoimmune thyroid diseases: etiology, pathogenesis and dermatologic manifestations. J Am Acad Dermatol. 2003;48:641-659. doi: 10.1067/mjd.2003.257
7. Franzotti AM, Avelar JCD, Cardoso TA, et al. Pityriasis rubra pilar and hypothyroidism. An Bras Dermatol. 2014;89:497-500. doi: 10.1590/abd1806-4841.20142994
8. Yaylali O, Kirac S, Yilmaz M, et al. Does hypothyroidism affect gastrointestinal motility? Gastroenterol Res Pract. 2009;2009:529802. doi: 10.1155/2009/529802
9. Patil AD. Link between hypothyroidism and small intestinal bacterial overgrowth. Indian J Endocrinol Metab. 2014;18:307-309.
10. Ono Y, Ono S, Yasunaga H, et al. Clinical characteristics and outcomes of myxedema coma: analysis of a national inpatient database in Japan. J Epidemiol. 2017;27:117-122. doi: 10.1016/j.je.2016.04.002
11. Boomsma MJ, Bijl HP, Langendijk JA. Radiation-induced hypothyroidism in head and neck cancer patients: a systematic review. Radiother Oncol. 2011;99:1-5. doi: 10.1016/j.radonc.2011.03.002
12. Boelaert K, Newby PR, Simmonds MJ, et al. Prevalence and relative risk of other autoimmune diseases in subjects with autoimmune thyroid disease. Am J Med. 2010;123:183.e1-e9. doi: 10.1016/j.amjmed.2009.06.030
13. Cheserek MJ, Wu G-R, Ntazinda A, et al. Association between thyroid hormones, lipids and oxidative stress markers in subclinical hypothyroidism. J Med Biochem. 2015;34:323-331. doi: 10.2478/jomb-2014-0044
14. Zha K, Zuo C, Wang A, et al. LDL in patients with subclinical hypothyroidism shows increased lipid peroxidation. Lipids Health Dis. 2015;14:95. doi: 10.1186/s12944-015-0092-4
15. Tejovathi B, Suchitra MM, Suresh V, et al. Association of lipid oxidation with endothelial dysfunction in patients with overt hypothyroidism. Exp Clin Endocrinol Diabetes. 2013;121:306-309. doi: 10.1055/s-0032-1333298
16. LeFevre ML; U.S. Preventive Services Task Force. Screening for thyroid dysfunction: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015;162:641-650. doi: 10.7326/M15-0483
17. Chaker L, Bianco AC, Jonklaas J, et al. Hypothyroidism. Lancet. 2017;390:1550-1562. doi: 10.1016/S0140-6736(17)30703-1
18. Vaidya B, Pearce SHS. Management of hypothyroidism in adults. BMJ. 2008;337:a801. doi: 10.1136/bmj.a801
19. Iyer PC, Cabanillas ME, Waguespack SG, et al. Immune-related thyroiditis with immune checkpoint inhibitors. Thyroid. 2018;28:1243-1251. doi: 10.1089/thy.2018.0116
20. Garber JR, Cobin RH, Gharib H, et al; American Association Of Clinical Endocrinologists And American Thyroid Association Taskforce On Hypothyroidism In Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid. 2012;22:1200-1235. doi: 10.1089/thy.2012.0205
21. Maiden MJ, Torpy DJ. Thyroid hormones in critical illness. Crit Care Clin. 2019;35:375-388. doi: 10.1016/j.ccc.2018.11.012
22. Peeters RP. Subclinical hypothyroidism. N Engl J Med. 2017;376:2556-2565. doi: 10.1056/NEJMcp1611144
23. Benvenga S, Carlé A. Levothyroxine formulations: pharmacological and clinical implications of generic substitution. Adv Ther. 2019;36(suppl 2):59-71. doi: 10.1007/s12325-019-01079-1
24. Jonklaas J, Bianco AC, Bauer AJ, et al; . Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association Task Force on Thyroid Hormone Replacement. Thyroid. 2014;24:1670-1751. doi: 10.1089/thy.2014.0028
25. Engler D, Burger AG. The deiodination of the iodothyronines and of their derivatives in man. Endocr Rev. 1984;5:151-184. doi: 10.1210/edrv-5-2-151
26. Ettleson MD, Bianco AC. Individualized therapy for hypothyroidism: is T4 enough for everyone? J Clin Endocrinol Metab. 2020;105:e3090-e3104. doi: 10.1210/clinem/dgaa430
27. Slayden TA, Shakir MKM, Hoang TD. A bull in a pill shop: alpha-gal allergy complicating treatment options for postprocedural hypothyroidism. AACE Clin Case Rep. 2020;6:e101-e104. doi: 10.4158/ACCR-2019-0495
28. Chamorro-Pareja N, Carrillo-Martin I, Haehn DA, et al. Self-reported allergy to thyroid replacement therapy: a multicenter retrospective chart review. Endocr Pract. 2020;26:761-767. doi: 10.4158/EP-2019-0488
29. Shakir MKM, Turton D, Aprill BS, et al. Anemia: a cause of intolerance to thyroxine sodium. Mayo Clin Proc. 2000;75:189-192.
30. Jonklaas J, Bianco AC, Cappola AR, et al. Evidence-based use of levothyroxine/liothyronine combinations in treating hypothyroidism: a consensus document. Thyroid. 2021;31:156-182. doi: 10.1089/thy.2020.0720
31. Appelhof BC, Fliers E, Wekking EM, et al. Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J Clin Endocrinol Metab. 2005;90:2666-2674. doi: 10.1210/jc.2004-2111
32. Escobar-Morreale HF, Botella-Carretero JI, M, et al. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann Intern Med. 2005;142:412-424. doi: 10.7326/0003-4819-142-6-200503150-00007
33. Hoang TD, Olsen CH, Mai VQ, et al. Desiccated thyroid extract compared with levothyroxine in the treatment of hypothyroidism: a randomized, double-blind, crossover study. J Clin Endocrinol Metab. 2013;98:1982-1990. doi: 10.1210/jc.2012-4107
34. Shakir MKM, Brooks DI, McAninch EA, et al. Comparative effectiveness of levothyroxine, desiccated thyroid extract, and levothyroxine+liothyronine in hypothyroidism. J Clin Endocrinol Metab. 2021;106:e4400-e4413. doi: 10.1210/clinem/dgab478
35. Valizadeh M, Seyyed-Majidi MR, Hajibeigloo H, et al. Efficacy of combined levothyroxine and liothyronine as compared with levothyroxine monotherapy in primary hypothyroidism: a randomized controlled trial. Endocr Res. 2009;34:80-89. doi: 10.1080/07435800903156340
36. Walsh JP, Shiels L, Lim EM, et al. Combined thyroxine/liothyronine treatment does not improve well-being, quality of life, or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J Clin Endocrinol Metab. 2003;88:4543-4550. doi: 10.1210/jc.2003-030249
37. Rodriguez T, Lavis VR, Meininger JC, et al. Substitution of liothyronine at a 1:5 ratio for a portion of levothyroxine: effect on fatigue, symptoms of depression, and working memory versus treatment with levothyroxine alone. Endocr Pract. 2005;11:223-233. doi: 10.4158/EP.11.4.223
38. Sawka AM, Gerstein HC, Marriott MJ, et al. Does a combination regimen of thyroxine (T4) and 3,5,3’-triiodothyronine improve depressive symptoms better than T4 alone in patients with hypothyroidism? Results of a double-blind, randomized, controlled trial. J Clin Endocrinol Metab. 2003;88:4551-4555. doi: 10.1210/jc.2003-030139
39. Clyde PW, Harari AE, Getka EJ, et al. Combined levothyroxine plus liothyronine compared with levothyroxine alone in primary hypothyroidism: a randomized controlled trial. JAMA. 2003;290:2952-2958. doi: 10.1001/jama.290.22.2952
40. Duntas LH. Selenium and the thyroid: a close-knit connection. J Clin Endocrinol Metab. 2010;95:5180-5188. doi: 10.1210/jc.2010-0191
41. Winther KH, Wichman JEM, Bonnema SJ, et al. Insufficient documentation for clinical efficacy of selenium supplementation in chronic autoimmune thyroiditis, based on a systematic review and meta-analysis. Endocrine. 2017;55:376-385. doi: 10.1007/s12020-016-1098-z
42. Parva NR, Tadepalli S, Singh P, et al. Prevalence of vitamin D deficiency and associated risk factors in the US population (2011-2012). Cureus. 2018;10:e2741. doi: 10.7759/cureus.2741
43. Wang J, Lv S, Chen G, et al. Meta-analysis of the association between vitamin D and autoimmune thyroid disease. Nutrients. 2015,7:2485-2498. doi: 10.3390/nu7042485
44. Wilson MM, Reedy J, Krebs-Smith SM. American diet quality: where it is, where it is heading, and what it could be. J Acad Nutr Diet. 2016;116:302-310.e1. doi: 10.1016/j.jand.2015.09.020
45. Babiker A, Alawi A, Al Atawi M, et al. The role of micronutrients in thyroid dysfunction. Sudan J Paediatr. 2020;20:13-19. doi: 10.24911/SJP.106-1587138942
46. Knezevic J, Starchl C, Tmava Berisha A, et al. Thyroid-gut-axis: How does the microbiota influence thyroid function? Nutrients. 2020;12:1769. doi: 10.3390/nu12061769
47. Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78:34-44. doi: 10.1017/S0029665118001192
48. Chakrabarti SK, Ghosh S, Banerjee S, et al. Oxidative stress in hypothyroid patients and the role of antioxidant supplementation. Indian J Endocrinol Metab. 2016;20:674-678. doi: 10.4103/2230-8210.190555
49. Tseng F-Y, Lin W-Y, Lin C-C, et al. Subclinical hypothyroidism is associated with increased risk for all-cause and cardiovascular mortality in adults. J Am Coll Cardiol. 2012;60:730-737. doi: 10.1016/j.jacc.2012.03.047
50. Roberts LM, Pattison H, Roalfe A, et al. Is subclinical thyroid dysfunction in the elderly associated with depression or cognitive dysfunction? Ann Intern Med. 2006;145:573-581. doi: 10.7326/0003-4819-145-8-200610170-00006
51. Gussekloo J, van Exel E, de Craen AJM, et al. Thyroid status, disability and cognitive function, and survival in old age. JAMA. 2004;292:2591-2599. doi: 10.1001/jama.292.21.2591
52. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism: a systematic review and meta-analysis. JAMA. 2018;320:1349-1359. doi: 10.1001/jama.2018.13770
53. Monzani F, Dardano A, Caraccio N. Does treating subclinical hypothyroidism improve markers of cardiovascular risk? Treat Endocrinol. 2006;5:65-81. doi: 10.2165/00024677-200605020-00001
54. Duntas LH. Does celiac disease trigger autoimmune thyroiditis? Nat Rev Endocrinol. 2009;5:190-191. doi: 10.1038/nrendo.2009.46
55. Lerner A, Jeremias P, Matthias T. Gut-thyroid axis and celiac disease. Endocr Connect. 2017;6:R52-R58. doi: 10.1530/EC-17-0021
56. Janegova A, Janega P, Rychly B, et al. The role of Epstein-Barr virus infection in the development of autoimmune thyroid diseases. Endokrynol Pol. 2015;66:132-136. doi: 10.5603/EP.2015.0020
57. Brent GA. Environmental exposures and autoimmune thyroid disease. Thyroid. 2010;20:755-761. doi: 10.1089/thy.2010.1636
58. Valko M, Leibfritz D, Moncol J, et al. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44-84. doi: 10.1016/j.biocel.2006.07.001
59. Garber JR, Cobin RH, Gharib H, et al; American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028. doi: 10.4158/EP12280.GL
60. Welsh KJ, Soldin SJ. Diagnosis of endocrine disease: How reliable are free thyroid and total T3 hormone assays? Eur J Endocrinol. 2016;175:R255-R263. doi: 10.1530/EJE-16-0193
61. Grossman A, Feldhamer I, Meyerovitch J. Treatment with levothyroxin in subclinical hypothyroidism is associated with increased mortality in the elderly. Eur J Intern Med. 2018;50:65-68. doi: 10.1016/j.ejim.2017.11.010
62. Pearce SHS, Brabant G, Duntas LH, et al. 2013 ETA Guideline: management of subclinical hypothyroidism. Eur Thyroid J. 2013;2:215-228. doi: 10.1159/000356507
1. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499. doi: 10.1210/jcem.87.2.8182
2. Vanderpump MPJ. The epidemiology of thyroid disease. Br Med Bull. 2011;99:39-51. doi: 10.1093/bmb/ldr030
3. Canaris GJ, Manowitz NR, Mayor G, et al. The Colorado thyroid disease prevalence study. Arch Intern Med. 2000;160:526-534. doi: 10.1001/archinte.160.4.526
4. Persani L. Central hypothyroidism: pathogenic, diagnostic, and therapeutic challenges. J Clin Endocrinol Metab. 2012;97:3068-3078. doi: 10.1210/jc.2012-1616
5. Almandoz JP, Gharib H. Hypothyroidism: etiology, diagnosis, and management. Med Clin North Am. 2012;96:203-221. doi: 10.1016/j.mcna.2012.01.005
6. Ai J, Leonhardt JM, Heymann WR. Autoimmune thyroid diseases: etiology, pathogenesis and dermatologic manifestations. J Am Acad Dermatol. 2003;48:641-659. doi: 10.1067/mjd.2003.257
7. Franzotti AM, Avelar JCD, Cardoso TA, et al. Pityriasis rubra pilar and hypothyroidism. An Bras Dermatol. 2014;89:497-500. doi: 10.1590/abd1806-4841.20142994
8. Yaylali O, Kirac S, Yilmaz M, et al. Does hypothyroidism affect gastrointestinal motility? Gastroenterol Res Pract. 2009;2009:529802. doi: 10.1155/2009/529802
9. Patil AD. Link between hypothyroidism and small intestinal bacterial overgrowth. Indian J Endocrinol Metab. 2014;18:307-309.
10. Ono Y, Ono S, Yasunaga H, et al. Clinical characteristics and outcomes of myxedema coma: analysis of a national inpatient database in Japan. J Epidemiol. 2017;27:117-122. doi: 10.1016/j.je.2016.04.002
11. Boomsma MJ, Bijl HP, Langendijk JA. Radiation-induced hypothyroidism in head and neck cancer patients: a systematic review. Radiother Oncol. 2011;99:1-5. doi: 10.1016/j.radonc.2011.03.002
12. Boelaert K, Newby PR, Simmonds MJ, et al. Prevalence and relative risk of other autoimmune diseases in subjects with autoimmune thyroid disease. Am J Med. 2010;123:183.e1-e9. doi: 10.1016/j.amjmed.2009.06.030
13. Cheserek MJ, Wu G-R, Ntazinda A, et al. Association between thyroid hormones, lipids and oxidative stress markers in subclinical hypothyroidism. J Med Biochem. 2015;34:323-331. doi: 10.2478/jomb-2014-0044
14. Zha K, Zuo C, Wang A, et al. LDL in patients with subclinical hypothyroidism shows increased lipid peroxidation. Lipids Health Dis. 2015;14:95. doi: 10.1186/s12944-015-0092-4
15. Tejovathi B, Suchitra MM, Suresh V, et al. Association of lipid oxidation with endothelial dysfunction in patients with overt hypothyroidism. Exp Clin Endocrinol Diabetes. 2013;121:306-309. doi: 10.1055/s-0032-1333298
16. LeFevre ML; U.S. Preventive Services Task Force. Screening for thyroid dysfunction: U.S. Preventive Services Task Force recommendation statement. Ann Intern Med. 2015;162:641-650. doi: 10.7326/M15-0483
17. Chaker L, Bianco AC, Jonklaas J, et al. Hypothyroidism. Lancet. 2017;390:1550-1562. doi: 10.1016/S0140-6736(17)30703-1
18. Vaidya B, Pearce SHS. Management of hypothyroidism in adults. BMJ. 2008;337:a801. doi: 10.1136/bmj.a801
19. Iyer PC, Cabanillas ME, Waguespack SG, et al. Immune-related thyroiditis with immune checkpoint inhibitors. Thyroid. 2018;28:1243-1251. doi: 10.1089/thy.2018.0116
20. Garber JR, Cobin RH, Gharib H, et al; American Association Of Clinical Endocrinologists And American Thyroid Association Taskforce On Hypothyroidism In Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid. 2012;22:1200-1235. doi: 10.1089/thy.2012.0205
21. Maiden MJ, Torpy DJ. Thyroid hormones in critical illness. Crit Care Clin. 2019;35:375-388. doi: 10.1016/j.ccc.2018.11.012
22. Peeters RP. Subclinical hypothyroidism. N Engl J Med. 2017;376:2556-2565. doi: 10.1056/NEJMcp1611144
23. Benvenga S, Carlé A. Levothyroxine formulations: pharmacological and clinical implications of generic substitution. Adv Ther. 2019;36(suppl 2):59-71. doi: 10.1007/s12325-019-01079-1
24. Jonklaas J, Bianco AC, Bauer AJ, et al; . Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association Task Force on Thyroid Hormone Replacement. Thyroid. 2014;24:1670-1751. doi: 10.1089/thy.2014.0028
25. Engler D, Burger AG. The deiodination of the iodothyronines and of their derivatives in man. Endocr Rev. 1984;5:151-184. doi: 10.1210/edrv-5-2-151
26. Ettleson MD, Bianco AC. Individualized therapy for hypothyroidism: is T4 enough for everyone? J Clin Endocrinol Metab. 2020;105:e3090-e3104. doi: 10.1210/clinem/dgaa430
27. Slayden TA, Shakir MKM, Hoang TD. A bull in a pill shop: alpha-gal allergy complicating treatment options for postprocedural hypothyroidism. AACE Clin Case Rep. 2020;6:e101-e104. doi: 10.4158/ACCR-2019-0495
28. Chamorro-Pareja N, Carrillo-Martin I, Haehn DA, et al. Self-reported allergy to thyroid replacement therapy: a multicenter retrospective chart review. Endocr Pract. 2020;26:761-767. doi: 10.4158/EP-2019-0488
29. Shakir MKM, Turton D, Aprill BS, et al. Anemia: a cause of intolerance to thyroxine sodium. Mayo Clin Proc. 2000;75:189-192.
30. Jonklaas J, Bianco AC, Cappola AR, et al. Evidence-based use of levothyroxine/liothyronine combinations in treating hypothyroidism: a consensus document. Thyroid. 2021;31:156-182. doi: 10.1089/thy.2020.0720
31. Appelhof BC, Fliers E, Wekking EM, et al. Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J Clin Endocrinol Metab. 2005;90:2666-2674. doi: 10.1210/jc.2004-2111
32. Escobar-Morreale HF, Botella-Carretero JI, M, et al. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann Intern Med. 2005;142:412-424. doi: 10.7326/0003-4819-142-6-200503150-00007
33. Hoang TD, Olsen CH, Mai VQ, et al. Desiccated thyroid extract compared with levothyroxine in the treatment of hypothyroidism: a randomized, double-blind, crossover study. J Clin Endocrinol Metab. 2013;98:1982-1990. doi: 10.1210/jc.2012-4107
34. Shakir MKM, Brooks DI, McAninch EA, et al. Comparative effectiveness of levothyroxine, desiccated thyroid extract, and levothyroxine+liothyronine in hypothyroidism. J Clin Endocrinol Metab. 2021;106:e4400-e4413. doi: 10.1210/clinem/dgab478
35. Valizadeh M, Seyyed-Majidi MR, Hajibeigloo H, et al. Efficacy of combined levothyroxine and liothyronine as compared with levothyroxine monotherapy in primary hypothyroidism: a randomized controlled trial. Endocr Res. 2009;34:80-89. doi: 10.1080/07435800903156340
36. Walsh JP, Shiels L, Lim EM, et al. Combined thyroxine/liothyronine treatment does not improve well-being, quality of life, or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J Clin Endocrinol Metab. 2003;88:4543-4550. doi: 10.1210/jc.2003-030249
37. Rodriguez T, Lavis VR, Meininger JC, et al. Substitution of liothyronine at a 1:5 ratio for a portion of levothyroxine: effect on fatigue, symptoms of depression, and working memory versus treatment with levothyroxine alone. Endocr Pract. 2005;11:223-233. doi: 10.4158/EP.11.4.223
38. Sawka AM, Gerstein HC, Marriott MJ, et al. Does a combination regimen of thyroxine (T4) and 3,5,3’-triiodothyronine improve depressive symptoms better than T4 alone in patients with hypothyroidism? Results of a double-blind, randomized, controlled trial. J Clin Endocrinol Metab. 2003;88:4551-4555. doi: 10.1210/jc.2003-030139
39. Clyde PW, Harari AE, Getka EJ, et al. Combined levothyroxine plus liothyronine compared with levothyroxine alone in primary hypothyroidism: a randomized controlled trial. JAMA. 2003;290:2952-2958. doi: 10.1001/jama.290.22.2952
40. Duntas LH. Selenium and the thyroid: a close-knit connection. J Clin Endocrinol Metab. 2010;95:5180-5188. doi: 10.1210/jc.2010-0191
41. Winther KH, Wichman JEM, Bonnema SJ, et al. Insufficient documentation for clinical efficacy of selenium supplementation in chronic autoimmune thyroiditis, based on a systematic review and meta-analysis. Endocrine. 2017;55:376-385. doi: 10.1007/s12020-016-1098-z
42. Parva NR, Tadepalli S, Singh P, et al. Prevalence of vitamin D deficiency and associated risk factors in the US population (2011-2012). Cureus. 2018;10:e2741. doi: 10.7759/cureus.2741
43. Wang J, Lv S, Chen G, et al. Meta-analysis of the association between vitamin D and autoimmune thyroid disease. Nutrients. 2015,7:2485-2498. doi: 10.3390/nu7042485
44. Wilson MM, Reedy J, Krebs-Smith SM. American diet quality: where it is, where it is heading, and what it could be. J Acad Nutr Diet. 2016;116:302-310.e1. doi: 10.1016/j.jand.2015.09.020
45. Babiker A, Alawi A, Al Atawi M, et al. The role of micronutrients in thyroid dysfunction. Sudan J Paediatr. 2020;20:13-19. doi: 10.24911/SJP.106-1587138942
46. Knezevic J, Starchl C, Tmava Berisha A, et al. Thyroid-gut-axis: How does the microbiota influence thyroid function? Nutrients. 2020;12:1769. doi: 10.3390/nu12061769
47. Rayman MP. Multiple nutritional factors and thyroid disease, with particular reference to autoimmune thyroid disease. Proc Nutr Soc. 2019;78:34-44. doi: 10.1017/S0029665118001192
48. Chakrabarti SK, Ghosh S, Banerjee S, et al. Oxidative stress in hypothyroid patients and the role of antioxidant supplementation. Indian J Endocrinol Metab. 2016;20:674-678. doi: 10.4103/2230-8210.190555
49. Tseng F-Y, Lin W-Y, Lin C-C, et al. Subclinical hypothyroidism is associated with increased risk for all-cause and cardiovascular mortality in adults. J Am Coll Cardiol. 2012;60:730-737. doi: 10.1016/j.jacc.2012.03.047
50. Roberts LM, Pattison H, Roalfe A, et al. Is subclinical thyroid dysfunction in the elderly associated with depression or cognitive dysfunction? Ann Intern Med. 2006;145:573-581. doi: 10.7326/0003-4819-145-8-200610170-00006
51. Gussekloo J, van Exel E, de Craen AJM, et al. Thyroid status, disability and cognitive function, and survival in old age. JAMA. 2004;292:2591-2599. doi: 10.1001/jama.292.21.2591
52. Feller M, Snel M, Moutzouri E, et al. Association of thyroid hormone therapy with quality of life and thyroid-related symptoms in patients with subclinical hypothyroidism: a systematic review and meta-analysis. JAMA. 2018;320:1349-1359. doi: 10.1001/jama.2018.13770
53. Monzani F, Dardano A, Caraccio N. Does treating subclinical hypothyroidism improve markers of cardiovascular risk? Treat Endocrinol. 2006;5:65-81. doi: 10.2165/00024677-200605020-00001
54. Duntas LH. Does celiac disease trigger autoimmune thyroiditis? Nat Rev Endocrinol. 2009;5:190-191. doi: 10.1038/nrendo.2009.46
55. Lerner A, Jeremias P, Matthias T. Gut-thyroid axis and celiac disease. Endocr Connect. 2017;6:R52-R58. doi: 10.1530/EC-17-0021
56. Janegova A, Janega P, Rychly B, et al. The role of Epstein-Barr virus infection in the development of autoimmune thyroid diseases. Endokrynol Pol. 2015;66:132-136. doi: 10.5603/EP.2015.0020
57. Brent GA. Environmental exposures and autoimmune thyroid disease. Thyroid. 2010;20:755-761. doi: 10.1089/thy.2010.1636
58. Valko M, Leibfritz D, Moncol J, et al. Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol. 2007;39:44-84. doi: 10.1016/j.biocel.2006.07.001
59. Garber JR, Cobin RH, Gharib H, et al; American Association of Clinical Endocrinologists and American Thyroid Association Taskforce on Hypothyroidism in Adults. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028. doi: 10.4158/EP12280.GL
60. Welsh KJ, Soldin SJ. Diagnosis of endocrine disease: How reliable are free thyroid and total T3 hormone assays? Eur J Endocrinol. 2016;175:R255-R263. doi: 10.1530/EJE-16-0193
61. Grossman A, Feldhamer I, Meyerovitch J. Treatment with levothyroxin in subclinical hypothyroidism is associated with increased mortality in the elderly. Eur J Intern Med. 2018;50:65-68. doi: 10.1016/j.ejim.2017.11.010
62. Pearce SHS, Brabant G, Duntas LH, et al. 2013 ETA Guideline: management of subclinical hypothyroidism. Eur Thyroid J. 2013;2:215-228. doi: 10.1159/000356507
PRACTICE RECOMMENDATIONS
› Prescribe levothyroxine (LT4) to maintain thyroid-stimulating hormone (TSH) at 4 to 7 mIU/L in select patients with primary hypothyroidism for whom that range of the serum TSH level can be considered appropriate (ie, those older than 65 years and those who have underlying coronary artery disease or another debilitating chronic disorder). A
› Counsel all women of childbearing age with primary hypothyroidism that they need to have their dosage of LT4 increased as soon as pregnancy is suspected. A
› Keep in mind that treating hypothyroidism is not always necessary in older patients who have subclinical disease and a serum TSH level < 10 mIU/L. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
PLA testing brings nuance to the diagnosis of early-stage melanoma
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
BOSTON – Although
One such test, the Pigmented Lesional Assay (PLA) uses adhesive patches applied to lesions of concern at the bedside to extract RNA from the stratum corneum to help determine the risk for melanoma.
At the annual meeting of the American Academy of Dermatology, Caroline C. Kim, MD, director of melanoma and pigmented lesion clinics at Newton Wellesley Dermatology, Wellesley Hills, Mass., and Tufts Medical Center, Boston, spoke about the PLA, which uses genetic expression profiling to measure the expression level of specific genes that are associated with melanoma: PRAME (preferentially expressed antigen in melanoma) and LINC00518 (LINC). There are four possible results of the test: Aberrant expression of both LINC and PRAME (high risk); aberrant expression of a single gene (moderate risk); aberrant expression of neither gene (low risk); or inconclusive.
Validation data have shown a sensitivity of 91% and a specificity of 69% for the PLA, with a 99% negative predictive value; so a lesion that tested negative by PLA has a less than 1% chance of being melanoma. In addition, a study published in 2020 found that the addition of TERT (telomerase reverse transcriptase) mutation analyses increased the sensitivity of the PLA to 97%.
While the high negative predictive value is helpful to consider in clinical scenarios to rule-out melanoma for borderline lesions, one must consider the positive predictive value as well and how this may impact clinical care, Dr. Kim said. In a study examining outcomes of 381 lesions, 51 were PLA positive (single or double) and were biopsied, of which 19 (37%) revealed a melanoma diagnosis. In a large U.S. registry study of 3,418 lesions, 324 lesions that were PLA double positive were biopsied, with 18.7% revealing a melanoma diagnosis.
“No test is perfect, and this applies to PLA, even if you get a double-positive or double-negative test result,” Dr. Kim said. “You want to make sure that patients are aware of false positives and negatives. However, PLA could be an additional piece of data to inform your decision to proceed with biopsy on select borderline suspicious pigmented lesions. More studies are needed to better understand the approach to single- and double-positive PLA results.”
The PLA kit contains adhesive patches and supplies and a FedEx envelope for return to DermTech, the test’s manufacturer, for processing. The patches can be applied to lesions at least 4 mm in diameter; multiple kits are recommended for those greater than 16 mm in diameter. The test is not validated for lesions located on mucous membranes, palms, soles, nails, or on ulcerated or bleeding lesions, nor for those that have been previously biopsied. It is also not validated for use in pediatric patients or in those with skin types IV or higher. Results are returned in 2-3 days. If insurance does not cover the test, the cost to the patient is approximately $50 per lesion or a maximum of $150, according to Dr. Kim.
Use in clinical practice
In Dr. Kim’s clinical experience, the PLA can be considered for suspicious pigmented lesions on cosmetically sensitive areas and for suspicious lesions in areas difficult to biopsy or excise. For example, she discussed the case of a 72-year-old woman with a family history of melanoma, who presented to her clinic with a longstanding pigmented lesion on her right upper and lower eyelids that had previously been treated with laser. She had undergone multiple prior biopsies over 12 years, which caused mild to moderate atypical melanocytic proliferation. The PLA result was double negative for PRAME and LINC in her upper and lower eyelid, “which provided reassurance to the patient,” Dr. Kim said. The patient continues to be followed closely for any clinical changes.
Another patient, a 67-year-old woman, was referred to Dr. Kim from out of state for a teledermatology visit early in the COVID-19 pandemic. The patient had a lesion on her right calf that was hard, raised, and pink, did not resemble other lesions on her body, and had been present for a few weeks. “Her husband had recently passed away from brain cancer and she was very concerned about melanoma,” Dr. Kim recalled. “She lived alone, and the adult son was with her during the teledermatology call to assist. The patient asked about the PLA test, and given her difficulty going to a medical office at the time, we agreed to help her with this test.” The patient and her son arranged another teledermatology visit with Dr. Kim after receiving the kit in the mail from DermTech, and Dr. Kim coached them on how to properly administer the test. The results came back as PRAME negative and LINC positive. A biopsy with a local provider was recommended and the pathology results showed an inflamed seborrheic keratosis.
“This case exemplifies a false-positive result. We should be sure to make patients aware of this possibility,” Dr. Kim said.
Incorporating PLA into clinical practice requires certain workflow considerations, with paperwork to fill out in addition to performing the adhesive test, collection of insurance information, mailing the kit via FedEx, retrieving the results, and following up with the patient, said Dr. Kim. “For select borderline pigmented lesions, I discuss the rationale of the test with patients, the possibility of false-positive and false-negative results and the need to return for a biopsy when there is positive result. Clinical follow-up is recommended for negative results. There is also the possibility of charge to the patient if the test is not covered by their insurance.”
Skin biopsy still the gold standard
Despite the availability of the PLA as an assessment tool, Dr. Kim emphasized that skin biopsy remains the gold standard for diagnosing melanoma. “Future prospective randomized clinical trials are needed to examine the role of genetic expression profiling in staging and managing patients,” she said.
In 2019, she and her colleagues surveyed 42 pigmented lesion experts in the United States about why they ordered one of three molecular tests on the market or not and how results affected patient treatment. The proportion of clinicians who ordered the tests ranged from 21% to 29%. The top 2 reasons respondents chose for not ordering the PLA test specifically were: “Feel that further validation studies are necessary” (20%) and “do not feel it would be useful in my practice” (18%).
Results of a larger follow-up survey on usage patterns of PLA of both pigmented lesion experts and general clinicians on this topic are expected to be published shortly.
Dr. Kim reported having no disclosures related to her presentation.
AT AAD 22
Neighborhood-level data sheds new light on racial and ethnic diversity in MS
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
SEATTLE – These populations often have more severe disease, likely driven by socioeconomic factors and health care access, according to a new study that examined neighborhood-level data and disease severity in the United States.
“It has previously been thought that MS is less common among non-European Caucasian White populations, driven partly by the well-known association of incidence with latitude. It is abundantly clear at this point that this idea is not true,” said Christopher Orlando, MD, during a presentation at the 2022 annual meeting of the American Academy of Neurology.
He noted that several U.S. studies with large sample sizes have shown greater disease severity and a higher disability burden among Hispanic and Black patients. “Black patients in particular appear to have a higher incidence of disease and a greater proportion of progressive disease phenotypes,” said Dr. Orlando.
Race and ethnicity are unlikely explanations for this disparity, according to Dr. Orlando. “While much remains to be discovered of the genetic underpinnings of MS, what we do know does not support the idea that minorities would have a predilection to more severe disease. For example, the well-known high-risk allele HLA DRB1*1501 appears to have a lower frequency in African populations, compared with European [populations].”
Instead, evidence suggests that interrelated social causes include access to resources, environmental exposures, and psychosocial stress. “These affect health via a number of pathways including direct physical injury, allostatic load, and access to health care,” said Dr. Orlando.
Probing racial and ethnic disparities
Previous studies that corrected for social determinants of health such as socioeconomic and insurance status reduce the association between MS disability and race, but they do not completely explain it.
To get a better understanding of the impacts of these factors, researchers have used neighborhood-level data combined with information on socioeconomic status and social deprivation to identify associations with MS severity.
At the conference, Dr. Orlando presented a new study that is the first to use this methodology in the United States, and it is the first to apply it to the study of racial and ethnic disparities in MS.
The study confirmed more severe disability in Hispanic and Black patients than in White patients. Clinical factors associated with more severe disease were similar across the three groups, with some small differences among individual traits. “More stark differences appeared when we compared social determinants of health. Hispanic patients were less likely to speak English as a primary language or to complete 12 years of education. Black patients were less likely to live in a rural county and more likely to be unemployed. One particularly stark difference was in the number of unemployed specifically due to their MS, with only 1 White patient [1.1%], 7 Hispanic patients [7.8%] and 27 Black patients [31.0%],” said Dr. Orlando.
The researchers found that Black and Hispanic patients tend to live in more vulnerable neighborhoods than White patients. The researchers found no significant association between social vulnerability index (SVI) values and MS severity, though there was an association in a separate analysis that only included White patients. The SVI uses 15 measures taken from the U.S. Census to identify communities that might require additional support during natural disasters.
“It would appear that the sheer complexity both in variety and magnitude of the social determinants of health are such that by far the stronger association is with race and ethnicity, which are surrogates for any number of social determinants and societal inequities,” said Dr. Orlando.
What drives the inequity?
Dr. Orlando acknowledged that some might wonder if these results indicate a true biologically intrinsic factor such as genetic predisposition. “I want to warn against that kind of thinking in the strongest possible terms. It is implausible on several levels. It’s not biologically plausible based on our understanding that race and ethnicity are not genetic constructs. And it’s also not numerically plausible based on these data,” said Dr. Orlando.
While some of the drivers of this inequity have been partially examined, many have not been studied. “As long as this is the case, our ability to fulfill our roles as physicians will be limited in several important ways. Our ability to assess our patients’ individual risk will be missing key information, which will limit the efficacy of shared decision-making, which of course is the cornerstone of MS treatment. In addition, we will continue to struggle to include minority patients in our research studies, and the very design and results of those studies may be misguided, as we will either fail to include these populations, or we will fail to adjust for important confounders,” he said.
New answers, new questions
The neighborhood-level data examined by Dr. Orlando’s group “brings extra information in terms of the negative impact of social determinants of health. The disparity seen in neighborhood living is quite striking,” said Lilyana Amezcua, MD, who served as a discussant for Dr. Orlando’s presentation. The study reinforces findings of her own group in Hispanic and Latinx individuals with MS. Some comorbidities are more common among these groups, which is exacerbated by poor health access.
“We have noted that almost 30% of them also have this comorbidity of hypertension, but what is also observed is that only 7% of them are aware [that they have hypertension],” said Dr. Amezcua, who is an associate professor of neurology at the University of Southern California, Los Angeles.
The findings should prompt further research to understand the impact of systemic racism and neighborhood factors, such as disinvestment in the public and private sectors, underresourced hospitals and clinics, and negative infrastructure. “We need to start discussing the (patient’s) environment so we can better understand the community resources they may have available, as well as create innovative transitional care services. We need to also recognize and accept that structural racism and imbalanced distribution of resources and neighborhoods does restrict educational and economic opportunities, as well as health care access and the safety of these marginalized communities,” said Dr. Amezcua.
Dr. Amezcua has consulted for, received speaking fees from, or served on steering committees or advisory boards for Biogen Idec, Novartis, Genentech, and EMD Serono. She has received research support from the Bristol-Myers Squibb Foundation and Biogen Idec. Dr. Orlando has no relevant financial disclosures.
AT AAN 2022
Central Centrifugal Cicatricial Alopecia
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.
Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia (CCCA).1 Central centrifugal cicatricial alopecia (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
Central centrifugal cicatricial alopecia predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. Central centrifugal cicatricial alopecia has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
• Central centrifugal cicatricial alopecia begins in the central scalp (crown area, vertex) and spreads centrifugally.
• Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
• Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
• Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
• Central centrifugal cicatricial alopecia can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral anti-inflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
- Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/j.1600-0560.2001 .280701.x
- Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/archderm.147.12.1453
- Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111 /1346-8138.14217
- Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
- Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
- Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
- Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056 /NEJMe1900042
- Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
- Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j .jdcr.2019.12.008
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.
Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia (CCCA).1 Central centrifugal cicatricial alopecia (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
Central centrifugal cicatricial alopecia predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. Central centrifugal cicatricial alopecia has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
• Central centrifugal cicatricial alopecia begins in the central scalp (crown area, vertex) and spreads centrifugally.
• Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
• Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
• Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
• Central centrifugal cicatricial alopecia can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral anti-inflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
THE PRESENTATION
A Early central centrifugal cicatricial alopecia with a small central patch of hair loss in a 45-year-old Black woman.
B Late central centrifugal cicatricial alopecia with a large central patch of hair loss in a 43-year-old Black woman.
Scarring alopecia is a collection of hair loss disorders including chronic cutaneous lupus erythematosus (discoid lupus), lichen planopilaris, dissecting cellulitis, acne keloidalis, and central centrifugal cicatricial alopecia (CCCA).1 Central centrifugal cicatricial alopecia (formerly hot comb alopecia or follicular degeneration syndrome) is a progressive, scarring, inflammatory alopecia and represents the most common form of scarring alopecia in women of African descent. It results in permanent destruction of hair follicles.
Epidemiology
Central centrifugal cicatricial alopecia predominantly affects women of African descent but also may affect men. The prevalence of CCCA in those of African descent has varied in the literature. Khumalo2 reported a prevalence of 1.2% for women younger than 50 years and 6.7% in women older than 50 years. Central centrifugal cicatricial alopecia has been reported in other ethnic groups, such as those of Asian descent.3
Historically, hair care practices that are more common in those of African descent, such as high-tension hairstyles as well as heat and chemical hair relaxers, were implicated in the development of CCCA. However, the causes of CCCA are most likely multifactorial, including family history, genetic mutations, and hair care practices.4-7PADI3 mutations likely predispose some women to CCCA. Mutations in PADI3, which encodes peptidyl arginine deiminase 3 (an enzyme that modifies proteins crucial for the formation of hair shafts), were found in some patients with CCCA.8 Moreover, other genetic defects also likely play a role.7
Key clinical features
Early recognition is key for patients with CCCA.
• Central centrifugal cicatricial alopecia begins in the central scalp (crown area, vertex) and spreads centrifugally.
• Scalp symptoms such as tenderness, pain, a tingling or crawling sensation, and itching may occur.9 Some patients may not have any symptoms at all, and hair loss may progress painlessly.
• Central hair breakage—forme fruste CCCA—may be a presenting sign of CCCA.9
• Loss of follicular ostia and mottled hypopigmented and hyperpigmented macules are common findings.6
• Central centrifugal cicatricial alopecia can be diagnosed clinically and by histopathology.
Worth noting
Patients may experience hair loss and scalp symptoms for years before seeking medical evaluation. In some cultures, hair breakage or itching on the top of the scalp may be viewed as a normal occurrence in life.
It is important to set patient expectations that CCCA is a scarring alopecia, and the initial goal often is to maintain the patient's existing hair. However, hair and areas responding to treatment should still be treated. Without any intervention, the resulting scarring from CCCA may permanently scar follicles on the entire scalp.
Due to the inflammatory nature of CCCA, potent topical corticosteroids (eg, clobetasol propionate), intralesional corticosteroids (eg, triamcinolone acetonide), and oral anti-inflammatory agents (eg, doxycycline) are utilized in the treatment of CCCA. Minoxidil is another treatment option. Adjuvant therapies such as topical metformin also have been tried.10 Importantly, treatment of CCCA may halt further permanent destruction of hair follicles, but scalp symptoms may reappear periodically and require re-treatment with anti-inflammatory agents.
Health care highlight
Thorough scalp examination and awareness of clinical features of CCCA may prompt earlier diagnosis and prevent future severe permanent alopecia. Clinicians should encourage patients with suggestive signs or symptoms of CCCA to seek care from a dermatologist.
- Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/j.1600-0560.2001 .280701.x
- Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/archderm.147.12.1453
- Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111 /1346-8138.14217
- Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
- Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
- Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
- Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056 /NEJMe1900042
- Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
- Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j .jdcr.2019.12.008
- Sperling LC. Scarring alopecia and the dermatopathologist. J Cutan Pathol. 2001;28:333-342. doi:10.1034/j.1600-0560.2001 .280701.x
- Khumalo NP. Prevalence of central centrifugal cicatricial alopecia. Arch Dermatol. 2011;147:1453-1454. doi:10.1001/archderm.147.12.1453
- Su HJ, Cheng AY, Liu CH, et al. Primary scarring alopecia: a retrospective study of 89 patients in Taiwan [published online January 16, 2018]. J Dermatol. 2018;45:450-455. doi:10.1111 /1346-8138.14217
- Sperling LC, Cowper SE. The histopathology of primary cicatricial alopecia. Semin Cutan Med Surg. 2006;25:41-50
- Dlova NC, Forder M. Central centrifugal cicatricial alopecia: possible familial aetiology in two African families from South Africa. Int J Dermatol. 2012;51(supp 1):17-20, 20-23.
- Ogunleye TA, Quinn CR, McMichael A. Alopecia. In: Taylor SC, Kelly AP, Lim HW, et al, eds. Dermatology for Skin of Color. McGraw Hill; 2016:253-264.
- Uitto J. Genetic susceptibility to alopecia [published online February 13, 2019]. N Engl J Med. 2019;380:873-876. doi:10.1056 /NEJMe1900042
- Malki L, Sarig O, Romano MT, et al. Variant PADI3 in central centrifugal cicatricial alopecia. N Engl J Med. 2019;380:833-841.
- Callender VD, Wright DR, Davis EC, et al. Hair breakage as a presenting sign of early or occult central centrifugal cicatricial alopecia: clinicopathologic findings in 9 patients. Arch Dermatol. 2012;148:1047-1052.
- Araoye EF, Thomas JAL, Aguh CU. Hair regrowth in 2 patients with recalcitrant central centrifugal cicatricial alopecia after use of topical metformin. JAAD Case Rep. 2020;6:106-108. doi:10.1016/j .jdcr.2019.12.008
FDA okays first sublingual med for agitation in serious mental illness
This is the first FDA-approved, orally dissolving, self-administered sublingual treatment for this indication. With a demonstrated onset of action as early as 20 minutes, it shows a high response rate in patients at both 120-mcg and 180-mcg doses.
An estimated 7.3 million individuals in the United States are diagnosed with schizophrenia or bipolar disorders, and up to one-quarter of them experience episodes of agitation that can occur 10-17 times annually. These episodes represent a significant burden for patients, caregivers, and the health care system.
“There are large numbers of patients who experience agitation associated with schizophrenia and bipolar disorders, and this condition has been a long-standing challenge for health care professionals to treat,” said John Krystal, MD, the Robert L. McNeil Jr. Professor of Translational Research and chair of the department of psychiatry at Yale University, New Haven, Conn.
“The approval of Igalmi, a self-administered film with a desirable onset of action, represents a milestone moment. It provides health care teams with an innovative tool to help control agitation. As clinicians, we welcome this much-needed new oral treatment option,” he added.
“Igalmi is the first new acute treatment for schizophrenia or bipolar disorder–associated agitation in nearly a decade and represents a differentiated approach to helping patients manage this difficult and debilitating symptom,” said Vimal Mehta, PhD, CEO of BioXcel Therapeutics.
The FDA approval of Igalmi is based on data from two pivotal randomized, double-blinded, placebo-controlled, parallel-group, phase 3 trials that evaluated Igalmi for the acute treatment of agitation associated with schizophrenia (SERENITY I) or bipolar I or II disorder (SERENITY II).
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were somnolence, paresthesia or oral hypoesthesia, dizziness, dry mouth, hypotension, and orthostatic hypotension. All adverse drug reactions were mild to moderate in severity. While Igalmi was not associated with any treatment-related serious adverse effects in phase 3 studies, it may cause notable side effects, including hypotension, orthostatic hypotension, bradycardia, QT interval prolongation, and somnolence.
As previously reported by this news organization, data from the phase 3 SERENITY II trial that evaluated Igalmi in bipolar disorders were published in JAMA.
A version of this article first appeared on Medscape.com.
This is the first FDA-approved, orally dissolving, self-administered sublingual treatment for this indication. With a demonstrated onset of action as early as 20 minutes, it shows a high response rate in patients at both 120-mcg and 180-mcg doses.
An estimated 7.3 million individuals in the United States are diagnosed with schizophrenia or bipolar disorders, and up to one-quarter of them experience episodes of agitation that can occur 10-17 times annually. These episodes represent a significant burden for patients, caregivers, and the health care system.
“There are large numbers of patients who experience agitation associated with schizophrenia and bipolar disorders, and this condition has been a long-standing challenge for health care professionals to treat,” said John Krystal, MD, the Robert L. McNeil Jr. Professor of Translational Research and chair of the department of psychiatry at Yale University, New Haven, Conn.
“The approval of Igalmi, a self-administered film with a desirable onset of action, represents a milestone moment. It provides health care teams with an innovative tool to help control agitation. As clinicians, we welcome this much-needed new oral treatment option,” he added.
“Igalmi is the first new acute treatment for schizophrenia or bipolar disorder–associated agitation in nearly a decade and represents a differentiated approach to helping patients manage this difficult and debilitating symptom,” said Vimal Mehta, PhD, CEO of BioXcel Therapeutics.
The FDA approval of Igalmi is based on data from two pivotal randomized, double-blinded, placebo-controlled, parallel-group, phase 3 trials that evaluated Igalmi for the acute treatment of agitation associated with schizophrenia (SERENITY I) or bipolar I or II disorder (SERENITY II).
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were somnolence, paresthesia or oral hypoesthesia, dizziness, dry mouth, hypotension, and orthostatic hypotension. All adverse drug reactions were mild to moderate in severity. While Igalmi was not associated with any treatment-related serious adverse effects in phase 3 studies, it may cause notable side effects, including hypotension, orthostatic hypotension, bradycardia, QT interval prolongation, and somnolence.
As previously reported by this news organization, data from the phase 3 SERENITY II trial that evaluated Igalmi in bipolar disorders were published in JAMA.
A version of this article first appeared on Medscape.com.
This is the first FDA-approved, orally dissolving, self-administered sublingual treatment for this indication. With a demonstrated onset of action as early as 20 minutes, it shows a high response rate in patients at both 120-mcg and 180-mcg doses.
An estimated 7.3 million individuals in the United States are diagnosed with schizophrenia or bipolar disorders, and up to one-quarter of them experience episodes of agitation that can occur 10-17 times annually. These episodes represent a significant burden for patients, caregivers, and the health care system.
“There are large numbers of patients who experience agitation associated with schizophrenia and bipolar disorders, and this condition has been a long-standing challenge for health care professionals to treat,” said John Krystal, MD, the Robert L. McNeil Jr. Professor of Translational Research and chair of the department of psychiatry at Yale University, New Haven, Conn.
“The approval of Igalmi, a self-administered film with a desirable onset of action, represents a milestone moment. It provides health care teams with an innovative tool to help control agitation. As clinicians, we welcome this much-needed new oral treatment option,” he added.
“Igalmi is the first new acute treatment for schizophrenia or bipolar disorder–associated agitation in nearly a decade and represents a differentiated approach to helping patients manage this difficult and debilitating symptom,” said Vimal Mehta, PhD, CEO of BioXcel Therapeutics.
The FDA approval of Igalmi is based on data from two pivotal randomized, double-blinded, placebo-controlled, parallel-group, phase 3 trials that evaluated Igalmi for the acute treatment of agitation associated with schizophrenia (SERENITY I) or bipolar I or II disorder (SERENITY II).
The most common adverse reactions (incidence ≥5% and at least twice the rate of placebo) were somnolence, paresthesia or oral hypoesthesia, dizziness, dry mouth, hypotension, and orthostatic hypotension. All adverse drug reactions were mild to moderate in severity. While Igalmi was not associated with any treatment-related serious adverse effects in phase 3 studies, it may cause notable side effects, including hypotension, orthostatic hypotension, bradycardia, QT interval prolongation, and somnolence.
As previously reported by this news organization, data from the phase 3 SERENITY II trial that evaluated Igalmi in bipolar disorders were published in JAMA.
A version of this article first appeared on Medscape.com.
Analysis boosts fluvoxamine for COVID, but what’s the evidence?
a new systematic review and meta-analysis has found. But outside experts differ over whether the evidence from just three studies is strong enough to warrant adding the drug to the COVID-19 armamentarium.
The report, published online in JAMA Network Open, looked at three studies and estimated that the drug could reduce the relative risk of hospitalization by around 25% (likelihood of moderate effect, 81.6%-91.8%), depending on the type of analysis used.
“This research might be valuable, but the jury remains out until several other adequately powered and designed trials are completed,” said infectious disease specialist Carl J. Fichtenbaum, MD, of the University of Cincinnati, who’s familiar with the findings. “I’m not sure how useful this is given we have several antiviral agents available. Why would we choose this over Paxlovid, remdesivir, or molnupiravir?”
According to Dr. Fichtenbaum, researchers began focusing on fluvoxamine after case reports about patients improving while on the medication. This led to further interest, he said, boosted by the drug’s known ability to dampen the immune system.
A Silicon Valley investor and antivaccine activist named Steve Kirsch has been pushing the drug along with the debunked treatment hydroxychloroquine. He’s accused the government of a cover-up of fluvoxamine’s worth, according to MIT Technology Review, and he wrote a commentary that referred to the drug as “the fast, easy, safe, simple, low-cost solution to COVID that works 100% of the time that nobody wants to talk about.”
For the new analysis, researchers examined three randomized clinical trials with a total of 2,196 participants. The most extensive trial, the TOGETHER study in Brazil (n = 1,497), focused on an unusual outcome: It linked the drug to a 32% reduction in relative risk of patients with COVID-19 being hospitalized in an ED for fewer than 6 hours or transferred to a tertiary hospital because of the disease.
Another study, the STOP COVID 2 trial in the United States and Canada (n = 547), was stopped because too few patients could be recruited to provide useful results. The initial phase of this trial, STOP COVID 1 (n = 152), was also included in the analysis.
All participants in the three studies were unvaccinated. Their median age was 46-50 years, 55%-72% were women, and 44%-56% were obese. Most were multiracial due to the high number of participants from Brazil.
“In the Bayesian analyses, the pooled risk ratio in favor of fluvoxamine was 0.78 (95% confidence interval, 0.58-1.08) for the weakly neutral prior and 0.73 (95% CI, 0.53-1.01) for the moderately optimistic prior,” the researchers reported, referring to a reduction in risk of hospitalization. “In the frequentist meta-analysis, the pooled risk ratio in favor of fluvoxamine was 0.75 (95% CI, 0.58-0.97; I2, 0.2%).”
Two of the authors of the new analysis were also coauthors of the TOGETHER trial and both STOP COVID trials.
Corresponding author Emily G. McDonald, MD, division of experimental medicine at McGill University, Montreal, said in an interview that the findings show fluvoxamine “very likely reduces hospitalization in high-risk outpatient adults with COVID-19. This effect varies depending on your baseline risk of developing complications in the first place.”
Dr. McDonald added that “fluvoxamine is an option to reduce hospitalizations in high-risk adults. It is likely effective, is inexpensive, and has a long safety track record.” She also noted that “not all countries have access to Paxlovid, and some people have drug interactions that preclude its use. Existing monoclonals are not effective with newer variants.”
The drug’s apparent anti-inflammatory properties seem to be key, she said. According to her, the next steps should be “testing lower doses to see if they remain effective, following patients long term to see what impact there is on long COVID symptoms, testing related medications in the drug class to see if they also show an effect, and testing in vaccinated people and with newer variants.”
In an interview, biostatistician James Watson, PhD, of the Mahidol Oxford Tropical Medicine Research Unit, Bangkok, Thailand, and Nuffield department of medicine, University of Oxford, England, said the findings of the analysis are “not overwhelming data.”
He noted the TOGETHER study’s unusual focus on ED visits that latest fewer than 6 hours, which he described as “not a very objective endpoint.” The new meta-analysis focused instead on “outcome data on emergency department visits lasting more than 24 hours and used this as a more representative proxy for hospital admission than an ED visit alone.”
Dr. Fichtenbaum also highlighted the odd endpoint. “Most of us would have chosen something like use of oxygen, requirement for ventilation, or death,” he said. “There are many reasons why people go to the ED. This endpoint is not very strong.”
He also noted that the three studies “are very different in design and endpoints.”
Jeffrey S. Morris, PhD, a biostatistician at the University of Pennsylvania, Philadelphia, offered a different perspective about the findings in an interview. “There’s good evidence that it helps some,” he said, and may reduce hospitalizations by 10%. “If the pill is super cheap and toxicity is very acceptable, it’s not adding additional risk. Most clinicians would say that: ‘If I’m reducing risk by 10%, it’s worthwhile.’ ”
No funding was reported. Two authors report having a patent application filed by Washington University for methods of treating COVID-19 during the conduct of the study. Dr. Watson is an investigator for studies analyzing antiviral drugs and Prozac as COVID-19 treatments. Dr. Fichtenbaum and Dr. Morris disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new systematic review and meta-analysis has found. But outside experts differ over whether the evidence from just three studies is strong enough to warrant adding the drug to the COVID-19 armamentarium.
The report, published online in JAMA Network Open, looked at three studies and estimated that the drug could reduce the relative risk of hospitalization by around 25% (likelihood of moderate effect, 81.6%-91.8%), depending on the type of analysis used.
“This research might be valuable, but the jury remains out until several other adequately powered and designed trials are completed,” said infectious disease specialist Carl J. Fichtenbaum, MD, of the University of Cincinnati, who’s familiar with the findings. “I’m not sure how useful this is given we have several antiviral agents available. Why would we choose this over Paxlovid, remdesivir, or molnupiravir?”
According to Dr. Fichtenbaum, researchers began focusing on fluvoxamine after case reports about patients improving while on the medication. This led to further interest, he said, boosted by the drug’s known ability to dampen the immune system.
A Silicon Valley investor and antivaccine activist named Steve Kirsch has been pushing the drug along with the debunked treatment hydroxychloroquine. He’s accused the government of a cover-up of fluvoxamine’s worth, according to MIT Technology Review, and he wrote a commentary that referred to the drug as “the fast, easy, safe, simple, low-cost solution to COVID that works 100% of the time that nobody wants to talk about.”
For the new analysis, researchers examined three randomized clinical trials with a total of 2,196 participants. The most extensive trial, the TOGETHER study in Brazil (n = 1,497), focused on an unusual outcome: It linked the drug to a 32% reduction in relative risk of patients with COVID-19 being hospitalized in an ED for fewer than 6 hours or transferred to a tertiary hospital because of the disease.
Another study, the STOP COVID 2 trial in the United States and Canada (n = 547), was stopped because too few patients could be recruited to provide useful results. The initial phase of this trial, STOP COVID 1 (n = 152), was also included in the analysis.
All participants in the three studies were unvaccinated. Their median age was 46-50 years, 55%-72% were women, and 44%-56% were obese. Most were multiracial due to the high number of participants from Brazil.
“In the Bayesian analyses, the pooled risk ratio in favor of fluvoxamine was 0.78 (95% confidence interval, 0.58-1.08) for the weakly neutral prior and 0.73 (95% CI, 0.53-1.01) for the moderately optimistic prior,” the researchers reported, referring to a reduction in risk of hospitalization. “In the frequentist meta-analysis, the pooled risk ratio in favor of fluvoxamine was 0.75 (95% CI, 0.58-0.97; I2, 0.2%).”
Two of the authors of the new analysis were also coauthors of the TOGETHER trial and both STOP COVID trials.
Corresponding author Emily G. McDonald, MD, division of experimental medicine at McGill University, Montreal, said in an interview that the findings show fluvoxamine “very likely reduces hospitalization in high-risk outpatient adults with COVID-19. This effect varies depending on your baseline risk of developing complications in the first place.”
Dr. McDonald added that “fluvoxamine is an option to reduce hospitalizations in high-risk adults. It is likely effective, is inexpensive, and has a long safety track record.” She also noted that “not all countries have access to Paxlovid, and some people have drug interactions that preclude its use. Existing monoclonals are not effective with newer variants.”
The drug’s apparent anti-inflammatory properties seem to be key, she said. According to her, the next steps should be “testing lower doses to see if they remain effective, following patients long term to see what impact there is on long COVID symptoms, testing related medications in the drug class to see if they also show an effect, and testing in vaccinated people and with newer variants.”
In an interview, biostatistician James Watson, PhD, of the Mahidol Oxford Tropical Medicine Research Unit, Bangkok, Thailand, and Nuffield department of medicine, University of Oxford, England, said the findings of the analysis are “not overwhelming data.”
He noted the TOGETHER study’s unusual focus on ED visits that latest fewer than 6 hours, which he described as “not a very objective endpoint.” The new meta-analysis focused instead on “outcome data on emergency department visits lasting more than 24 hours and used this as a more representative proxy for hospital admission than an ED visit alone.”
Dr. Fichtenbaum also highlighted the odd endpoint. “Most of us would have chosen something like use of oxygen, requirement for ventilation, or death,” he said. “There are many reasons why people go to the ED. This endpoint is not very strong.”
He also noted that the three studies “are very different in design and endpoints.”
Jeffrey S. Morris, PhD, a biostatistician at the University of Pennsylvania, Philadelphia, offered a different perspective about the findings in an interview. “There’s good evidence that it helps some,” he said, and may reduce hospitalizations by 10%. “If the pill is super cheap and toxicity is very acceptable, it’s not adding additional risk. Most clinicians would say that: ‘If I’m reducing risk by 10%, it’s worthwhile.’ ”
No funding was reported. Two authors report having a patent application filed by Washington University for methods of treating COVID-19 during the conduct of the study. Dr. Watson is an investigator for studies analyzing antiviral drugs and Prozac as COVID-19 treatments. Dr. Fichtenbaum and Dr. Morris disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
a new systematic review and meta-analysis has found. But outside experts differ over whether the evidence from just three studies is strong enough to warrant adding the drug to the COVID-19 armamentarium.
The report, published online in JAMA Network Open, looked at three studies and estimated that the drug could reduce the relative risk of hospitalization by around 25% (likelihood of moderate effect, 81.6%-91.8%), depending on the type of analysis used.
“This research might be valuable, but the jury remains out until several other adequately powered and designed trials are completed,” said infectious disease specialist Carl J. Fichtenbaum, MD, of the University of Cincinnati, who’s familiar with the findings. “I’m not sure how useful this is given we have several antiviral agents available. Why would we choose this over Paxlovid, remdesivir, or molnupiravir?”
According to Dr. Fichtenbaum, researchers began focusing on fluvoxamine after case reports about patients improving while on the medication. This led to further interest, he said, boosted by the drug’s known ability to dampen the immune system.
A Silicon Valley investor and antivaccine activist named Steve Kirsch has been pushing the drug along with the debunked treatment hydroxychloroquine. He’s accused the government of a cover-up of fluvoxamine’s worth, according to MIT Technology Review, and he wrote a commentary that referred to the drug as “the fast, easy, safe, simple, low-cost solution to COVID that works 100% of the time that nobody wants to talk about.”
For the new analysis, researchers examined three randomized clinical trials with a total of 2,196 participants. The most extensive trial, the TOGETHER study in Brazil (n = 1,497), focused on an unusual outcome: It linked the drug to a 32% reduction in relative risk of patients with COVID-19 being hospitalized in an ED for fewer than 6 hours or transferred to a tertiary hospital because of the disease.
Another study, the STOP COVID 2 trial in the United States and Canada (n = 547), was stopped because too few patients could be recruited to provide useful results. The initial phase of this trial, STOP COVID 1 (n = 152), was also included in the analysis.
All participants in the three studies were unvaccinated. Their median age was 46-50 years, 55%-72% were women, and 44%-56% were obese. Most were multiracial due to the high number of participants from Brazil.
“In the Bayesian analyses, the pooled risk ratio in favor of fluvoxamine was 0.78 (95% confidence interval, 0.58-1.08) for the weakly neutral prior and 0.73 (95% CI, 0.53-1.01) for the moderately optimistic prior,” the researchers reported, referring to a reduction in risk of hospitalization. “In the frequentist meta-analysis, the pooled risk ratio in favor of fluvoxamine was 0.75 (95% CI, 0.58-0.97; I2, 0.2%).”
Two of the authors of the new analysis were also coauthors of the TOGETHER trial and both STOP COVID trials.
Corresponding author Emily G. McDonald, MD, division of experimental medicine at McGill University, Montreal, said in an interview that the findings show fluvoxamine “very likely reduces hospitalization in high-risk outpatient adults with COVID-19. This effect varies depending on your baseline risk of developing complications in the first place.”
Dr. McDonald added that “fluvoxamine is an option to reduce hospitalizations in high-risk adults. It is likely effective, is inexpensive, and has a long safety track record.” She also noted that “not all countries have access to Paxlovid, and some people have drug interactions that preclude its use. Existing monoclonals are not effective with newer variants.”
The drug’s apparent anti-inflammatory properties seem to be key, she said. According to her, the next steps should be “testing lower doses to see if they remain effective, following patients long term to see what impact there is on long COVID symptoms, testing related medications in the drug class to see if they also show an effect, and testing in vaccinated people and with newer variants.”
In an interview, biostatistician James Watson, PhD, of the Mahidol Oxford Tropical Medicine Research Unit, Bangkok, Thailand, and Nuffield department of medicine, University of Oxford, England, said the findings of the analysis are “not overwhelming data.”
He noted the TOGETHER study’s unusual focus on ED visits that latest fewer than 6 hours, which he described as “not a very objective endpoint.” The new meta-analysis focused instead on “outcome data on emergency department visits lasting more than 24 hours and used this as a more representative proxy for hospital admission than an ED visit alone.”
Dr. Fichtenbaum also highlighted the odd endpoint. “Most of us would have chosen something like use of oxygen, requirement for ventilation, or death,” he said. “There are many reasons why people go to the ED. This endpoint is not very strong.”
He also noted that the three studies “are very different in design and endpoints.”
Jeffrey S. Morris, PhD, a biostatistician at the University of Pennsylvania, Philadelphia, offered a different perspective about the findings in an interview. “There’s good evidence that it helps some,” he said, and may reduce hospitalizations by 10%. “If the pill is super cheap and toxicity is very acceptable, it’s not adding additional risk. Most clinicians would say that: ‘If I’m reducing risk by 10%, it’s worthwhile.’ ”
No funding was reported. Two authors report having a patent application filed by Washington University for methods of treating COVID-19 during the conduct of the study. Dr. Watson is an investigator for studies analyzing antiviral drugs and Prozac as COVID-19 treatments. Dr. Fichtenbaum and Dr. Morris disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
COVID cases rising in about half of states
About half the states have reported increases in COVID cases fueled by the Omicron subvariant, Axios reported. Alaska, Vermont, and Rhode Island had the highest increases, with more than 20 new cases per 100,000 people.
Nationally, the statistics are encouraging, with the 7-day average of daily cases around 26,000 on April 6, down from around 41,000 on March 6, according to the Centers for Disease Control and Prevention. The number of deaths has dropped to an average of around 600 a day, down 34% from 2 weeks ago.
National health officials have said some spots would have a lot of COVID cases.
“Looking across the country, we see that 95% of counties are reporting low COVID-19 community levels, which represent over 97% of the U.S. population,” CDC Director Rochelle Walensky, MD, said April 5 at a White House news briefing.
“If we look more closely at the local level, we find a handful of counties where we are seeing increases in both cases and markers of more severe disease, like hospitalizations and in-patient bed capacity, which have resulted in an increased COVID-19 community level in some areas.”
Meanwhile, the Commonwealth Fund issued a report April 8 saying the U.S. vaccine program had prevented an estimated 2.2 million deaths and 17 million hospitalizations.
If the vaccine program didn’t exist, the United States would have had another 66 million COVID infections and spent about $900 billion more on health care, the foundation said.
The United States has reported about 982,000 COVID-related deaths so far with about 80 million COVID cases, according to the CDC.
“Our findings highlight the profound and ongoing impact of the vaccination program in reducing infections, hospitalizations, and deaths,” the Commonwealth Fund said.
“Investing in vaccination programs also has produced substantial cost savings – approximately the size of one-fifth of annual national health expenditures – by dramatically reducing the amount spent on COVID-19 hospitalizations.”
A version of this article first appeared on WebMD.com.
About half the states have reported increases in COVID cases fueled by the Omicron subvariant, Axios reported. Alaska, Vermont, and Rhode Island had the highest increases, with more than 20 new cases per 100,000 people.
Nationally, the statistics are encouraging, with the 7-day average of daily cases around 26,000 on April 6, down from around 41,000 on March 6, according to the Centers for Disease Control and Prevention. The number of deaths has dropped to an average of around 600 a day, down 34% from 2 weeks ago.
National health officials have said some spots would have a lot of COVID cases.
“Looking across the country, we see that 95% of counties are reporting low COVID-19 community levels, which represent over 97% of the U.S. population,” CDC Director Rochelle Walensky, MD, said April 5 at a White House news briefing.
“If we look more closely at the local level, we find a handful of counties where we are seeing increases in both cases and markers of more severe disease, like hospitalizations and in-patient bed capacity, which have resulted in an increased COVID-19 community level in some areas.”
Meanwhile, the Commonwealth Fund issued a report April 8 saying the U.S. vaccine program had prevented an estimated 2.2 million deaths and 17 million hospitalizations.
If the vaccine program didn’t exist, the United States would have had another 66 million COVID infections and spent about $900 billion more on health care, the foundation said.
The United States has reported about 982,000 COVID-related deaths so far with about 80 million COVID cases, according to the CDC.
“Our findings highlight the profound and ongoing impact of the vaccination program in reducing infections, hospitalizations, and deaths,” the Commonwealth Fund said.
“Investing in vaccination programs also has produced substantial cost savings – approximately the size of one-fifth of annual national health expenditures – by dramatically reducing the amount spent on COVID-19 hospitalizations.”
A version of this article first appeared on WebMD.com.
About half the states have reported increases in COVID cases fueled by the Omicron subvariant, Axios reported. Alaska, Vermont, and Rhode Island had the highest increases, with more than 20 new cases per 100,000 people.
Nationally, the statistics are encouraging, with the 7-day average of daily cases around 26,000 on April 6, down from around 41,000 on March 6, according to the Centers for Disease Control and Prevention. The number of deaths has dropped to an average of around 600 a day, down 34% from 2 weeks ago.
National health officials have said some spots would have a lot of COVID cases.
“Looking across the country, we see that 95% of counties are reporting low COVID-19 community levels, which represent over 97% of the U.S. population,” CDC Director Rochelle Walensky, MD, said April 5 at a White House news briefing.
“If we look more closely at the local level, we find a handful of counties where we are seeing increases in both cases and markers of more severe disease, like hospitalizations and in-patient bed capacity, which have resulted in an increased COVID-19 community level in some areas.”
Meanwhile, the Commonwealth Fund issued a report April 8 saying the U.S. vaccine program had prevented an estimated 2.2 million deaths and 17 million hospitalizations.
If the vaccine program didn’t exist, the United States would have had another 66 million COVID infections and spent about $900 billion more on health care, the foundation said.
The United States has reported about 982,000 COVID-related deaths so far with about 80 million COVID cases, according to the CDC.
“Our findings highlight the profound and ongoing impact of the vaccination program in reducing infections, hospitalizations, and deaths,” the Commonwealth Fund said.
“Investing in vaccination programs also has produced substantial cost savings – approximately the size of one-fifth of annual national health expenditures – by dramatically reducing the amount spent on COVID-19 hospitalizations.”
A version of this article first appeared on WebMD.com.