User login
Reimbursement for Mohs Surgery Not Keeping Up With Health Care Costs
PHOENIX — Medicare cuts, which are expected to continue, have exacerbated this issue even further.
“This ongoing downward trend in inflation-adjusted reimbursement may lead to delayed patient access to quality dermatology surgical care,” said lead study author Lily Park, DO, a resident in the Department of Dermatology, Larkin Community Hospital, Miami. “This trend will lead to reduced access.”
Dr. Park emphasized that reimbursement for Mohs surgery has also been further affected by the multiple surgery reduction rule, which is where Medicare pays less for the second and subsequent procedures performed during the same patient encounter. Reductions may be calculated in several ways, depending on what kind of procedure or service is involved.
“The Mohs surgery community needs to be aware of this financial trend and actively engage with healthcare policymakers to ensure the establishment of a sustainable payment infrastructure,” she said.
Dr. Park presented the study results at the annual meeting of the American College of Mohs Surgery.
The landscape of healthcare economics continues to evolve and has been further complicated by rising inflation. In addition, a 2% cut to the Medicare payment conversion factor was implemented in 2023, followed by a further 3.37% cut in early 2024 — which was cut by half in March 2024, with an additional cut expected this year, she noted. “This has presented more challenges for dermatologic surgeons who are already dealing with the rising healthcare costs.”
However, despite these financial challenges, there is a lack of research on physician reimbursement for dermatologic procedures, including surgery.
Decreased Reimbursement for All Procedures
Dr. Park and colleagues analyzed trends in Medicare reimbursement rates for Mohs micrographic surgery and several other common dermatologic procedures. Beginning with 2007, they calculated the inflation-adjusted values for each year’s non-facility prices for all codes except Current Procedural Terminology (CPT) codes 11102 and 11104. For those two codes, inflation-adjusted prices were based on the prices in 2019, the year when the codes were first introduced. The authors estimated the inflation adjusted value for each year based on the non-facility price for 2007, and the difference between the rate of inflation and the change in physician reimbursement over time was calculated.
The six most commonly performed procedures in 2023, ranked in descending order of frequency, were identified as CPT 17000, used for the removal of actinic keratosis; CPT 11102, used for biopsy of skin; CPT 17110, used for the destruction of benign lesions such as seborrheic keratoses and warts; CPT 17311, used for the destruction of malignant lesions (including Mohs surgery); CPT 11104, which is also related to biopsy of skin; and CPT 10060, used for incision and drainage of abscess.
Their analysis showed that all CPT codes experienced a decline when compared with their inflation-adjusted values. Both Mohs surgery (17311) and benign destruction of premalignant lesions (17000) showed a decrease of 46% in reimbursement during an inflation-adjusted 18-year time span between 2007 and 2024.
When adjusted for inflation, Dr. Park noted, reimbursement for CPT 17311 and 17000 should actually be increased by 42% and 41% in 2024, respectively. The greatest declines in reimbursement were seen during the last 4 years.
“Future legislation, such as H.R.2474, a bill proposing inflation-based updates to physician pay, would aid us in the future if implemented,” said Dr. Park.
Dangerous Trend
The finding that payments have declined for many common dermatologic procedures since 2007 “is particularly important given the rising cost of healthcare delivery,” said Jesse M. Lewin, MD, who was asked to comment on the study results. “The administrative burden of electronic medical records, filing, and following up insurance claims has necessitated the employment of more non-physician staff to support these tasks,” he told this news organization.
“Declining reimbursement for Mohs surgery and other cancer-related procedures is a dangerous trend, as the ultimate impact will be the effect it has on quality and accessibility of skin cancer care for patients,” added Dr. Lewin, chief of Mohs micrographic and dermatologic surgery and vice chair of surgical operations at the Icahn School of Medicine at Mount Sinai, New York City. “This is an important study that reinforces physician engagement in healthcare policy and legislation to advocate for our specialty and patients.”
The study was independently supported. Dr. Park and Dr. Lewin, who was not involved with the study, reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
PHOENIX — Medicare cuts, which are expected to continue, have exacerbated this issue even further.
“This ongoing downward trend in inflation-adjusted reimbursement may lead to delayed patient access to quality dermatology surgical care,” said lead study author Lily Park, DO, a resident in the Department of Dermatology, Larkin Community Hospital, Miami. “This trend will lead to reduced access.”
Dr. Park emphasized that reimbursement for Mohs surgery has also been further affected by the multiple surgery reduction rule, which is where Medicare pays less for the second and subsequent procedures performed during the same patient encounter. Reductions may be calculated in several ways, depending on what kind of procedure or service is involved.
“The Mohs surgery community needs to be aware of this financial trend and actively engage with healthcare policymakers to ensure the establishment of a sustainable payment infrastructure,” she said.
Dr. Park presented the study results at the annual meeting of the American College of Mohs Surgery.
The landscape of healthcare economics continues to evolve and has been further complicated by rising inflation. In addition, a 2% cut to the Medicare payment conversion factor was implemented in 2023, followed by a further 3.37% cut in early 2024 — which was cut by half in March 2024, with an additional cut expected this year, she noted. “This has presented more challenges for dermatologic surgeons who are already dealing with the rising healthcare costs.”
However, despite these financial challenges, there is a lack of research on physician reimbursement for dermatologic procedures, including surgery.
Decreased Reimbursement for All Procedures
Dr. Park and colleagues analyzed trends in Medicare reimbursement rates for Mohs micrographic surgery and several other common dermatologic procedures. Beginning with 2007, they calculated the inflation-adjusted values for each year’s non-facility prices for all codes except Current Procedural Terminology (CPT) codes 11102 and 11104. For those two codes, inflation-adjusted prices were based on the prices in 2019, the year when the codes were first introduced. The authors estimated the inflation adjusted value for each year based on the non-facility price for 2007, and the difference between the rate of inflation and the change in physician reimbursement over time was calculated.
The six most commonly performed procedures in 2023, ranked in descending order of frequency, were identified as CPT 17000, used for the removal of actinic keratosis; CPT 11102, used for biopsy of skin; CPT 17110, used for the destruction of benign lesions such as seborrheic keratoses and warts; CPT 17311, used for the destruction of malignant lesions (including Mohs surgery); CPT 11104, which is also related to biopsy of skin; and CPT 10060, used for incision and drainage of abscess.
Their analysis showed that all CPT codes experienced a decline when compared with their inflation-adjusted values. Both Mohs surgery (17311) and benign destruction of premalignant lesions (17000) showed a decrease of 46% in reimbursement during an inflation-adjusted 18-year time span between 2007 and 2024.
When adjusted for inflation, Dr. Park noted, reimbursement for CPT 17311 and 17000 should actually be increased by 42% and 41% in 2024, respectively. The greatest declines in reimbursement were seen during the last 4 years.
“Future legislation, such as H.R.2474, a bill proposing inflation-based updates to physician pay, would aid us in the future if implemented,” said Dr. Park.
Dangerous Trend
The finding that payments have declined for many common dermatologic procedures since 2007 “is particularly important given the rising cost of healthcare delivery,” said Jesse M. Lewin, MD, who was asked to comment on the study results. “The administrative burden of electronic medical records, filing, and following up insurance claims has necessitated the employment of more non-physician staff to support these tasks,” he told this news organization.
“Declining reimbursement for Mohs surgery and other cancer-related procedures is a dangerous trend, as the ultimate impact will be the effect it has on quality and accessibility of skin cancer care for patients,” added Dr. Lewin, chief of Mohs micrographic and dermatologic surgery and vice chair of surgical operations at the Icahn School of Medicine at Mount Sinai, New York City. “This is an important study that reinforces physician engagement in healthcare policy and legislation to advocate for our specialty and patients.”
The study was independently supported. Dr. Park and Dr. Lewin, who was not involved with the study, reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
PHOENIX — Medicare cuts, which are expected to continue, have exacerbated this issue even further.
“This ongoing downward trend in inflation-adjusted reimbursement may lead to delayed patient access to quality dermatology surgical care,” said lead study author Lily Park, DO, a resident in the Department of Dermatology, Larkin Community Hospital, Miami. “This trend will lead to reduced access.”
Dr. Park emphasized that reimbursement for Mohs surgery has also been further affected by the multiple surgery reduction rule, which is where Medicare pays less for the second and subsequent procedures performed during the same patient encounter. Reductions may be calculated in several ways, depending on what kind of procedure or service is involved.
“The Mohs surgery community needs to be aware of this financial trend and actively engage with healthcare policymakers to ensure the establishment of a sustainable payment infrastructure,” she said.
Dr. Park presented the study results at the annual meeting of the American College of Mohs Surgery.
The landscape of healthcare economics continues to evolve and has been further complicated by rising inflation. In addition, a 2% cut to the Medicare payment conversion factor was implemented in 2023, followed by a further 3.37% cut in early 2024 — which was cut by half in March 2024, with an additional cut expected this year, she noted. “This has presented more challenges for dermatologic surgeons who are already dealing with the rising healthcare costs.”
However, despite these financial challenges, there is a lack of research on physician reimbursement for dermatologic procedures, including surgery.
Decreased Reimbursement for All Procedures
Dr. Park and colleagues analyzed trends in Medicare reimbursement rates for Mohs micrographic surgery and several other common dermatologic procedures. Beginning with 2007, they calculated the inflation-adjusted values for each year’s non-facility prices for all codes except Current Procedural Terminology (CPT) codes 11102 and 11104. For those two codes, inflation-adjusted prices were based on the prices in 2019, the year when the codes were first introduced. The authors estimated the inflation adjusted value for each year based on the non-facility price for 2007, and the difference between the rate of inflation and the change in physician reimbursement over time was calculated.
The six most commonly performed procedures in 2023, ranked in descending order of frequency, were identified as CPT 17000, used for the removal of actinic keratosis; CPT 11102, used for biopsy of skin; CPT 17110, used for the destruction of benign lesions such as seborrheic keratoses and warts; CPT 17311, used for the destruction of malignant lesions (including Mohs surgery); CPT 11104, which is also related to biopsy of skin; and CPT 10060, used for incision and drainage of abscess.
Their analysis showed that all CPT codes experienced a decline when compared with their inflation-adjusted values. Both Mohs surgery (17311) and benign destruction of premalignant lesions (17000) showed a decrease of 46% in reimbursement during an inflation-adjusted 18-year time span between 2007 and 2024.
When adjusted for inflation, Dr. Park noted, reimbursement for CPT 17311 and 17000 should actually be increased by 42% and 41% in 2024, respectively. The greatest declines in reimbursement were seen during the last 4 years.
“Future legislation, such as H.R.2474, a bill proposing inflation-based updates to physician pay, would aid us in the future if implemented,” said Dr. Park.
Dangerous Trend
The finding that payments have declined for many common dermatologic procedures since 2007 “is particularly important given the rising cost of healthcare delivery,” said Jesse M. Lewin, MD, who was asked to comment on the study results. “The administrative burden of electronic medical records, filing, and following up insurance claims has necessitated the employment of more non-physician staff to support these tasks,” he told this news organization.
“Declining reimbursement for Mohs surgery and other cancer-related procedures is a dangerous trend, as the ultimate impact will be the effect it has on quality and accessibility of skin cancer care for patients,” added Dr. Lewin, chief of Mohs micrographic and dermatologic surgery and vice chair of surgical operations at the Icahn School of Medicine at Mount Sinai, New York City. “This is an important study that reinforces physician engagement in healthcare policy and legislation to advocate for our specialty and patients.”
The study was independently supported. Dr. Park and Dr. Lewin, who was not involved with the study, reported no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM ACMS 2024
Will the Federal Non-Compete Ban Take Effect?
(with very limited exceptions). The final rule will not go into effect until 120 days after its publication in the Federal Register, which took place on May 7, and numerous legal challenges appear to be on the horizon.
The principal components of the rule are as follows:
- After the effective date, most non-compete agreements (which prevent departing employees from signing with a new employer for a defined period within a specific geographic area) are banned nationwide.
- The rule exempts certain “senior executives,” ie individuals who earn more than $151,164 annually and serve in policy-making positions.
- There is another major exception for non-competes connected with a sale of a business.
- While not explicitly stated, the rule arguably exempts non-profits, tax-exempt hospitals, and other tax-exempt entities.
- Employers must provide verbal and written notice to employees regarding existing agreements, which would be voided under the rule.
The final rule is the latest skirmish in an ongoing, years-long debate. Twelve states have already put non-compete bans in place, according to a recent paper, and they may serve as a harbinger of things to come should the federal ban go into effect. Each state rule varies in its specifics as states respond to local market conditions. While some states ban all non-compete agreements outright, others limit them based on variables, such as income and employment circumstances. Of course, should the federal ban take effect, it will supersede whatever rules the individual states have in place.
In drafting the rule, the FTC reasoned that non-compete clauses constitute restraint of trade, and eliminating them could potentially increase worker earnings as well as lower health care costs by billions of dollars. In its statements on the proposed ban, the FTC claimed that it could lower health spending across the board by almost $150 billion per year and return $300 million to workers each year in earnings. The agency cited a large body of research that non-competes make it harder for workers to move between jobs and can raise prices for goods and services, while suppressing wages for workers and inhibiting the creation of new businesses.
Most physicians affected by non-compete agreements heavily favor the new rule, because it would give them more control over their careers and expand their practice and income opportunities. It would allow them to get a new job with a competing organization, bucking a long-standing trend that hospitals and health care systems have heavily relied on to keep staff in place.
The rule would, however, keep in place “non-solicitation” rules that many health care organizations have put in place. That means that if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to join him or her at the new employment venue.
Within that clause, however, the FTC has specified that if such non-solicitation agreement has the “equivalent effect” of a non-compete, the agency would deem it such. That means, even if that rule stands, it could be contested and may be interpreted as violating the non-compete provision. So, there is value in reading all the fine print should the rule move forward.
Physicians in independent practices who employ physician assistants and nurse practitioners have expressed concerns that their expensively trained employees might be tempted to accept a nearby, higher-paying position. The “non-solicitation” clause would theoretically prevent them from taking patients and co-workers with them — unless it were successfully contested. Many questions remain.
Further complicating the non-compete ban issue is how it might impact nonprofit institutions. Most hospitals structured as nonprofits would theoretically be exempt from the rule, although it is not specifically stated in the rule itself, because the FTC Act gives the Commission jurisdiction over for-profit companies only. This would obviously create an unfair advantage for nonprofits, who could continue writing non-compete clauses with impunity.
All of these questions may be moot, of course, because a number of powerful entities with deep pockets have lined up in opposition to the rule. Some of them have even questioned the FTC’s authority to pass the rule at all, on the grounds that Section 5 of the FTC Act does not give it the authority to police labor markets. A lawsuit has already been filed by the US Chamber of Commerce. Other large groups in opposition are the American Medical Group Association, the American Hospital Association, and numerous large hospital and healthcare networks.
Only time will tell whether this issue will be regulated on a national level or remain the purview of each individual state.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
(with very limited exceptions). The final rule will not go into effect until 120 days after its publication in the Federal Register, which took place on May 7, and numerous legal challenges appear to be on the horizon.
The principal components of the rule are as follows:
- After the effective date, most non-compete agreements (which prevent departing employees from signing with a new employer for a defined period within a specific geographic area) are banned nationwide.
- The rule exempts certain “senior executives,” ie individuals who earn more than $151,164 annually and serve in policy-making positions.
- There is another major exception for non-competes connected with a sale of a business.
- While not explicitly stated, the rule arguably exempts non-profits, tax-exempt hospitals, and other tax-exempt entities.
- Employers must provide verbal and written notice to employees regarding existing agreements, which would be voided under the rule.
The final rule is the latest skirmish in an ongoing, years-long debate. Twelve states have already put non-compete bans in place, according to a recent paper, and they may serve as a harbinger of things to come should the federal ban go into effect. Each state rule varies in its specifics as states respond to local market conditions. While some states ban all non-compete agreements outright, others limit them based on variables, such as income and employment circumstances. Of course, should the federal ban take effect, it will supersede whatever rules the individual states have in place.
In drafting the rule, the FTC reasoned that non-compete clauses constitute restraint of trade, and eliminating them could potentially increase worker earnings as well as lower health care costs by billions of dollars. In its statements on the proposed ban, the FTC claimed that it could lower health spending across the board by almost $150 billion per year and return $300 million to workers each year in earnings. The agency cited a large body of research that non-competes make it harder for workers to move between jobs and can raise prices for goods and services, while suppressing wages for workers and inhibiting the creation of new businesses.
Most physicians affected by non-compete agreements heavily favor the new rule, because it would give them more control over their careers and expand their practice and income opportunities. It would allow them to get a new job with a competing organization, bucking a long-standing trend that hospitals and health care systems have heavily relied on to keep staff in place.
The rule would, however, keep in place “non-solicitation” rules that many health care organizations have put in place. That means that if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to join him or her at the new employment venue.
Within that clause, however, the FTC has specified that if such non-solicitation agreement has the “equivalent effect” of a non-compete, the agency would deem it such. That means, even if that rule stands, it could be contested and may be interpreted as violating the non-compete provision. So, there is value in reading all the fine print should the rule move forward.
Physicians in independent practices who employ physician assistants and nurse practitioners have expressed concerns that their expensively trained employees might be tempted to accept a nearby, higher-paying position. The “non-solicitation” clause would theoretically prevent them from taking patients and co-workers with them — unless it were successfully contested. Many questions remain.
Further complicating the non-compete ban issue is how it might impact nonprofit institutions. Most hospitals structured as nonprofits would theoretically be exempt from the rule, although it is not specifically stated in the rule itself, because the FTC Act gives the Commission jurisdiction over for-profit companies only. This would obviously create an unfair advantage for nonprofits, who could continue writing non-compete clauses with impunity.
All of these questions may be moot, of course, because a number of powerful entities with deep pockets have lined up in opposition to the rule. Some of them have even questioned the FTC’s authority to pass the rule at all, on the grounds that Section 5 of the FTC Act does not give it the authority to police labor markets. A lawsuit has already been filed by the US Chamber of Commerce. Other large groups in opposition are the American Medical Group Association, the American Hospital Association, and numerous large hospital and healthcare networks.
Only time will tell whether this issue will be regulated on a national level or remain the purview of each individual state.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
(with very limited exceptions). The final rule will not go into effect until 120 days after its publication in the Federal Register, which took place on May 7, and numerous legal challenges appear to be on the horizon.
The principal components of the rule are as follows:
- After the effective date, most non-compete agreements (which prevent departing employees from signing with a new employer for a defined period within a specific geographic area) are banned nationwide.
- The rule exempts certain “senior executives,” ie individuals who earn more than $151,164 annually and serve in policy-making positions.
- There is another major exception for non-competes connected with a sale of a business.
- While not explicitly stated, the rule arguably exempts non-profits, tax-exempt hospitals, and other tax-exempt entities.
- Employers must provide verbal and written notice to employees regarding existing agreements, which would be voided under the rule.
The final rule is the latest skirmish in an ongoing, years-long debate. Twelve states have already put non-compete bans in place, according to a recent paper, and they may serve as a harbinger of things to come should the federal ban go into effect. Each state rule varies in its specifics as states respond to local market conditions. While some states ban all non-compete agreements outright, others limit them based on variables, such as income and employment circumstances. Of course, should the federal ban take effect, it will supersede whatever rules the individual states have in place.
In drafting the rule, the FTC reasoned that non-compete clauses constitute restraint of trade, and eliminating them could potentially increase worker earnings as well as lower health care costs by billions of dollars. In its statements on the proposed ban, the FTC claimed that it could lower health spending across the board by almost $150 billion per year and return $300 million to workers each year in earnings. The agency cited a large body of research that non-competes make it harder for workers to move between jobs and can raise prices for goods and services, while suppressing wages for workers and inhibiting the creation of new businesses.
Most physicians affected by non-compete agreements heavily favor the new rule, because it would give them more control over their careers and expand their practice and income opportunities. It would allow them to get a new job with a competing organization, bucking a long-standing trend that hospitals and health care systems have heavily relied on to keep staff in place.
The rule would, however, keep in place “non-solicitation” rules that many health care organizations have put in place. That means that if a physician leaves an employer, he or she cannot reach out to former patients and colleagues to bring them along or invite them to join him or her at the new employment venue.
Within that clause, however, the FTC has specified that if such non-solicitation agreement has the “equivalent effect” of a non-compete, the agency would deem it such. That means, even if that rule stands, it could be contested and may be interpreted as violating the non-compete provision. So, there is value in reading all the fine print should the rule move forward.
Physicians in independent practices who employ physician assistants and nurse practitioners have expressed concerns that their expensively trained employees might be tempted to accept a nearby, higher-paying position. The “non-solicitation” clause would theoretically prevent them from taking patients and co-workers with them — unless it were successfully contested. Many questions remain.
Further complicating the non-compete ban issue is how it might impact nonprofit institutions. Most hospitals structured as nonprofits would theoretically be exempt from the rule, although it is not specifically stated in the rule itself, because the FTC Act gives the Commission jurisdiction over for-profit companies only. This would obviously create an unfair advantage for nonprofits, who could continue writing non-compete clauses with impunity.
All of these questions may be moot, of course, because a number of powerful entities with deep pockets have lined up in opposition to the rule. Some of them have even questioned the FTC’s authority to pass the rule at all, on the grounds that Section 5 of the FTC Act does not give it the authority to police labor markets. A lawsuit has already been filed by the US Chamber of Commerce. Other large groups in opposition are the American Medical Group Association, the American Hospital Association, and numerous large hospital and healthcare networks.
Only time will tell whether this issue will be regulated on a national level or remain the purview of each individual state.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at dermnews@mdedge.com.
Fluoride, Water, and Kids’ Brains: It’s Complicated
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.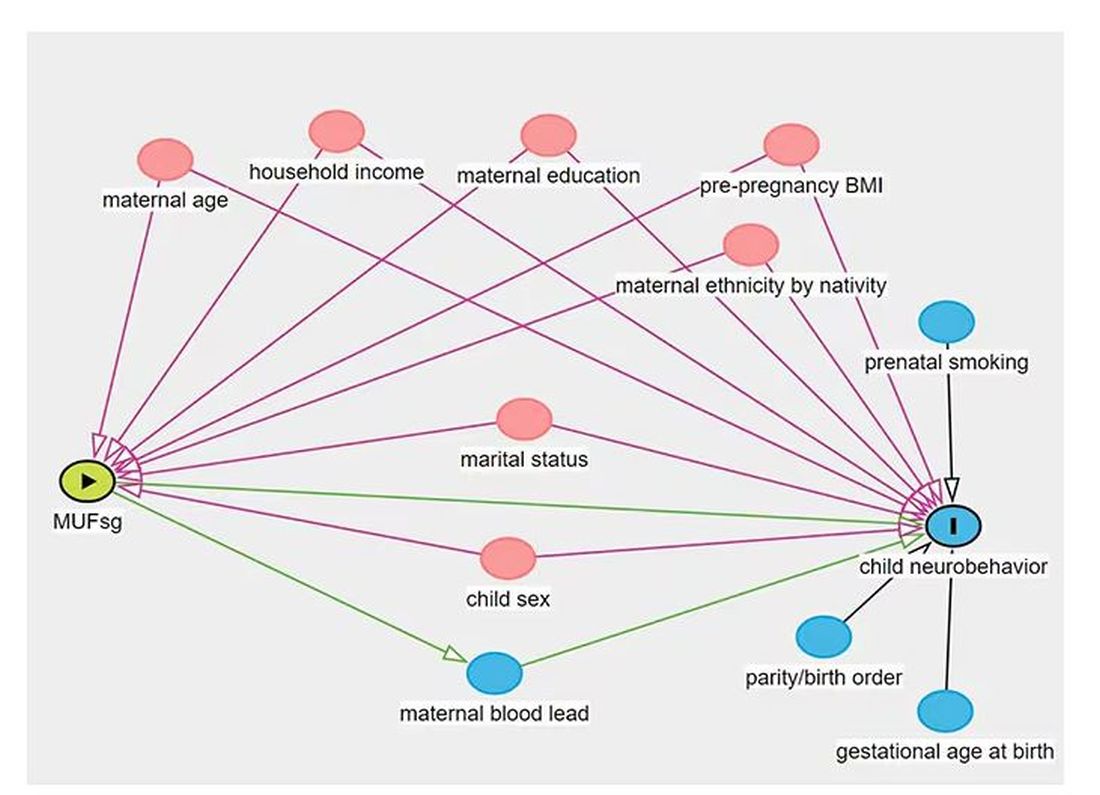
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
This transcript has been edited for clarity.
I recently looked back at my folder full of these medical study commentaries, this weekly video series we call Impact Factor, and realized that I’ve been doing this for a long time. More than 400 articles, believe it or not.
I’ve learned a lot in that time — about medicine, of course — but also about how people react to certain topics. If you’ve been with me this whole time, or even for just a chunk of it, you’ll know that I tend to take a measured approach to most topics. No one study is ever truly definitive, after all. But regardless of how even-keeled I may be, there are some topics that I just know in advance are going to be a bit divisive: studies about gun control; studies about vitamin D; and, of course, studies about fluoride.
Shall We Shake This Hornet’s Nest?
The fluoridation of the US water system began in 1945 with the goal of reducing cavities in the population. The CDC named water fluoridation one of the 10 great public health achievements of the 20th century, along with such inarguable achievements as the recognition of tobacco as a health hazard.
But fluoridation has never been without its detractors. One problem is that the spectrum of beliefs about the potential harm of fluoridation is huge. On one end, you have science-based concerns such as the recognition that excessive fluoride intake can cause fluorosis and stain tooth enamel. I’ll note that the EPA regulates fluoride levels — there is a fair amount of naturally occurring fluoride in water tables around the world — to prevent this. And, of course, on the other end of the spectrum, you have beliefs that are essentially conspiracy theories: “They” add fluoride to the water supply to control us.
The challenge for me is that when one “side” of a scientific debate includes the crazy theories, it can be hard to discuss that whole spectrum, since there are those who will see evidence of any adverse fluoride effect as confirmation that the conspiracy theory is true.
I can’t help this. So I’ll just say this up front: I am about to tell you about a study that shows some potential risk from fluoride exposure. I will tell you up front that there are some significant caveats to the study that call the results into question. And I will tell you up front that no one is controlling your mind, or my mind, with fluoride; they do it with social media.
Let’s Dive Into These Shark-Infested, Fluoridated Waters
We’re talking about the study, “Maternal Urinary Fluoride and Child Neurobehavior at Age 36 Months,” which appears in JAMA Network Open.
It’s a study of 229 mother-child pairs from the Los Angeles area. The moms had their urinary fluoride level measured once before 30 weeks of gestation. A neurobehavioral battery called the Preschool Child Behavior Checklist was administered to the children at age 36 months.
The main thing you’ll hear about this study — in headlines, Facebook posts, and manifestos locked in drawers somewhere — is the primary result: A 0.68-mg/L increase in urinary fluoride in the mothers, about 25 percentile points, was associated with a doubling of the risk for neurobehavioral problems in their kids when they were 3 years old.
Yikes.
But this is not a randomized trial. Researchers didn’t randomly assign some women to have high fluoride intake and some women to have low fluoride intake. They knew that other factors that might lead to neurobehavioral problems could also lead to higher fluoride intake. They represent these factors in what’s known as a directed acyclic graph, as seen here, and account for them statistically using a regression equation.
Not represented here are neighborhood characteristics. Los Angeles does not have uniformly fluoridated water, and neurobehavioral problems in kids are strongly linked to stressors in their environments. Fluoride level could be an innocent bystander.
I’m really just describing the classic issue of correlation versus causation here, the bane of all observational research and — let’s be honest — a bit of a crutch that allows us to disregard the results of studies we don’t like, provided the study wasn’t a randomized trial.
But I have a deeper issue with this study than the old “failure to adjust for relevant confounders” thing, as important as that is.
The exposure of interest in this study is maternal urinary fluoride, as measured in a spot sample. It’s not often that I get to go deep on nephrology in this space, but let’s think about that for a second. Let’s assume for a moment that fluoride is toxic to the developing fetal brain, the main concern raised by the results of the study. How would that work? Presumably, mom would be ingesting fluoride from various sources (like the water supply), and that fluoride would get into her blood, and from her blood across the placenta to the baby’s blood, and into the baby’s brain.
Is Urinary Fluoride a Good Measure of Blood Fluoride?
It’s not great. Empirically, we have data that tell us that levels of urine fluoride are not all that similar to levels of serum fluoride. In 2014, a study investigated the correlation between urine and serum fluoride in a cohort of 60 schoolchildren and found a correlation coefficient of around 0.5.
Why isn’t urine fluoride a great proxy for serum fluoride? The most obvious reason is the urine concentration. Human urine concentration can range from about 50 mmol to 1200 mmol (a 24-fold difference) depending on hydration status. Over the course of 24 hours, for example, the amount of fluoride you put out in your urine may be fairly stable in relation to intake, but for a spot urine sample it would be wildly variable. The authors know this, of course, and so they divide the measured urine fluoride by the specific gravity of the urine to give a sort of “dilution adjusted” value. That’s what is actually used in this study. But specific gravity is, itself, an imperfect measure of how dilute the urine is.
This is something that comes up a lot in urinary biomarker research and it’s not that hard to get around. The best thing would be to just measure blood levels of fluoride. The second best option is 24-hour fluoride excretion. After that, the next best thing would be to adjust the spot concentration by other markers of urinary dilution — creatinine or osmolality — as sensitivity analyses. Any of these approaches would lend credence to the results of the study.
Urinary fluoride excretion is pH dependent. The more acidic the urine, the less fluoride is excreted. Many things — including, importantly, diet — affect urine pH. And it is not a stretch to think that diet may also affect the developing fetus. Neither urine pH nor dietary habits were accounted for in this study.
So, here we are. We have an observational study suggesting a harm that may be associated with fluoride. There may be a causal link here, in which case we need further studies to weigh the harm against the more well-established public health benefit. Or, this is all correlation — an illusion created by the limitations of observational data, and the unique challenges of estimating intake from a single urine sample. In other words, this study has something for everyone, fluoride boosters and skeptics alike. Let the arguments begin. But, if possible, leave me out of it.
Dr. Wilson is associate professor of medicine and public health and director of the Clinical and Translational Research Accelerator at Yale University, New Haven, Conn. He has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Ob.Gyns. Can Help Patients Manage Weight With Anti-Obesity Medications
SAN FRANCISCO — An estimated two out of five adult women in the United States have obesity, and given the potential challenges of losing pregnancy weight postpartum or staving off the weight gain associated with menopause, women are likely to be receptive toward weight management help from their ob.gyns. A whole new armamentarium of anti-obesity medications has become available in the past decade, providing physicians and patients with more treatment options.
Ob.gyns. are therefore well-poised to offer counseling and treatment for obesity management for their patients, Johanna G. Finkle, MD, clinical assistant professor of obstetrics and gynecology and a weight management specialist at the University of Kansas Heath System, told attendees at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. Dr. Finkle provided an extensive overview of what ob.gyns. need to know if they are interested in prescribing anti-obesity medications or simply providing their patients with information about the available drugs.
Kitila S. Heyward, MD, an ob.gyn. at Atrium Health in Monroe, North Carolina, who attended the talk, tries to prescribe anti-obesity medications but has run into roadblocks that Dr. Finkle’s talk helped her understand how to overcome.
“I thought it was very helpful because [I] and one of my midwives, in practice, have been trying to get things prescribed, and we can’t figure out the loopholes,” Dr. Heyward said. “Also, the failure rates are really helpful to us so that we know how to counsel people.”
Even for clinicians who aren’t prescribing these medications, Dr. Heyward said the talk was illuminating. “It offered a better understanding of the medications that your patients are on and how it can affect things like birth control, management of surgery, pregnancy, and things along those lines from a clinical day-by-day standpoint,” she said.
Starting With the Basics
Dr. Finkle began by emphasizing the importance of using patient-first language in discussing obesity, which means using terms such as “weight, excess weight, overweight, body mass index,” and “affected by obesity” instead of “obese, morbidly obese, heaviness, or large.” She also cited the Obesity Medicine Association’s definition of obesity: “a chronic, relapsing and treatable multifactorial, neurobehavioral disease, wherein an increase in body fat promotes adipose tissue dysfunction and abnormal fat mass physical forces, resulting in adverse metabolic, biomechanical, and psychosocial health consequences.”
Though Dr. Finkle acknowledged the limitations of relying on BMI for defining obesity, it remains the standard tool in current practice, with a BMI of 25-29.9 defining overweight and a BMI of 30 or greater defining obesity. Other diagnostic criteria for obesity in women, however, include a percentage body fat over 32% or a waist circumference of more than 35 inches.
“Women are at risk for weight gain through their entire lifespan” Dr. Finkle said, and in women with polycystic ovarian syndrome, 60%-80% have pre-obesity or obesity. In menopause, the triple threat of decreased estrogen, decreased activity, and changes in diet all contribute to obesity risk and no evidence suggests that hormone therapy can prevent weight gain.
Healthy nutrition, physical activity, and behavioral modification remain key pillars of weight management, but interventions such as surgery or medications are also important tools, she said.
“One size does not fit all in terms of treatment,” Dr. Finkle said. ”When I talk to a patient, I think about other medical complications that I can treat with these medications.”
Women for whom anti-obesity medications may be indicated are those with a BMI of 30 or greater, and those with a BMI of at least 27 along with at least one obesity-related comorbidity, such as hypertension, high cholesterol, diabetes, or sleep apnea. The goal of treating obesity with medication is at least a 5%-10% reduction of body weight.
Three Pharmacotherapy Categories
Dr. Finkle reviewed three basic categories of anti-obesity medications: Food and Drug Administration–approved short-term and long-term medications and then off-label drugs that can also aid in healthy weight loss. Short-term options include phentermine, diethylpropion, phendimetrazine, and benzphetamine. Long-term options include orlistat, phentermine/topiramate ER, naltrexone HCl/bupropion HCl ER, and the three GLP-1 receptor agonist drugs, liraglutide, semaglutide, and tirzepatide.
The short-term medications are stimulants that increase satiety, but adverse effects can include tachycardia, hypertension, insomnia, dry mouth, constipation, and diarrhea.
These medications are contraindicated for anyone with uncontrolled hypertension, hyperthyroidism, cardiovascular disease, MAOI use, glaucoma, or history of substance use. The goal is a 5% weight loss in 3 months, and 3 months is the maximum prescribing term.
Then Dr. Finkle reviewed the side effects and contraindications for the oral long-term medications. Orlistat, which can aid in up to 5% weight loss, can result in oily stools and fecal incontinence and is contraindicated for people with chronic malabsorption or cholestasis.
Phentermine/topiramate ER, which can aid in up to 10% weight loss, can result in hypertension, paresthesia, or constipation, and is contraindicated for those with glaucoma, hyperthyroidism, and kidney stones. After the starting dose of 3.75 mg/23 mg, Dr. Finkle increases patients’ dose every 2 weeks, ”but if they’re not tolerating it, if they’re having significant side effects, or they’re losing weight, you do not increase the medication.”
Side effects of naltrexone HCl/bupropion HCl ER, which can lead to 5%-6% weight loss, can include hypertension, suicidal ideation, and glaucoma, and it’s contraindicated in those taking opioids or with a history of seizures or anorexia.
The GLP-1 Receptor Agonists
Next Dr. Finkle discussed the newest but most effective medications, the GLP-1 agonists liraglutide, semaglutide, and tirzepatide. The main contraindications for these drugs are a personal or family history of medullary thyroid cancer, multiple endocrine neoplasia type II syndrome, or any hypersensitivity to this drug class. The two main serious risks are pancreatitis — a 1% risk — and gallstones. Though Dr. Finkle included suicidal ideation as a potential risk of these drugs, the most recent evidence suggests there is no link between suicidal ideation and GLP-1 agonists. The most common side effects are nausea, vomiting, diarrhea, constipation, dyspepsia, and an increased heart rate, though these eventually resolve.
“We always start low with these medications,” Dr. Finkle said, and then titrate the dose up each week, “but if they are having awful side effects, just stay on that dose longer.”
The mechanisms of all three drugs for treating obesity are similar; they work to curb central satiety and slow gastric emptying, though they also have additional mechanisms with benefits for blood glucose levels and for the liver and heart.
- Liraglutide, the first of these drugs approved, is a daily subcutaneous injection that starts at a dose of 0.6 mg and goes up to 3 mg. Patients should lose 4% of weight in 16 weeks or else they are non-responders, Dr. Finkle said.
- Semaglutide, a GLP-1 agonist given as a weekly subcutaneous injection, starts at a dose of 0.25 mg and goes up to 2.4 mg; patients should expect a 5% weight loss in 16 weeks if they are responders. Long term, however, patients lose up to an average 15% of body weight with semaglutide; a third of patients lost more than 20% of body weight in clinical trials, compared with 7%-8% body weight loss with liraglutide. An additional benefit of semaglutide is a 20% reduction in risk of cardiovascular disease.
- Tirzepatide is a combined GLP-1 and GIP agonist, also delivered as a weekly subcutaneous injection, that should result in an estimated 5% weight loss in 16 weeks for responders. But tirzepatide is the most effective of the three, with 91% of patients losing at least 5% body weight and more than half of patients (56%) losing at least 20%.
The big drawbacks to the GLP-1 agonists, however, are their high cost, common lack of insurance coverage, and continued shortages. Dr. Finkle recommended using manufacturer coupons, comparison shopping on Good Rx, and appealing prior authorization requirements to help patients pay for the GLP-1 agonists.
“Drug availability is my second problem. There’s not enough drug,” she said, and her patients often have to call around to different pharmacies to find out which ones are carrying the drug and at what doses. She will sometimes switch their doses as needed based on availability.
It’s also important for physicians to be aware of guidance from the American Society of Anesthesiologists regarding GLP-1 agonist use prior to surgery because of their slowed gastric-emptying mechanism. To reduce the risk of aspiration, patients undergoing general anesthesia should not take liraglutide on the day of surgery, and semaglutide and tirzepatide should be held for 1 week prior to the procedure. New research in JAMA Surgery, however, suggests holding these medications for longer than a week may be wiser.
Getting Patients Started
All the short-term and long-term medications are contraindicated during pregnancy and breastfeeding, Dr. Finkle said. Animal studies with GLP-1 agonists suggest adverse fetal effects when used during pregnancy, but the limited data in human studies so far have not shown a risk of major malformations. Dr. Finkle said the recommendations for now are to stop all GLP-1 receptor agonist drugs 2 months before patients attempt to become pregnant and not to begin them again until after they are no longer breastfeeding.
Finally, Dr. Finkle reviewed off-label medications that can result in modest weight loss, including topiramate, phentermine (not to be used for longer than 12 weeks), bupropion, naltrexone, and metformin. Metformin is likely to result in only 2% weight loss, but it may enhance the effects of GLP-1s, she said.
For ob.gyns. who want to get their patients started on one of these medications, Dr. Finkle first recommends asking patients if it’s okay to discuss their weight. ”Studies show that if you just ask permission to discuss someone’s weight, they go on to lose weight and lose more than someone who has never been asked,” Dr. Finkle said. Then she takes a history.
”When I see a patient, I ask, ‘Tell me why you’re here today,’ ” Dr. Finkle said.
This gives me a lot of insight as to why they’re coming in and it helps me understand where they’re at in terms of other things, such as depression or anxiety with weight, and it helps me to tailor my treatment.”
A full medical history is important for learning about potential contraindications or picking medications that might help with other conditions, such as topiramate for migraines. Finally, Dr. Finkle advises a lab screening with a comprehensive metabolic panel, lipid panel, HbA1c, and vitamin D.
“The [comprehensive metabolic panel] allows me to know about creatinine and liver function,” she said. If these are elevated, she will still prescribe GLP-1s but will monitor the values more closely. “Then I discuss options with the patient. They may be eligible for bariatric surgery or medications. We talk about lifestyle behavioral management, and then I go through the medications and we set goals.”
Goals include nutrition and exercise; start modest and have them work their way up by doing activities they enjoy. In addition, patients taking GLP-1s need to eat enough protein — 80 to 100 grams a day, though she starts them at 60 grams — and do regular muscle strengthening since they can lose muscle mass.
Indications for referral to an obesity medicine specialist are a history of gastric bypass/sleeve surgery, having type 2 diabetes, having an eating disorder, or having failed one of these anti-obesity medications.
Finally, Dr. Finkle reviewed medications that can cause weight gain: medroxyprogesterone acetate for birth control; beta blockers for hypertension or migraine; the antidepressants amitriptyline, paroxetine, venlafaxine, and trazodone; the mood stabilizers gabapentin, lithium, valproate, and carbamazepine; and diphenhydramine and zolpidem for sleep.
No external funding was used for the talk. Dr. Finkle and Dr. Heyward had no disclosures.
SAN FRANCISCO — An estimated two out of five adult women in the United States have obesity, and given the potential challenges of losing pregnancy weight postpartum or staving off the weight gain associated with menopause, women are likely to be receptive toward weight management help from their ob.gyns. A whole new armamentarium of anti-obesity medications has become available in the past decade, providing physicians and patients with more treatment options.
Ob.gyns. are therefore well-poised to offer counseling and treatment for obesity management for their patients, Johanna G. Finkle, MD, clinical assistant professor of obstetrics and gynecology and a weight management specialist at the University of Kansas Heath System, told attendees at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. Dr. Finkle provided an extensive overview of what ob.gyns. need to know if they are interested in prescribing anti-obesity medications or simply providing their patients with information about the available drugs.
Kitila S. Heyward, MD, an ob.gyn. at Atrium Health in Monroe, North Carolina, who attended the talk, tries to prescribe anti-obesity medications but has run into roadblocks that Dr. Finkle’s talk helped her understand how to overcome.
“I thought it was very helpful because [I] and one of my midwives, in practice, have been trying to get things prescribed, and we can’t figure out the loopholes,” Dr. Heyward said. “Also, the failure rates are really helpful to us so that we know how to counsel people.”
Even for clinicians who aren’t prescribing these medications, Dr. Heyward said the talk was illuminating. “It offered a better understanding of the medications that your patients are on and how it can affect things like birth control, management of surgery, pregnancy, and things along those lines from a clinical day-by-day standpoint,” she said.
Starting With the Basics
Dr. Finkle began by emphasizing the importance of using patient-first language in discussing obesity, which means using terms such as “weight, excess weight, overweight, body mass index,” and “affected by obesity” instead of “obese, morbidly obese, heaviness, or large.” She also cited the Obesity Medicine Association’s definition of obesity: “a chronic, relapsing and treatable multifactorial, neurobehavioral disease, wherein an increase in body fat promotes adipose tissue dysfunction and abnormal fat mass physical forces, resulting in adverse metabolic, biomechanical, and psychosocial health consequences.”
Though Dr. Finkle acknowledged the limitations of relying on BMI for defining obesity, it remains the standard tool in current practice, with a BMI of 25-29.9 defining overweight and a BMI of 30 or greater defining obesity. Other diagnostic criteria for obesity in women, however, include a percentage body fat over 32% or a waist circumference of more than 35 inches.
“Women are at risk for weight gain through their entire lifespan” Dr. Finkle said, and in women with polycystic ovarian syndrome, 60%-80% have pre-obesity or obesity. In menopause, the triple threat of decreased estrogen, decreased activity, and changes in diet all contribute to obesity risk and no evidence suggests that hormone therapy can prevent weight gain.
Healthy nutrition, physical activity, and behavioral modification remain key pillars of weight management, but interventions such as surgery or medications are also important tools, she said.
“One size does not fit all in terms of treatment,” Dr. Finkle said. ”When I talk to a patient, I think about other medical complications that I can treat with these medications.”
Women for whom anti-obesity medications may be indicated are those with a BMI of 30 or greater, and those with a BMI of at least 27 along with at least one obesity-related comorbidity, such as hypertension, high cholesterol, diabetes, or sleep apnea. The goal of treating obesity with medication is at least a 5%-10% reduction of body weight.
Three Pharmacotherapy Categories
Dr. Finkle reviewed three basic categories of anti-obesity medications: Food and Drug Administration–approved short-term and long-term medications and then off-label drugs that can also aid in healthy weight loss. Short-term options include phentermine, diethylpropion, phendimetrazine, and benzphetamine. Long-term options include orlistat, phentermine/topiramate ER, naltrexone HCl/bupropion HCl ER, and the three GLP-1 receptor agonist drugs, liraglutide, semaglutide, and tirzepatide.
The short-term medications are stimulants that increase satiety, but adverse effects can include tachycardia, hypertension, insomnia, dry mouth, constipation, and diarrhea.
These medications are contraindicated for anyone with uncontrolled hypertension, hyperthyroidism, cardiovascular disease, MAOI use, glaucoma, or history of substance use. The goal is a 5% weight loss in 3 months, and 3 months is the maximum prescribing term.
Then Dr. Finkle reviewed the side effects and contraindications for the oral long-term medications. Orlistat, which can aid in up to 5% weight loss, can result in oily stools and fecal incontinence and is contraindicated for people with chronic malabsorption or cholestasis.
Phentermine/topiramate ER, which can aid in up to 10% weight loss, can result in hypertension, paresthesia, or constipation, and is contraindicated for those with glaucoma, hyperthyroidism, and kidney stones. After the starting dose of 3.75 mg/23 mg, Dr. Finkle increases patients’ dose every 2 weeks, ”but if they’re not tolerating it, if they’re having significant side effects, or they’re losing weight, you do not increase the medication.”
Side effects of naltrexone HCl/bupropion HCl ER, which can lead to 5%-6% weight loss, can include hypertension, suicidal ideation, and glaucoma, and it’s contraindicated in those taking opioids or with a history of seizures or anorexia.
The GLP-1 Receptor Agonists
Next Dr. Finkle discussed the newest but most effective medications, the GLP-1 agonists liraglutide, semaglutide, and tirzepatide. The main contraindications for these drugs are a personal or family history of medullary thyroid cancer, multiple endocrine neoplasia type II syndrome, or any hypersensitivity to this drug class. The two main serious risks are pancreatitis — a 1% risk — and gallstones. Though Dr. Finkle included suicidal ideation as a potential risk of these drugs, the most recent evidence suggests there is no link between suicidal ideation and GLP-1 agonists. The most common side effects are nausea, vomiting, diarrhea, constipation, dyspepsia, and an increased heart rate, though these eventually resolve.
“We always start low with these medications,” Dr. Finkle said, and then titrate the dose up each week, “but if they are having awful side effects, just stay on that dose longer.”
The mechanisms of all three drugs for treating obesity are similar; they work to curb central satiety and slow gastric emptying, though they also have additional mechanisms with benefits for blood glucose levels and for the liver and heart.
- Liraglutide, the first of these drugs approved, is a daily subcutaneous injection that starts at a dose of 0.6 mg and goes up to 3 mg. Patients should lose 4% of weight in 16 weeks or else they are non-responders, Dr. Finkle said.
- Semaglutide, a GLP-1 agonist given as a weekly subcutaneous injection, starts at a dose of 0.25 mg and goes up to 2.4 mg; patients should expect a 5% weight loss in 16 weeks if they are responders. Long term, however, patients lose up to an average 15% of body weight with semaglutide; a third of patients lost more than 20% of body weight in clinical trials, compared with 7%-8% body weight loss with liraglutide. An additional benefit of semaglutide is a 20% reduction in risk of cardiovascular disease.
- Tirzepatide is a combined GLP-1 and GIP agonist, also delivered as a weekly subcutaneous injection, that should result in an estimated 5% weight loss in 16 weeks for responders. But tirzepatide is the most effective of the three, with 91% of patients losing at least 5% body weight and more than half of patients (56%) losing at least 20%.
The big drawbacks to the GLP-1 agonists, however, are their high cost, common lack of insurance coverage, and continued shortages. Dr. Finkle recommended using manufacturer coupons, comparison shopping on Good Rx, and appealing prior authorization requirements to help patients pay for the GLP-1 agonists.
“Drug availability is my second problem. There’s not enough drug,” she said, and her patients often have to call around to different pharmacies to find out which ones are carrying the drug and at what doses. She will sometimes switch their doses as needed based on availability.
It’s also important for physicians to be aware of guidance from the American Society of Anesthesiologists regarding GLP-1 agonist use prior to surgery because of their slowed gastric-emptying mechanism. To reduce the risk of aspiration, patients undergoing general anesthesia should not take liraglutide on the day of surgery, and semaglutide and tirzepatide should be held for 1 week prior to the procedure. New research in JAMA Surgery, however, suggests holding these medications for longer than a week may be wiser.
Getting Patients Started
All the short-term and long-term medications are contraindicated during pregnancy and breastfeeding, Dr. Finkle said. Animal studies with GLP-1 agonists suggest adverse fetal effects when used during pregnancy, but the limited data in human studies so far have not shown a risk of major malformations. Dr. Finkle said the recommendations for now are to stop all GLP-1 receptor agonist drugs 2 months before patients attempt to become pregnant and not to begin them again until after they are no longer breastfeeding.
Finally, Dr. Finkle reviewed off-label medications that can result in modest weight loss, including topiramate, phentermine (not to be used for longer than 12 weeks), bupropion, naltrexone, and metformin. Metformin is likely to result in only 2% weight loss, but it may enhance the effects of GLP-1s, she said.
For ob.gyns. who want to get their patients started on one of these medications, Dr. Finkle first recommends asking patients if it’s okay to discuss their weight. ”Studies show that if you just ask permission to discuss someone’s weight, they go on to lose weight and lose more than someone who has never been asked,” Dr. Finkle said. Then she takes a history.
”When I see a patient, I ask, ‘Tell me why you’re here today,’ ” Dr. Finkle said.
This gives me a lot of insight as to why they’re coming in and it helps me understand where they’re at in terms of other things, such as depression or anxiety with weight, and it helps me to tailor my treatment.”
A full medical history is important for learning about potential contraindications or picking medications that might help with other conditions, such as topiramate for migraines. Finally, Dr. Finkle advises a lab screening with a comprehensive metabolic panel, lipid panel, HbA1c, and vitamin D.
“The [comprehensive metabolic panel] allows me to know about creatinine and liver function,” she said. If these are elevated, she will still prescribe GLP-1s but will monitor the values more closely. “Then I discuss options with the patient. They may be eligible for bariatric surgery or medications. We talk about lifestyle behavioral management, and then I go through the medications and we set goals.”
Goals include nutrition and exercise; start modest and have them work their way up by doing activities they enjoy. In addition, patients taking GLP-1s need to eat enough protein — 80 to 100 grams a day, though she starts them at 60 grams — and do regular muscle strengthening since they can lose muscle mass.
Indications for referral to an obesity medicine specialist are a history of gastric bypass/sleeve surgery, having type 2 diabetes, having an eating disorder, or having failed one of these anti-obesity medications.
Finally, Dr. Finkle reviewed medications that can cause weight gain: medroxyprogesterone acetate for birth control; beta blockers for hypertension or migraine; the antidepressants amitriptyline, paroxetine, venlafaxine, and trazodone; the mood stabilizers gabapentin, lithium, valproate, and carbamazepine; and diphenhydramine and zolpidem for sleep.
No external funding was used for the talk. Dr. Finkle and Dr. Heyward had no disclosures.
SAN FRANCISCO — An estimated two out of five adult women in the United States have obesity, and given the potential challenges of losing pregnancy weight postpartum or staving off the weight gain associated with menopause, women are likely to be receptive toward weight management help from their ob.gyns. A whole new armamentarium of anti-obesity medications has become available in the past decade, providing physicians and patients with more treatment options.
Ob.gyns. are therefore well-poised to offer counseling and treatment for obesity management for their patients, Johanna G. Finkle, MD, clinical assistant professor of obstetrics and gynecology and a weight management specialist at the University of Kansas Heath System, told attendees at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. Dr. Finkle provided an extensive overview of what ob.gyns. need to know if they are interested in prescribing anti-obesity medications or simply providing their patients with information about the available drugs.
Kitila S. Heyward, MD, an ob.gyn. at Atrium Health in Monroe, North Carolina, who attended the talk, tries to prescribe anti-obesity medications but has run into roadblocks that Dr. Finkle’s talk helped her understand how to overcome.
“I thought it was very helpful because [I] and one of my midwives, in practice, have been trying to get things prescribed, and we can’t figure out the loopholes,” Dr. Heyward said. “Also, the failure rates are really helpful to us so that we know how to counsel people.”
Even for clinicians who aren’t prescribing these medications, Dr. Heyward said the talk was illuminating. “It offered a better understanding of the medications that your patients are on and how it can affect things like birth control, management of surgery, pregnancy, and things along those lines from a clinical day-by-day standpoint,” she said.
Starting With the Basics
Dr. Finkle began by emphasizing the importance of using patient-first language in discussing obesity, which means using terms such as “weight, excess weight, overweight, body mass index,” and “affected by obesity” instead of “obese, morbidly obese, heaviness, or large.” She also cited the Obesity Medicine Association’s definition of obesity: “a chronic, relapsing and treatable multifactorial, neurobehavioral disease, wherein an increase in body fat promotes adipose tissue dysfunction and abnormal fat mass physical forces, resulting in adverse metabolic, biomechanical, and psychosocial health consequences.”
Though Dr. Finkle acknowledged the limitations of relying on BMI for defining obesity, it remains the standard tool in current practice, with a BMI of 25-29.9 defining overweight and a BMI of 30 or greater defining obesity. Other diagnostic criteria for obesity in women, however, include a percentage body fat over 32% or a waist circumference of more than 35 inches.
“Women are at risk for weight gain through their entire lifespan” Dr. Finkle said, and in women with polycystic ovarian syndrome, 60%-80% have pre-obesity or obesity. In menopause, the triple threat of decreased estrogen, decreased activity, and changes in diet all contribute to obesity risk and no evidence suggests that hormone therapy can prevent weight gain.
Healthy nutrition, physical activity, and behavioral modification remain key pillars of weight management, but interventions such as surgery or medications are also important tools, she said.
“One size does not fit all in terms of treatment,” Dr. Finkle said. ”When I talk to a patient, I think about other medical complications that I can treat with these medications.”
Women for whom anti-obesity medications may be indicated are those with a BMI of 30 or greater, and those with a BMI of at least 27 along with at least one obesity-related comorbidity, such as hypertension, high cholesterol, diabetes, or sleep apnea. The goal of treating obesity with medication is at least a 5%-10% reduction of body weight.
Three Pharmacotherapy Categories
Dr. Finkle reviewed three basic categories of anti-obesity medications: Food and Drug Administration–approved short-term and long-term medications and then off-label drugs that can also aid in healthy weight loss. Short-term options include phentermine, diethylpropion, phendimetrazine, and benzphetamine. Long-term options include orlistat, phentermine/topiramate ER, naltrexone HCl/bupropion HCl ER, and the three GLP-1 receptor agonist drugs, liraglutide, semaglutide, and tirzepatide.
The short-term medications are stimulants that increase satiety, but adverse effects can include tachycardia, hypertension, insomnia, dry mouth, constipation, and diarrhea.
These medications are contraindicated for anyone with uncontrolled hypertension, hyperthyroidism, cardiovascular disease, MAOI use, glaucoma, or history of substance use. The goal is a 5% weight loss in 3 months, and 3 months is the maximum prescribing term.
Then Dr. Finkle reviewed the side effects and contraindications for the oral long-term medications. Orlistat, which can aid in up to 5% weight loss, can result in oily stools and fecal incontinence and is contraindicated for people with chronic malabsorption or cholestasis.
Phentermine/topiramate ER, which can aid in up to 10% weight loss, can result in hypertension, paresthesia, or constipation, and is contraindicated for those with glaucoma, hyperthyroidism, and kidney stones. After the starting dose of 3.75 mg/23 mg, Dr. Finkle increases patients’ dose every 2 weeks, ”but if they’re not tolerating it, if they’re having significant side effects, or they’re losing weight, you do not increase the medication.”
Side effects of naltrexone HCl/bupropion HCl ER, which can lead to 5%-6% weight loss, can include hypertension, suicidal ideation, and glaucoma, and it’s contraindicated in those taking opioids or with a history of seizures or anorexia.
The GLP-1 Receptor Agonists
Next Dr. Finkle discussed the newest but most effective medications, the GLP-1 agonists liraglutide, semaglutide, and tirzepatide. The main contraindications for these drugs are a personal or family history of medullary thyroid cancer, multiple endocrine neoplasia type II syndrome, or any hypersensitivity to this drug class. The two main serious risks are pancreatitis — a 1% risk — and gallstones. Though Dr. Finkle included suicidal ideation as a potential risk of these drugs, the most recent evidence suggests there is no link between suicidal ideation and GLP-1 agonists. The most common side effects are nausea, vomiting, diarrhea, constipation, dyspepsia, and an increased heart rate, though these eventually resolve.
“We always start low with these medications,” Dr. Finkle said, and then titrate the dose up each week, “but if they are having awful side effects, just stay on that dose longer.”
The mechanisms of all three drugs for treating obesity are similar; they work to curb central satiety and slow gastric emptying, though they also have additional mechanisms with benefits for blood glucose levels and for the liver and heart.
- Liraglutide, the first of these drugs approved, is a daily subcutaneous injection that starts at a dose of 0.6 mg and goes up to 3 mg. Patients should lose 4% of weight in 16 weeks or else they are non-responders, Dr. Finkle said.
- Semaglutide, a GLP-1 agonist given as a weekly subcutaneous injection, starts at a dose of 0.25 mg and goes up to 2.4 mg; patients should expect a 5% weight loss in 16 weeks if they are responders. Long term, however, patients lose up to an average 15% of body weight with semaglutide; a third of patients lost more than 20% of body weight in clinical trials, compared with 7%-8% body weight loss with liraglutide. An additional benefit of semaglutide is a 20% reduction in risk of cardiovascular disease.
- Tirzepatide is a combined GLP-1 and GIP agonist, also delivered as a weekly subcutaneous injection, that should result in an estimated 5% weight loss in 16 weeks for responders. But tirzepatide is the most effective of the three, with 91% of patients losing at least 5% body weight and more than half of patients (56%) losing at least 20%.
The big drawbacks to the GLP-1 agonists, however, are their high cost, common lack of insurance coverage, and continued shortages. Dr. Finkle recommended using manufacturer coupons, comparison shopping on Good Rx, and appealing prior authorization requirements to help patients pay for the GLP-1 agonists.
“Drug availability is my second problem. There’s not enough drug,” she said, and her patients often have to call around to different pharmacies to find out which ones are carrying the drug and at what doses. She will sometimes switch their doses as needed based on availability.
It’s also important for physicians to be aware of guidance from the American Society of Anesthesiologists regarding GLP-1 agonist use prior to surgery because of their slowed gastric-emptying mechanism. To reduce the risk of aspiration, patients undergoing general anesthesia should not take liraglutide on the day of surgery, and semaglutide and tirzepatide should be held for 1 week prior to the procedure. New research in JAMA Surgery, however, suggests holding these medications for longer than a week may be wiser.
Getting Patients Started
All the short-term and long-term medications are contraindicated during pregnancy and breastfeeding, Dr. Finkle said. Animal studies with GLP-1 agonists suggest adverse fetal effects when used during pregnancy, but the limited data in human studies so far have not shown a risk of major malformations. Dr. Finkle said the recommendations for now are to stop all GLP-1 receptor agonist drugs 2 months before patients attempt to become pregnant and not to begin them again until after they are no longer breastfeeding.
Finally, Dr. Finkle reviewed off-label medications that can result in modest weight loss, including topiramate, phentermine (not to be used for longer than 12 weeks), bupropion, naltrexone, and metformin. Metformin is likely to result in only 2% weight loss, but it may enhance the effects of GLP-1s, she said.
For ob.gyns. who want to get their patients started on one of these medications, Dr. Finkle first recommends asking patients if it’s okay to discuss their weight. ”Studies show that if you just ask permission to discuss someone’s weight, they go on to lose weight and lose more than someone who has never been asked,” Dr. Finkle said. Then she takes a history.
”When I see a patient, I ask, ‘Tell me why you’re here today,’ ” Dr. Finkle said.
This gives me a lot of insight as to why they’re coming in and it helps me understand where they’re at in terms of other things, such as depression or anxiety with weight, and it helps me to tailor my treatment.”
A full medical history is important for learning about potential contraindications or picking medications that might help with other conditions, such as topiramate for migraines. Finally, Dr. Finkle advises a lab screening with a comprehensive metabolic panel, lipid panel, HbA1c, and vitamin D.
“The [comprehensive metabolic panel] allows me to know about creatinine and liver function,” she said. If these are elevated, she will still prescribe GLP-1s but will monitor the values more closely. “Then I discuss options with the patient. They may be eligible for bariatric surgery or medications. We talk about lifestyle behavioral management, and then I go through the medications and we set goals.”
Goals include nutrition and exercise; start modest and have them work their way up by doing activities they enjoy. In addition, patients taking GLP-1s need to eat enough protein — 80 to 100 grams a day, though she starts them at 60 grams — and do regular muscle strengthening since they can lose muscle mass.
Indications for referral to an obesity medicine specialist are a history of gastric bypass/sleeve surgery, having type 2 diabetes, having an eating disorder, or having failed one of these anti-obesity medications.
Finally, Dr. Finkle reviewed medications that can cause weight gain: medroxyprogesterone acetate for birth control; beta blockers for hypertension or migraine; the antidepressants amitriptyline, paroxetine, venlafaxine, and trazodone; the mood stabilizers gabapentin, lithium, valproate, and carbamazepine; and diphenhydramine and zolpidem for sleep.
No external funding was used for the talk. Dr. Finkle and Dr. Heyward had no disclosures.
FROM ACOG 2024
Recent Evidence for Home Phototherapy Benefits May Improve Access for Patients with Psoriasis
Supporters of home phototherapy for patients with plaque and guttate psoriasis had plenty to cheer about at the annual meeting of the American Academy of Dermatology (AAD) in March. There, Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania in Philadelphia, presented results from the LITE study, a trial that tested the hypothesis that narrowband ultraviolet B phototherapy of psoriasis at home is noninferior to office treatment, based on outcomes that matter to patients, clinicians, and payers. While smaller studies have drawn similar conclusions,
The co-primary outcomes in the LITE study were a Physician’s Global Assessment (PGA) score of 0/1 (clear, almost clear) and a Dermatology Life Quality Index (DLQI) score of 5 or less (small, no effect on health-related quality of life).
Dr. Gelfand and colleagues at 42 sites in the United States enrolled 783 patients aged 12 years and older who had plaque or guttate psoriasis and were candidates for phototherapy at home or in an office setting. Following 12 weeks of treatment, 25.6% of patients in the office-based phototherapy group achieved a PGA score of 0/1 compared with 32.8% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data). Similarly, 33.6% of patients in the office-based phototherapy group achieved a DLQI score of 5 or less compared with 52.4% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data).
A Safe and Effective Option
“I think that it’s important for physicians, insurance companies, and patients with psoriasis to understand that this is a very safe and effective form of therapy,” Craig A. Elmets, MD, professor of dermatology at The University of Alabama at Birmingham, said in an interview. “For people who are not interested in systemic medications or who have contraindications to systemic medications, phototherapy would be ideal,” added Dr. Elmets, first author of the joint AAD–National Psoriasis Foundation (NPF) guidelines for the management and treatment of psoriasis with phototherapy, published in 2019.
Factors beyond efficacy support the role of home phototherapy, Dr. Gelfand said, including the fact that it costs 10-100 times less than biologics for psoriasis and that office-based phototherapy is not available in 90% of counties in the United States. However, insurance coverage of home phototherapy “is highly variable because until the LITE study, there was no large-scale US data to support its use,” he told this news organization.
“Also, insurance companies are broken up into two parts: Durable medical goods and the medical side such as pharmacy costs, and they are siloed. The durable medical goods side views phototherapy as expensive, while the pharmacy side views it as dirt cheap. This is part of the problem with our health system. A lot of things are siloed and don’t make any sense,” said Dr. Gelfand, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania. By working with the NPF and payers, he added, “we’re hoping ... to transform the way insurance companies think about covering home phototherapy.”
In the meantime, he and Dr. Elmets shared practical ways to optimize access to home phototherapy for psoriasis patients:
Have the discussion. Patients “rarely bring this up as an option,” Dr. Elmets said, so the onus is on clinicians to talk about it. In his view, the ideal candidate “is averse to using systemic agents but whose disease is beyond the point where topical medicines alone will work. One of the advantages of phototherapy is that it doesn’t have immunosuppressive effects.”
Clinicians and patients can learn about the efficacy and safety of phototherapy for psoriasis, including home-based options, on the NPF’s web site and by reading the 2019 joint AAD-NPF guidelines.
Shared decision-making is key. “When a patient comes in, I’ll discuss what their treatment options are and [we] will decide upon a course of action based on their unique needs and preferences [and] if it’s medically appropriate, meaning they have the type of psoriasis likely to respond to phototherapy,” Dr. Gelfand said. A patient with psoriasis mainly on the fingernails or genitals “is not a good candidate for phototherapy. If it’s on the trunk or extremities, that patient would be a good candidate.”
Home phototherapy candidates also must be willing and able to operate a machine and have dedicated space in their dwelling for it (most units are about the size of a door). Patients also have to be reliable, follow directions, and come back in person for follow-up appointments “so we can assess their response to treatment and fine-tune things as necessary and make sure they’re not developing any skin damage,” Dr. Gelfand said.
Educate yourself about existing options. Home phototherapy units from manufacturers such as Daavlin, National Biological Corporation, and SolRx range between $1200 and $6000 in cost, Dr. Gelfand said. He and his colleagues used the Daavlin 7 series in the LITE study. That unit features an integrated dosimetry system that delivers the correct dose of energy based on parameters that the prescribing clinician recommends. Settings are based on the patient’s skin type and how much the prescriber wants to increase the dose for each treatment. “The machine does the rest,” he said. “It knows what dose to give, so they get the same dosing as they would in an office situation.”
Smaller home-based phototherapy units designed to treat the hands and feet are available. So are handheld units to treat the scalp. “These can be a nice option for patients who have a few spots, but if the disease is moderate to severe, then it’s going to be pretty laborious to [use them],” Dr. Elmets said.
Remember that phototherapy is not a cure-all. According to the joint AAD-NPF guidelines, most phototherapy regimens require treatments two to three times per week for 10-14 weeks. Once patients achieve their home phototherapy treatment goal, Dr. Elmets often recommends treatments one to two times per week for maintenance.
“Patients with psoriasis have a lifetime condition,” he noted. “There are certainly cases where people have gone on phototherapy, cleared, and then stopped for a period of time. If they flare up, they can always go back to phototherapy. Usually, people who are on phototherapy use some type of topical agents to touch up areas that are resistant.”
Expect pushback from insurers on coverage. While Medicare and some integrated health plans cover home phototherapy, expect to spend time writing letters or placing phone calls to insurance companies to convince them why they should cover home phototherapy for candidate psoriasis patients. “Usually there’s a lot of letter writing and a long delay in getting approval,” Dr. Elmets said.
Dr. Elmets and Dr. Gelfand reported no relevant financial relationships. The LITE study was funded by the Patient-Centered Outcomes Research Institute. Research partners included the National Psoriasis Foundation and Daavlin, which provided the home phototherapy machines and covered the cost of shipping the devices.
A version of this article appeared on Medscape.com.
Supporters of home phototherapy for patients with plaque and guttate psoriasis had plenty to cheer about at the annual meeting of the American Academy of Dermatology (AAD) in March. There, Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania in Philadelphia, presented results from the LITE study, a trial that tested the hypothesis that narrowband ultraviolet B phototherapy of psoriasis at home is noninferior to office treatment, based on outcomes that matter to patients, clinicians, and payers. While smaller studies have drawn similar conclusions,
The co-primary outcomes in the LITE study were a Physician’s Global Assessment (PGA) score of 0/1 (clear, almost clear) and a Dermatology Life Quality Index (DLQI) score of 5 or less (small, no effect on health-related quality of life).
Dr. Gelfand and colleagues at 42 sites in the United States enrolled 783 patients aged 12 years and older who had plaque or guttate psoriasis and were candidates for phototherapy at home or in an office setting. Following 12 weeks of treatment, 25.6% of patients in the office-based phototherapy group achieved a PGA score of 0/1 compared with 32.8% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data). Similarly, 33.6% of patients in the office-based phototherapy group achieved a DLQI score of 5 or less compared with 52.4% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data).
A Safe and Effective Option
“I think that it’s important for physicians, insurance companies, and patients with psoriasis to understand that this is a very safe and effective form of therapy,” Craig A. Elmets, MD, professor of dermatology at The University of Alabama at Birmingham, said in an interview. “For people who are not interested in systemic medications or who have contraindications to systemic medications, phototherapy would be ideal,” added Dr. Elmets, first author of the joint AAD–National Psoriasis Foundation (NPF) guidelines for the management and treatment of psoriasis with phototherapy, published in 2019.
Factors beyond efficacy support the role of home phototherapy, Dr. Gelfand said, including the fact that it costs 10-100 times less than biologics for psoriasis and that office-based phototherapy is not available in 90% of counties in the United States. However, insurance coverage of home phototherapy “is highly variable because until the LITE study, there was no large-scale US data to support its use,” he told this news organization.
“Also, insurance companies are broken up into two parts: Durable medical goods and the medical side such as pharmacy costs, and they are siloed. The durable medical goods side views phototherapy as expensive, while the pharmacy side views it as dirt cheap. This is part of the problem with our health system. A lot of things are siloed and don’t make any sense,” said Dr. Gelfand, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania. By working with the NPF and payers, he added, “we’re hoping ... to transform the way insurance companies think about covering home phototherapy.”
In the meantime, he and Dr. Elmets shared practical ways to optimize access to home phototherapy for psoriasis patients:
Have the discussion. Patients “rarely bring this up as an option,” Dr. Elmets said, so the onus is on clinicians to talk about it. In his view, the ideal candidate “is averse to using systemic agents but whose disease is beyond the point where topical medicines alone will work. One of the advantages of phototherapy is that it doesn’t have immunosuppressive effects.”
Clinicians and patients can learn about the efficacy and safety of phototherapy for psoriasis, including home-based options, on the NPF’s web site and by reading the 2019 joint AAD-NPF guidelines.
Shared decision-making is key. “When a patient comes in, I’ll discuss what their treatment options are and [we] will decide upon a course of action based on their unique needs and preferences [and] if it’s medically appropriate, meaning they have the type of psoriasis likely to respond to phototherapy,” Dr. Gelfand said. A patient with psoriasis mainly on the fingernails or genitals “is not a good candidate for phototherapy. If it’s on the trunk or extremities, that patient would be a good candidate.”
Home phototherapy candidates also must be willing and able to operate a machine and have dedicated space in their dwelling for it (most units are about the size of a door). Patients also have to be reliable, follow directions, and come back in person for follow-up appointments “so we can assess their response to treatment and fine-tune things as necessary and make sure they’re not developing any skin damage,” Dr. Gelfand said.
Educate yourself about existing options. Home phototherapy units from manufacturers such as Daavlin, National Biological Corporation, and SolRx range between $1200 and $6000 in cost, Dr. Gelfand said. He and his colleagues used the Daavlin 7 series in the LITE study. That unit features an integrated dosimetry system that delivers the correct dose of energy based on parameters that the prescribing clinician recommends. Settings are based on the patient’s skin type and how much the prescriber wants to increase the dose for each treatment. “The machine does the rest,” he said. “It knows what dose to give, so they get the same dosing as they would in an office situation.”
Smaller home-based phototherapy units designed to treat the hands and feet are available. So are handheld units to treat the scalp. “These can be a nice option for patients who have a few spots, but if the disease is moderate to severe, then it’s going to be pretty laborious to [use them],” Dr. Elmets said.
Remember that phototherapy is not a cure-all. According to the joint AAD-NPF guidelines, most phototherapy regimens require treatments two to three times per week for 10-14 weeks. Once patients achieve their home phototherapy treatment goal, Dr. Elmets often recommends treatments one to two times per week for maintenance.
“Patients with psoriasis have a lifetime condition,” he noted. “There are certainly cases where people have gone on phototherapy, cleared, and then stopped for a period of time. If they flare up, they can always go back to phototherapy. Usually, people who are on phototherapy use some type of topical agents to touch up areas that are resistant.”
Expect pushback from insurers on coverage. While Medicare and some integrated health plans cover home phototherapy, expect to spend time writing letters or placing phone calls to insurance companies to convince them why they should cover home phototherapy for candidate psoriasis patients. “Usually there’s a lot of letter writing and a long delay in getting approval,” Dr. Elmets said.
Dr. Elmets and Dr. Gelfand reported no relevant financial relationships. The LITE study was funded by the Patient-Centered Outcomes Research Institute. Research partners included the National Psoriasis Foundation and Daavlin, which provided the home phototherapy machines and covered the cost of shipping the devices.
A version of this article appeared on Medscape.com.
Supporters of home phototherapy for patients with plaque and guttate psoriasis had plenty to cheer about at the annual meeting of the American Academy of Dermatology (AAD) in March. There, Joel M. Gelfand, MD, professor of dermatology and epidemiology at the University of Pennsylvania in Philadelphia, presented results from the LITE study, a trial that tested the hypothesis that narrowband ultraviolet B phototherapy of psoriasis at home is noninferior to office treatment, based on outcomes that matter to patients, clinicians, and payers. While smaller studies have drawn similar conclusions,
The co-primary outcomes in the LITE study were a Physician’s Global Assessment (PGA) score of 0/1 (clear, almost clear) and a Dermatology Life Quality Index (DLQI) score of 5 or less (small, no effect on health-related quality of life).
Dr. Gelfand and colleagues at 42 sites in the United States enrolled 783 patients aged 12 years and older who had plaque or guttate psoriasis and were candidates for phototherapy at home or in an office setting. Following 12 weeks of treatment, 25.6% of patients in the office-based phototherapy group achieved a PGA score of 0/1 compared with 32.8% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data). Similarly, 33.6% of patients in the office-based phototherapy group achieved a DLQI score of 5 or less compared with 52.4% of patients in the home-based phototherapy group (P > .0001 for noninferiority, non-response imputation for missing data).
A Safe and Effective Option
“I think that it’s important for physicians, insurance companies, and patients with psoriasis to understand that this is a very safe and effective form of therapy,” Craig A. Elmets, MD, professor of dermatology at The University of Alabama at Birmingham, said in an interview. “For people who are not interested in systemic medications or who have contraindications to systemic medications, phototherapy would be ideal,” added Dr. Elmets, first author of the joint AAD–National Psoriasis Foundation (NPF) guidelines for the management and treatment of psoriasis with phototherapy, published in 2019.
Factors beyond efficacy support the role of home phototherapy, Dr. Gelfand said, including the fact that it costs 10-100 times less than biologics for psoriasis and that office-based phototherapy is not available in 90% of counties in the United States. However, insurance coverage of home phototherapy “is highly variable because until the LITE study, there was no large-scale US data to support its use,” he told this news organization.
“Also, insurance companies are broken up into two parts: Durable medical goods and the medical side such as pharmacy costs, and they are siloed. The durable medical goods side views phototherapy as expensive, while the pharmacy side views it as dirt cheap. This is part of the problem with our health system. A lot of things are siloed and don’t make any sense,” said Dr. Gelfand, director of the Psoriasis and Phototherapy Treatment Center at the University of Pennsylvania. By working with the NPF and payers, he added, “we’re hoping ... to transform the way insurance companies think about covering home phototherapy.”
In the meantime, he and Dr. Elmets shared practical ways to optimize access to home phototherapy for psoriasis patients:
Have the discussion. Patients “rarely bring this up as an option,” Dr. Elmets said, so the onus is on clinicians to talk about it. In his view, the ideal candidate “is averse to using systemic agents but whose disease is beyond the point where topical medicines alone will work. One of the advantages of phototherapy is that it doesn’t have immunosuppressive effects.”
Clinicians and patients can learn about the efficacy and safety of phototherapy for psoriasis, including home-based options, on the NPF’s web site and by reading the 2019 joint AAD-NPF guidelines.
Shared decision-making is key. “When a patient comes in, I’ll discuss what their treatment options are and [we] will decide upon a course of action based on their unique needs and preferences [and] if it’s medically appropriate, meaning they have the type of psoriasis likely to respond to phototherapy,” Dr. Gelfand said. A patient with psoriasis mainly on the fingernails or genitals “is not a good candidate for phototherapy. If it’s on the trunk or extremities, that patient would be a good candidate.”
Home phototherapy candidates also must be willing and able to operate a machine and have dedicated space in their dwelling for it (most units are about the size of a door). Patients also have to be reliable, follow directions, and come back in person for follow-up appointments “so we can assess their response to treatment and fine-tune things as necessary and make sure they’re not developing any skin damage,” Dr. Gelfand said.
Educate yourself about existing options. Home phototherapy units from manufacturers such as Daavlin, National Biological Corporation, and SolRx range between $1200 and $6000 in cost, Dr. Gelfand said. He and his colleagues used the Daavlin 7 series in the LITE study. That unit features an integrated dosimetry system that delivers the correct dose of energy based on parameters that the prescribing clinician recommends. Settings are based on the patient’s skin type and how much the prescriber wants to increase the dose for each treatment. “The machine does the rest,” he said. “It knows what dose to give, so they get the same dosing as they would in an office situation.”
Smaller home-based phototherapy units designed to treat the hands and feet are available. So are handheld units to treat the scalp. “These can be a nice option for patients who have a few spots, but if the disease is moderate to severe, then it’s going to be pretty laborious to [use them],” Dr. Elmets said.
Remember that phototherapy is not a cure-all. According to the joint AAD-NPF guidelines, most phototherapy regimens require treatments two to three times per week for 10-14 weeks. Once patients achieve their home phototherapy treatment goal, Dr. Elmets often recommends treatments one to two times per week for maintenance.
“Patients with psoriasis have a lifetime condition,” he noted. “There are certainly cases where people have gone on phototherapy, cleared, and then stopped for a period of time. If they flare up, they can always go back to phototherapy. Usually, people who are on phototherapy use some type of topical agents to touch up areas that are resistant.”
Expect pushback from insurers on coverage. While Medicare and some integrated health plans cover home phototherapy, expect to spend time writing letters or placing phone calls to insurance companies to convince them why they should cover home phototherapy for candidate psoriasis patients. “Usually there’s a lot of letter writing and a long delay in getting approval,” Dr. Elmets said.
Dr. Elmets and Dr. Gelfand reported no relevant financial relationships. The LITE study was funded by the Patient-Centered Outcomes Research Institute. Research partners included the National Psoriasis Foundation and Daavlin, which provided the home phototherapy machines and covered the cost of shipping the devices.
A version of this article appeared on Medscape.com.
Are Secondary Osteoporosis Causes Under-Investigated?
NEW ORLEANS — Postmenopausal women with osteoporosis may not be receiving all the recommended tests to rule out secondary causes of bone loss prior to treatment initiation, new research found.
In a single-center chart review of 150 postmenopausal women who had been diagnosed and treated for osteoporosis, most had received a complete blood cell count, basic metabolic panel, thyroid screening, and vitamin D testing. However, one in four had not been tested for a parathyroid hormone (PTH) level, and in nearly two thirds, a 24-hour urine calcium collection had not been ordered.
Overall, less than a third had received the complete workup for secondary osteoporosis causes as recommended by the American Association of Clinical Endocrinologists (AACE) and the Endocrine Society.
“An appropriate evaluation for secondary causes of osteoporosis is essential because it impacts different treatment options and modalities. We discovered low rates of complete testing for secondary causes of osteoporosis in our patient population prior to treatment initiation,” said Kajol Manglani, MD, an internal medicine resident at Georgetown University/MedStar Washington Hospital Center, Washington, DC, and colleagues, in a poster at the American Association of Clinical Endocrinology (AACE) annual meeting held on May 9-12, 2024.
First author Sheetal Bulchandani, MD, said in an interview, “It depends a lot on clinical judgment, but there are certain things that everybody with osteoporosis should be evaluated for. We looked for the things that all the guidelines recommend.”
Studies have suggested that up to 30% of postmenopausal women with osteoporosis have secondary causes, noted Dr. Bulchandani, who conducted the study as a postdoctoral fellow with colleagues at Georgetown University/MedStar Washington Hospital and is now in private endocrine practice in Petersburg, Virginia.
“It’s important not to assume that every woman who walks in with osteoporosis has postmenopausal osteoporosis. I think it would be appropriate to at least discuss with the patients what would warrant certain kinds of clinical workup. … If you don’t figure out if there is an underlying cause, you may end up using an unnecessary medication,” Dr. Bulchandani said.
Are You Missing Something Treatable?
For example, she said, if the patient has underlying hyperparathyroidism and is treated with osteoporosis medications, “you might not see the desired or expected outcome in their bone density.”
Asked to comment, Rachel Pessah-Pollack, MD, clinical associate professor at the Holman Division of Endocrinology, Diabetes, and Metabolism at New York University School of Medicine, New York City, told this news organization, “Certainly, if you have patients who have osteoporosis, it’s important to take a good history and consider secondary causes of bone loss because you may find a treatable etiology that actually can improve their bone density without even starting on a medication.”
Dr. Pessah-Pollack, who was an author of the 2020 AACE/American College of Endocrinology 2020 Clinical Practice Guidelines for the Diagnosis and Treatment of Osteoporosis, said a 24-hour urine calcium collection, not a spot calcium check, is “super important because you’re looking to see if there’s any evidence of hypercalciuria or malabsorption that may be associated with higher rates of bone loss. … These may be a little more cumbersome and harder to get patients to do and more logistics to arrange. But clearly, if you pick up hypercalciuria, that is a potentially treatable etiology and can improve bone density as well.”
Another example, Dr. Pessah-Pollack said, is “if they have a low serum calcium level and high PTH, that would be a real reason to look for celiac disease. By not getting that PTH level, you may be missing that potential diagnosis. There is a wide range of additional causes of osteoporosis ranging from common conditions such as hyperthyroidism to rare conditions such as Cushing disease.”
Differences in Ordering Found Across Specialties
The 150 postmenopausal women were all receiving treatment with either alendronate, denosumab, or zoledronic acid. Their average age was 64.7 years, and 63% were seeing an endocrinologist.
Complete workups as per AACE and Endocrine Society guidelines had been performed in just 28% of those who saw an endocrinologist and 12.5% of patients seen by a rheumatologist, in contrast to 84% of those who saw the head of the hospital’s fracture prevention program.
Overall, across all specialties, just 28.67% had the complete recommended workup for secondary osteoporosis causes.
The most missed test was a 24-hour urine calcium collection, ordered for just 38% of the patients, while PTH was ordered for 73% and phosphorus for 80%. The rest were more commonly ordered: Thyroid-stimulating hormone level for 92.7%, complete blood cell count for 91.3%, basic metabolic panel for 100%, and vitamin D level for 96%.
The high rate of vitamin D testing is noteworthy, Dr. Pessah-Pollack said. “The fact that 96% of women are having vitamin D levels checked as part of an osteoporosis evaluation means that everybody’s aware about vitamin D deficiency, and people want to know what their vitamin D levels are. … That’s good because we want to identify vitamin D deficiency in our osteoporosis patients.”
But the low rate of complete secondary screening even by endocrinologists is concerning. “I look at this study as an opportunity for education that we can reinforce the importance of a secondary evaluation for our osteoporosis patients and really tailor which additional tests should be ordered for the individual patient,” Dr. Pessah-Pollack said.
In the poster, Dr. Bulchandani and colleagues wrote, “Further intervention will be aimed to ensure physicians undertake adequate evaluation before considering further treatment directions.” Possibilities that have been discussed include electronic health record alerts and educational materials for primary care providers, she told this news organization.
Dr. Manglani and Dr. Bulchandani had no disclosures. Dr. Pessah-Pollack is an advisor for Boehringer Ingelheim and Eli Lilly.
A version of this article appeared on Medscape.com.
NEW ORLEANS — Postmenopausal women with osteoporosis may not be receiving all the recommended tests to rule out secondary causes of bone loss prior to treatment initiation, new research found.
In a single-center chart review of 150 postmenopausal women who had been diagnosed and treated for osteoporosis, most had received a complete blood cell count, basic metabolic panel, thyroid screening, and vitamin D testing. However, one in four had not been tested for a parathyroid hormone (PTH) level, and in nearly two thirds, a 24-hour urine calcium collection had not been ordered.
Overall, less than a third had received the complete workup for secondary osteoporosis causes as recommended by the American Association of Clinical Endocrinologists (AACE) and the Endocrine Society.
“An appropriate evaluation for secondary causes of osteoporosis is essential because it impacts different treatment options and modalities. We discovered low rates of complete testing for secondary causes of osteoporosis in our patient population prior to treatment initiation,” said Kajol Manglani, MD, an internal medicine resident at Georgetown University/MedStar Washington Hospital Center, Washington, DC, and colleagues, in a poster at the American Association of Clinical Endocrinology (AACE) annual meeting held on May 9-12, 2024.
First author Sheetal Bulchandani, MD, said in an interview, “It depends a lot on clinical judgment, but there are certain things that everybody with osteoporosis should be evaluated for. We looked for the things that all the guidelines recommend.”
Studies have suggested that up to 30% of postmenopausal women with osteoporosis have secondary causes, noted Dr. Bulchandani, who conducted the study as a postdoctoral fellow with colleagues at Georgetown University/MedStar Washington Hospital and is now in private endocrine practice in Petersburg, Virginia.
“It’s important not to assume that every woman who walks in with osteoporosis has postmenopausal osteoporosis. I think it would be appropriate to at least discuss with the patients what would warrant certain kinds of clinical workup. … If you don’t figure out if there is an underlying cause, you may end up using an unnecessary medication,” Dr. Bulchandani said.
Are You Missing Something Treatable?
For example, she said, if the patient has underlying hyperparathyroidism and is treated with osteoporosis medications, “you might not see the desired or expected outcome in their bone density.”
Asked to comment, Rachel Pessah-Pollack, MD, clinical associate professor at the Holman Division of Endocrinology, Diabetes, and Metabolism at New York University School of Medicine, New York City, told this news organization, “Certainly, if you have patients who have osteoporosis, it’s important to take a good history and consider secondary causes of bone loss because you may find a treatable etiology that actually can improve their bone density without even starting on a medication.”
Dr. Pessah-Pollack, who was an author of the 2020 AACE/American College of Endocrinology 2020 Clinical Practice Guidelines for the Diagnosis and Treatment of Osteoporosis, said a 24-hour urine calcium collection, not a spot calcium check, is “super important because you’re looking to see if there’s any evidence of hypercalciuria or malabsorption that may be associated with higher rates of bone loss. … These may be a little more cumbersome and harder to get patients to do and more logistics to arrange. But clearly, if you pick up hypercalciuria, that is a potentially treatable etiology and can improve bone density as well.”
Another example, Dr. Pessah-Pollack said, is “if they have a low serum calcium level and high PTH, that would be a real reason to look for celiac disease. By not getting that PTH level, you may be missing that potential diagnosis. There is a wide range of additional causes of osteoporosis ranging from common conditions such as hyperthyroidism to rare conditions such as Cushing disease.”
Differences in Ordering Found Across Specialties
The 150 postmenopausal women were all receiving treatment with either alendronate, denosumab, or zoledronic acid. Their average age was 64.7 years, and 63% were seeing an endocrinologist.
Complete workups as per AACE and Endocrine Society guidelines had been performed in just 28% of those who saw an endocrinologist and 12.5% of patients seen by a rheumatologist, in contrast to 84% of those who saw the head of the hospital’s fracture prevention program.
Overall, across all specialties, just 28.67% had the complete recommended workup for secondary osteoporosis causes.
The most missed test was a 24-hour urine calcium collection, ordered for just 38% of the patients, while PTH was ordered for 73% and phosphorus for 80%. The rest were more commonly ordered: Thyroid-stimulating hormone level for 92.7%, complete blood cell count for 91.3%, basic metabolic panel for 100%, and vitamin D level for 96%.
The high rate of vitamin D testing is noteworthy, Dr. Pessah-Pollack said. “The fact that 96% of women are having vitamin D levels checked as part of an osteoporosis evaluation means that everybody’s aware about vitamin D deficiency, and people want to know what their vitamin D levels are. … That’s good because we want to identify vitamin D deficiency in our osteoporosis patients.”
But the low rate of complete secondary screening even by endocrinologists is concerning. “I look at this study as an opportunity for education that we can reinforce the importance of a secondary evaluation for our osteoporosis patients and really tailor which additional tests should be ordered for the individual patient,” Dr. Pessah-Pollack said.
In the poster, Dr. Bulchandani and colleagues wrote, “Further intervention will be aimed to ensure physicians undertake adequate evaluation before considering further treatment directions.” Possibilities that have been discussed include electronic health record alerts and educational materials for primary care providers, she told this news organization.
Dr. Manglani and Dr. Bulchandani had no disclosures. Dr. Pessah-Pollack is an advisor for Boehringer Ingelheim and Eli Lilly.
A version of this article appeared on Medscape.com.
NEW ORLEANS — Postmenopausal women with osteoporosis may not be receiving all the recommended tests to rule out secondary causes of bone loss prior to treatment initiation, new research found.
In a single-center chart review of 150 postmenopausal women who had been diagnosed and treated for osteoporosis, most had received a complete blood cell count, basic metabolic panel, thyroid screening, and vitamin D testing. However, one in four had not been tested for a parathyroid hormone (PTH) level, and in nearly two thirds, a 24-hour urine calcium collection had not been ordered.
Overall, less than a third had received the complete workup for secondary osteoporosis causes as recommended by the American Association of Clinical Endocrinologists (AACE) and the Endocrine Society.
“An appropriate evaluation for secondary causes of osteoporosis is essential because it impacts different treatment options and modalities. We discovered low rates of complete testing for secondary causes of osteoporosis in our patient population prior to treatment initiation,” said Kajol Manglani, MD, an internal medicine resident at Georgetown University/MedStar Washington Hospital Center, Washington, DC, and colleagues, in a poster at the American Association of Clinical Endocrinology (AACE) annual meeting held on May 9-12, 2024.
First author Sheetal Bulchandani, MD, said in an interview, “It depends a lot on clinical judgment, but there are certain things that everybody with osteoporosis should be evaluated for. We looked for the things that all the guidelines recommend.”
Studies have suggested that up to 30% of postmenopausal women with osteoporosis have secondary causes, noted Dr. Bulchandani, who conducted the study as a postdoctoral fellow with colleagues at Georgetown University/MedStar Washington Hospital and is now in private endocrine practice in Petersburg, Virginia.
“It’s important not to assume that every woman who walks in with osteoporosis has postmenopausal osteoporosis. I think it would be appropriate to at least discuss with the patients what would warrant certain kinds of clinical workup. … If you don’t figure out if there is an underlying cause, you may end up using an unnecessary medication,” Dr. Bulchandani said.
Are You Missing Something Treatable?
For example, she said, if the patient has underlying hyperparathyroidism and is treated with osteoporosis medications, “you might not see the desired or expected outcome in their bone density.”
Asked to comment, Rachel Pessah-Pollack, MD, clinical associate professor at the Holman Division of Endocrinology, Diabetes, and Metabolism at New York University School of Medicine, New York City, told this news organization, “Certainly, if you have patients who have osteoporosis, it’s important to take a good history and consider secondary causes of bone loss because you may find a treatable etiology that actually can improve their bone density without even starting on a medication.”
Dr. Pessah-Pollack, who was an author of the 2020 AACE/American College of Endocrinology 2020 Clinical Practice Guidelines for the Diagnosis and Treatment of Osteoporosis, said a 24-hour urine calcium collection, not a spot calcium check, is “super important because you’re looking to see if there’s any evidence of hypercalciuria or malabsorption that may be associated with higher rates of bone loss. … These may be a little more cumbersome and harder to get patients to do and more logistics to arrange. But clearly, if you pick up hypercalciuria, that is a potentially treatable etiology and can improve bone density as well.”
Another example, Dr. Pessah-Pollack said, is “if they have a low serum calcium level and high PTH, that would be a real reason to look for celiac disease. By not getting that PTH level, you may be missing that potential diagnosis. There is a wide range of additional causes of osteoporosis ranging from common conditions such as hyperthyroidism to rare conditions such as Cushing disease.”
Differences in Ordering Found Across Specialties
The 150 postmenopausal women were all receiving treatment with either alendronate, denosumab, or zoledronic acid. Their average age was 64.7 years, and 63% were seeing an endocrinologist.
Complete workups as per AACE and Endocrine Society guidelines had been performed in just 28% of those who saw an endocrinologist and 12.5% of patients seen by a rheumatologist, in contrast to 84% of those who saw the head of the hospital’s fracture prevention program.
Overall, across all specialties, just 28.67% had the complete recommended workup for secondary osteoporosis causes.
The most missed test was a 24-hour urine calcium collection, ordered for just 38% of the patients, while PTH was ordered for 73% and phosphorus for 80%. The rest were more commonly ordered: Thyroid-stimulating hormone level for 92.7%, complete blood cell count for 91.3%, basic metabolic panel for 100%, and vitamin D level for 96%.
The high rate of vitamin D testing is noteworthy, Dr. Pessah-Pollack said. “The fact that 96% of women are having vitamin D levels checked as part of an osteoporosis evaluation means that everybody’s aware about vitamin D deficiency, and people want to know what their vitamin D levels are. … That’s good because we want to identify vitamin D deficiency in our osteoporosis patients.”
But the low rate of complete secondary screening even by endocrinologists is concerning. “I look at this study as an opportunity for education that we can reinforce the importance of a secondary evaluation for our osteoporosis patients and really tailor which additional tests should be ordered for the individual patient,” Dr. Pessah-Pollack said.
In the poster, Dr. Bulchandani and colleagues wrote, “Further intervention will be aimed to ensure physicians undertake adequate evaluation before considering further treatment directions.” Possibilities that have been discussed include electronic health record alerts and educational materials for primary care providers, she told this news organization.
Dr. Manglani and Dr. Bulchandani had no disclosures. Dr. Pessah-Pollack is an advisor for Boehringer Ingelheim and Eli Lilly.
A version of this article appeared on Medscape.com.
Debate on pulmonary safety of gas stoves: Is the risk just hot air?
SAN DIEGO — While there is currently no smoking gun definitively showing that indoor nitrogen dioxide (NO2) concentrations from gas appliances are a cause of pulmonary diseases, the circumstantial evidence of the baleful effects of gas stoves on lung function is pretty compelling, said participants in a pro-con debate.
The debate was held at the American Thoracic Society’s international conference.
PRO: Gas stoves cause lung disease
Arguing for the “pro” side, John R. Balmes, MD of the University of California, San Francisco, and a physician member of the California Air Resources Board, began by admitting that “I would never have said gas stoves cause lung disease, but that’s what they assigned me.”
Gamely proceeding anyway, Dr. Balmes noted that natural gas — methane — is a potent greenhouse gas, and that cooking with natural gas leads to generation of NO2 with high peak concentrations in the home, especially in the kitchen, but in other rooms as well.
“We know that NO2 is an irritant gas that can cause bronchoconstriction, airway hyperresponsiveness and inflammation, and there’s increased risk of asthma and COPD exacerbations,” he said.
The US Environmental Protection Agency (EPA) outdoor ambient air standard for NO2 is 100 parts per billion (ppb) or lower, which are the levels needed to prevent asthma exacerbations. In separate meta-analyses there was a 1.05 rise in asthma incidence per every 2 ppb of NO2, and an increase of 1.07 in COPD incidence for every 5 ppb of NO2, Dr. Balmes noted.
The respiratory effects of gas stoves were revealed in a 2013 meta-analysis of 10 studies from North America and Europe, which showed a pooled odds ratio for current asthma of 1.34. Building on these data, authors of a 2022 paper estimated that 13% of childhood asthma could be prevented by elimination gas cooking.
Although the causative link is missing, the evidence is abundant that natural gas isn’t good for anyone, he acknowledged.
Con: More evidence needed
Arguing for the “con” side of the question, Meredith C. McCormack, MD, MHS, professor of medicine in the pulmonary and critical care division at Johns Hopkins University in Baltimore, said that “more definitive evidence is needed to define whether gas stoves cause lung disease.”
But Dr. McCormack didn’t let the natural gas industry off the hook, noting that a systematic review and meta-analysis of cooking with gas in high-, middle-, and low-income countries showed that domestic use of gas fuels vs. electric was associated with increased risk of asthma (1.11 overall), COPD (1.15), and pneumonia (1.26).
The link between gas and risk of asthma was significant only for adults, however, and the data on the risks for COPD and for pneumonia or other respiratory infections came almost exclusively from low-income countries, she noted.
Despite the lack of evidence for a causative link, however, Dr. McCormack pointed to evidence that indoor NO2 is an air pollutant that acts as a respiratory irritant, and that indoor NO2 levels in homes with gas stoves have been shown to be more than twice as high as those in homes with electric stoves.
Other evidence shows that indoor NO2 is associated with increased symptoms and use of rescue medications for children with asthma, and with shortness of breath, nocturnal symptoms, reduction in lung function, and exacerbations in COPD.
Still other studies have shown that exchanging a gas stove for an electric stove can reduce NO2 concentrations in the home by up to 50%, but there is still a need for clinical trial evidence of a health benefit for such an exchange, she said.
And even if a gas stove is swapped out for an electric or induction range, household members with asthma are exposed to other hazards, including second-hand smoke, cooking exhaust, candle or incense burning, outdoor particulate matter that finds its way indoors, mold, and mouse or cockroach allergens, she noted.
On common ground
Environmental interventions that can benefit all members of a household — not just those with obstructive pulmonary disease — include smoking cessation, charcoal filter-equipped air cleaners, stove hoods that vent outdoors, integrated pest management, hypoallergenic pillow and mattress covers, high efficiency particulate air (HEPA) vacuums, and mold and radon abatement.
Both Dr. Balmes and Dr. McCormack agreed in the end that gas stoves contribute to respiratory morbidity, and that both state and national policy changes are needed to support transition to cleaner indoor air, with financial incentives available for households with more modest incomes.
“For everyone, there is a climate-change mitigation imperative to transition away from gas appliances if we want to tackle the climate emergency,” Dr. Balmes said.
End indoor combustion
George D. Thurston, ScD, professor of medicine and population health at the NYU Grossman School of Medicine, who attended the debate, told Chest Physician that the participants talked about NO2 but didn’t touch on particulate pollution generated by gas stoves.
Burning natural gas produces particles that are very similar in composition to those produced by burning coal, oil, or diesel fuel, Dr. Thurston said, and he pointed out that interventions such as range hoods work only if they actually vent outdoors, and aren’t simply fans that recirculate the air within the home. And even when ventilation works as it should to move air out of the house, it only pumps it back into the atmosphere, where it contributes to climate change.
“We need combustion-free homes. That’s the unifying principle. We have to keep our eyes on that prize,” he said.
Dr. Balmes, Dr. McCormack, and Dr. Thurston all reported having no relevant disclosures.
SAN DIEGO — While there is currently no smoking gun definitively showing that indoor nitrogen dioxide (NO2) concentrations from gas appliances are a cause of pulmonary diseases, the circumstantial evidence of the baleful effects of gas stoves on lung function is pretty compelling, said participants in a pro-con debate.
The debate was held at the American Thoracic Society’s international conference.
PRO: Gas stoves cause lung disease
Arguing for the “pro” side, John R. Balmes, MD of the University of California, San Francisco, and a physician member of the California Air Resources Board, began by admitting that “I would never have said gas stoves cause lung disease, but that’s what they assigned me.”
Gamely proceeding anyway, Dr. Balmes noted that natural gas — methane — is a potent greenhouse gas, and that cooking with natural gas leads to generation of NO2 with high peak concentrations in the home, especially in the kitchen, but in other rooms as well.
“We know that NO2 is an irritant gas that can cause bronchoconstriction, airway hyperresponsiveness and inflammation, and there’s increased risk of asthma and COPD exacerbations,” he said.
The US Environmental Protection Agency (EPA) outdoor ambient air standard for NO2 is 100 parts per billion (ppb) or lower, which are the levels needed to prevent asthma exacerbations. In separate meta-analyses there was a 1.05 rise in asthma incidence per every 2 ppb of NO2, and an increase of 1.07 in COPD incidence for every 5 ppb of NO2, Dr. Balmes noted.
The respiratory effects of gas stoves were revealed in a 2013 meta-analysis of 10 studies from North America and Europe, which showed a pooled odds ratio for current asthma of 1.34. Building on these data, authors of a 2022 paper estimated that 13% of childhood asthma could be prevented by elimination gas cooking.
Although the causative link is missing, the evidence is abundant that natural gas isn’t good for anyone, he acknowledged.
Con: More evidence needed
Arguing for the “con” side of the question, Meredith C. McCormack, MD, MHS, professor of medicine in the pulmonary and critical care division at Johns Hopkins University in Baltimore, said that “more definitive evidence is needed to define whether gas stoves cause lung disease.”
But Dr. McCormack didn’t let the natural gas industry off the hook, noting that a systematic review and meta-analysis of cooking with gas in high-, middle-, and low-income countries showed that domestic use of gas fuels vs. electric was associated with increased risk of asthma (1.11 overall), COPD (1.15), and pneumonia (1.26).
The link between gas and risk of asthma was significant only for adults, however, and the data on the risks for COPD and for pneumonia or other respiratory infections came almost exclusively from low-income countries, she noted.
Despite the lack of evidence for a causative link, however, Dr. McCormack pointed to evidence that indoor NO2 is an air pollutant that acts as a respiratory irritant, and that indoor NO2 levels in homes with gas stoves have been shown to be more than twice as high as those in homes with electric stoves.
Other evidence shows that indoor NO2 is associated with increased symptoms and use of rescue medications for children with asthma, and with shortness of breath, nocturnal symptoms, reduction in lung function, and exacerbations in COPD.
Still other studies have shown that exchanging a gas stove for an electric stove can reduce NO2 concentrations in the home by up to 50%, but there is still a need for clinical trial evidence of a health benefit for such an exchange, she said.
And even if a gas stove is swapped out for an electric or induction range, household members with asthma are exposed to other hazards, including second-hand smoke, cooking exhaust, candle or incense burning, outdoor particulate matter that finds its way indoors, mold, and mouse or cockroach allergens, she noted.
On common ground
Environmental interventions that can benefit all members of a household — not just those with obstructive pulmonary disease — include smoking cessation, charcoal filter-equipped air cleaners, stove hoods that vent outdoors, integrated pest management, hypoallergenic pillow and mattress covers, high efficiency particulate air (HEPA) vacuums, and mold and radon abatement.
Both Dr. Balmes and Dr. McCormack agreed in the end that gas stoves contribute to respiratory morbidity, and that both state and national policy changes are needed to support transition to cleaner indoor air, with financial incentives available for households with more modest incomes.
“For everyone, there is a climate-change mitigation imperative to transition away from gas appliances if we want to tackle the climate emergency,” Dr. Balmes said.
End indoor combustion
George D. Thurston, ScD, professor of medicine and population health at the NYU Grossman School of Medicine, who attended the debate, told Chest Physician that the participants talked about NO2 but didn’t touch on particulate pollution generated by gas stoves.
Burning natural gas produces particles that are very similar in composition to those produced by burning coal, oil, or diesel fuel, Dr. Thurston said, and he pointed out that interventions such as range hoods work only if they actually vent outdoors, and aren’t simply fans that recirculate the air within the home. And even when ventilation works as it should to move air out of the house, it only pumps it back into the atmosphere, where it contributes to climate change.
“We need combustion-free homes. That’s the unifying principle. We have to keep our eyes on that prize,” he said.
Dr. Balmes, Dr. McCormack, and Dr. Thurston all reported having no relevant disclosures.
SAN DIEGO — While there is currently no smoking gun definitively showing that indoor nitrogen dioxide (NO2) concentrations from gas appliances are a cause of pulmonary diseases, the circumstantial evidence of the baleful effects of gas stoves on lung function is pretty compelling, said participants in a pro-con debate.
The debate was held at the American Thoracic Society’s international conference.
PRO: Gas stoves cause lung disease
Arguing for the “pro” side, John R. Balmes, MD of the University of California, San Francisco, and a physician member of the California Air Resources Board, began by admitting that “I would never have said gas stoves cause lung disease, but that’s what they assigned me.”
Gamely proceeding anyway, Dr. Balmes noted that natural gas — methane — is a potent greenhouse gas, and that cooking with natural gas leads to generation of NO2 with high peak concentrations in the home, especially in the kitchen, but in other rooms as well.
“We know that NO2 is an irritant gas that can cause bronchoconstriction, airway hyperresponsiveness and inflammation, and there’s increased risk of asthma and COPD exacerbations,” he said.
The US Environmental Protection Agency (EPA) outdoor ambient air standard for NO2 is 100 parts per billion (ppb) or lower, which are the levels needed to prevent asthma exacerbations. In separate meta-analyses there was a 1.05 rise in asthma incidence per every 2 ppb of NO2, and an increase of 1.07 in COPD incidence for every 5 ppb of NO2, Dr. Balmes noted.
The respiratory effects of gas stoves were revealed in a 2013 meta-analysis of 10 studies from North America and Europe, which showed a pooled odds ratio for current asthma of 1.34. Building on these data, authors of a 2022 paper estimated that 13% of childhood asthma could be prevented by elimination gas cooking.
Although the causative link is missing, the evidence is abundant that natural gas isn’t good for anyone, he acknowledged.
Con: More evidence needed
Arguing for the “con” side of the question, Meredith C. McCormack, MD, MHS, professor of medicine in the pulmonary and critical care division at Johns Hopkins University in Baltimore, said that “more definitive evidence is needed to define whether gas stoves cause lung disease.”
But Dr. McCormack didn’t let the natural gas industry off the hook, noting that a systematic review and meta-analysis of cooking with gas in high-, middle-, and low-income countries showed that domestic use of gas fuels vs. electric was associated with increased risk of asthma (1.11 overall), COPD (1.15), and pneumonia (1.26).
The link between gas and risk of asthma was significant only for adults, however, and the data on the risks for COPD and for pneumonia or other respiratory infections came almost exclusively from low-income countries, she noted.
Despite the lack of evidence for a causative link, however, Dr. McCormack pointed to evidence that indoor NO2 is an air pollutant that acts as a respiratory irritant, and that indoor NO2 levels in homes with gas stoves have been shown to be more than twice as high as those in homes with electric stoves.
Other evidence shows that indoor NO2 is associated with increased symptoms and use of rescue medications for children with asthma, and with shortness of breath, nocturnal symptoms, reduction in lung function, and exacerbations in COPD.
Still other studies have shown that exchanging a gas stove for an electric stove can reduce NO2 concentrations in the home by up to 50%, but there is still a need for clinical trial evidence of a health benefit for such an exchange, she said.
And even if a gas stove is swapped out for an electric or induction range, household members with asthma are exposed to other hazards, including second-hand smoke, cooking exhaust, candle or incense burning, outdoor particulate matter that finds its way indoors, mold, and mouse or cockroach allergens, she noted.
On common ground
Environmental interventions that can benefit all members of a household — not just those with obstructive pulmonary disease — include smoking cessation, charcoal filter-equipped air cleaners, stove hoods that vent outdoors, integrated pest management, hypoallergenic pillow and mattress covers, high efficiency particulate air (HEPA) vacuums, and mold and radon abatement.
Both Dr. Balmes and Dr. McCormack agreed in the end that gas stoves contribute to respiratory morbidity, and that both state and national policy changes are needed to support transition to cleaner indoor air, with financial incentives available for households with more modest incomes.
“For everyone, there is a climate-change mitigation imperative to transition away from gas appliances if we want to tackle the climate emergency,” Dr. Balmes said.
End indoor combustion
George D. Thurston, ScD, professor of medicine and population health at the NYU Grossman School of Medicine, who attended the debate, told Chest Physician that the participants talked about NO2 but didn’t touch on particulate pollution generated by gas stoves.
Burning natural gas produces particles that are very similar in composition to those produced by burning coal, oil, or diesel fuel, Dr. Thurston said, and he pointed out that interventions such as range hoods work only if they actually vent outdoors, and aren’t simply fans that recirculate the air within the home. And even when ventilation works as it should to move air out of the house, it only pumps it back into the atmosphere, where it contributes to climate change.
“We need combustion-free homes. That’s the unifying principle. We have to keep our eyes on that prize,” he said.
Dr. Balmes, Dr. McCormack, and Dr. Thurston all reported having no relevant disclosures.
FROM ATS 2024
Key Risk Factors for Hydroxychloroquine Retinopathy Described in Large Study
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
Older patients prescribed hydroxychloroquine (HCQ) have a higher risk of developing retinal damage from taking the medication, according to a new analysis.
In addition to known risk factors such as a higher weight-based HCQ dose and higher cumulative dose, researchers also found that female sex, chronic kidney disease stage III, and tamoxifen use were associated with HCQ retinopathy.
The findings provide “evidence for other key risk factors for hydroxychloroquine retinopathy beyond hydroxychloroquine exposure itself,” wrote April M. Jorge, MD, of the Division of Rheumatology, Allergy, and Immunology at Massachusetts General Hospital, Boston, and colleagues.
“It is the largest cohort study to date looking specifically at the association of [HCQ] retinopathy with risk factors,” Christina Weng, MD, MBA, professor of ophthalmology at Baylor College of Medicine, Houston, said in an interview. She was not involved with the research. Some of the associations, such as tamoxifen use, “have been suggested before in smaller studies, but never on this scale,” she said.
“It’s provided reinforcement of findings that we have seen from prior research and also some new glimpses into strengthening some associations that were identified, but not yet fully understood, in prior work,” she continued.
Study Details
Researchers identified patients in the Kaiser Permanente Northern California (KPNC), Oakland, California, health system who began taking HCQ between July 1, 1997, and December 14, 2014. To be included, patients needed to have at least 5 years of continuous enrollment in the KPNC system and at least one prescription for HCQ after more than 5 years of starting the drug. Patients were followed from HCQ initiation to their last retinopathy screening study, up to December 31, 2020.
The study was published May 9 in JAMA Network Open.
Of the 4677 users followed for the study, 83% were women, and the average age starting HCQ was 52. Most patients were White (58.1%), while 13.7% were Asian, 10.5% were Black, and 17.7% were Hispanic.
More than 60% of patients had an initial dose > 5 mg/kg/d, though the mean initial dose of HCQ was 4.4 mg/kg/d. After 5 years, only 34.4% of patients were using a daily dose over 5 mg/kg.
Of the entire cohort, 125 patients (2.7%) developed HCQ retinopathy. As expected, cumulative HCQ exposure was associated with a higher retinopathy risk: For every 100 g of HCQ cumulative exposure, risk rose by 64% (hazard ratio [HR], 1.64; 95% CI, 1.44-1.87).
Age was a significant risk factor for retinal damage from HCQ use. Individuals who began taking the drug at 65 years or older were nearly six times more likely to develop retinopathy than those who started HCQ when they were younger than 45. In people aged 55-64 years, this risk was nearly four times higher, and individuals aged 45-54 years when starting the drug were 2.5 times more likely to have retinal damage than those younger than 45.
Other risk factors were female sex (HR, 3.83; 95% CI, 1.86-7.89), chronic kidney disease stage III (HR, 1.95; 95% CI, 1.25-3.04), and tamoxifen use (HR, 3.43; 95% CI, 1.08-10.89), although only 17 patients were taking tamoxifen during the study.
Researchers also found that the type of HCQ retinopathy varied by race. Of the 125 cases in the cohort, 102 had a parafoveal pattern, and 23 had a pericentral pattern. Asian individuals were 15 times more likely, and Black individuals were more than 5 times more likely to develop this pericentral type than were White patients.
This association in Asian patients has also been found in previous studies, Dr. Weng said, and many eye practices now screen their Asian patients with a 30-2 Humphrey visual field — rather than the more commonly used 10-2 — to examine areas farther outside the center.
This study also found this association in Black patients, though only five Black patients developed HCQ retinopathy over the study period.
“More studies and larger studies will be very helpful in strengthening or dispelling some of the associations that have been seen here,” Dr. Weng said.
‘More Room for Personalized Medicine’
The team found a “relatively linear” relationship between HCQ dose and retinopathy risk, with higher daily doses correlating with higher incidence. While 2016 guidelines from the American Academy of Ophthalmology advise using < 5 mg/kg, “what we found is it’s not that straightforward [where there’s] just this one cutoff,” Dr. Jorge told this news organization. “It does seem like the higher the dose of medication per bodyweight and the longer duration of use, there is a higher risk of retinopathy.”
These findings leave “a bit more room for personalized medicine” with patients, she explained. “For an elderly female patient with CKD, aiming for a dose < 5 mg/kg might be more appropriate; however, a young male patient without any additional risk factors may be able to exceed 5 mg/kg and continue to have a low risk for HCQ retinopathy,” she said.
“For anyone, I think it really is more of an individual risk-benefit evaluation,” rather than strict cutoffs, she continued.
“Guidelines are just that: They’re guidelines,” added Dr. Weng, “and treatment plans should be tailored to each individual patient.”
As the study authors also discussed, “the goal is to treat the patient with the lowest dose that is still effective and also be mindful of the duration that a patient is left at higher doses,” Dr. Weng said. “But ultimately, we need to control these diseases, which can cause damage across multiple organ systems in the body. While it’s important to be aware of the potential retinopathy adverse events, we also don’t want physicians to feel restricted in their use of this very effective drug.”
The work of three coauthors on the current study was supported by a grant from the National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS). Dr. Jorge’s work on the study was supported by an award from the Rheumatology Research Foundation and a grant from NIAMS. Dr. Jorge reported clinical trial agreements with Bristol Myers Squibb and Cabaletta Bio outside of this study. Dr. Weng has served as a consultant for Allergan/AbbVie, Alcon, Apellis Pharmaceuticals, Alimera Sciences, DORC, Novartis, Genentech, Regeneron, RegenxBio, Iveric Bio, and EyePoint Pharmaceuticals. Dr. Weng disclosed financial relationships with Springer Publishers (royalties) and DRCR Retina Network, Alimera Sciences, and Applied Genetic Technologies Corporation (research).
A version of this article appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Belimumab Autoinjector Approved for Pediatric Lupus
The US Food and Drug Administration (FDA) has approved Benlysta (belimumab) autoinjector for patients aged 5 years or older with active systemic lupus erythematosus (SLE) on standard therapy. This is the first time that children with SLE can receive this treatment at home, according to a GSK press release.
Prior to this approval, pediatric patients aged 5 years or older could receive belimumab only intravenously via a 1-hour infusion in a hospital or clinic setting.
“Going to the doctor’s office once every 4 weeks can be a logistical hurdle for some children and their caregivers, so having the option to administer Benlysta in the comfort of their home provides much-needed flexibility,” Mary Crimmings, the interim CEO and senior vice president for marketing and communications at the Lupus Foundation of America, said in a statement.
An estimated 5000-10,000 children in the United States are living with SLE.
Belimumab is a B-lymphocyte stimulator–specific inhibitor approved for the treatment of active SLE and active lupus nephritis in patients aged 5 years or older receiving standard therapy. This approval of the subcutaneous administration of belimumab applies only to pediatric patients with SLE.
The 200-mg injection can be administered once every week for children who weigh ≥ 40 kg and should be given once every 2 weeks for children weighing between 15 and 40 kg.
The autoinjector “will be available immediately” for caregivers, the company announcement said.
“Patients are our top priority, and we are always working to innovate solutions that can improve lives and address unmet needs,” Court Horncastle, senior vice president and head of US specialty at GSK, said in the press release. “This approval for an at-home treatment is the first and only of its kind for children with lupus and is a testament to our continued commitment to the lupus community.”
A version of this article appeared on Medscape.com.
The US Food and Drug Administration (FDA) has approved Benlysta (belimumab) autoinjector for patients aged 5 years or older with active systemic lupus erythematosus (SLE) on standard therapy. This is the first time that children with SLE can receive this treatment at home, according to a GSK press release.
Prior to this approval, pediatric patients aged 5 years or older could receive belimumab only intravenously via a 1-hour infusion in a hospital or clinic setting.
“Going to the doctor’s office once every 4 weeks can be a logistical hurdle for some children and their caregivers, so having the option to administer Benlysta in the comfort of their home provides much-needed flexibility,” Mary Crimmings, the interim CEO and senior vice president for marketing and communications at the Lupus Foundation of America, said in a statement.
An estimated 5000-10,000 children in the United States are living with SLE.
Belimumab is a B-lymphocyte stimulator–specific inhibitor approved for the treatment of active SLE and active lupus nephritis in patients aged 5 years or older receiving standard therapy. This approval of the subcutaneous administration of belimumab applies only to pediatric patients with SLE.
The 200-mg injection can be administered once every week for children who weigh ≥ 40 kg and should be given once every 2 weeks for children weighing between 15 and 40 kg.
The autoinjector “will be available immediately” for caregivers, the company announcement said.
“Patients are our top priority, and we are always working to innovate solutions that can improve lives and address unmet needs,” Court Horncastle, senior vice president and head of US specialty at GSK, said in the press release. “This approval for an at-home treatment is the first and only of its kind for children with lupus and is a testament to our continued commitment to the lupus community.”
A version of this article appeared on Medscape.com.
The US Food and Drug Administration (FDA) has approved Benlysta (belimumab) autoinjector for patients aged 5 years or older with active systemic lupus erythematosus (SLE) on standard therapy. This is the first time that children with SLE can receive this treatment at home, according to a GSK press release.
Prior to this approval, pediatric patients aged 5 years or older could receive belimumab only intravenously via a 1-hour infusion in a hospital or clinic setting.
“Going to the doctor’s office once every 4 weeks can be a logistical hurdle for some children and their caregivers, so having the option to administer Benlysta in the comfort of their home provides much-needed flexibility,” Mary Crimmings, the interim CEO and senior vice president for marketing and communications at the Lupus Foundation of America, said in a statement.
An estimated 5000-10,000 children in the United States are living with SLE.
Belimumab is a B-lymphocyte stimulator–specific inhibitor approved for the treatment of active SLE and active lupus nephritis in patients aged 5 years or older receiving standard therapy. This approval of the subcutaneous administration of belimumab applies only to pediatric patients with SLE.
The 200-mg injection can be administered once every week for children who weigh ≥ 40 kg and should be given once every 2 weeks for children weighing between 15 and 40 kg.
The autoinjector “will be available immediately” for caregivers, the company announcement said.
“Patients are our top priority, and we are always working to innovate solutions that can improve lives and address unmet needs,” Court Horncastle, senior vice president and head of US specialty at GSK, said in the press release. “This approval for an at-home treatment is the first and only of its kind for children with lupus and is a testament to our continued commitment to the lupus community.”
A version of this article appeared on Medscape.com.
Genetic Test Can Predict Response to Semaglutide for Weight Loss
TOPLINE:
, new evidence reveals.
METHODOLOGY:
- A machine learning genetic risk score can identify people with the hungry gut obesity phenotype, which has been found to be associated with greater weight loss with the glucagon-like peptide 1 receptor agonists (GLP-1 RA) liraglutide and exenatide.
- For this study, researchers calculated the genetic risk score for 84 adults undergoing weight loss interventions at Mayo Clinic who were prescribed the GLP-1 RA semaglutide.
- Study participants were classified as the obesity phenotype hungry gut positive (n = 51) or hungry gut negative (n = 33).
- The researchers measured total body weight loss at 3, 6, 9, and 12 months and assessed the ability of the score to predict the response to semaglutide (defined as ≥ 5% of total body weight loss measured at 12 months).
TAKEAWAY:
- At 3 and 6 months, there were no significant differences in weight loss between the hungry gut positive and hungry gut negative groups.
- By 9 months, participants in the positive group lost 14.4% of their total body weight compared with 10.3% in case of participants in the negative group (P = .045).
- After a total of 12 months, the positive group lost 19.5% of their total body weight compared with 10.0% in case of participants in the negative group (P = .01).
- When used to predict the response to semaglutide, the area under the curve for the machine-learning genetic risk score was 0.76 (95% CI, 0.57-0.94; P = .04).
IN PRACTICE:
We can now tell with confidence who is going to respond to semaglutide, said Andres Acosta, MD, PhD, associate professor of medicine at Mayo Clinic. “For nonresponders, we can think about other interventions or medications that we have available.”
SOURCE:
This study was presented on May 20, 2024, at the annual Digestive Disease Week® (DDW) (Abstract 638).
LIMITATIONS:
Further prospective studies are needed to assess the validity of the test in a more diverse population and with different weight loss interventions.
DISCLOSURES:
This study was supported by a partnership between Mayo Clinic and Phenomix Sciences. Dr. Acosta is a cofounder of Phenomix Sciences.
A version of this article appeared on Medscape.com.
TOPLINE:
, new evidence reveals.
METHODOLOGY:
- A machine learning genetic risk score can identify people with the hungry gut obesity phenotype, which has been found to be associated with greater weight loss with the glucagon-like peptide 1 receptor agonists (GLP-1 RA) liraglutide and exenatide.
- For this study, researchers calculated the genetic risk score for 84 adults undergoing weight loss interventions at Mayo Clinic who were prescribed the GLP-1 RA semaglutide.
- Study participants were classified as the obesity phenotype hungry gut positive (n = 51) or hungry gut negative (n = 33).
- The researchers measured total body weight loss at 3, 6, 9, and 12 months and assessed the ability of the score to predict the response to semaglutide (defined as ≥ 5% of total body weight loss measured at 12 months).
TAKEAWAY:
- At 3 and 6 months, there were no significant differences in weight loss between the hungry gut positive and hungry gut negative groups.
- By 9 months, participants in the positive group lost 14.4% of their total body weight compared with 10.3% in case of participants in the negative group (P = .045).
- After a total of 12 months, the positive group lost 19.5% of their total body weight compared with 10.0% in case of participants in the negative group (P = .01).
- When used to predict the response to semaglutide, the area under the curve for the machine-learning genetic risk score was 0.76 (95% CI, 0.57-0.94; P = .04).
IN PRACTICE:
We can now tell with confidence who is going to respond to semaglutide, said Andres Acosta, MD, PhD, associate professor of medicine at Mayo Clinic. “For nonresponders, we can think about other interventions or medications that we have available.”
SOURCE:
This study was presented on May 20, 2024, at the annual Digestive Disease Week® (DDW) (Abstract 638).
LIMITATIONS:
Further prospective studies are needed to assess the validity of the test in a more diverse population and with different weight loss interventions.
DISCLOSURES:
This study was supported by a partnership between Mayo Clinic and Phenomix Sciences. Dr. Acosta is a cofounder of Phenomix Sciences.
A version of this article appeared on Medscape.com.
TOPLINE:
, new evidence reveals.
METHODOLOGY:
- A machine learning genetic risk score can identify people with the hungry gut obesity phenotype, which has been found to be associated with greater weight loss with the glucagon-like peptide 1 receptor agonists (GLP-1 RA) liraglutide and exenatide.
- For this study, researchers calculated the genetic risk score for 84 adults undergoing weight loss interventions at Mayo Clinic who were prescribed the GLP-1 RA semaglutide.
- Study participants were classified as the obesity phenotype hungry gut positive (n = 51) or hungry gut negative (n = 33).
- The researchers measured total body weight loss at 3, 6, 9, and 12 months and assessed the ability of the score to predict the response to semaglutide (defined as ≥ 5% of total body weight loss measured at 12 months).
TAKEAWAY:
- At 3 and 6 months, there were no significant differences in weight loss between the hungry gut positive and hungry gut negative groups.
- By 9 months, participants in the positive group lost 14.4% of their total body weight compared with 10.3% in case of participants in the negative group (P = .045).
- After a total of 12 months, the positive group lost 19.5% of their total body weight compared with 10.0% in case of participants in the negative group (P = .01).
- When used to predict the response to semaglutide, the area under the curve for the machine-learning genetic risk score was 0.76 (95% CI, 0.57-0.94; P = .04).
IN PRACTICE:
We can now tell with confidence who is going to respond to semaglutide, said Andres Acosta, MD, PhD, associate professor of medicine at Mayo Clinic. “For nonresponders, we can think about other interventions or medications that we have available.”
SOURCE:
This study was presented on May 20, 2024, at the annual Digestive Disease Week® (DDW) (Abstract 638).
LIMITATIONS:
Further prospective studies are needed to assess the validity of the test in a more diverse population and with different weight loss interventions.
DISCLOSURES:
This study was supported by a partnership between Mayo Clinic and Phenomix Sciences. Dr. Acosta is a cofounder of Phenomix Sciences.
A version of this article appeared on Medscape.com.










