User login
Erosive Esophagitis: 5 Things to Know
Erosive esophagitis (EE) is erosion of the esophageal epithelium due to chronic irritation. It can be caused by a number of factors but is primarily a result of gastroesophageal reflux disease (GERD). The main symptoms of EE are heartburn and regurgitation; other symptoms can include epigastric pain, odynophagia, dysphagia, nausea, chronic cough, dental erosion, laryngitis, and asthma. , including nonerosive esophagitis and Barrett esophagus (BE). EE occurs in approximately 30% of cases of GERD, and EE may evolve to BE in 1%-13% of cases.
Long-term management of EE focuses on relieving symptoms to allow the esophageal lining to heal, thereby reducing both acute symptoms and the risk for other complications. Management plans may incorporate lifestyle changes, such as dietary modifications and weight loss, alongside pharmacologic therapy. In extreme cases, surgery may be considered to repair a damaged esophagus and/or to prevent ongoing acid reflux. If left untreated, EE may progress, potentially leading to more serious conditions.
Here are five things to know about EE.
1. GERD is the main risk factor for EE, but not the only risk factor.
An estimated 1% of the population has EE. Risk factors other than GERD include:
Radiation therapy toxicity can cause acute or chronic EE. For individuals undergoing radiotherapy, radiation esophagitis is a relatively frequent complication. Acute esophagitis generally occurs in all patients taking radiation doses of 6000 cGy given in fractions of 1000 cGy per week. The risk is lower among patients on longer schedules and lower doses of radiotherapy.
Bacterial, viral, and fungal infections can cause EE. These include herpes, CMV, HIV, Helicobacter pylori, and Candida.
Food allergies, asthma, and eczema are associated with eosinophilic esophagitis, which disproportionately affects young men and has an estimated prevalence of 55 cases per 100,000 population.
Oral medication in pill form causes esophagitis at an estimated rate of 3.9 cases per 100,000 population per year. The mean age at diagnosis is 41.5 years. Oral bisphosphonates such as alendronate are the most common agents, along with antibiotics such as tetracycline, doxycycline, and clindamycin. There have also been reports of pill-induced esophagitis with NSAIDs, aspirin, ferrous sulfate, potassium chloride, and mexiletine.
Excessive vomiting can, in rare cases, cause esophagitis.
Certain autoimmune diseases can manifest as EE.
2. Proton pump inhibitors (PPIs) remain the preferred treatment for EE.
Several over-the-counter and prescription medications can be used to manage the symptoms of EE. PPIs are the preferred treatment both in the acute setting and for maintenance therapy. PPIs help to alleviate symptoms and promote healing of the esophageal lining by reducing the production of stomach acid. Options include omeprazole, lansoprazole, pantoprazole, rabeprazole, and esomeprazole. Many patients with EE require a dose that exceeds the FDA-approved dose for GERD. For instance, a 40-mg/d dosage of omeprazole is recommended in the latest guidelines, although the FDA-approved dosage is 20 mg/d.
H2-receptor antagonists, including famotidine, cimetidine, and nizatidine, may also be prescribed to reduce stomach acid production and promote healing in patients with EE due to GERD, but these agents are considered less efficacious than PPIs for either acute or maintenance therapy.
The potassium-competitive acid blocker (PCAB) vonoprazan is the latest agent to be indicated for EE and may provide more potent acid suppression for patients. A randomized comparative trial showed noninferiority compared with lansoprazole for healing and maintenance of healing of EE. In another randomized comparative study, the investigational PCAP fexuprazan was shown to be noninferior to the PPI esomeprazole in treating EE.
Mild GERD symptoms can be controlled by traditional antacids taken after each meal and at bedtime or with short-term use of prokinetic agents, which can help reduce acid reflux by improving esophageal and stomach motility and by increasing pressure to the lower esophageal sphincter. Gastric emptying is also accelerated by prokinetic agents. Long-term use is discouraged, as it may cause serious or life-threatening complications.
In patients who do not fully respond to PPI therapy, surgical therapy may be considered. Other candidates for surgery include younger patients, those who have difficulty adhering to treatment, postmenopausal women with osteoporosis, patients with cardiac conduction defects, and those for whom the cost of treatment is prohibitive. Surgery may also be warranted if there are extraesophageal manifestations of GERD, such as enamel erosion; respiratory issues (eg, coughing, wheezing, aspiration); or ear, nose, and throat manifestations (eg, hoarseness, sore throat, otitis media). For those who have progressed to BE, surgical intervention is also indicated.
The types of surgery for patients with EE have evolved to include both transthoracic and transabdominal fundoplication. Usually, a 360° transabdominal fundoplication is performed. General anesthesia is required for laparoscopic fundoplication, in which five small incisions are used to create a new valve at the level of the esophagogastric junction by wrapping the fundus of the stomach around the esophagus.
Laparoscopic insertion of a small band known as the LINX Reflux Management System is FDA approved to augment the lower esophageal sphincter. The system creates a natural barrier to reflux by placing a band consisting of titanium beads with magnetic cores around the esophagus just above the stomach. The magnetic bond is temporarily disrupted by swallowing, allowing food and liquid to pass.
Endoscopic therapies are another treatment option for certain patients who are not considered candidates for surgery or long-term therapy. Among the types of endoscopic procedures are radiofrequency therapy, suturing/plication, and mucosal ablation/resection techniques at the gastroesophageal junction. Full-thickness endoscopic suturing is an area of interest because this technique offers significant durability of the recreated lower esophageal sphincter.
3. PPI therapy for GERD should be stopped before endoscopy is performed to confirm a diagnosis of EE.
A clinical diagnosis of GERD can be made if the presenting symptoms are heartburn and regurgitation, without chest pain or alarm symptoms such as dysphagia, weight loss, or gastrointestinal bleeding. In this setting, once-daily PPIs are generally prescribed for 8 weeks to see if symptoms resolve. If symptoms have not resolved, a twice-daily PPI regimen may be prescribed. In patients who do not respond to PPIs, or for whom GERD returns after stopping therapy, an upper endoscopy with biopsy is recommended after 2-4 weeks off therapy to rule out other causes. Endoscopy should be the first step in diagnosis for individuals experiencing chest pain without heartburn; those in whom heart disease has been ruled out; individuals experiencing dysphagia, weight loss, or gastrointestinal bleeding; or those who have multiple risk factors for BE.
4. The most serious complication of EE is BE, which can lead to esophageal cancer.
Several complications can arise from EE. The most serious of these is BE, which can lead to esophageal adenocarcinoma. BE is characterized by the conversion of normal distal squamous esophageal epithelium to columnar epithelium. It has the potential to become malignant if it exhibits intestinal-type metaplasia. In the industrialized world, adenocarcinoma currently represents more than half of all esophageal cancers. The most common symptom of esophageal cancer is dysphagia. Other signs and symptoms include weight loss, hoarseness, chronic or intractable cough, bleeding, epigastric or retrosternal pain, frequent pneumonia, and, if metastatic, bone pain.
5. Lifestyle modifications can help control the symptoms of EE.
Guidelines recommend a number of lifestyle modification strategies to help control the symptoms of EE. Smoking cessation and weight loss are two evidence-based strategies for relieving symptoms of GERD and, ultimately, lowering the risk for esophageal cancer. One large prospective Norwegian cohort study (N = 29,610) found that stopping smoking improved GERD symptoms, but only in those with normal body mass index. In a smaller Japanese study (N = 191) specifically surveying people attempting smoking cessation, individuals who successfully stopped smoking had a 44% improvement in GERD symptoms at 1 year, vs an 18% improvement in those who continued to smoke, with no statistical difference between the success and failure groups based on patient body mass index (P = .60).
Other recommended strategies for nonpharmacologic management of EE symptoms include elevation of the head when lying down in bed and avoidance of lying down after eating, cessation of alcohol consumption, avoidance of food close to bedtime, and avoidance of trigger foods that can incite or worsen symptoms of acid reflux. Such trigger foods vary among individuals, but they often include fatty foods, coffee, chocolate, carbonated beverages, spicy foods, citrus fruits, and tomatoes.
Dr. Puerta has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Erosive esophagitis (EE) is erosion of the esophageal epithelium due to chronic irritation. It can be caused by a number of factors but is primarily a result of gastroesophageal reflux disease (GERD). The main symptoms of EE are heartburn and regurgitation; other symptoms can include epigastric pain, odynophagia, dysphagia, nausea, chronic cough, dental erosion, laryngitis, and asthma. , including nonerosive esophagitis and Barrett esophagus (BE). EE occurs in approximately 30% of cases of GERD, and EE may evolve to BE in 1%-13% of cases.
Long-term management of EE focuses on relieving symptoms to allow the esophageal lining to heal, thereby reducing both acute symptoms and the risk for other complications. Management plans may incorporate lifestyle changes, such as dietary modifications and weight loss, alongside pharmacologic therapy. In extreme cases, surgery may be considered to repair a damaged esophagus and/or to prevent ongoing acid reflux. If left untreated, EE may progress, potentially leading to more serious conditions.
Here are five things to know about EE.
1. GERD is the main risk factor for EE, but not the only risk factor.
An estimated 1% of the population has EE. Risk factors other than GERD include:
Radiation therapy toxicity can cause acute or chronic EE. For individuals undergoing radiotherapy, radiation esophagitis is a relatively frequent complication. Acute esophagitis generally occurs in all patients taking radiation doses of 6000 cGy given in fractions of 1000 cGy per week. The risk is lower among patients on longer schedules and lower doses of radiotherapy.
Bacterial, viral, and fungal infections can cause EE. These include herpes, CMV, HIV, Helicobacter pylori, and Candida.
Food allergies, asthma, and eczema are associated with eosinophilic esophagitis, which disproportionately affects young men and has an estimated prevalence of 55 cases per 100,000 population.
Oral medication in pill form causes esophagitis at an estimated rate of 3.9 cases per 100,000 population per year. The mean age at diagnosis is 41.5 years. Oral bisphosphonates such as alendronate are the most common agents, along with antibiotics such as tetracycline, doxycycline, and clindamycin. There have also been reports of pill-induced esophagitis with NSAIDs, aspirin, ferrous sulfate, potassium chloride, and mexiletine.
Excessive vomiting can, in rare cases, cause esophagitis.
Certain autoimmune diseases can manifest as EE.
2. Proton pump inhibitors (PPIs) remain the preferred treatment for EE.
Several over-the-counter and prescription medications can be used to manage the symptoms of EE. PPIs are the preferred treatment both in the acute setting and for maintenance therapy. PPIs help to alleviate symptoms and promote healing of the esophageal lining by reducing the production of stomach acid. Options include omeprazole, lansoprazole, pantoprazole, rabeprazole, and esomeprazole. Many patients with EE require a dose that exceeds the FDA-approved dose for GERD. For instance, a 40-mg/d dosage of omeprazole is recommended in the latest guidelines, although the FDA-approved dosage is 20 mg/d.
H2-receptor antagonists, including famotidine, cimetidine, and nizatidine, may also be prescribed to reduce stomach acid production and promote healing in patients with EE due to GERD, but these agents are considered less efficacious than PPIs for either acute or maintenance therapy.
The potassium-competitive acid blocker (PCAB) vonoprazan is the latest agent to be indicated for EE and may provide more potent acid suppression for patients. A randomized comparative trial showed noninferiority compared with lansoprazole for healing and maintenance of healing of EE. In another randomized comparative study, the investigational PCAP fexuprazan was shown to be noninferior to the PPI esomeprazole in treating EE.
Mild GERD symptoms can be controlled by traditional antacids taken after each meal and at bedtime or with short-term use of prokinetic agents, which can help reduce acid reflux by improving esophageal and stomach motility and by increasing pressure to the lower esophageal sphincter. Gastric emptying is also accelerated by prokinetic agents. Long-term use is discouraged, as it may cause serious or life-threatening complications.
In patients who do not fully respond to PPI therapy, surgical therapy may be considered. Other candidates for surgery include younger patients, those who have difficulty adhering to treatment, postmenopausal women with osteoporosis, patients with cardiac conduction defects, and those for whom the cost of treatment is prohibitive. Surgery may also be warranted if there are extraesophageal manifestations of GERD, such as enamel erosion; respiratory issues (eg, coughing, wheezing, aspiration); or ear, nose, and throat manifestations (eg, hoarseness, sore throat, otitis media). For those who have progressed to BE, surgical intervention is also indicated.
The types of surgery for patients with EE have evolved to include both transthoracic and transabdominal fundoplication. Usually, a 360° transabdominal fundoplication is performed. General anesthesia is required for laparoscopic fundoplication, in which five small incisions are used to create a new valve at the level of the esophagogastric junction by wrapping the fundus of the stomach around the esophagus.
Laparoscopic insertion of a small band known as the LINX Reflux Management System is FDA approved to augment the lower esophageal sphincter. The system creates a natural barrier to reflux by placing a band consisting of titanium beads with magnetic cores around the esophagus just above the stomach. The magnetic bond is temporarily disrupted by swallowing, allowing food and liquid to pass.
Endoscopic therapies are another treatment option for certain patients who are not considered candidates for surgery or long-term therapy. Among the types of endoscopic procedures are radiofrequency therapy, suturing/plication, and mucosal ablation/resection techniques at the gastroesophageal junction. Full-thickness endoscopic suturing is an area of interest because this technique offers significant durability of the recreated lower esophageal sphincter.
3. PPI therapy for GERD should be stopped before endoscopy is performed to confirm a diagnosis of EE.
A clinical diagnosis of GERD can be made if the presenting symptoms are heartburn and regurgitation, without chest pain or alarm symptoms such as dysphagia, weight loss, or gastrointestinal bleeding. In this setting, once-daily PPIs are generally prescribed for 8 weeks to see if symptoms resolve. If symptoms have not resolved, a twice-daily PPI regimen may be prescribed. In patients who do not respond to PPIs, or for whom GERD returns after stopping therapy, an upper endoscopy with biopsy is recommended after 2-4 weeks off therapy to rule out other causes. Endoscopy should be the first step in diagnosis for individuals experiencing chest pain without heartburn; those in whom heart disease has been ruled out; individuals experiencing dysphagia, weight loss, or gastrointestinal bleeding; or those who have multiple risk factors for BE.
4. The most serious complication of EE is BE, which can lead to esophageal cancer.
Several complications can arise from EE. The most serious of these is BE, which can lead to esophageal adenocarcinoma. BE is characterized by the conversion of normal distal squamous esophageal epithelium to columnar epithelium. It has the potential to become malignant if it exhibits intestinal-type metaplasia. In the industrialized world, adenocarcinoma currently represents more than half of all esophageal cancers. The most common symptom of esophageal cancer is dysphagia. Other signs and symptoms include weight loss, hoarseness, chronic or intractable cough, bleeding, epigastric or retrosternal pain, frequent pneumonia, and, if metastatic, bone pain.
5. Lifestyle modifications can help control the symptoms of EE.
Guidelines recommend a number of lifestyle modification strategies to help control the symptoms of EE. Smoking cessation and weight loss are two evidence-based strategies for relieving symptoms of GERD and, ultimately, lowering the risk for esophageal cancer. One large prospective Norwegian cohort study (N = 29,610) found that stopping smoking improved GERD symptoms, but only in those with normal body mass index. In a smaller Japanese study (N = 191) specifically surveying people attempting smoking cessation, individuals who successfully stopped smoking had a 44% improvement in GERD symptoms at 1 year, vs an 18% improvement in those who continued to smoke, with no statistical difference between the success and failure groups based on patient body mass index (P = .60).
Other recommended strategies for nonpharmacologic management of EE symptoms include elevation of the head when lying down in bed and avoidance of lying down after eating, cessation of alcohol consumption, avoidance of food close to bedtime, and avoidance of trigger foods that can incite or worsen symptoms of acid reflux. Such trigger foods vary among individuals, but they often include fatty foods, coffee, chocolate, carbonated beverages, spicy foods, citrus fruits, and tomatoes.
Dr. Puerta has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Erosive esophagitis (EE) is erosion of the esophageal epithelium due to chronic irritation. It can be caused by a number of factors but is primarily a result of gastroesophageal reflux disease (GERD). The main symptoms of EE are heartburn and regurgitation; other symptoms can include epigastric pain, odynophagia, dysphagia, nausea, chronic cough, dental erosion, laryngitis, and asthma. , including nonerosive esophagitis and Barrett esophagus (BE). EE occurs in approximately 30% of cases of GERD, and EE may evolve to BE in 1%-13% of cases.
Long-term management of EE focuses on relieving symptoms to allow the esophageal lining to heal, thereby reducing both acute symptoms and the risk for other complications. Management plans may incorporate lifestyle changes, such as dietary modifications and weight loss, alongside pharmacologic therapy. In extreme cases, surgery may be considered to repair a damaged esophagus and/or to prevent ongoing acid reflux. If left untreated, EE may progress, potentially leading to more serious conditions.
Here are five things to know about EE.
1. GERD is the main risk factor for EE, but not the only risk factor.
An estimated 1% of the population has EE. Risk factors other than GERD include:
Radiation therapy toxicity can cause acute or chronic EE. For individuals undergoing radiotherapy, radiation esophagitis is a relatively frequent complication. Acute esophagitis generally occurs in all patients taking radiation doses of 6000 cGy given in fractions of 1000 cGy per week. The risk is lower among patients on longer schedules and lower doses of radiotherapy.
Bacterial, viral, and fungal infections can cause EE. These include herpes, CMV, HIV, Helicobacter pylori, and Candida.
Food allergies, asthma, and eczema are associated with eosinophilic esophagitis, which disproportionately affects young men and has an estimated prevalence of 55 cases per 100,000 population.
Oral medication in pill form causes esophagitis at an estimated rate of 3.9 cases per 100,000 population per year. The mean age at diagnosis is 41.5 years. Oral bisphosphonates such as alendronate are the most common agents, along with antibiotics such as tetracycline, doxycycline, and clindamycin. There have also been reports of pill-induced esophagitis with NSAIDs, aspirin, ferrous sulfate, potassium chloride, and mexiletine.
Excessive vomiting can, in rare cases, cause esophagitis.
Certain autoimmune diseases can manifest as EE.
2. Proton pump inhibitors (PPIs) remain the preferred treatment for EE.
Several over-the-counter and prescription medications can be used to manage the symptoms of EE. PPIs are the preferred treatment both in the acute setting and for maintenance therapy. PPIs help to alleviate symptoms and promote healing of the esophageal lining by reducing the production of stomach acid. Options include omeprazole, lansoprazole, pantoprazole, rabeprazole, and esomeprazole. Many patients with EE require a dose that exceeds the FDA-approved dose for GERD. For instance, a 40-mg/d dosage of omeprazole is recommended in the latest guidelines, although the FDA-approved dosage is 20 mg/d.
H2-receptor antagonists, including famotidine, cimetidine, and nizatidine, may also be prescribed to reduce stomach acid production and promote healing in patients with EE due to GERD, but these agents are considered less efficacious than PPIs for either acute or maintenance therapy.
The potassium-competitive acid blocker (PCAB) vonoprazan is the latest agent to be indicated for EE and may provide more potent acid suppression for patients. A randomized comparative trial showed noninferiority compared with lansoprazole for healing and maintenance of healing of EE. In another randomized comparative study, the investigational PCAP fexuprazan was shown to be noninferior to the PPI esomeprazole in treating EE.
Mild GERD symptoms can be controlled by traditional antacids taken after each meal and at bedtime or with short-term use of prokinetic agents, which can help reduce acid reflux by improving esophageal and stomach motility and by increasing pressure to the lower esophageal sphincter. Gastric emptying is also accelerated by prokinetic agents. Long-term use is discouraged, as it may cause serious or life-threatening complications.
In patients who do not fully respond to PPI therapy, surgical therapy may be considered. Other candidates for surgery include younger patients, those who have difficulty adhering to treatment, postmenopausal women with osteoporosis, patients with cardiac conduction defects, and those for whom the cost of treatment is prohibitive. Surgery may also be warranted if there are extraesophageal manifestations of GERD, such as enamel erosion; respiratory issues (eg, coughing, wheezing, aspiration); or ear, nose, and throat manifestations (eg, hoarseness, sore throat, otitis media). For those who have progressed to BE, surgical intervention is also indicated.
The types of surgery for patients with EE have evolved to include both transthoracic and transabdominal fundoplication. Usually, a 360° transabdominal fundoplication is performed. General anesthesia is required for laparoscopic fundoplication, in which five small incisions are used to create a new valve at the level of the esophagogastric junction by wrapping the fundus of the stomach around the esophagus.
Laparoscopic insertion of a small band known as the LINX Reflux Management System is FDA approved to augment the lower esophageal sphincter. The system creates a natural barrier to reflux by placing a band consisting of titanium beads with magnetic cores around the esophagus just above the stomach. The magnetic bond is temporarily disrupted by swallowing, allowing food and liquid to pass.
Endoscopic therapies are another treatment option for certain patients who are not considered candidates for surgery or long-term therapy. Among the types of endoscopic procedures are radiofrequency therapy, suturing/plication, and mucosal ablation/resection techniques at the gastroesophageal junction. Full-thickness endoscopic suturing is an area of interest because this technique offers significant durability of the recreated lower esophageal sphincter.
3. PPI therapy for GERD should be stopped before endoscopy is performed to confirm a diagnosis of EE.
A clinical diagnosis of GERD can be made if the presenting symptoms are heartburn and regurgitation, without chest pain or alarm symptoms such as dysphagia, weight loss, or gastrointestinal bleeding. In this setting, once-daily PPIs are generally prescribed for 8 weeks to see if symptoms resolve. If symptoms have not resolved, a twice-daily PPI regimen may be prescribed. In patients who do not respond to PPIs, or for whom GERD returns after stopping therapy, an upper endoscopy with biopsy is recommended after 2-4 weeks off therapy to rule out other causes. Endoscopy should be the first step in diagnosis for individuals experiencing chest pain without heartburn; those in whom heart disease has been ruled out; individuals experiencing dysphagia, weight loss, or gastrointestinal bleeding; or those who have multiple risk factors for BE.
4. The most serious complication of EE is BE, which can lead to esophageal cancer.
Several complications can arise from EE. The most serious of these is BE, which can lead to esophageal adenocarcinoma. BE is characterized by the conversion of normal distal squamous esophageal epithelium to columnar epithelium. It has the potential to become malignant if it exhibits intestinal-type metaplasia. In the industrialized world, adenocarcinoma currently represents more than half of all esophageal cancers. The most common symptom of esophageal cancer is dysphagia. Other signs and symptoms include weight loss, hoarseness, chronic or intractable cough, bleeding, epigastric or retrosternal pain, frequent pneumonia, and, if metastatic, bone pain.
5. Lifestyle modifications can help control the symptoms of EE.
Guidelines recommend a number of lifestyle modification strategies to help control the symptoms of EE. Smoking cessation and weight loss are two evidence-based strategies for relieving symptoms of GERD and, ultimately, lowering the risk for esophageal cancer. One large prospective Norwegian cohort study (N = 29,610) found that stopping smoking improved GERD symptoms, but only in those with normal body mass index. In a smaller Japanese study (N = 191) specifically surveying people attempting smoking cessation, individuals who successfully stopped smoking had a 44% improvement in GERD symptoms at 1 year, vs an 18% improvement in those who continued to smoke, with no statistical difference between the success and failure groups based on patient body mass index (P = .60).
Other recommended strategies for nonpharmacologic management of EE symptoms include elevation of the head when lying down in bed and avoidance of lying down after eating, cessation of alcohol consumption, avoidance of food close to bedtime, and avoidance of trigger foods that can incite or worsen symptoms of acid reflux. Such trigger foods vary among individuals, but they often include fatty foods, coffee, chocolate, carbonated beverages, spicy foods, citrus fruits, and tomatoes.
Dr. Puerta has disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Centrifugally Spreading Lymphocutaneous Sporotrichosis: A Rare Cutaneous Manifestation
To the Editor:
Sporotrichosis refers to a subacute to chronic fungal infection that usually involves the cutaneous and subcutaneous tissues and is caused by the introduction of Sporothrix, a dimorphic fungus, through the skin. We present a case of chronic atypical lymphocutaneous sporotrichosis.
A 46-year-old man presented to the outpatient dermatology clinic for follow-up for a rash on the right leg that spread to the thigh and became painful and pruritic. It initially developed 8 years prior to the current presentation after he sustained trauma to the leg from an electroshock weapon. One year prior to the current presentation, he had presented to the emergency department and was prescribed doxycycline 100 mg twice daily for 7 days as well as bacitracin ointment. He also was instructed to follow up with dermatology, but a lack of health insurance and other socioeconomic barriers prevented him from seeking dermatologic care. Nine months later, he again presented to the emergency department due to a motor vehicle accident. Computed tomography (CT) of the right leg revealed exophytic dermal masses, inflammatory stranding of the subcutaneous tissue, and right inguinal lymph nodes measuring up to 1.4 cm; there was no osteoarticular involvement. At that time, the patient was applying gentian violet to the skin lesions and taking hydroxyzine 50 mg 3 times daily as needed for pruritus with minimal relief. Financial support was provided for follow-up with dermatology, which occurred almost 5 months later.
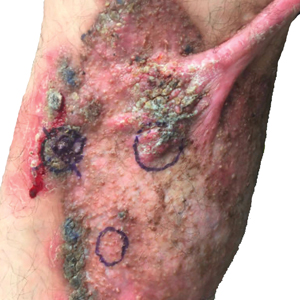
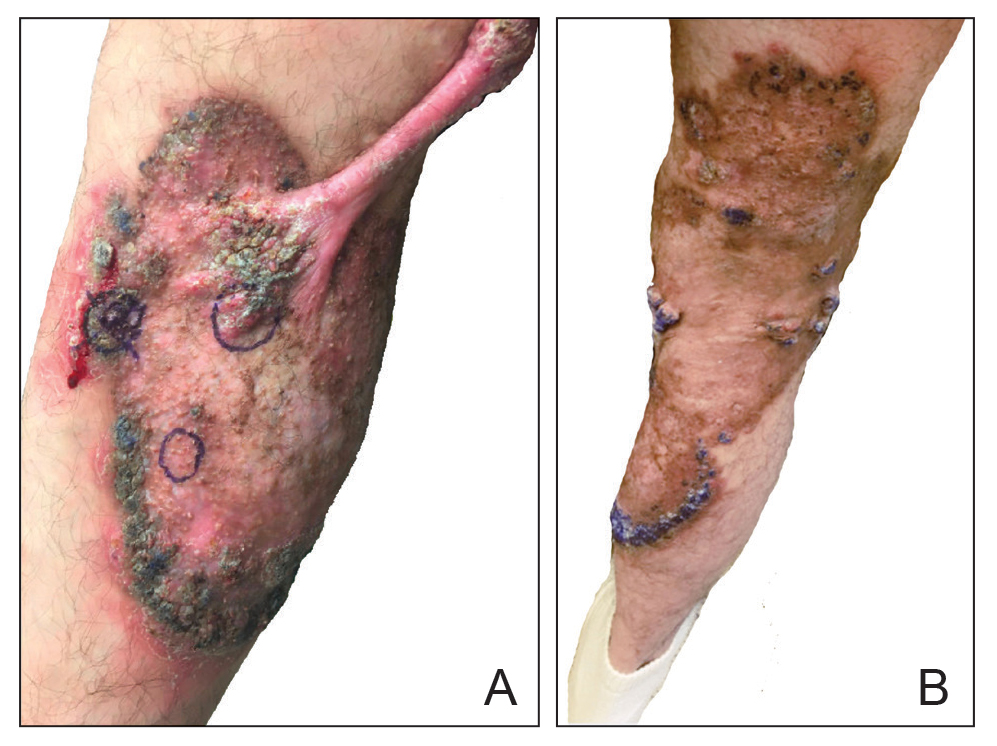
At the current presentation, physical examination revealed a large annular plaque with verrucous, scaly, erythematous borders and a hypopigmented atrophic center extending from the medial aspect of the right leg to the posterior thigh. Numerous pink, scaly, crusted nodules were scattered primarily along the periphery, with some evidence of draining sinus tracts. In addition, a fibrotic pink linear plaque extended from the medial right leg to the popliteal fossa, consistent with a keloid. Violet staining along the periphery of the lesion also was appreciated secondary to the application of topical gentian violet (Figure 1).
Based on the chronic history and morphology, a diagnosis of a chronic fungal or atypical mycobacterial infection was favored. In particular, chromoblastomycosis, cutaneous tuberculosis (eg, scrofuloderma, lupus vulgaris, tuberculosis verrucosa cutis), and atypical mycobacterial infection were highest on the differential, as these conditions often exhibit annular, nodular, verrucous, and/or atrophic lesions. The nodularity, crusting, and draining sinus tracts also raised the possibility of mycetoma. Given the extension of the lesion from the lower to upper leg, a sporotrichoid infection also was considered but was thought to be less likely based on the annular configuration.
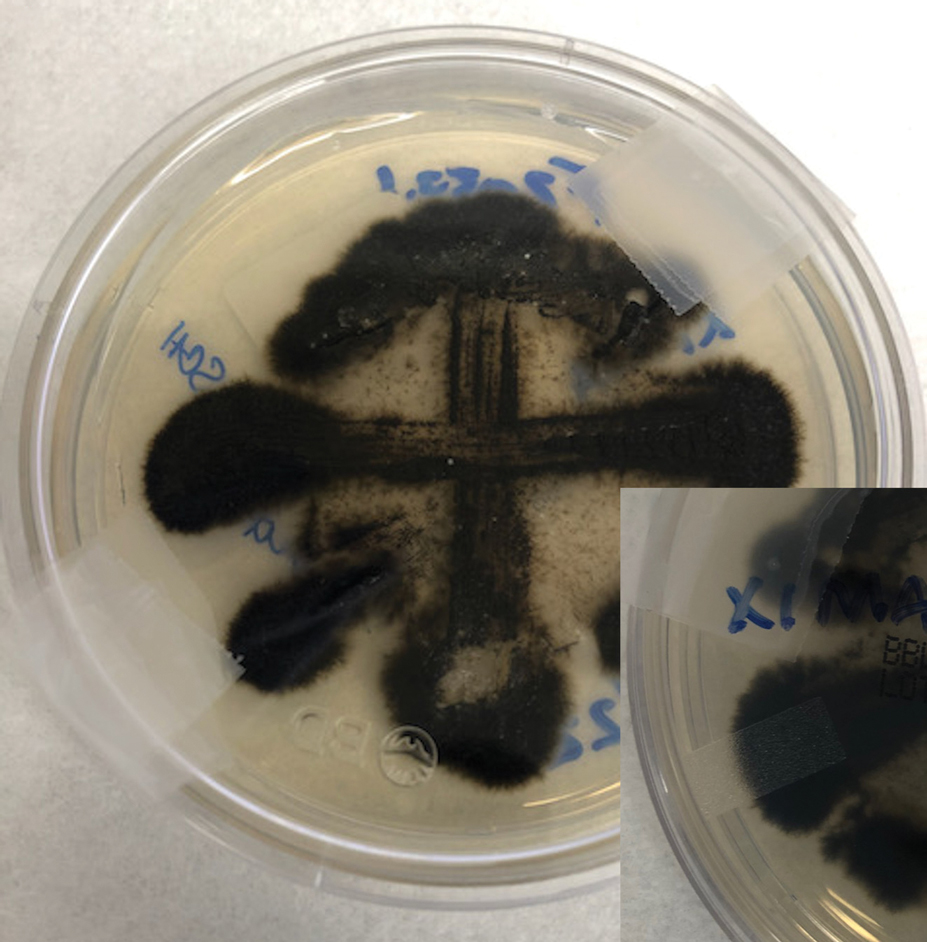
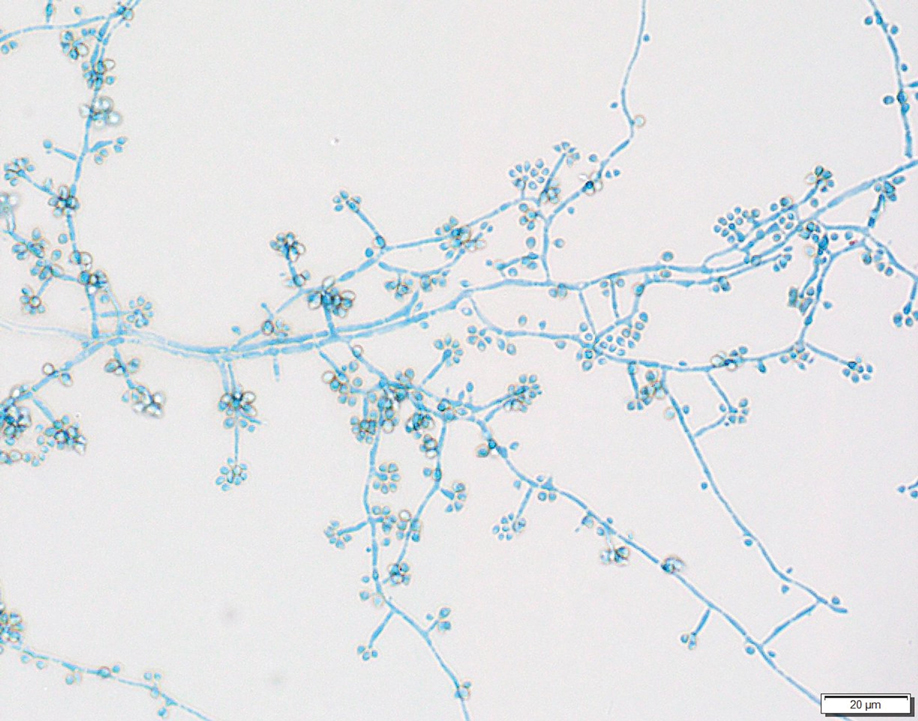
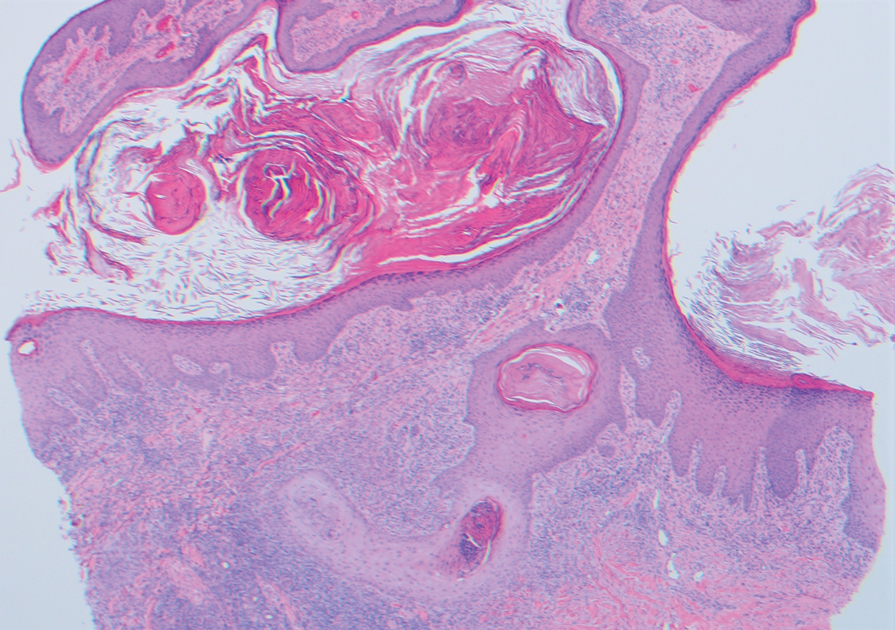
Two 4-mm punch biopsies were taken from a peripheral nodule—one for routine histology and another for bacterial, fungal, and mycobacterial cultures. An interferon-gamma release assay also was ordered to evaluate for immune responses indicative of prior Mycobacterium tuberculosis infection, but the patient did not obtain this for unknown reasons. Histology demonstrated pseudoepitheliomatous hyperplasia and necrotizing granulomas, which suggested an infectious etiology, but no organisms were identified on tissue staining and all cultures were negative for growth at 6 weeks. The patient was asked to return at that point, and 4 additional scouting biopsies were performed and sent for routine histology, M tuberculosis nucleic acid amplification testing, and microbiologic cultures (ie, bacterial, mycobacterial, fungal, nocardia, actinomycetes). Within 1 week, a filamentous organism with pigmentation visible on the front and back of a Sabouraud dextrose agar plate was identified on fungal culture (Figure 2). Microscopic evaluation of this mold with lactophenol blue stain revealed thin septate hyphae with conidiophores arising at right angles that bore clusters of microconidia (Figure 3). Sequencing analysis ultimately identified this organism as Sporothrix schenckii. Routine histology demonstrated pseudoepitheliomatous hyperplasia with scattered intraepidermal collections of neutrophils (Figure 4). The dermis showed a dense, superficial, and deep infiltrate composed of lymphocytes, histiocytes, and plasma cells with occasional neutrophils and eosinophils. A Grocott-Gomori methenamine-silver stain revealed a cluster of ovoid yeast forms within the stratum corneum (Figure 5). The patient was referred to infectious disease for follow-up and treatment.
The patient later visited a community clinic providing dermatologic care for patients without insurance. He was started on itraconazole 200 mg daily for a total of 6 months until dermatologic clearance of the cutaneous lesions was observed. He was followed by the clinic with laboratory tests including a liver function test. At follow-up 8 months later, a repeat biopsy was performed to ensure histologic clearance of the sporotrichosis, which revealed a dermal scar and no evidence of residual infection.
Sporothrix schenckii was first isolated in 1898 by Benjamin Schenck, a student at Johns Hopkins Medicine (Baltimore, Maryland), and identified by a mycologist as sporotricha.1 Species within the genus Sporothrix are unique in that the fungi are both dimorphic (growing as a mold at 25 °C but as a yeast at 37 °C) and dematiaceous (dark pigmentation from melanin is visible on inspection of the anterior and reverse sides of culture plates). Infection usually occurs when cutaneous or subcutaneous tissues are exposed to the fungus via microabrasions; activities thought to contribute to exposure include gardening, agricultural work, animal husbandry, and feline scratches.2 Although skin trauma frequently is considered the primary route of infection, patient recall is variable, with one study noting that only 37.7% of patients recalled trauma and another study similarly demonstrating a patient recall rate of 25%.3,4
Lymphocutaneous sporotrichosis is the most common presentation of the fungal infection,5 and clinical cases may be classified into 1 of 4 categories: (1) lymphangitic lesions—papules at the site of inoculation with spread along the lymphatic channels; (2) localized (fixed) cutaneous lesions—1 or 2 lesions at the inoculation site; (3) disseminated (multifocal) cutaneous lesions; and (4) extracutaneous lesions.6 Extracutaneous manifestations of this infection most notably have been reported as pulmonary disease through inhalation of conidia or through dissemination in immunocompromised hosts.7 Our patient’s infection was categorized as lymphangitic lesions due to spread from the lower to upper leg, albeit in a highly atypical, annular fashion. A review of systems was otherwise negative, and CT ruled out osteoarticular involvement.

In addition to socioeconomic barriers, several factors contributed to a delayed diagnosis in this patient including the annular presentation with central hypopigmentation and atrophy, negative initial microbiological cultures and lack of visualization of organisms on histopathology, and the consequent need for repeat biopsies. For lymphocutaneous sporotrichosis, the typical presentation consists of a papule or ulcerated nodule at the site of inoculation with subsequent linear spread along lymphatic channels. This classic sporotrichoid pattern is a key diagnostic clue for identifying sporotrichosis but was absent at the time our patient presented for medical care. Rather, the sporotrichoid spread seemed to have occurred in a centrifugal fashion up the leg. Few case reports have documented an annular presentation of lymphocutaneous sporotrichosis,8-13 and one report described central atrophy and hypopigmentation.10 Pain and pruritus, which were present in our patient, rarely are documented.9 Finally, the diagnosis of cutaneous fungal infections may require multiple biopsies due to the variable abundance of viable organisms in tissue specimens as well as the fastidious growth characteristics of these organisms. Furthermore, sensitivity often is low for both fungal and mycobacterial cultures, and cultures may take days to weeks to yield growth.14,15 For these reasons, empiric therapy and repeat biopsies often are pursued if clinical suspicion is high enough.16 Our patient returned for multiple scouting biopsies after the initial tissue culture was negative and was even considered for empiric treatment against Mycobacterium prior to positive fungal cultures.
Another unique aspect of our case was the presence of a keloid. It is difficult to know if this keloid was secondary to the trauma the patient sustained in the inciting incident or formed from the fungal infection. Interestingly, it has been hypothesized that fungal infections may contribute to keloid and hypertrophic scar formation.17 In a case series of 3 patients with either keloids or hypertrophic scars and concomitant tinea infection, there was notable improvement in the appearance of the scars 2 weeks after beginning itraconazole therapy.17 However, it is not yet known if a fungal infection can contribute to the pathogenesis of keloid formation.
As with other aspects of this case, the length of time the patient went without diagnosis and treatment was unusual and may help explain the atypical presentation. Although the incubation period for S schenckii can vary, most reports identify patients as seeking medical attention within 1 year of rash onset.18-20 In our case, the patient was not diagnosed until 8 years after his symptoms began, requiring multiple referrals, multiple health system touchpoints, and an institution-specific financial aid program. As such, this case also highlights the potential need for a multidisciplinary team approach when caring for patients with poor access to health care.
In conclusion, this case illustrates a unique presentation of lymphocutaneous sporotrichosis that may mimic other chronic infections and result in delayed diagnosis. Although lymphangitic sporotrichosis generally is recognized as having a linear distribution, mounting evidence from this report and others suggests an annular presentation also is possible. Pruritus or pain is rare but should not preclude a diagnosis of sporotrichosis if present. For patients with limited access to health care resources, it is especially important to involve multiple members of the health care team, including social workers and specialists, to prevent a protracted and severe course of disease.
- Schenck BR. On refractory subcutaneous abscesses caused by a fungus possibly related to the sporotricha. Bulletin of the Johns Hopkins Hospital. 1898;93:286-290.
- de Lima Barros MB, de Almeida Paes R, Schubach AO. Sporothrix schenckii and sporotrichosis. Clin Microbiol Rev. 2011;24:633-654. doi:10.1128/CMR.00007-11
- Crevasse L, Ellner PD. An outbreak of sporotrichosis in florida. J Am Med Assoc. 1960;173:29-33. doi:10.1001/jama.1960.03020190031006
- Mayorga R, Cáceres A, Toriello C, et al. An endemic area of sporotrichosis in Guatemala [in French]. Sabouraudia. 1978;16:185-198.
- Morris-Jones R. Sporotrichosis. Clin Exp Dermatol. 2002;27:427-431. doi:10.1046/j.1365-2230.2002.01087.x
- Sampaio SA, Da Lacaz CS. Clinical and statistical studies on sporotrichosis in Sao Paulo (Brazil). Article in German. Hautarzt. 1959;10:490-493.
- Ramos-e-Silva M, Vasconcelos C, Carneiro S, et al. Sporotrichosis. Clin Dermatol. 2007;25:181-187. doi:10.1016/j.clindermatol.2006.05.006
- Williams BA, Jennings TA, Rushing EC, et al. Sporotrichosis on the face of a 7-year-old boy following a bicycle accident. Pediatr Dermatol. 2013;30:E246-E247. doi:10.1111/j.1525-1470.2011.01696.x
- Vaishampayan SS, Borde P. An unusual presentation of sporotrichosis. Indian J Dermatol. 2013;58:409. doi:10.4103/0019-5154.117350
- Qin J, Zhang J. Sporotrichosis. N Engl J Med. 2019;380:771. doi:10.1056/NEJMicm1809179
- Patel A, Mudenda V, Lakhi S, et al. A 27-year-old severely immunosuppressed female with misleading clinical features of disseminated cutaneous sporotrichosis. Case Rep Dermatol Med. 2016;2016:1-4. doi:10.1155/2016/9403690
- de Oliveira-Esteves ICMR, Almeida Rosa da Silva G, Eyer-Silva WA, et al. Rapidly progressive disseminated sporotrichosis as the first presentation of HIV infection in a patient with a very low CD4 cell count. Case Rep Infect Dis. 2017;2017:4713140. doi:10.1155/2017/4713140
- Singh S, Bachaspatimayum R, Meetei U, et al. Terbinafine in fixed cutaneous sporotrichosis: a case series. J Clin Diagnostic Res. 2018;12:FR01-FR03. doi:10.7860/JCDR/2018/25315.12223
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280. doi:10.1128/CMR.00053-10
- Peters F, Batinica M, Plum G, et al. Bug or no bug: challenges in diagnosing cutaneous mycobacterial infections. J Ger Soc Dermatol. 2016;14:1227-1236. doi:10.1111/ddg.13001
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973. doi:10.1155/2018/7201973
- Okada E, Maruyama Y. Are keloids and hypertrophic scars caused by fungal infection? . Plast Reconstr Surg. 2007;120:814-815. doi:10.1097/01.prs.0000278813.23244.3f
- Pappas PG, Tellez I, Deep AE, et al. Sporotrichosis in Peru: description of an area of hyperendemicity. Clin Infect Dis. 2000;30:65-70. doi:10.1086/313607
- McGuinness SL, Boyd R, Kidd S, et al. Epidemiological investigation of an outbreak of cutaneous sporotrichosis, Northern Territory, Australia. BMC Infect Dis. 2016;16:1-7. doi:10.1186/s12879-016-1338-0
- Rojas FD, Fernández MS, Lucchelli JM, et al. Cavitary pulmonary sporotrichosis: case report and literature review. Mycopathologia. 2017;182:1119-1123. doi:10.1007/s11046-017-0197-6
To the Editor:
Sporotrichosis refers to a subacute to chronic fungal infection that usually involves the cutaneous and subcutaneous tissues and is caused by the introduction of Sporothrix, a dimorphic fungus, through the skin. We present a case of chronic atypical lymphocutaneous sporotrichosis.
A 46-year-old man presented to the outpatient dermatology clinic for follow-up for a rash on the right leg that spread to the thigh and became painful and pruritic. It initially developed 8 years prior to the current presentation after he sustained trauma to the leg from an electroshock weapon. One year prior to the current presentation, he had presented to the emergency department and was prescribed doxycycline 100 mg twice daily for 7 days as well as bacitracin ointment. He also was instructed to follow up with dermatology, but a lack of health insurance and other socioeconomic barriers prevented him from seeking dermatologic care. Nine months later, he again presented to the emergency department due to a motor vehicle accident. Computed tomography (CT) of the right leg revealed exophytic dermal masses, inflammatory stranding of the subcutaneous tissue, and right inguinal lymph nodes measuring up to 1.4 cm; there was no osteoarticular involvement. At that time, the patient was applying gentian violet to the skin lesions and taking hydroxyzine 50 mg 3 times daily as needed for pruritus with minimal relief. Financial support was provided for follow-up with dermatology, which occurred almost 5 months later.
At the current presentation, physical examination revealed a large annular plaque with verrucous, scaly, erythematous borders and a hypopigmented atrophic center extending from the medial aspect of the right leg to the posterior thigh. Numerous pink, scaly, crusted nodules were scattered primarily along the periphery, with some evidence of draining sinus tracts. In addition, a fibrotic pink linear plaque extended from the medial right leg to the popliteal fossa, consistent with a keloid. Violet staining along the periphery of the lesion also was appreciated secondary to the application of topical gentian violet (Figure 1).

Based on the chronic history and morphology, a diagnosis of a chronic fungal or atypical mycobacterial infection was favored. In particular, chromoblastomycosis, cutaneous tuberculosis (eg, scrofuloderma, lupus vulgaris, tuberculosis verrucosa cutis), and atypical mycobacterial infection were highest on the differential, as these conditions often exhibit annular, nodular, verrucous, and/or atrophic lesions. The nodularity, crusting, and draining sinus tracts also raised the possibility of mycetoma. Given the extension of the lesion from the lower to upper leg, a sporotrichoid infection also was considered but was thought to be less likely based on the annular configuration.
Two 4-mm punch biopsies were taken from a peripheral nodule—one for routine histology and another for bacterial, fungal, and mycobacterial cultures. An interferon-gamma release assay also was ordered to evaluate for immune responses indicative of prior Mycobacterium tuberculosis infection, but the patient did not obtain this for unknown reasons. Histology demonstrated pseudoepitheliomatous hyperplasia and necrotizing granulomas, which suggested an infectious etiology, but no organisms were identified on tissue staining and all cultures were negative for growth at 6 weeks. The patient was asked to return at that point, and 4 additional scouting biopsies were performed and sent for routine histology, M tuberculosis nucleic acid amplification testing, and microbiologic cultures (ie, bacterial, mycobacterial, fungal, nocardia, actinomycetes). Within 1 week, a filamentous organism with pigmentation visible on the front and back of a Sabouraud dextrose agar plate was identified on fungal culture (Figure 2). Microscopic evaluation of this mold with lactophenol blue stain revealed thin septate hyphae with conidiophores arising at right angles that bore clusters of microconidia (Figure 3). Sequencing analysis ultimately identified this organism as Sporothrix schenckii. Routine histology demonstrated pseudoepitheliomatous hyperplasia with scattered intraepidermal collections of neutrophils (Figure 4). The dermis showed a dense, superficial, and deep infiltrate composed of lymphocytes, histiocytes, and plasma cells with occasional neutrophils and eosinophils. A Grocott-Gomori methenamine-silver stain revealed a cluster of ovoid yeast forms within the stratum corneum (Figure 5). The patient was referred to infectious disease for follow-up and treatment.

The patient later visited a community clinic providing dermatologic care for patients without insurance. He was started on itraconazole 200 mg daily for a total of 6 months until dermatologic clearance of the cutaneous lesions was observed. He was followed by the clinic with laboratory tests including a liver function test. At follow-up 8 months later, a repeat biopsy was performed to ensure histologic clearance of the sporotrichosis, which revealed a dermal scar and no evidence of residual infection.

Sporothrix schenckii was first isolated in 1898 by Benjamin Schenck, a student at Johns Hopkins Medicine (Baltimore, Maryland), and identified by a mycologist as sporotricha.1 Species within the genus Sporothrix are unique in that the fungi are both dimorphic (growing as a mold at 25 °C but as a yeast at 37 °C) and dematiaceous (dark pigmentation from melanin is visible on inspection of the anterior and reverse sides of culture plates). Infection usually occurs when cutaneous or subcutaneous tissues are exposed to the fungus via microabrasions; activities thought to contribute to exposure include gardening, agricultural work, animal husbandry, and feline scratches.2 Although skin trauma frequently is considered the primary route of infection, patient recall is variable, with one study noting that only 37.7% of patients recalled trauma and another study similarly demonstrating a patient recall rate of 25%.3,4

Lymphocutaneous sporotrichosis is the most common presentation of the fungal infection,5 and clinical cases may be classified into 1 of 4 categories: (1) lymphangitic lesions—papules at the site of inoculation with spread along the lymphatic channels; (2) localized (fixed) cutaneous lesions—1 or 2 lesions at the inoculation site; (3) disseminated (multifocal) cutaneous lesions; and (4) extracutaneous lesions.6 Extracutaneous manifestations of this infection most notably have been reported as pulmonary disease through inhalation of conidia or through dissemination in immunocompromised hosts.7 Our patient’s infection was categorized as lymphangitic lesions due to spread from the lower to upper leg, albeit in a highly atypical, annular fashion. A review of systems was otherwise negative, and CT ruled out osteoarticular involvement.

In addition to socioeconomic barriers, several factors contributed to a delayed diagnosis in this patient including the annular presentation with central hypopigmentation and atrophy, negative initial microbiological cultures and lack of visualization of organisms on histopathology, and the consequent need for repeat biopsies. For lymphocutaneous sporotrichosis, the typical presentation consists of a papule or ulcerated nodule at the site of inoculation with subsequent linear spread along lymphatic channels. This classic sporotrichoid pattern is a key diagnostic clue for identifying sporotrichosis but was absent at the time our patient presented for medical care. Rather, the sporotrichoid spread seemed to have occurred in a centrifugal fashion up the leg. Few case reports have documented an annular presentation of lymphocutaneous sporotrichosis,8-13 and one report described central atrophy and hypopigmentation.10 Pain and pruritus, which were present in our patient, rarely are documented.9 Finally, the diagnosis of cutaneous fungal infections may require multiple biopsies due to the variable abundance of viable organisms in tissue specimens as well as the fastidious growth characteristics of these organisms. Furthermore, sensitivity often is low for both fungal and mycobacterial cultures, and cultures may take days to weeks to yield growth.14,15 For these reasons, empiric therapy and repeat biopsies often are pursued if clinical suspicion is high enough.16 Our patient returned for multiple scouting biopsies after the initial tissue culture was negative and was even considered for empiric treatment against Mycobacterium prior to positive fungal cultures.
Another unique aspect of our case was the presence of a keloid. It is difficult to know if this keloid was secondary to the trauma the patient sustained in the inciting incident or formed from the fungal infection. Interestingly, it has been hypothesized that fungal infections may contribute to keloid and hypertrophic scar formation.17 In a case series of 3 patients with either keloids or hypertrophic scars and concomitant tinea infection, there was notable improvement in the appearance of the scars 2 weeks after beginning itraconazole therapy.17 However, it is not yet known if a fungal infection can contribute to the pathogenesis of keloid formation.
As with other aspects of this case, the length of time the patient went without diagnosis and treatment was unusual and may help explain the atypical presentation. Although the incubation period for S schenckii can vary, most reports identify patients as seeking medical attention within 1 year of rash onset.18-20 In our case, the patient was not diagnosed until 8 years after his symptoms began, requiring multiple referrals, multiple health system touchpoints, and an institution-specific financial aid program. As such, this case also highlights the potential need for a multidisciplinary team approach when caring for patients with poor access to health care.
In conclusion, this case illustrates a unique presentation of lymphocutaneous sporotrichosis that may mimic other chronic infections and result in delayed diagnosis. Although lymphangitic sporotrichosis generally is recognized as having a linear distribution, mounting evidence from this report and others suggests an annular presentation also is possible. Pruritus or pain is rare but should not preclude a diagnosis of sporotrichosis if present. For patients with limited access to health care resources, it is especially important to involve multiple members of the health care team, including social workers and specialists, to prevent a protracted and severe course of disease.
To the Editor:
Sporotrichosis refers to a subacute to chronic fungal infection that usually involves the cutaneous and subcutaneous tissues and is caused by the introduction of Sporothrix, a dimorphic fungus, through the skin. We present a case of chronic atypical lymphocutaneous sporotrichosis.
A 46-year-old man presented to the outpatient dermatology clinic for follow-up for a rash on the right leg that spread to the thigh and became painful and pruritic. It initially developed 8 years prior to the current presentation after he sustained trauma to the leg from an electroshock weapon. One year prior to the current presentation, he had presented to the emergency department and was prescribed doxycycline 100 mg twice daily for 7 days as well as bacitracin ointment. He also was instructed to follow up with dermatology, but a lack of health insurance and other socioeconomic barriers prevented him from seeking dermatologic care. Nine months later, he again presented to the emergency department due to a motor vehicle accident. Computed tomography (CT) of the right leg revealed exophytic dermal masses, inflammatory stranding of the subcutaneous tissue, and right inguinal lymph nodes measuring up to 1.4 cm; there was no osteoarticular involvement. At that time, the patient was applying gentian violet to the skin lesions and taking hydroxyzine 50 mg 3 times daily as needed for pruritus with minimal relief. Financial support was provided for follow-up with dermatology, which occurred almost 5 months later.
At the current presentation, physical examination revealed a large annular plaque with verrucous, scaly, erythematous borders and a hypopigmented atrophic center extending from the medial aspect of the right leg to the posterior thigh. Numerous pink, scaly, crusted nodules were scattered primarily along the periphery, with some evidence of draining sinus tracts. In addition, a fibrotic pink linear plaque extended from the medial right leg to the popliteal fossa, consistent with a keloid. Violet staining along the periphery of the lesion also was appreciated secondary to the application of topical gentian violet (Figure 1).

Based on the chronic history and morphology, a diagnosis of a chronic fungal or atypical mycobacterial infection was favored. In particular, chromoblastomycosis, cutaneous tuberculosis (eg, scrofuloderma, lupus vulgaris, tuberculosis verrucosa cutis), and atypical mycobacterial infection were highest on the differential, as these conditions often exhibit annular, nodular, verrucous, and/or atrophic lesions. The nodularity, crusting, and draining sinus tracts also raised the possibility of mycetoma. Given the extension of the lesion from the lower to upper leg, a sporotrichoid infection also was considered but was thought to be less likely based on the annular configuration.
Two 4-mm punch biopsies were taken from a peripheral nodule—one for routine histology and another for bacterial, fungal, and mycobacterial cultures. An interferon-gamma release assay also was ordered to evaluate for immune responses indicative of prior Mycobacterium tuberculosis infection, but the patient did not obtain this for unknown reasons. Histology demonstrated pseudoepitheliomatous hyperplasia and necrotizing granulomas, which suggested an infectious etiology, but no organisms were identified on tissue staining and all cultures were negative for growth at 6 weeks. The patient was asked to return at that point, and 4 additional scouting biopsies were performed and sent for routine histology, M tuberculosis nucleic acid amplification testing, and microbiologic cultures (ie, bacterial, mycobacterial, fungal, nocardia, actinomycetes). Within 1 week, a filamentous organism with pigmentation visible on the front and back of a Sabouraud dextrose agar plate was identified on fungal culture (Figure 2). Microscopic evaluation of this mold with lactophenol blue stain revealed thin septate hyphae with conidiophores arising at right angles that bore clusters of microconidia (Figure 3). Sequencing analysis ultimately identified this organism as Sporothrix schenckii. Routine histology demonstrated pseudoepitheliomatous hyperplasia with scattered intraepidermal collections of neutrophils (Figure 4). The dermis showed a dense, superficial, and deep infiltrate composed of lymphocytes, histiocytes, and plasma cells with occasional neutrophils and eosinophils. A Grocott-Gomori methenamine-silver stain revealed a cluster of ovoid yeast forms within the stratum corneum (Figure 5). The patient was referred to infectious disease for follow-up and treatment.

The patient later visited a community clinic providing dermatologic care for patients without insurance. He was started on itraconazole 200 mg daily for a total of 6 months until dermatologic clearance of the cutaneous lesions was observed. He was followed by the clinic with laboratory tests including a liver function test. At follow-up 8 months later, a repeat biopsy was performed to ensure histologic clearance of the sporotrichosis, which revealed a dermal scar and no evidence of residual infection.

Sporothrix schenckii was first isolated in 1898 by Benjamin Schenck, a student at Johns Hopkins Medicine (Baltimore, Maryland), and identified by a mycologist as sporotricha.1 Species within the genus Sporothrix are unique in that the fungi are both dimorphic (growing as a mold at 25 °C but as a yeast at 37 °C) and dematiaceous (dark pigmentation from melanin is visible on inspection of the anterior and reverse sides of culture plates). Infection usually occurs when cutaneous or subcutaneous tissues are exposed to the fungus via microabrasions; activities thought to contribute to exposure include gardening, agricultural work, animal husbandry, and feline scratches.2 Although skin trauma frequently is considered the primary route of infection, patient recall is variable, with one study noting that only 37.7% of patients recalled trauma and another study similarly demonstrating a patient recall rate of 25%.3,4

Lymphocutaneous sporotrichosis is the most common presentation of the fungal infection,5 and clinical cases may be classified into 1 of 4 categories: (1) lymphangitic lesions—papules at the site of inoculation with spread along the lymphatic channels; (2) localized (fixed) cutaneous lesions—1 or 2 lesions at the inoculation site; (3) disseminated (multifocal) cutaneous lesions; and (4) extracutaneous lesions.6 Extracutaneous manifestations of this infection most notably have been reported as pulmonary disease through inhalation of conidia or through dissemination in immunocompromised hosts.7 Our patient’s infection was categorized as lymphangitic lesions due to spread from the lower to upper leg, albeit in a highly atypical, annular fashion. A review of systems was otherwise negative, and CT ruled out osteoarticular involvement.

In addition to socioeconomic barriers, several factors contributed to a delayed diagnosis in this patient including the annular presentation with central hypopigmentation and atrophy, negative initial microbiological cultures and lack of visualization of organisms on histopathology, and the consequent need for repeat biopsies. For lymphocutaneous sporotrichosis, the typical presentation consists of a papule or ulcerated nodule at the site of inoculation with subsequent linear spread along lymphatic channels. This classic sporotrichoid pattern is a key diagnostic clue for identifying sporotrichosis but was absent at the time our patient presented for medical care. Rather, the sporotrichoid spread seemed to have occurred in a centrifugal fashion up the leg. Few case reports have documented an annular presentation of lymphocutaneous sporotrichosis,8-13 and one report described central atrophy and hypopigmentation.10 Pain and pruritus, which were present in our patient, rarely are documented.9 Finally, the diagnosis of cutaneous fungal infections may require multiple biopsies due to the variable abundance of viable organisms in tissue specimens as well as the fastidious growth characteristics of these organisms. Furthermore, sensitivity often is low for both fungal and mycobacterial cultures, and cultures may take days to weeks to yield growth.14,15 For these reasons, empiric therapy and repeat biopsies often are pursued if clinical suspicion is high enough.16 Our patient returned for multiple scouting biopsies after the initial tissue culture was negative and was even considered for empiric treatment against Mycobacterium prior to positive fungal cultures.
Another unique aspect of our case was the presence of a keloid. It is difficult to know if this keloid was secondary to the trauma the patient sustained in the inciting incident or formed from the fungal infection. Interestingly, it has been hypothesized that fungal infections may contribute to keloid and hypertrophic scar formation.17 In a case series of 3 patients with either keloids or hypertrophic scars and concomitant tinea infection, there was notable improvement in the appearance of the scars 2 weeks after beginning itraconazole therapy.17 However, it is not yet known if a fungal infection can contribute to the pathogenesis of keloid formation.
As with other aspects of this case, the length of time the patient went without diagnosis and treatment was unusual and may help explain the atypical presentation. Although the incubation period for S schenckii can vary, most reports identify patients as seeking medical attention within 1 year of rash onset.18-20 In our case, the patient was not diagnosed until 8 years after his symptoms began, requiring multiple referrals, multiple health system touchpoints, and an institution-specific financial aid program. As such, this case also highlights the potential need for a multidisciplinary team approach when caring for patients with poor access to health care.
In conclusion, this case illustrates a unique presentation of lymphocutaneous sporotrichosis that may mimic other chronic infections and result in delayed diagnosis. Although lymphangitic sporotrichosis generally is recognized as having a linear distribution, mounting evidence from this report and others suggests an annular presentation also is possible. Pruritus or pain is rare but should not preclude a diagnosis of sporotrichosis if present. For patients with limited access to health care resources, it is especially important to involve multiple members of the health care team, including social workers and specialists, to prevent a protracted and severe course of disease.
- Schenck BR. On refractory subcutaneous abscesses caused by a fungus possibly related to the sporotricha. Bulletin of the Johns Hopkins Hospital. 1898;93:286-290.
- de Lima Barros MB, de Almeida Paes R, Schubach AO. Sporothrix schenckii and sporotrichosis. Clin Microbiol Rev. 2011;24:633-654. doi:10.1128/CMR.00007-11
- Crevasse L, Ellner PD. An outbreak of sporotrichosis in florida. J Am Med Assoc. 1960;173:29-33. doi:10.1001/jama.1960.03020190031006
- Mayorga R, Cáceres A, Toriello C, et al. An endemic area of sporotrichosis in Guatemala [in French]. Sabouraudia. 1978;16:185-198.
- Morris-Jones R. Sporotrichosis. Clin Exp Dermatol. 2002;27:427-431. doi:10.1046/j.1365-2230.2002.01087.x
- Sampaio SA, Da Lacaz CS. Clinical and statistical studies on sporotrichosis in Sao Paulo (Brazil). Article in German. Hautarzt. 1959;10:490-493.
- Ramos-e-Silva M, Vasconcelos C, Carneiro S, et al. Sporotrichosis. Clin Dermatol. 2007;25:181-187. doi:10.1016/j.clindermatol.2006.05.006
- Williams BA, Jennings TA, Rushing EC, et al. Sporotrichosis on the face of a 7-year-old boy following a bicycle accident. Pediatr Dermatol. 2013;30:E246-E247. doi:10.1111/j.1525-1470.2011.01696.x
- Vaishampayan SS, Borde P. An unusual presentation of sporotrichosis. Indian J Dermatol. 2013;58:409. doi:10.4103/0019-5154.117350
- Qin J, Zhang J. Sporotrichosis. N Engl J Med. 2019;380:771. doi:10.1056/NEJMicm1809179
- Patel A, Mudenda V, Lakhi S, et al. A 27-year-old severely immunosuppressed female with misleading clinical features of disseminated cutaneous sporotrichosis. Case Rep Dermatol Med. 2016;2016:1-4. doi:10.1155/2016/9403690
- de Oliveira-Esteves ICMR, Almeida Rosa da Silva G, Eyer-Silva WA, et al. Rapidly progressive disseminated sporotrichosis as the first presentation of HIV infection in a patient with a very low CD4 cell count. Case Rep Infect Dis. 2017;2017:4713140. doi:10.1155/2017/4713140
- Singh S, Bachaspatimayum R, Meetei U, et al. Terbinafine in fixed cutaneous sporotrichosis: a case series. J Clin Diagnostic Res. 2018;12:FR01-FR03. doi:10.7860/JCDR/2018/25315.12223
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280. doi:10.1128/CMR.00053-10
- Peters F, Batinica M, Plum G, et al. Bug or no bug: challenges in diagnosing cutaneous mycobacterial infections. J Ger Soc Dermatol. 2016;14:1227-1236. doi:10.1111/ddg.13001
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973. doi:10.1155/2018/7201973
- Okada E, Maruyama Y. Are keloids and hypertrophic scars caused by fungal infection? . Plast Reconstr Surg. 2007;120:814-815. doi:10.1097/01.prs.0000278813.23244.3f
- Pappas PG, Tellez I, Deep AE, et al. Sporotrichosis in Peru: description of an area of hyperendemicity. Clin Infect Dis. 2000;30:65-70. doi:10.1086/313607
- McGuinness SL, Boyd R, Kidd S, et al. Epidemiological investigation of an outbreak of cutaneous sporotrichosis, Northern Territory, Australia. BMC Infect Dis. 2016;16:1-7. doi:10.1186/s12879-016-1338-0
- Rojas FD, Fernández MS, Lucchelli JM, et al. Cavitary pulmonary sporotrichosis: case report and literature review. Mycopathologia. 2017;182:1119-1123. doi:10.1007/s11046-017-0197-6
- Schenck BR. On refractory subcutaneous abscesses caused by a fungus possibly related to the sporotricha. Bulletin of the Johns Hopkins Hospital. 1898;93:286-290.
- de Lima Barros MB, de Almeida Paes R, Schubach AO. Sporothrix schenckii and sporotrichosis. Clin Microbiol Rev. 2011;24:633-654. doi:10.1128/CMR.00007-11
- Crevasse L, Ellner PD. An outbreak of sporotrichosis in florida. J Am Med Assoc. 1960;173:29-33. doi:10.1001/jama.1960.03020190031006
- Mayorga R, Cáceres A, Toriello C, et al. An endemic area of sporotrichosis in Guatemala [in French]. Sabouraudia. 1978;16:185-198.
- Morris-Jones R. Sporotrichosis. Clin Exp Dermatol. 2002;27:427-431. doi:10.1046/j.1365-2230.2002.01087.x
- Sampaio SA, Da Lacaz CS. Clinical and statistical studies on sporotrichosis in Sao Paulo (Brazil). Article in German. Hautarzt. 1959;10:490-493.
- Ramos-e-Silva M, Vasconcelos C, Carneiro S, et al. Sporotrichosis. Clin Dermatol. 2007;25:181-187. doi:10.1016/j.clindermatol.2006.05.006
- Williams BA, Jennings TA, Rushing EC, et al. Sporotrichosis on the face of a 7-year-old boy following a bicycle accident. Pediatr Dermatol. 2013;30:E246-E247. doi:10.1111/j.1525-1470.2011.01696.x
- Vaishampayan SS, Borde P. An unusual presentation of sporotrichosis. Indian J Dermatol. 2013;58:409. doi:10.4103/0019-5154.117350
- Qin J, Zhang J. Sporotrichosis. N Engl J Med. 2019;380:771. doi:10.1056/NEJMicm1809179
- Patel A, Mudenda V, Lakhi S, et al. A 27-year-old severely immunosuppressed female with misleading clinical features of disseminated cutaneous sporotrichosis. Case Rep Dermatol Med. 2016;2016:1-4. doi:10.1155/2016/9403690
- de Oliveira-Esteves ICMR, Almeida Rosa da Silva G, Eyer-Silva WA, et al. Rapidly progressive disseminated sporotrichosis as the first presentation of HIV infection in a patient with a very low CD4 cell count. Case Rep Infect Dis. 2017;2017:4713140. doi:10.1155/2017/4713140
- Singh S, Bachaspatimayum R, Meetei U, et al. Terbinafine in fixed cutaneous sporotrichosis: a case series. J Clin Diagnostic Res. 2018;12:FR01-FR03. doi:10.7860/JCDR/2018/25315.12223
- Guarner J, Brandt ME. Histopathologic diagnosis of fungal infections in the 21st century. Clin Microbiol Rev. 2011;24:247-280. doi:10.1128/CMR.00053-10
- Peters F, Batinica M, Plum G, et al. Bug or no bug: challenges in diagnosing cutaneous mycobacterial infections. J Ger Soc Dermatol. 2016;14:1227-1236. doi:10.1111/ddg.13001
- Khadka P, Koirala S, Thapaliya J. Cutaneous tuberculosis: clinicopathologic arrays and diagnostic challenges. Dermatol Res Pract. 2018;2018:7201973. doi:10.1155/2018/7201973
- Okada E, Maruyama Y. Are keloids and hypertrophic scars caused by fungal infection? . Plast Reconstr Surg. 2007;120:814-815. doi:10.1097/01.prs.0000278813.23244.3f
- Pappas PG, Tellez I, Deep AE, et al. Sporotrichosis in Peru: description of an area of hyperendemicity. Clin Infect Dis. 2000;30:65-70. doi:10.1086/313607
- McGuinness SL, Boyd R, Kidd S, et al. Epidemiological investigation of an outbreak of cutaneous sporotrichosis, Northern Territory, Australia. BMC Infect Dis. 2016;16:1-7. doi:10.1186/s12879-016-1338-0
- Rojas FD, Fernández MS, Lucchelli JM, et al. Cavitary pulmonary sporotrichosis: case report and literature review. Mycopathologia. 2017;182:1119-1123. doi:10.1007/s11046-017-0197-6
Practice Points
- An atypical presentation of lymphocutaneous sporotrichosis may pose challenges to timely diagnosis and treatment.
- Although lymphocutaneous sporotrichosis spreads most commonly in a linear fashion along lymphatic channels, an annular configuration is possible.
- Initial tissue cultures and histopathology of lymphocutaneous sporotrichosis may not yield a diagnosis, necessitating repeat biopsies when clinical suspicion is high.
Empowering ICU physicians in MCS critical care
Pulmonary Vascular and Cardiovascular Network
Cardiovascular Medicine and Surgery Section
Intensive care physicians around the nation are pivotal in improving shock-related patient outcomes. Most intensive care units (academic and nonacademic) are currently managed by intensive care physicians, and a large majority of these physicians are either pulmonary/critical care, emergency medicine critical care, surgery critical care, or medicine/critical care.
There is lack of systematic training in cardiogenic shock across the board in these specialties as it relates to management of patients supported on extracorporeal membrane oxygenation (ECMO), left ventricular assist devices (LVADs), percutaneous devices, and intermediate devices such as centrimag devices.
By integrating comprehensive systematic training on cardiogenic shock recognition and management into educational initiatives, fellowship programs that are noncardiology-based can empower health care providers to make informed decisions and expedite life-saving interventions for patients in need of advanced cardiac support. Furthermore, the next generation of intensive care physicians may require ongoing education in the cardiac space, including additional training in point-of-care ultrasound, transesophageal echocardiography (TEE), and advanced hemodynamics, including management of alarms related to percutaneous and durable devices. Through continuous education and training both at conferences and at the simulation center in Glenview, Illinois, CHEST is especially suited to aid intensive care physicians to navigate the evolving landscape of mechanical circulatory support critical care and improve outcomes for patients in need of mechanical circulatory support.
Pulmonary Vascular and Cardiovascular Network
Cardiovascular Medicine and Surgery Section
Intensive care physicians around the nation are pivotal in improving shock-related patient outcomes. Most intensive care units (academic and nonacademic) are currently managed by intensive care physicians, and a large majority of these physicians are either pulmonary/critical care, emergency medicine critical care, surgery critical care, or medicine/critical care.
There is lack of systematic training in cardiogenic shock across the board in these specialties as it relates to management of patients supported on extracorporeal membrane oxygenation (ECMO), left ventricular assist devices (LVADs), percutaneous devices, and intermediate devices such as centrimag devices.
By integrating comprehensive systematic training on cardiogenic shock recognition and management into educational initiatives, fellowship programs that are noncardiology-based can empower health care providers to make informed decisions and expedite life-saving interventions for patients in need of advanced cardiac support. Furthermore, the next generation of intensive care physicians may require ongoing education in the cardiac space, including additional training in point-of-care ultrasound, transesophageal echocardiography (TEE), and advanced hemodynamics, including management of alarms related to percutaneous and durable devices. Through continuous education and training both at conferences and at the simulation center in Glenview, Illinois, CHEST is especially suited to aid intensive care physicians to navigate the evolving landscape of mechanical circulatory support critical care and improve outcomes for patients in need of mechanical circulatory support.
Pulmonary Vascular and Cardiovascular Network
Cardiovascular Medicine and Surgery Section
Intensive care physicians around the nation are pivotal in improving shock-related patient outcomes. Most intensive care units (academic and nonacademic) are currently managed by intensive care physicians, and a large majority of these physicians are either pulmonary/critical care, emergency medicine critical care, surgery critical care, or medicine/critical care.
There is lack of systematic training in cardiogenic shock across the board in these specialties as it relates to management of patients supported on extracorporeal membrane oxygenation (ECMO), left ventricular assist devices (LVADs), percutaneous devices, and intermediate devices such as centrimag devices.
By integrating comprehensive systematic training on cardiogenic shock recognition and management into educational initiatives, fellowship programs that are noncardiology-based can empower health care providers to make informed decisions and expedite life-saving interventions for patients in need of advanced cardiac support. Furthermore, the next generation of intensive care physicians may require ongoing education in the cardiac space, including additional training in point-of-care ultrasound, transesophageal echocardiography (TEE), and advanced hemodynamics, including management of alarms related to percutaneous and durable devices. Through continuous education and training both at conferences and at the simulation center in Glenview, Illinois, CHEST is especially suited to aid intensive care physicians to navigate the evolving landscape of mechanical circulatory support critical care and improve outcomes for patients in need of mechanical circulatory support.
What Does Health Equity in Dermatology Look Like?
SAN DIEGO —.
It also means embracing diversity, which she defined as diversity of thinking. “If you look at the literature, diversity in higher education and health profession training settings is associated with better educational outcomes for all students,” Dr. Treadwell, professor emeritus of dermatology and pediatrics at Indiana University School of Medicine, Indianapolis, said in a presentation on health equity during the plenary session at the annual meeting of the American Academy of Dermatology. “Each person brings a variety of experiences and perspectives. This provides a wide range of opinions and different ways to look at things. Racial and ethnic minority providers can help health organization reduce cultural and linguistic barriers and improve cultural competence.”
Such efforts matter, she continued, because according to the United States Census, Black individuals make up 13.6% of the population, while Latinx individuals represent 19.1% of the population. “So, melanin matters,” she said. “If you look at a dermatology textbook, a high percentage [of cases] are identified as Caucasian individuals, which results in an overrepresentation of Caucasians in photographs. That can result in delayed or missed diagnoses [in different skin types]. If you are contributing to cases in textbooks, make sure you have a variety of different skin types so that individuals who are referring to the textbooks will be more equipped.”
Practicing dermatologists can support diversity by offering opportunities to underrepresented in medicine (URM) students, “African-American students, Hispanic students, and Native American students,” said Dr. Treadwell, who was chief of pediatric dermatology at Riley Hospital for Children in Indianapolis from 1987 to 2004. “You also want to be encouraging,” she said.
Dermatologists can also support diversity by providing precepting opportunities, “because many [medical] students may not have connections and networks. Providing those opportunities is important,” she said. Another way to help is to be a mentor to young dermatologists. “I certainly have had mentors in my career who have been very helpful,” she said. “They’ve given me advice about things I was not familiar with.”
Dr. Treadwell suggested the Skin of Color Society as an organization that can assist with networking, mentoring, and research efforts. She also cited the Society for Pediatric Dermatology’s Equity, Diversity, and Inclusion Committee, formed in 2020. One of its initiatives was assembling a special issue of Pediatric Dermatology dedicated to DEI issues, which was published in November 2021.
Dr. Treadwell concluded her presentation by encouraging dermatologists to find ways to care for uninsured or underinsured patients, particularly those with skin of color. This might involve work at a county hospital “to provide access, to serve the patients ... and helping to decrease some the issues in terms of health equity,” she said.
Dr. Treadwell reported having no relevant disclosures. At the plenary session, she presented the John Kenney Jr., MD Lifetime Achievement Award and Lectureship.
SAN DIEGO —.
It also means embracing diversity, which she defined as diversity of thinking. “If you look at the literature, diversity in higher education and health profession training settings is associated with better educational outcomes for all students,” Dr. Treadwell, professor emeritus of dermatology and pediatrics at Indiana University School of Medicine, Indianapolis, said in a presentation on health equity during the plenary session at the annual meeting of the American Academy of Dermatology. “Each person brings a variety of experiences and perspectives. This provides a wide range of opinions and different ways to look at things. Racial and ethnic minority providers can help health organization reduce cultural and linguistic barriers and improve cultural competence.”
Such efforts matter, she continued, because according to the United States Census, Black individuals make up 13.6% of the population, while Latinx individuals represent 19.1% of the population. “So, melanin matters,” she said. “If you look at a dermatology textbook, a high percentage [of cases] are identified as Caucasian individuals, which results in an overrepresentation of Caucasians in photographs. That can result in delayed or missed diagnoses [in different skin types]. If you are contributing to cases in textbooks, make sure you have a variety of different skin types so that individuals who are referring to the textbooks will be more equipped.”
Practicing dermatologists can support diversity by offering opportunities to underrepresented in medicine (URM) students, “African-American students, Hispanic students, and Native American students,” said Dr. Treadwell, who was chief of pediatric dermatology at Riley Hospital for Children in Indianapolis from 1987 to 2004. “You also want to be encouraging,” she said.
Dermatologists can also support diversity by providing precepting opportunities, “because many [medical] students may not have connections and networks. Providing those opportunities is important,” she said. Another way to help is to be a mentor to young dermatologists. “I certainly have had mentors in my career who have been very helpful,” she said. “They’ve given me advice about things I was not familiar with.”
Dr. Treadwell suggested the Skin of Color Society as an organization that can assist with networking, mentoring, and research efforts. She also cited the Society for Pediatric Dermatology’s Equity, Diversity, and Inclusion Committee, formed in 2020. One of its initiatives was assembling a special issue of Pediatric Dermatology dedicated to DEI issues, which was published in November 2021.
Dr. Treadwell concluded her presentation by encouraging dermatologists to find ways to care for uninsured or underinsured patients, particularly those with skin of color. This might involve work at a county hospital “to provide access, to serve the patients ... and helping to decrease some the issues in terms of health equity,” she said.
Dr. Treadwell reported having no relevant disclosures. At the plenary session, she presented the John Kenney Jr., MD Lifetime Achievement Award and Lectureship.
SAN DIEGO —.
It also means embracing diversity, which she defined as diversity of thinking. “If you look at the literature, diversity in higher education and health profession training settings is associated with better educational outcomes for all students,” Dr. Treadwell, professor emeritus of dermatology and pediatrics at Indiana University School of Medicine, Indianapolis, said in a presentation on health equity during the plenary session at the annual meeting of the American Academy of Dermatology. “Each person brings a variety of experiences and perspectives. This provides a wide range of opinions and different ways to look at things. Racial and ethnic minority providers can help health organization reduce cultural and linguistic barriers and improve cultural competence.”
Such efforts matter, she continued, because according to the United States Census, Black individuals make up 13.6% of the population, while Latinx individuals represent 19.1% of the population. “So, melanin matters,” she said. “If you look at a dermatology textbook, a high percentage [of cases] are identified as Caucasian individuals, which results in an overrepresentation of Caucasians in photographs. That can result in delayed or missed diagnoses [in different skin types]. If you are contributing to cases in textbooks, make sure you have a variety of different skin types so that individuals who are referring to the textbooks will be more equipped.”
Practicing dermatologists can support diversity by offering opportunities to underrepresented in medicine (URM) students, “African-American students, Hispanic students, and Native American students,” said Dr. Treadwell, who was chief of pediatric dermatology at Riley Hospital for Children in Indianapolis from 1987 to 2004. “You also want to be encouraging,” she said.
Dermatologists can also support diversity by providing precepting opportunities, “because many [medical] students may not have connections and networks. Providing those opportunities is important,” she said. Another way to help is to be a mentor to young dermatologists. “I certainly have had mentors in my career who have been very helpful,” she said. “They’ve given me advice about things I was not familiar with.”
Dr. Treadwell suggested the Skin of Color Society as an organization that can assist with networking, mentoring, and research efforts. She also cited the Society for Pediatric Dermatology’s Equity, Diversity, and Inclusion Committee, formed in 2020. One of its initiatives was assembling a special issue of Pediatric Dermatology dedicated to DEI issues, which was published in November 2021.
Dr. Treadwell concluded her presentation by encouraging dermatologists to find ways to care for uninsured or underinsured patients, particularly those with skin of color. This might involve work at a county hospital “to provide access, to serve the patients ... and helping to decrease some the issues in terms of health equity,” she said.
Dr. Treadwell reported having no relevant disclosures. At the plenary session, she presented the John Kenney Jr., MD Lifetime Achievement Award and Lectureship.
FROM AAD 2024
Eosinophilic Pustular Folliculitis in the Setting of Untreated Chronic Lymphocytic Leukemia
To the Editor:
Eosinophilic pustular folliculitis (EPF) is a noninfectious dermatosis that typically manifests as recurrent follicular papulopustules that generally affect the face and occasionally the trunk and arms. There are several subtypes of EPF: classic EPF (Ofuji disease), infancy-associated EPF, and immunosuppression-associated EPF.1,2 We report a rare case of EPF in the setting of untreated chronic lymphocytic leukemia (CLL), a subtype of immunosuppression-associated EPF that has been associated with hematologic malignancy EPF (HM-EPF).3-5
A 69-year-old woman presented with diffusely scattered, pruritic, erythematous, erosive lesions on the back, arms, legs, and forehead (Figure 1) of 4 months’ duration, as well as an ulcerative lesion on the left third toe due to a suspected insect bite. She had a history of untreated CLL that was diagnosed 2 years prior. The patient was empirically started on clindamycin for presumed infection of the toe. A punch biopsy of the left wrist revealed superficial and deep dermal perivascular and interstitial inflammatory infiltrates composed of lymphocytes, histiocytes, and numerous eosinophils in association with edema and necrosis. Histopathology was overall most consistent with an exuberant arthropod reaction; however, at 2-week follow-up, the patient reported that the pustular lesions improved upon starting antibiotics, which raised concerns for a bacterial process. The patient initially was continued on clindamycin given subjective improvement but was later switched to daptomycin, as she developed clindamycin-resistant methicillin-resistant Staphylococcus aureus osteomyelitis from the necrotic toe.
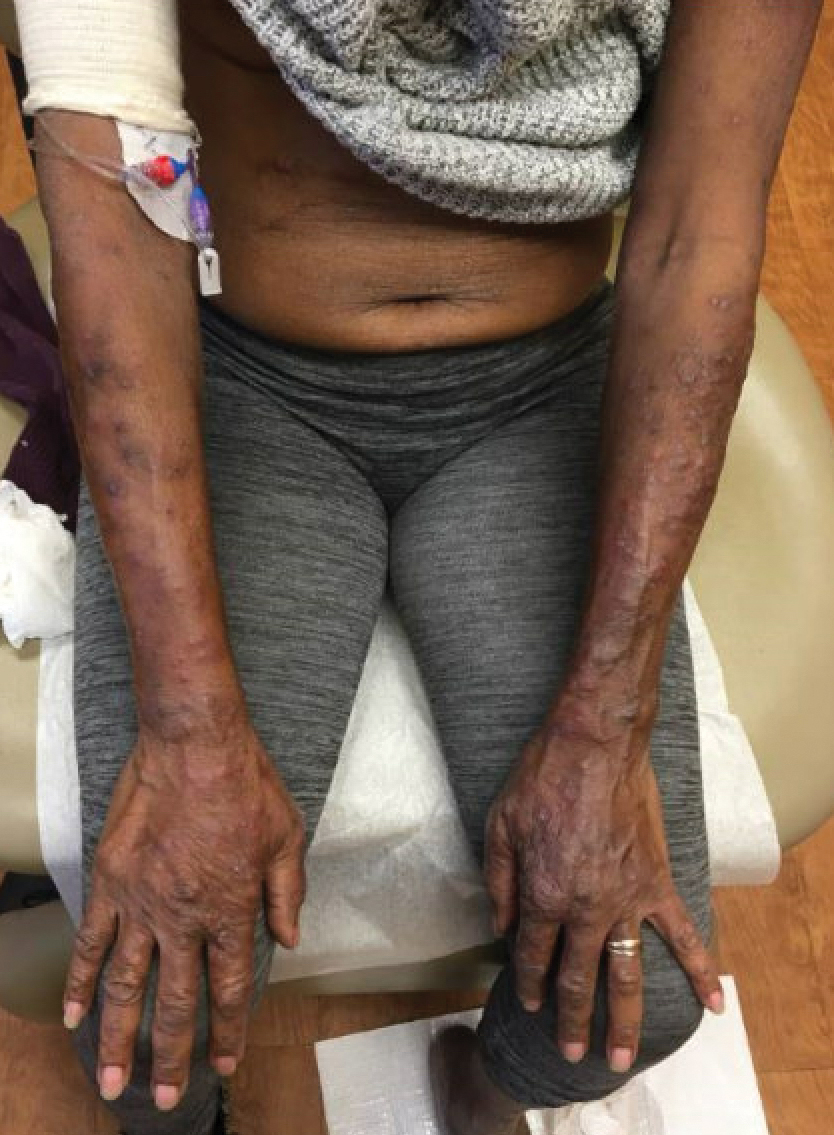
A month later, the patient returned with new papules and pustules on the arms and trunk. A repeat biopsy showed notable dermal collections comprised predominantly of neutrophils and eosinophils as well as involvement of follicular structures by dense inflammation (Figure 2). Immunohistochemistry demonstrated a predominant population of small CD3+ T cells, which raised concern for cutaneous T-cell lymphoma. However, retention of CD5 expression made this less likely. Few scattered CD20+ B cells with limited CD23 reactivity and without CD5 co-expression were detected, which ruled out cutaneous involvement of the patient’s CLL. Bacterial culture and Grocott methenamine-silver, Gram, acid-fast bacilli, and periodic acid-Schiff stains were negative. Polymerase chain reaction testing for varicella-zoster virus and herpes simplex virus also were negative. Thus, a diagnosis of EPF secondary to CLL was favored, as an infectious process also was unlikely. The patient was started on triamcinolone cream 0.1% with gradual improvement.
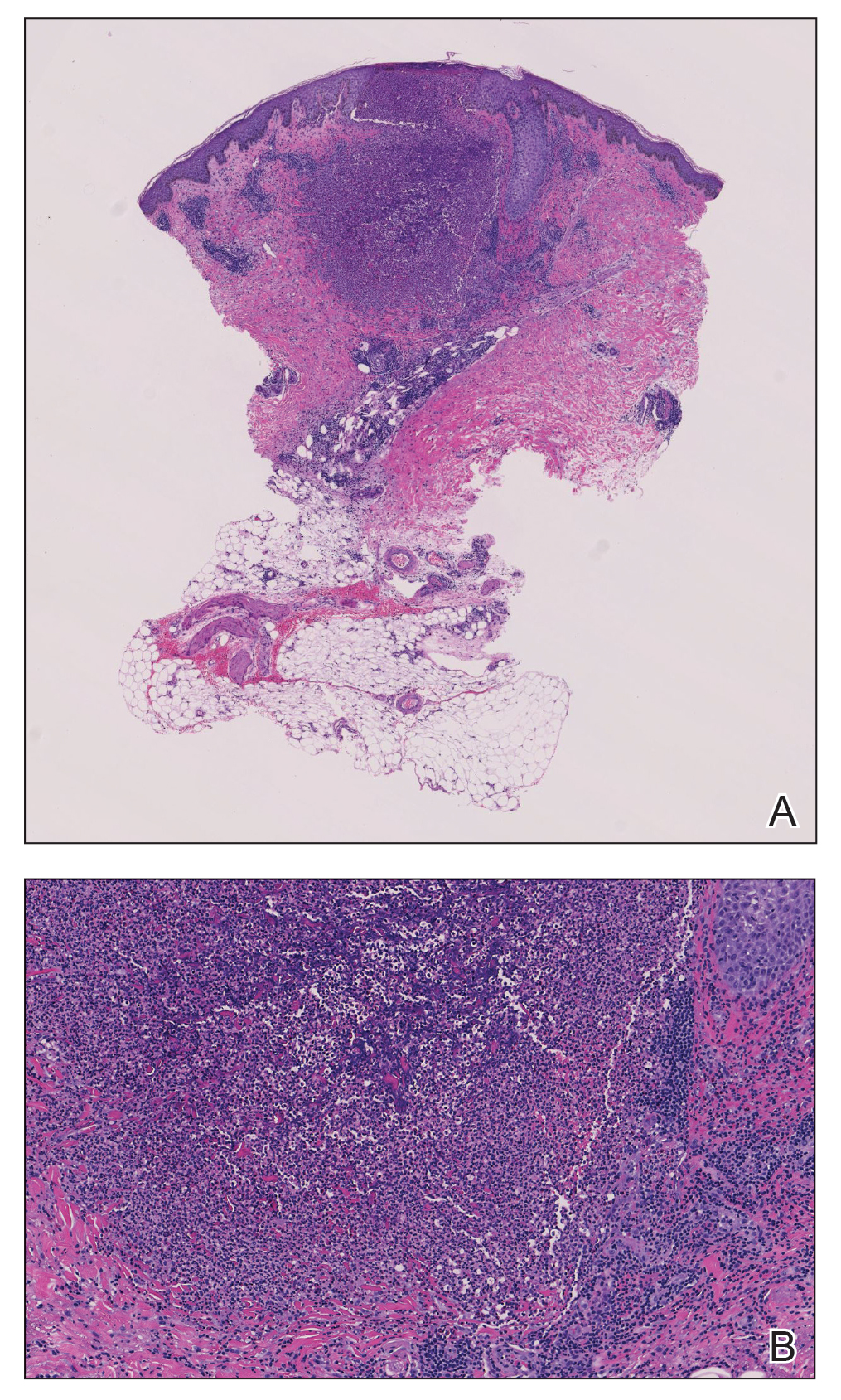
Cases of HM-EPF predominantly have been reported in patients who have undergone chemotherapy, bone marrow transplantation, or hematopoietic stem cell transplantation. Furthermore, a vast majority of these cases have been reported in older males.3-16 In a retrospective study of more than 750 patients with established CLL, Agnew et al7 identified 125 different skin complications in 40 patients. Of this subset, only a small number (2/40) were associated with eosinophilic folliculitis, with 1 case noted in a middle-aged woman with a history of CLL treatment.7 Moreover, Motaparthi et al4 reported 3 additional cases of HM-EPF, with all patients identified as middle-aged men who were treated with chemotherapy for underlying CLL. Our patient represents a case of EPF in the context of untreated CLL in a woman.
Although topical corticosteroids remain the first-line treatment for EPF, a survey study conducted across 67 hospitals in Japan indicated that antibiotics were moderately or highly effective in 79% of EPF patients (n=143).17 This association may explain the subjective improvement reported by our patient upon starting clindamycin. Furthermore, in HIV-associated EPF, high-dose cetirizine, itraconazole, and metronidazole have been successful when topical therapies have failed.18 Although the precise pathogenesis of EPF is unknown, histopathologic features, clinical appearance, and identification of the accurate EPF subtype can still prove valuable in informing empiric treatment strategies. Consequently, the initial histopathologic diagnosis of an arthropod bite reaction in our patient highlights the importance of clinical correlation and additional ancillary studies in the determination of EPF vs other inflammatory dermatoses that manifest microscopically with lymphocytic infiltrates, prominent eosinophils, and follicular involvement.4 The histopathologic features of EPF demonstrate considerable overlap with eosinophilic dermatosis of hematologic malignancy (also known as eosinophilic dermatosis of myeloproliferative disease). It is suspected that eosinophilic dermatosis of hematologic malignancy and EPF may exist on a spectrum, and additional cases may improve categorization of these entities.19
In conclusion, this report adds to the medical practitioner’s awareness of EPF manifestations in patients with underlying CLL, an infrequently reported subtype of HM-EPF.
- Fujiyama T, Tokura Y. Clinical and histopathological differential diagnosis of eosinophilic pustular folliculitis. J Dermatol. 2013;40:419-423. doi:10.1111/1346-8138.12125
- Katoh M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. doi:10.1111/1346-8138.12008
- Takamura S, Teraki Y. Eosinophilic pustular folliculitis associated with hematological disorders: a report of two cases and review of Japanese literature. J Dermatol. 2016;43:432-435. doi: 10.1111/1346-8138.13088
- Motaparthi K, Kapil J, Hsu S. Eosinophilic folliculitis in association with chronic lymphocytic leukemia: a clinicopathologic series. JAAD Case Rep. 2017;3:263-268. doi:10.1016/j.jdcr.2017.03.007
- Lambert J, Berneman Z, Dockx P, et al. Eosinophilic pustular folliculitis and B-cell chronic lymphatic leukaemia. Dermatology. 1994;189(suppl 2):58-59. doi:10.1159/000246994
- Patrizi A, Chieregato C, Visani G, et al. Leukaemia-associated eosinophilic folliculitis (Ofuji’s disease). J Eur Acad Dermatol Venereol. 2004;18:596-598. doi:10.1111/j.1468-3083.2004.00982.x
- Agnew KL, Ruchlemer R, Catovsky D, et al. Cutaneous findings in chronic lymphocytic leukaemia. Br J Dermatol. 2004;150:1129-1135. doi:10.1111/j.1365-2133.2004.05982.x
- Zitelli K, Fernandes N, Adams BB. Eosinophilic folliculitis occurring after stem cell transplant for acute lymphoblastic leukemia: a case report and review. Int J Dermatol. 2015;54:785-789. doi:10.1111/j.1365-2133.2004.05982.x
- Goiriz R, Guhl-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36. doi:10.1111/j.1600-0560.2006.00725.x
- Bhandare PC, Ghodge RR, Bhobe MR, et al. Eosinophilic pustular folliculitis post chemotherapy in a patient of non-Hodgkins lymphoma: a case report. Indian J Dermatol. 2015;60:521. doi:10.4103/0019-5154.164432
- Sugaya M, Suga H, Miyagaki T, et al. Eosinophilic pustular folliculitis associated with Sézary syndrome. Clin Exp Dermatol. 2014;39:536-538. doi:10.1111/ced.12315
- Keida T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplantation. J Dermatol. 2004;31:21-26. doi:10.1111/j.1346-8138.2004.tb00499.x
- Ota M, Shimizu T, Hashino S, et al. Eosinophilic folliculitis in a patient after allogeneic bone marrow transplantation: case report and review of the literature. Am J Hematol. 2004;76:295-296. doi:10.1002/ajh.20080
- Vassallo C, Ciocca O, Arcaini L, et al. Eosinophilic folliculitis occurring in a patient affected by Hodgkin lymphoma. Int J Dermatol. 2002;41:298-300. doi:10.1046/j.1365-4362.2002.01356_6.x
- Evans TR, Mansi JL, Bull R, et al. Eosinophilic folliculitis occurring after bone marrow autograft in a patient with non-Hodgkin’s lymphoma. Cancer. 1994;73:2512-2514. doi:10.1002/1097-0142(19940515)73:10<2512::aid-cncr2820731010>3.0.co;2-s
- Patrizi A, Di Lernia V, Neri I, et al. Eosinophilic pustular folliculitis (Ofuji’s disease) and non-Hodgkin lymphoma. Acta Derm Venereol. 1992;72:146-147.
- Ono S, Yamamoto Y, Otsuka A, et al. Evaluation of the effectiveness of antibiotics against eosinophilic pustular folliculitis. Case Rep Dermatol. 2013;5:144-147. doi:10.1159/000351330
- Ellis E, Scheinfeld N. Eosinophilic pustular folliculitis. Am J Clin Dermatol. 2004;5:189-197. doi:10.2165/00128071-200405030-00007
- Bailey CAR, Laurain DA, Sheinbein DM, et al. Eosinophilic folliculitis, eosinophilic dermatosis of hematologic malignancy and acneiform follicular mucinosis: two case reports and a review of the literature highlighting the spectrum of histopathology. J Cutan Pathol. 2021;48:439-450. doi:10.1111/cup.13932
To the Editor:
Eosinophilic pustular folliculitis (EPF) is a noninfectious dermatosis that typically manifests as recurrent follicular papulopustules that generally affect the face and occasionally the trunk and arms. There are several subtypes of EPF: classic EPF (Ofuji disease), infancy-associated EPF, and immunosuppression-associated EPF.1,2 We report a rare case of EPF in the setting of untreated chronic lymphocytic leukemia (CLL), a subtype of immunosuppression-associated EPF that has been associated with hematologic malignancy EPF (HM-EPF).3-5
A 69-year-old woman presented with diffusely scattered, pruritic, erythematous, erosive lesions on the back, arms, legs, and forehead (Figure 1) of 4 months’ duration, as well as an ulcerative lesion on the left third toe due to a suspected insect bite. She had a history of untreated CLL that was diagnosed 2 years prior. The patient was empirically started on clindamycin for presumed infection of the toe. A punch biopsy of the left wrist revealed superficial and deep dermal perivascular and interstitial inflammatory infiltrates composed of lymphocytes, histiocytes, and numerous eosinophils in association with edema and necrosis. Histopathology was overall most consistent with an exuberant arthropod reaction; however, at 2-week follow-up, the patient reported that the pustular lesions improved upon starting antibiotics, which raised concerns for a bacterial process. The patient initially was continued on clindamycin given subjective improvement but was later switched to daptomycin, as she developed clindamycin-resistant methicillin-resistant Staphylococcus aureus osteomyelitis from the necrotic toe.

A month later, the patient returned with new papules and pustules on the arms and trunk. A repeat biopsy showed notable dermal collections comprised predominantly of neutrophils and eosinophils as well as involvement of follicular structures by dense inflammation (Figure 2). Immunohistochemistry demonstrated a predominant population of small CD3+ T cells, which raised concern for cutaneous T-cell lymphoma. However, retention of CD5 expression made this less likely. Few scattered CD20+ B cells with limited CD23 reactivity and without CD5 co-expression were detected, which ruled out cutaneous involvement of the patient’s CLL. Bacterial culture and Grocott methenamine-silver, Gram, acid-fast bacilli, and periodic acid-Schiff stains were negative. Polymerase chain reaction testing for varicella-zoster virus and herpes simplex virus also were negative. Thus, a diagnosis of EPF secondary to CLL was favored, as an infectious process also was unlikely. The patient was started on triamcinolone cream 0.1% with gradual improvement.

Cases of HM-EPF predominantly have been reported in patients who have undergone chemotherapy, bone marrow transplantation, or hematopoietic stem cell transplantation. Furthermore, a vast majority of these cases have been reported in older males.3-16 In a retrospective study of more than 750 patients with established CLL, Agnew et al7 identified 125 different skin complications in 40 patients. Of this subset, only a small number (2/40) were associated with eosinophilic folliculitis, with 1 case noted in a middle-aged woman with a history of CLL treatment.7 Moreover, Motaparthi et al4 reported 3 additional cases of HM-EPF, with all patients identified as middle-aged men who were treated with chemotherapy for underlying CLL. Our patient represents a case of EPF in the context of untreated CLL in a woman.
Although topical corticosteroids remain the first-line treatment for EPF, a survey study conducted across 67 hospitals in Japan indicated that antibiotics were moderately or highly effective in 79% of EPF patients (n=143).17 This association may explain the subjective improvement reported by our patient upon starting clindamycin. Furthermore, in HIV-associated EPF, high-dose cetirizine, itraconazole, and metronidazole have been successful when topical therapies have failed.18 Although the precise pathogenesis of EPF is unknown, histopathologic features, clinical appearance, and identification of the accurate EPF subtype can still prove valuable in informing empiric treatment strategies. Consequently, the initial histopathologic diagnosis of an arthropod bite reaction in our patient highlights the importance of clinical correlation and additional ancillary studies in the determination of EPF vs other inflammatory dermatoses that manifest microscopically with lymphocytic infiltrates, prominent eosinophils, and follicular involvement.4 The histopathologic features of EPF demonstrate considerable overlap with eosinophilic dermatosis of hematologic malignancy (also known as eosinophilic dermatosis of myeloproliferative disease). It is suspected that eosinophilic dermatosis of hematologic malignancy and EPF may exist on a spectrum, and additional cases may improve categorization of these entities.19
In conclusion, this report adds to the medical practitioner’s awareness of EPF manifestations in patients with underlying CLL, an infrequently reported subtype of HM-EPF.
To the Editor:
Eosinophilic pustular folliculitis (EPF) is a noninfectious dermatosis that typically manifests as recurrent follicular papulopustules that generally affect the face and occasionally the trunk and arms. There are several subtypes of EPF: classic EPF (Ofuji disease), infancy-associated EPF, and immunosuppression-associated EPF.1,2 We report a rare case of EPF in the setting of untreated chronic lymphocytic leukemia (CLL), a subtype of immunosuppression-associated EPF that has been associated with hematologic malignancy EPF (HM-EPF).3-5
A 69-year-old woman presented with diffusely scattered, pruritic, erythematous, erosive lesions on the back, arms, legs, and forehead (Figure 1) of 4 months’ duration, as well as an ulcerative lesion on the left third toe due to a suspected insect bite. She had a history of untreated CLL that was diagnosed 2 years prior. The patient was empirically started on clindamycin for presumed infection of the toe. A punch biopsy of the left wrist revealed superficial and deep dermal perivascular and interstitial inflammatory infiltrates composed of lymphocytes, histiocytes, and numerous eosinophils in association with edema and necrosis. Histopathology was overall most consistent with an exuberant arthropod reaction; however, at 2-week follow-up, the patient reported that the pustular lesions improved upon starting antibiotics, which raised concerns for a bacterial process. The patient initially was continued on clindamycin given subjective improvement but was later switched to daptomycin, as she developed clindamycin-resistant methicillin-resistant Staphylococcus aureus osteomyelitis from the necrotic toe.

A month later, the patient returned with new papules and pustules on the arms and trunk. A repeat biopsy showed notable dermal collections comprised predominantly of neutrophils and eosinophils as well as involvement of follicular structures by dense inflammation (Figure 2). Immunohistochemistry demonstrated a predominant population of small CD3+ T cells, which raised concern for cutaneous T-cell lymphoma. However, retention of CD5 expression made this less likely. Few scattered CD20+ B cells with limited CD23 reactivity and without CD5 co-expression were detected, which ruled out cutaneous involvement of the patient’s CLL. Bacterial culture and Grocott methenamine-silver, Gram, acid-fast bacilli, and periodic acid-Schiff stains were negative. Polymerase chain reaction testing for varicella-zoster virus and herpes simplex virus also were negative. Thus, a diagnosis of EPF secondary to CLL was favored, as an infectious process also was unlikely. The patient was started on triamcinolone cream 0.1% with gradual improvement.

Cases of HM-EPF predominantly have been reported in patients who have undergone chemotherapy, bone marrow transplantation, or hematopoietic stem cell transplantation. Furthermore, a vast majority of these cases have been reported in older males.3-16 In a retrospective study of more than 750 patients with established CLL, Agnew et al7 identified 125 different skin complications in 40 patients. Of this subset, only a small number (2/40) were associated with eosinophilic folliculitis, with 1 case noted in a middle-aged woman with a history of CLL treatment.7 Moreover, Motaparthi et al4 reported 3 additional cases of HM-EPF, with all patients identified as middle-aged men who were treated with chemotherapy for underlying CLL. Our patient represents a case of EPF in the context of untreated CLL in a woman.
Although topical corticosteroids remain the first-line treatment for EPF, a survey study conducted across 67 hospitals in Japan indicated that antibiotics were moderately or highly effective in 79% of EPF patients (n=143).17 This association may explain the subjective improvement reported by our patient upon starting clindamycin. Furthermore, in HIV-associated EPF, high-dose cetirizine, itraconazole, and metronidazole have been successful when topical therapies have failed.18 Although the precise pathogenesis of EPF is unknown, histopathologic features, clinical appearance, and identification of the accurate EPF subtype can still prove valuable in informing empiric treatment strategies. Consequently, the initial histopathologic diagnosis of an arthropod bite reaction in our patient highlights the importance of clinical correlation and additional ancillary studies in the determination of EPF vs other inflammatory dermatoses that manifest microscopically with lymphocytic infiltrates, prominent eosinophils, and follicular involvement.4 The histopathologic features of EPF demonstrate considerable overlap with eosinophilic dermatosis of hematologic malignancy (also known as eosinophilic dermatosis of myeloproliferative disease). It is suspected that eosinophilic dermatosis of hematologic malignancy and EPF may exist on a spectrum, and additional cases may improve categorization of these entities.19
In conclusion, this report adds to the medical practitioner’s awareness of EPF manifestations in patients with underlying CLL, an infrequently reported subtype of HM-EPF.
- Fujiyama T, Tokura Y. Clinical and histopathological differential diagnosis of eosinophilic pustular folliculitis. J Dermatol. 2013;40:419-423. doi:10.1111/1346-8138.12125
- Katoh M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. doi:10.1111/1346-8138.12008
- Takamura S, Teraki Y. Eosinophilic pustular folliculitis associated with hematological disorders: a report of two cases and review of Japanese literature. J Dermatol. 2016;43:432-435. doi: 10.1111/1346-8138.13088
- Motaparthi K, Kapil J, Hsu S. Eosinophilic folliculitis in association with chronic lymphocytic leukemia: a clinicopathologic series. JAAD Case Rep. 2017;3:263-268. doi:10.1016/j.jdcr.2017.03.007
- Lambert J, Berneman Z, Dockx P, et al. Eosinophilic pustular folliculitis and B-cell chronic lymphatic leukaemia. Dermatology. 1994;189(suppl 2):58-59. doi:10.1159/000246994
- Patrizi A, Chieregato C, Visani G, et al. Leukaemia-associated eosinophilic folliculitis (Ofuji’s disease). J Eur Acad Dermatol Venereol. 2004;18:596-598. doi:10.1111/j.1468-3083.2004.00982.x
- Agnew KL, Ruchlemer R, Catovsky D, et al. Cutaneous findings in chronic lymphocytic leukaemia. Br J Dermatol. 2004;150:1129-1135. doi:10.1111/j.1365-2133.2004.05982.x
- Zitelli K, Fernandes N, Adams BB. Eosinophilic folliculitis occurring after stem cell transplant for acute lymphoblastic leukemia: a case report and review. Int J Dermatol. 2015;54:785-789. doi:10.1111/j.1365-2133.2004.05982.x
- Goiriz R, Guhl-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36. doi:10.1111/j.1600-0560.2006.00725.x
- Bhandare PC, Ghodge RR, Bhobe MR, et al. Eosinophilic pustular folliculitis post chemotherapy in a patient of non-Hodgkins lymphoma: a case report. Indian J Dermatol. 2015;60:521. doi:10.4103/0019-5154.164432
- Sugaya M, Suga H, Miyagaki T, et al. Eosinophilic pustular folliculitis associated with Sézary syndrome. Clin Exp Dermatol. 2014;39:536-538. doi:10.1111/ced.12315
- Keida T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplantation. J Dermatol. 2004;31:21-26. doi:10.1111/j.1346-8138.2004.tb00499.x
- Ota M, Shimizu T, Hashino S, et al. Eosinophilic folliculitis in a patient after allogeneic bone marrow transplantation: case report and review of the literature. Am J Hematol. 2004;76:295-296. doi:10.1002/ajh.20080
- Vassallo C, Ciocca O, Arcaini L, et al. Eosinophilic folliculitis occurring in a patient affected by Hodgkin lymphoma. Int J Dermatol. 2002;41:298-300. doi:10.1046/j.1365-4362.2002.01356_6.x
- Evans TR, Mansi JL, Bull R, et al. Eosinophilic folliculitis occurring after bone marrow autograft in a patient with non-Hodgkin’s lymphoma. Cancer. 1994;73:2512-2514. doi:10.1002/1097-0142(19940515)73:10<2512::aid-cncr2820731010>3.0.co;2-s
- Patrizi A, Di Lernia V, Neri I, et al. Eosinophilic pustular folliculitis (Ofuji’s disease) and non-Hodgkin lymphoma. Acta Derm Venereol. 1992;72:146-147.
- Ono S, Yamamoto Y, Otsuka A, et al. Evaluation of the effectiveness of antibiotics against eosinophilic pustular folliculitis. Case Rep Dermatol. 2013;5:144-147. doi:10.1159/000351330
- Ellis E, Scheinfeld N. Eosinophilic pustular folliculitis. Am J Clin Dermatol. 2004;5:189-197. doi:10.2165/00128071-200405030-00007
- Bailey CAR, Laurain DA, Sheinbein DM, et al. Eosinophilic folliculitis, eosinophilic dermatosis of hematologic malignancy and acneiform follicular mucinosis: two case reports and a review of the literature highlighting the spectrum of histopathology. J Cutan Pathol. 2021;48:439-450. doi:10.1111/cup.13932
- Fujiyama T, Tokura Y. Clinical and histopathological differential diagnosis of eosinophilic pustular folliculitis. J Dermatol. 2013;40:419-423. doi:10.1111/1346-8138.12125
- Katoh M, Nomura T, Miyachi Y, et al. Eosinophilic pustular folliculitis: a review of the Japanese published works. J Dermatol. 2013;40:15-20. doi:10.1111/1346-8138.12008
- Takamura S, Teraki Y. Eosinophilic pustular folliculitis associated with hematological disorders: a report of two cases and review of Japanese literature. J Dermatol. 2016;43:432-435. doi: 10.1111/1346-8138.13088
- Motaparthi K, Kapil J, Hsu S. Eosinophilic folliculitis in association with chronic lymphocytic leukemia: a clinicopathologic series. JAAD Case Rep. 2017;3:263-268. doi:10.1016/j.jdcr.2017.03.007
- Lambert J, Berneman Z, Dockx P, et al. Eosinophilic pustular folliculitis and B-cell chronic lymphatic leukaemia. Dermatology. 1994;189(suppl 2):58-59. doi:10.1159/000246994
- Patrizi A, Chieregato C, Visani G, et al. Leukaemia-associated eosinophilic folliculitis (Ofuji’s disease). J Eur Acad Dermatol Venereol. 2004;18:596-598. doi:10.1111/j.1468-3083.2004.00982.x
- Agnew KL, Ruchlemer R, Catovsky D, et al. Cutaneous findings in chronic lymphocytic leukaemia. Br J Dermatol. 2004;150:1129-1135. doi:10.1111/j.1365-2133.2004.05982.x
- Zitelli K, Fernandes N, Adams BB. Eosinophilic folliculitis occurring after stem cell transplant for acute lymphoblastic leukemia: a case report and review. Int J Dermatol. 2015;54:785-789. doi:10.1111/j.1365-2133.2004.05982.x
- Goiriz R, Guhl-Millán G, Peñas PF, et al. Eosinophilic folliculitis following allogeneic peripheral blood stem cell transplantation: case report and review. J Cutan Pathol. 2007;34(suppl 1):33-36. doi:10.1111/j.1600-0560.2006.00725.x
- Bhandare PC, Ghodge RR, Bhobe MR, et al. Eosinophilic pustular folliculitis post chemotherapy in a patient of non-Hodgkins lymphoma: a case report. Indian J Dermatol. 2015;60:521. doi:10.4103/0019-5154.164432
- Sugaya M, Suga H, Miyagaki T, et al. Eosinophilic pustular folliculitis associated with Sézary syndrome. Clin Exp Dermatol. 2014;39:536-538. doi:10.1111/ced.12315
- Keida T, Hayashi N, Kawashima M. Eosinophilic pustular folliculitis following autologous peripheral blood stem-cell transplantation. J Dermatol. 2004;31:21-26. doi:10.1111/j.1346-8138.2004.tb00499.x
- Ota M, Shimizu T, Hashino S, et al. Eosinophilic folliculitis in a patient after allogeneic bone marrow transplantation: case report and review of the literature. Am J Hematol. 2004;76:295-296. doi:10.1002/ajh.20080
- Vassallo C, Ciocca O, Arcaini L, et al. Eosinophilic folliculitis occurring in a patient affected by Hodgkin lymphoma. Int J Dermatol. 2002;41:298-300. doi:10.1046/j.1365-4362.2002.01356_6.x
- Evans TR, Mansi JL, Bull R, et al. Eosinophilic folliculitis occurring after bone marrow autograft in a patient with non-Hodgkin’s lymphoma. Cancer. 1994;73:2512-2514. doi:10.1002/1097-0142(19940515)73:10<2512::aid-cncr2820731010>3.0.co;2-s
- Patrizi A, Di Lernia V, Neri I, et al. Eosinophilic pustular folliculitis (Ofuji’s disease) and non-Hodgkin lymphoma. Acta Derm Venereol. 1992;72:146-147.
- Ono S, Yamamoto Y, Otsuka A, et al. Evaluation of the effectiveness of antibiotics against eosinophilic pustular folliculitis. Case Rep Dermatol. 2013;5:144-147. doi:10.1159/000351330
- Ellis E, Scheinfeld N. Eosinophilic pustular folliculitis. Am J Clin Dermatol. 2004;5:189-197. doi:10.2165/00128071-200405030-00007
- Bailey CAR, Laurain DA, Sheinbein DM, et al. Eosinophilic folliculitis, eosinophilic dermatosis of hematologic malignancy and acneiform follicular mucinosis: two case reports and a review of the literature highlighting the spectrum of histopathology. J Cutan Pathol. 2021;48:439-450. doi:10.1111/cup.13932
Practice Points
- Eosinophilic pustular folliculitis (EPF) is associated with an immunosuppressed state, as in patients with underlying hematologic malignancy.
- Topical corticosteroids remain the first-line treatment for EPF; however, antimicrobial agents have been used with moderate success when topical therapies have failed.
Papulosquamous Dermatophytid Reaction in a Child With Tinea Capitis
To the Editor:
Tinea capitis is a common childhood infection seen worldwide and is more prevalent in children of African descent.1 Treatment can be effective; however, the diagnosis may be delayed due to variability in presentation, camouflage of scalp scale with ointment, and the diagnostic experience of the provider. A common complication of tinea capitis is the dermatophytid (id) reaction, which commonly manifests as multiple 1- to 2-mm monomorphic papules. We report a case of a papulosquamous variant of an id reaction secondary to tinea capitis.
An 8-year-old African American child presented with annular hyperpigmented patches on the face and trunk of several months’ duration. There was no preceding fever, illness, scalp pruritus, or alopecia according to the patient’s mother. The hyperpigmented patches persisted despite use of hydrocortisone and antifungal creams prescribed by a primary care provider. A fungal culture of a scalp specimen was negative. Physical examination during the initial dermatology visit revealed multiple annular hyperpigmented patches on the trunk and extremities. No plaques were evident; however, the mother reported that when the lesions first developed, they were raised and mildly pruritic. The patient was prescribed triamcinolone ointment 0.1% twice daily as needed for itching, and sun protection was emphasized.
At the follow-up visit weeks later, the patient’s mother reported that the ointment had helped the lesions resolve faster, but new lesions continued to appear. Physical examination at this visit was notable for scattered hyperpigmented patches, annular hyperpigmented plaques, and erythematous plaques on the trunk, arms, and legs, in addition to papulosquamous plaques and hyperpigmented patches on the forehead (Figure 1). Suspicion for tinea capitis was discussed, a repeat scalp fungal culture was performed, and oral terbinafine 250 mg once daily was started empirically. The culture was positive for Trichophyton tonsurans supporting the diagnosis of concomitant tinea capitis. The rash resolved with terbinafine, and annular patches of postinflammatory hyperpigmentation remained.
Dermatophytid reactions are immunologically mediated, disseminated, eczematous eruptions occurring after cutaneous infections or inflammatory skin conditions. Reactions occur days to weeks after exposure to antigens of dermatophytes causing tinea pedis or capitis.2
Common culprits include Microsporum canis and T tonsurans.3 Dermatophytid reactions with tinea capitis exhibit morphologic variability including a symmetric distribution of grouped or diffuse,4 pruritic, erythematous or flesh-colored, follicular papules on the trunk, with or without progression to the face, torso, upper extremities, and/or lower extremities.3 Other reported manifestations include erythema multiforme, erythema nodosum,3 or lupuslike lesions, and crops of dyshidrotic vesicles on the hands in the setting of Trichophyton mentagrophytes–induced tinea pedis.5
The papulosquamous variant id reaction should be considered in a wider differential that includes psoriasis, nummular eczema, and pityriasis rosea. Unlike psoriasis, the id reaction is not chronic and responds to systemic antifungal therapy. Nummular eczema can be ruled out, though not entirely, by a lack of personal or family history of atopy. The characteristic cleavage lines of pityriasis rosea on the trunk are absent in patients with an id reaction, and there would be no preceding illness or herald patches seen in the id reaction.
Tinea capitis may cause a variety of id manifestations, including the papulosquamous phenotype. This case addresses practice gaps that may lead to delayed diagnosis. It also highlights the importance of recognizing uncommon morphologies, performing repeat cultures of the scalp after a negative fungal culture, and lowering the threshold of suspicion for tinea capitis in the appropriate age group and demographic, specifically pediatric patients of African descent.
- Sharma V, Silverberg NB, Howard R, et al. Do hair care practices affect the acquisition of tinea capitis? a case-control study. Arch Pediatr Adolesc Med. 2001;155:818-821.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:e453-e457.
- Mayser P. Dermatophyte: current situation [in German]. Hautarzt. 2017;68:316-323.
- Nowicki R. Allergic phenomena in the course of dermatomycoses [in Polish]. Pol Merkur Lekarski. 2003;14:532-534.
5. Boralevi F, Léauté-Labrèze C, Roul S, et al. Lupus-erythematosus-like eruption induced by Trichophyton mentagrophytes infection. Dermatology. 2003;206:303-306.
To the Editor:
Tinea capitis is a common childhood infection seen worldwide and is more prevalent in children of African descent.1 Treatment can be effective; however, the diagnosis may be delayed due to variability in presentation, camouflage of scalp scale with ointment, and the diagnostic experience of the provider. A common complication of tinea capitis is the dermatophytid (id) reaction, which commonly manifests as multiple 1- to 2-mm monomorphic papules. We report a case of a papulosquamous variant of an id reaction secondary to tinea capitis.
An 8-year-old African American child presented with annular hyperpigmented patches on the face and trunk of several months’ duration. There was no preceding fever, illness, scalp pruritus, or alopecia according to the patient’s mother. The hyperpigmented patches persisted despite use of hydrocortisone and antifungal creams prescribed by a primary care provider. A fungal culture of a scalp specimen was negative. Physical examination during the initial dermatology visit revealed multiple annular hyperpigmented patches on the trunk and extremities. No plaques were evident; however, the mother reported that when the lesions first developed, they were raised and mildly pruritic. The patient was prescribed triamcinolone ointment 0.1% twice daily as needed for itching, and sun protection was emphasized.
At the follow-up visit weeks later, the patient’s mother reported that the ointment had helped the lesions resolve faster, but new lesions continued to appear. Physical examination at this visit was notable for scattered hyperpigmented patches, annular hyperpigmented plaques, and erythematous plaques on the trunk, arms, and legs, in addition to papulosquamous plaques and hyperpigmented patches on the forehead (Figure 1). Suspicion for tinea capitis was discussed, a repeat scalp fungal culture was performed, and oral terbinafine 250 mg once daily was started empirically. The culture was positive for Trichophyton tonsurans supporting the diagnosis of concomitant tinea capitis. The rash resolved with terbinafine, and annular patches of postinflammatory hyperpigmentation remained.

Dermatophytid reactions are immunologically mediated, disseminated, eczematous eruptions occurring after cutaneous infections or inflammatory skin conditions. Reactions occur days to weeks after exposure to antigens of dermatophytes causing tinea pedis or capitis.2
Common culprits include Microsporum canis and T tonsurans.3 Dermatophytid reactions with tinea capitis exhibit morphologic variability including a symmetric distribution of grouped or diffuse,4 pruritic, erythematous or flesh-colored, follicular papules on the trunk, with or without progression to the face, torso, upper extremities, and/or lower extremities.3 Other reported manifestations include erythema multiforme, erythema nodosum,3 or lupuslike lesions, and crops of dyshidrotic vesicles on the hands in the setting of Trichophyton mentagrophytes–induced tinea pedis.5
The papulosquamous variant id reaction should be considered in a wider differential that includes psoriasis, nummular eczema, and pityriasis rosea. Unlike psoriasis, the id reaction is not chronic and responds to systemic antifungal therapy. Nummular eczema can be ruled out, though not entirely, by a lack of personal or family history of atopy. The characteristic cleavage lines of pityriasis rosea on the trunk are absent in patients with an id reaction, and there would be no preceding illness or herald patches seen in the id reaction.
Tinea capitis may cause a variety of id manifestations, including the papulosquamous phenotype. This case addresses practice gaps that may lead to delayed diagnosis. It also highlights the importance of recognizing uncommon morphologies, performing repeat cultures of the scalp after a negative fungal culture, and lowering the threshold of suspicion for tinea capitis in the appropriate age group and demographic, specifically pediatric patients of African descent.
To the Editor:
Tinea capitis is a common childhood infection seen worldwide and is more prevalent in children of African descent.1 Treatment can be effective; however, the diagnosis may be delayed due to variability in presentation, camouflage of scalp scale with ointment, and the diagnostic experience of the provider. A common complication of tinea capitis is the dermatophytid (id) reaction, which commonly manifests as multiple 1- to 2-mm monomorphic papules. We report a case of a papulosquamous variant of an id reaction secondary to tinea capitis.
An 8-year-old African American child presented with annular hyperpigmented patches on the face and trunk of several months’ duration. There was no preceding fever, illness, scalp pruritus, or alopecia according to the patient’s mother. The hyperpigmented patches persisted despite use of hydrocortisone and antifungal creams prescribed by a primary care provider. A fungal culture of a scalp specimen was negative. Physical examination during the initial dermatology visit revealed multiple annular hyperpigmented patches on the trunk and extremities. No plaques were evident; however, the mother reported that when the lesions first developed, they were raised and mildly pruritic. The patient was prescribed triamcinolone ointment 0.1% twice daily as needed for itching, and sun protection was emphasized.
At the follow-up visit weeks later, the patient’s mother reported that the ointment had helped the lesions resolve faster, but new lesions continued to appear. Physical examination at this visit was notable for scattered hyperpigmented patches, annular hyperpigmented plaques, and erythematous plaques on the trunk, arms, and legs, in addition to papulosquamous plaques and hyperpigmented patches on the forehead (Figure 1). Suspicion for tinea capitis was discussed, a repeat scalp fungal culture was performed, and oral terbinafine 250 mg once daily was started empirically. The culture was positive for Trichophyton tonsurans supporting the diagnosis of concomitant tinea capitis. The rash resolved with terbinafine, and annular patches of postinflammatory hyperpigmentation remained.

Dermatophytid reactions are immunologically mediated, disseminated, eczematous eruptions occurring after cutaneous infections or inflammatory skin conditions. Reactions occur days to weeks after exposure to antigens of dermatophytes causing tinea pedis or capitis.2
Common culprits include Microsporum canis and T tonsurans.3 Dermatophytid reactions with tinea capitis exhibit morphologic variability including a symmetric distribution of grouped or diffuse,4 pruritic, erythematous or flesh-colored, follicular papules on the trunk, with or without progression to the face, torso, upper extremities, and/or lower extremities.3 Other reported manifestations include erythema multiforme, erythema nodosum,3 or lupuslike lesions, and crops of dyshidrotic vesicles on the hands in the setting of Trichophyton mentagrophytes–induced tinea pedis.5
The papulosquamous variant id reaction should be considered in a wider differential that includes psoriasis, nummular eczema, and pityriasis rosea. Unlike psoriasis, the id reaction is not chronic and responds to systemic antifungal therapy. Nummular eczema can be ruled out, though not entirely, by a lack of personal or family history of atopy. The characteristic cleavage lines of pityriasis rosea on the trunk are absent in patients with an id reaction, and there would be no preceding illness or herald patches seen in the id reaction.
Tinea capitis may cause a variety of id manifestations, including the papulosquamous phenotype. This case addresses practice gaps that may lead to delayed diagnosis. It also highlights the importance of recognizing uncommon morphologies, performing repeat cultures of the scalp after a negative fungal culture, and lowering the threshold of suspicion for tinea capitis in the appropriate age group and demographic, specifically pediatric patients of African descent.
- Sharma V, Silverberg NB, Howard R, et al. Do hair care practices affect the acquisition of tinea capitis? a case-control study. Arch Pediatr Adolesc Med. 2001;155:818-821.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:e453-e457.
- Mayser P. Dermatophyte: current situation [in German]. Hautarzt. 2017;68:316-323.
- Nowicki R. Allergic phenomena in the course of dermatomycoses [in Polish]. Pol Merkur Lekarski. 2003;14:532-534.
5. Boralevi F, Léauté-Labrèze C, Roul S, et al. Lupus-erythematosus-like eruption induced by Trichophyton mentagrophytes infection. Dermatology. 2003;206:303-306.
- Sharma V, Silverberg NB, Howard R, et al. Do hair care practices affect the acquisition of tinea capitis? a case-control study. Arch Pediatr Adolesc Med. 2001;155:818-821.
- Cheng N, Rucker Wright D, Cohen BA. Dermatophytid in tinea capitis: rarely reported common phenomenon with clinical implications. Pediatrics. 2011;128:e453-e457.
- Mayser P. Dermatophyte: current situation [in German]. Hautarzt. 2017;68:316-323.
- Nowicki R. Allergic phenomena in the course of dermatomycoses [in Polish]. Pol Merkur Lekarski. 2003;14:532-534.
5. Boralevi F, Léauté-Labrèze C, Roul S, et al. Lupus-erythematosus-like eruption induced by Trichophyton mentagrophytes infection. Dermatology. 2003;206:303-306.
Practice Points
- Dermatophytid (id) reactions can manifest as papulosquamous eruptions after cutaneous infections or inflammatory skin conditions.
- High clinical suspicion for id reaction in patients of the appropriate age group and demographic—pediatric patients of African descent—is imperative for reaching the correct diagnosis.
- Repeat cultures of the scalp may be indicated in patients with high clinical probability for an id reaction despite a negative fungal culture or empiric systemic treatment.
The Role of Dermatology in Identifying and Reporting a Primary Varicella Outbreak
To the Editor:
Cases of primary varicella-zoster virus (VZV) are relatively uncommon in the United States since the introduction of the varicella vaccine in 1995, with an overall decline in cases of more than 97%.1 Prior to the vaccine, 70% of hospitalizations occurred in children; subsequently, hospitalizations among the pediatric population (aged ≤20 years) declined by 97%. Compared to children, adults and immunocompromised patients with VZV infection may present with more severe disease and experience more complications.1
Most children in the United States are vaccinated against VZV, with 90.3% receiving at least 1 dose by 24 months of age.2 However, many countries do not implement universal varicella vaccination for infants.3 As a result, physicians should remember to include primary varicella in the differential when clinically correlated, especially when evaluating patients who have immigrated to the United States or who may be living in unvaccinated communities. We report 2 cases of primary VZV manifesting in adults to remind readers of the salient clinical features of this disease and how dermatologists play a critical role in early and accurate identification of diseases that can have wide-reaching public health implications.
A 26-year-old man with no relevant medical history presented to the emergency department with an itchy and painful rash of 5 days’ duration that began on the trunk and spread to the face, lips, feet, hands, arms, and legs. He also reported shortness of breath, cough, and chills, and he had a temperature of 100.8 °F (38.2 °C). Physical examination revealed numerous erythematous papules and vesiculopustules, some with central umbilication and some with overlying gold crusts (Figure 1).
Later that day, a 47-year-old man with no relevant medical history presented to the same emergency department with a rash along with self-reported fever and sore throat of 3 days’ duration. Physical examination found innumerable erythematous vesicopustules scattered on the face, scalp, neck, trunk, arms, and legs, some with a “dew drop on a rose petal” appearance and some with overlying hemorrhagic crust (Figure 2).
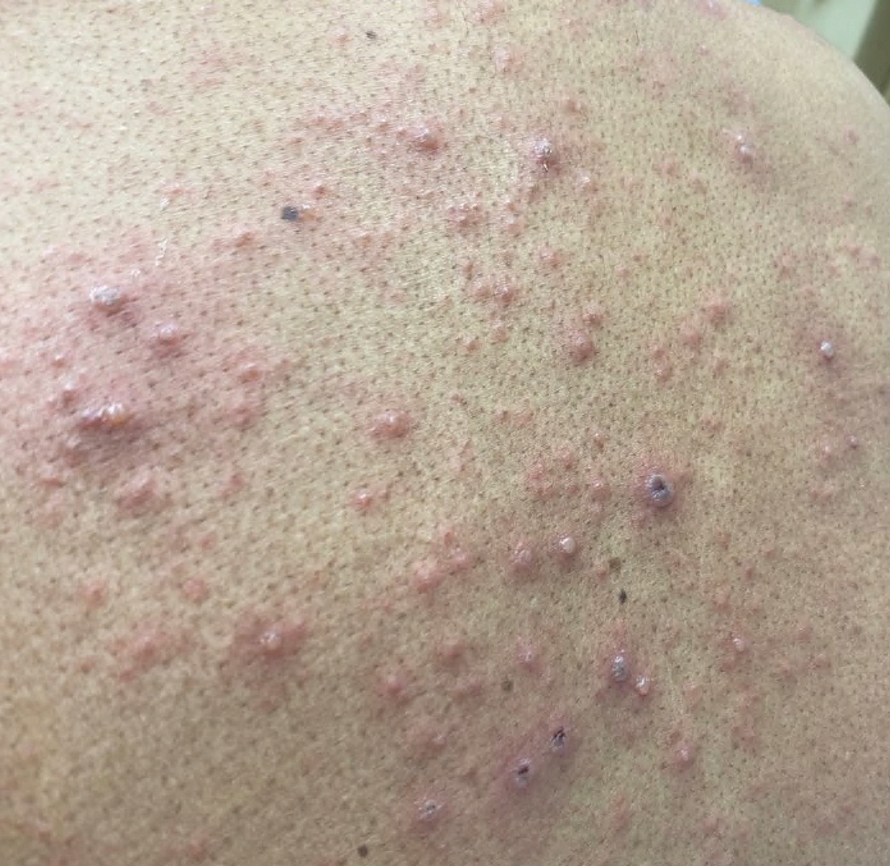
Although infection was of primary concern for the first patient, the presentation of the second patient prompted specific concern for primary VZV infection in both patients, who were placed on airborne and contact isolation precautions.
Skin biopsies from both patients showed acantholytic blisters, hair follicle necrosis, and marked dermal inflammation (Figure 3). Herpetic viral changes were seen in keratinocytes, with steel-grey nuclei, multinucleated keratinocytes, and chromatin margination. An immunostain for VZV was diffusely positive, and VZV antibody IgG was positive (Figure 4).
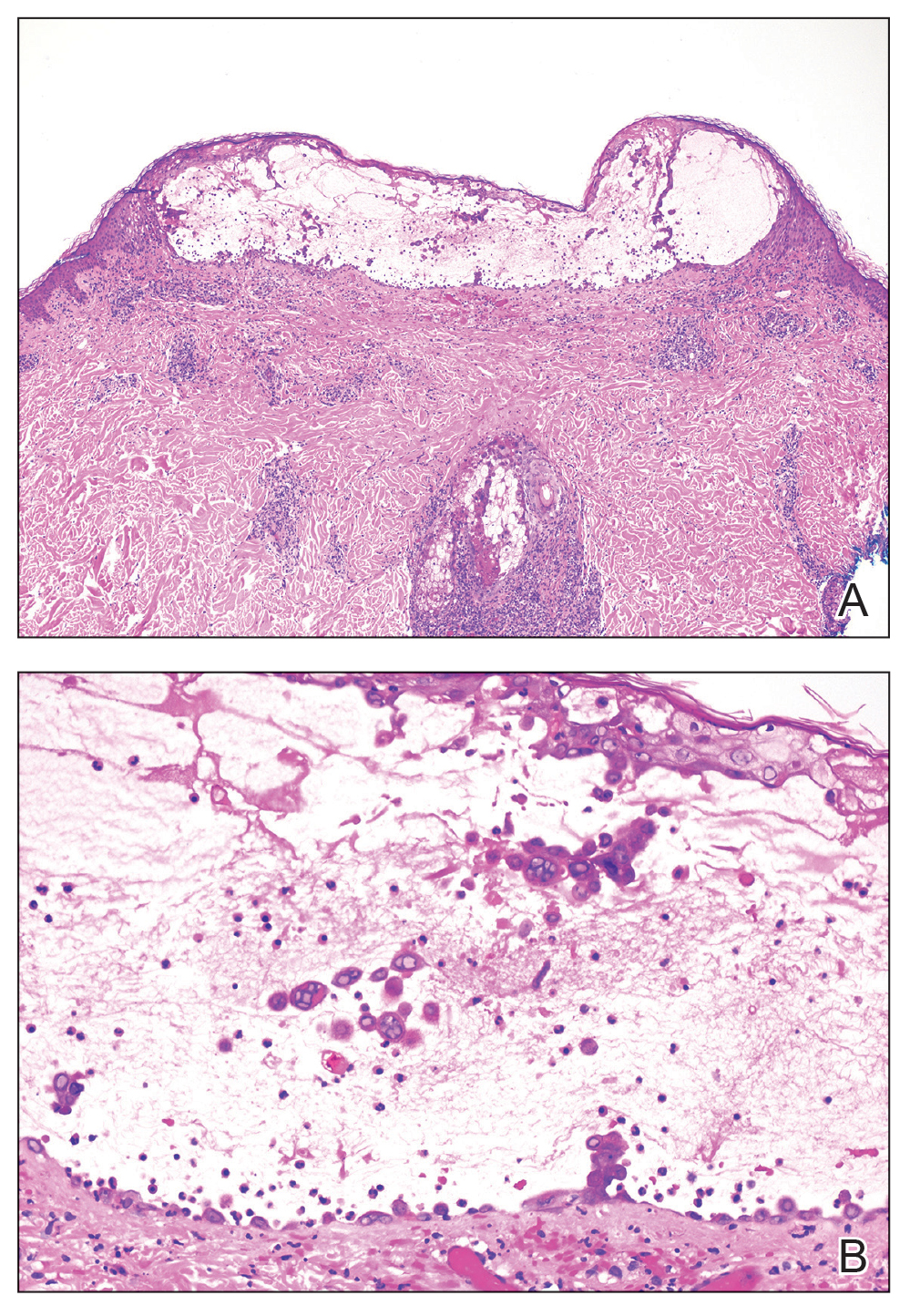
Upon additional questioning, both patients reported recent exposure to VZV-like illnesses in family members without a history of international travel. Neither of the patients was sure of their vaccination status or prior infection history. Both patients received intravenous acyclovir 10 mg/kg administered every 8 hours. Both patients experienced improvement and were discharged after 3 days on oral valacyclovir (1 g 3 times daily for a 7-day treatment course).
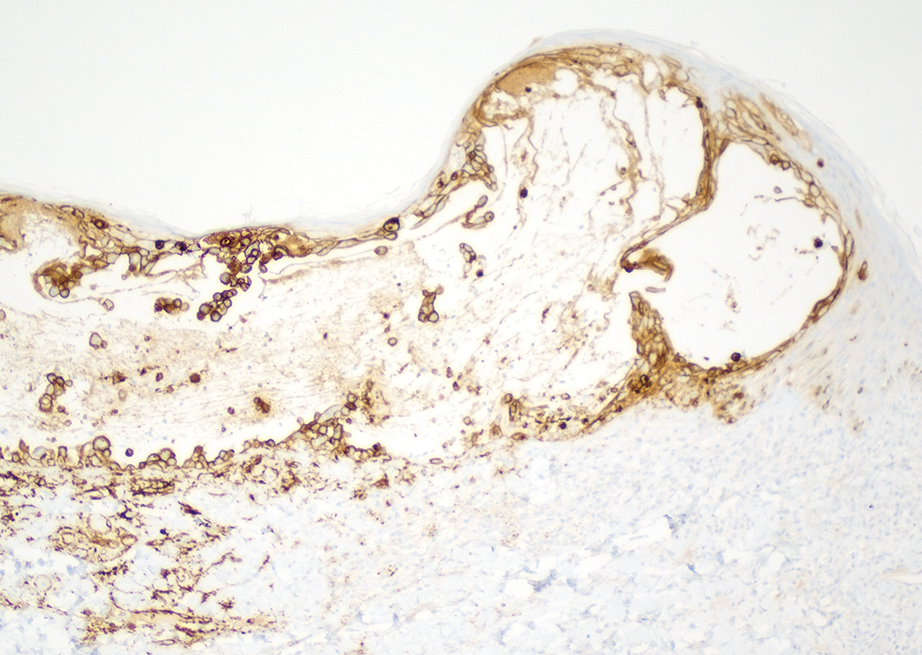
The similar presentation and timing of these 2 VZV cases caused concern for an unidentified community outbreak. The infection control team was notified; additionally, per hospital protocol the state health department was alerted as well as the clinicians and staff of the hospital with a request to be vigilant for further cases.
Despite high vaccination rates in the United States, outbreaks of varicella still occur, particularly among unvaccinated individuals, and a robust and efficient response is necessary to control the spread of such outbreaks.4 Many states, including Arkansas where our cases occurred, have laws mandating report of VZV cases to the department of health.5 Dermatologists play an important role in reporting cases, aiding in diagnosis through recognition of the physical examination findings, obtaining appropriate biopsy, and recommending additional laboratory testing.
Typical skin manifestations include a pruritic rash of macules, papules, vesicles, and crusted lesions distributed throughout the trunk, face, arms, and legs. Because new lesions appear over several days, they will be in different stages of healing, resulting in the simultaneous presence of papules, vesicles, and crusted lesions.6 This unique characteristic helps distinguish VZV from other skin diseases such as smallpox or mpox (monkeypox), which generally show lesions in similar stages of evolution.
Biopsy also can aid in identification. Viruses in the herpes family reveal similar histopathologic characteristics, including acantholysis and vesicle formation, intranuclear inclusions with margination of chromatin, multinucleation, and nuclear molding.7 Immunohistochemistry can be used to differentiate VZV from herpes simplex virus; however, neither microscopic examination nor immunohistochemistry distinguish primary VZV infection from herpes zoster (HZ).8
The mpox rash progresses more slowly than a VZV rash and has a centrifugal rather than central distribution that can involve the palms and soles. Lymphadenopathy is a characteristic finding in mpox.9 Rickettsialpox is distinguished from VZV primarily by the appearance of brown or black eschar after the original papulovesicular lesions dry out.10 Atypical hand, foot, and mouth disease can manifest in adults as widespread papulovesicular lesions. This form is associated with coxsackievirus A6 and may require direct fluorescent antibody assay or polymerase chain reaction of keratinocytes to rule out VZV.11
Herpes zoster occurs in older adults with a history of primary VZV.6 It manifests as vesicular lesions confined to 1 or 2 adjacent dermatomes vs the diffuse spread of VZV over the entire body. However, HZ can become disseminated in immunocompromised individuals, making it difficult to clinically distinguish from VZV.6 Serology can be helpful, as high IgM titers indicate an acute primary VZV infection. Subsequently increased IgG titers steadily wane over time and spike during reactivation.12
Dermatology and infectious disease consultations in our cases yielded a preliminary diagnosis through physical examination that was confirmed by biopsy and subsequent laboratory testing, which allowed for a swift response by the infection control team including isolation precautions to control a potential outbreak. Patients with VZV should remain in respiratory isolation until all lesions have crusted over.6
Individuals who had face-to-face indoor contact for at least 5 minutes or who shared a living space with an infected individual should be assessed for VZV immunity, which is defined as confirmed prior immunization or infection.5,13 Lack of VZV immunity requires postexposure prophylaxis—active immunization for the immunocompetent and passive immunization for the immunocompromised.13 Ultimately, no additional cases were reported in the community where our patients resided.
Immunocompetent children with primary VZV require supportive care only. Oral antiviral therapy is the treatment of choice for immunocompetent adults or anyone at increased risk for complications, while intravenous antivirals are recommended for the immunocompromised or those with VZV-related complications.14 A similar approach is used for HZ. Uncomplicated cases are treated with oral antivirals, and complicated cases (eg, HZ ophthalmicus) are treated with intravenous antivirals.15 Commonly used antivirals include acyclovir, valacyclovir, and famciclovir.14
Our cases highlight the ongoing risk for varicella outbreaks in unvaccinated or undervaccinated communities. Physician vigilance is necessary, and dermatology plays a particularly important role in swift and accurate detection of VZV, as demonstrated in our cases by the recognition of classic physical examination findings of erythematous and vesicular papules in each of the patients. Because primary VZV infection can result in life-threatening complications including hepatitis, encephalitis, and pancreatitis, prompt identification and initiation of therapy is important.6 Similarly, quick notification of public health officials about detected primary VZV cases is vital to containing potential community outbreaks.
- Centers for Disease Control and Prevention. Chickenpox (varicella) for healthcare professionals. Published October 21, 2022. Accessed March 6, 2024. https://www.cdc.gov/chickenpox/hcp/index.html#vaccination-impact
- National Center for Health Statistics. Immunization. Published June 13, 2023. Accessed March 6, 2024. https://www.cdc.gov/nchs/fastats/immunize.htm
- Lee YH, Choe YJ, Lee J, et al. Global varicella vaccination programs. Clin Exp Pediatr. 2022;65:555. doi:10.3345/CEP.2021.01564
- Leung J, Lopez AS, Marin M. Changing epidemiology of varicella outbreaks in the United States during the Varicella Vaccination Program, 1995–2019. J Infect Dis. 2022;226(suppl 4):S400-S406.
- Arkansas Department of Health. Rules Pertaining to Reportable Diseases. Published September 11, 2023. Accessed March 6, 2024. https://www.healthy.arkansas.gov/images/uploads/rules/ReportableDiseaseList.pdf
- Pergam S, Limaye A; The AST Infectious Diseases Community of Practice. Varicella zoster virus (VZV). Am J Transplant. 2009;9(suppl 4):S108-S115. doi:10.1111/J.1600-9143.2009.02901.X
- Hoyt B, Bhawan J. Histological spectrum of cutaneous herpes infections. Am J Dermatopathol. 2014;36:609-619. doi:10.1097/DAD.0000000000000148
- Oumarou Hama H, Aboudharam G, Barbieri R, et al. Immunohistochemical diagnosis of human infectious diseases: a review. Diagn Pathol. 2022;17. doi:10.1186/S13000-022-01197-5
- World Health Organization. Mpox (monkeypox). Published April 18, 2023. Accessed March 7, 2024. https://www.who.int/news-room/fact-sheets/detail/monkeypox
- Akram SM, Jamil RT, Gossman W. Rickettsia akari (Rickettsialpox). StatPearls [Internet]. Updated May 8, 2023. Accessed February 29, 2024. https://www.ncbi.nlm.nih.gov/books/NBK448081/
- Lott JP, Liu K, Landry ML, et al. Atypical hand-foot-mouth disease associated with coxsackievirus A6 infection. J Am Acad Dermatol. 2013;69:736. doi:10.1016/J.JAAD.2013.07.024
- Petrun B, Williams V, Brice S. Disseminated varicella-zoster virus in an immunocompetent adult. Dermatol Online J. 2015;21. doi:10.5070/D3213022343
- Kimberlin D, Barnett E, Lynfield R, et al. Exposure to specific pathogens. In: Red Book: 2021-2024 Report of the Committee of Infectious Disease. 32nd ed. American Academy of Pediatrics; 2021:1007-1009.
- Treatment of varicella (chickenpox) infection. UpToDate [Internet]. Updated February 7, 2024. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-varicella-chickenpox-infection
- Treatment of herpes zoster in the immunocompetent host. UpToDate [Internet]. Updated November 29, 2023. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-herpes-zoster
To the Editor:
Cases of primary varicella-zoster virus (VZV) are relatively uncommon in the United States since the introduction of the varicella vaccine in 1995, with an overall decline in cases of more than 97%.1 Prior to the vaccine, 70% of hospitalizations occurred in children; subsequently, hospitalizations among the pediatric population (aged ≤20 years) declined by 97%. Compared to children, adults and immunocompromised patients with VZV infection may present with more severe disease and experience more complications.1
Most children in the United States are vaccinated against VZV, with 90.3% receiving at least 1 dose by 24 months of age.2 However, many countries do not implement universal varicella vaccination for infants.3 As a result, physicians should remember to include primary varicella in the differential when clinically correlated, especially when evaluating patients who have immigrated to the United States or who may be living in unvaccinated communities. We report 2 cases of primary VZV manifesting in adults to remind readers of the salient clinical features of this disease and how dermatologists play a critical role in early and accurate identification of diseases that can have wide-reaching public health implications.
A 26-year-old man with no relevant medical history presented to the emergency department with an itchy and painful rash of 5 days’ duration that began on the trunk and spread to the face, lips, feet, hands, arms, and legs. He also reported shortness of breath, cough, and chills, and he had a temperature of 100.8 °F (38.2 °C). Physical examination revealed numerous erythematous papules and vesiculopustules, some with central umbilication and some with overlying gold crusts (Figure 1).

Later that day, a 47-year-old man with no relevant medical history presented to the same emergency department with a rash along with self-reported fever and sore throat of 3 days’ duration. Physical examination found innumerable erythematous vesicopustules scattered on the face, scalp, neck, trunk, arms, and legs, some with a “dew drop on a rose petal” appearance and some with overlying hemorrhagic crust (Figure 2).

Although infection was of primary concern for the first patient, the presentation of the second patient prompted specific concern for primary VZV infection in both patients, who were placed on airborne and contact isolation precautions.
Skin biopsies from both patients showed acantholytic blisters, hair follicle necrosis, and marked dermal inflammation (Figure 3). Herpetic viral changes were seen in keratinocytes, with steel-grey nuclei, multinucleated keratinocytes, and chromatin margination. An immunostain for VZV was diffusely positive, and VZV antibody IgG was positive (Figure 4).

Upon additional questioning, both patients reported recent exposure to VZV-like illnesses in family members without a history of international travel. Neither of the patients was sure of their vaccination status or prior infection history. Both patients received intravenous acyclovir 10 mg/kg administered every 8 hours. Both patients experienced improvement and were discharged after 3 days on oral valacyclovir (1 g 3 times daily for a 7-day treatment course).

The similar presentation and timing of these 2 VZV cases caused concern for an unidentified community outbreak. The infection control team was notified; additionally, per hospital protocol the state health department was alerted as well as the clinicians and staff of the hospital with a request to be vigilant for further cases.
Despite high vaccination rates in the United States, outbreaks of varicella still occur, particularly among unvaccinated individuals, and a robust and efficient response is necessary to control the spread of such outbreaks.4 Many states, including Arkansas where our cases occurred, have laws mandating report of VZV cases to the department of health.5 Dermatologists play an important role in reporting cases, aiding in diagnosis through recognition of the physical examination findings, obtaining appropriate biopsy, and recommending additional laboratory testing.
Typical skin manifestations include a pruritic rash of macules, papules, vesicles, and crusted lesions distributed throughout the trunk, face, arms, and legs. Because new lesions appear over several days, they will be in different stages of healing, resulting in the simultaneous presence of papules, vesicles, and crusted lesions.6 This unique characteristic helps distinguish VZV from other skin diseases such as smallpox or mpox (monkeypox), which generally show lesions in similar stages of evolution.
Biopsy also can aid in identification. Viruses in the herpes family reveal similar histopathologic characteristics, including acantholysis and vesicle formation, intranuclear inclusions with margination of chromatin, multinucleation, and nuclear molding.7 Immunohistochemistry can be used to differentiate VZV from herpes simplex virus; however, neither microscopic examination nor immunohistochemistry distinguish primary VZV infection from herpes zoster (HZ).8
The mpox rash progresses more slowly than a VZV rash and has a centrifugal rather than central distribution that can involve the palms and soles. Lymphadenopathy is a characteristic finding in mpox.9 Rickettsialpox is distinguished from VZV primarily by the appearance of brown or black eschar after the original papulovesicular lesions dry out.10 Atypical hand, foot, and mouth disease can manifest in adults as widespread papulovesicular lesions. This form is associated with coxsackievirus A6 and may require direct fluorescent antibody assay or polymerase chain reaction of keratinocytes to rule out VZV.11
Herpes zoster occurs in older adults with a history of primary VZV.6 It manifests as vesicular lesions confined to 1 or 2 adjacent dermatomes vs the diffuse spread of VZV over the entire body. However, HZ can become disseminated in immunocompromised individuals, making it difficult to clinically distinguish from VZV.6 Serology can be helpful, as high IgM titers indicate an acute primary VZV infection. Subsequently increased IgG titers steadily wane over time and spike during reactivation.12
Dermatology and infectious disease consultations in our cases yielded a preliminary diagnosis through physical examination that was confirmed by biopsy and subsequent laboratory testing, which allowed for a swift response by the infection control team including isolation precautions to control a potential outbreak. Patients with VZV should remain in respiratory isolation until all lesions have crusted over.6
Individuals who had face-to-face indoor contact for at least 5 minutes or who shared a living space with an infected individual should be assessed for VZV immunity, which is defined as confirmed prior immunization or infection.5,13 Lack of VZV immunity requires postexposure prophylaxis—active immunization for the immunocompetent and passive immunization for the immunocompromised.13 Ultimately, no additional cases were reported in the community where our patients resided.
Immunocompetent children with primary VZV require supportive care only. Oral antiviral therapy is the treatment of choice for immunocompetent adults or anyone at increased risk for complications, while intravenous antivirals are recommended for the immunocompromised or those with VZV-related complications.14 A similar approach is used for HZ. Uncomplicated cases are treated with oral antivirals, and complicated cases (eg, HZ ophthalmicus) are treated with intravenous antivirals.15 Commonly used antivirals include acyclovir, valacyclovir, and famciclovir.14
Our cases highlight the ongoing risk for varicella outbreaks in unvaccinated or undervaccinated communities. Physician vigilance is necessary, and dermatology plays a particularly important role in swift and accurate detection of VZV, as demonstrated in our cases by the recognition of classic physical examination findings of erythematous and vesicular papules in each of the patients. Because primary VZV infection can result in life-threatening complications including hepatitis, encephalitis, and pancreatitis, prompt identification and initiation of therapy is important.6 Similarly, quick notification of public health officials about detected primary VZV cases is vital to containing potential community outbreaks.
To the Editor:
Cases of primary varicella-zoster virus (VZV) are relatively uncommon in the United States since the introduction of the varicella vaccine in 1995, with an overall decline in cases of more than 97%.1 Prior to the vaccine, 70% of hospitalizations occurred in children; subsequently, hospitalizations among the pediatric population (aged ≤20 years) declined by 97%. Compared to children, adults and immunocompromised patients with VZV infection may present with more severe disease and experience more complications.1
Most children in the United States are vaccinated against VZV, with 90.3% receiving at least 1 dose by 24 months of age.2 However, many countries do not implement universal varicella vaccination for infants.3 As a result, physicians should remember to include primary varicella in the differential when clinically correlated, especially when evaluating patients who have immigrated to the United States or who may be living in unvaccinated communities. We report 2 cases of primary VZV manifesting in adults to remind readers of the salient clinical features of this disease and how dermatologists play a critical role in early and accurate identification of diseases that can have wide-reaching public health implications.
A 26-year-old man with no relevant medical history presented to the emergency department with an itchy and painful rash of 5 days’ duration that began on the trunk and spread to the face, lips, feet, hands, arms, and legs. He also reported shortness of breath, cough, and chills, and he had a temperature of 100.8 °F (38.2 °C). Physical examination revealed numerous erythematous papules and vesiculopustules, some with central umbilication and some with overlying gold crusts (Figure 1).

Later that day, a 47-year-old man with no relevant medical history presented to the same emergency department with a rash along with self-reported fever and sore throat of 3 days’ duration. Physical examination found innumerable erythematous vesicopustules scattered on the face, scalp, neck, trunk, arms, and legs, some with a “dew drop on a rose petal” appearance and some with overlying hemorrhagic crust (Figure 2).

Although infection was of primary concern for the first patient, the presentation of the second patient prompted specific concern for primary VZV infection in both patients, who were placed on airborne and contact isolation precautions.
Skin biopsies from both patients showed acantholytic blisters, hair follicle necrosis, and marked dermal inflammation (Figure 3). Herpetic viral changes were seen in keratinocytes, with steel-grey nuclei, multinucleated keratinocytes, and chromatin margination. An immunostain for VZV was diffusely positive, and VZV antibody IgG was positive (Figure 4).

Upon additional questioning, both patients reported recent exposure to VZV-like illnesses in family members without a history of international travel. Neither of the patients was sure of their vaccination status or prior infection history. Both patients received intravenous acyclovir 10 mg/kg administered every 8 hours. Both patients experienced improvement and were discharged after 3 days on oral valacyclovir (1 g 3 times daily for a 7-day treatment course).

The similar presentation and timing of these 2 VZV cases caused concern for an unidentified community outbreak. The infection control team was notified; additionally, per hospital protocol the state health department was alerted as well as the clinicians and staff of the hospital with a request to be vigilant for further cases.
Despite high vaccination rates in the United States, outbreaks of varicella still occur, particularly among unvaccinated individuals, and a robust and efficient response is necessary to control the spread of such outbreaks.4 Many states, including Arkansas where our cases occurred, have laws mandating report of VZV cases to the department of health.5 Dermatologists play an important role in reporting cases, aiding in diagnosis through recognition of the physical examination findings, obtaining appropriate biopsy, and recommending additional laboratory testing.
Typical skin manifestations include a pruritic rash of macules, papules, vesicles, and crusted lesions distributed throughout the trunk, face, arms, and legs. Because new lesions appear over several days, they will be in different stages of healing, resulting in the simultaneous presence of papules, vesicles, and crusted lesions.6 This unique characteristic helps distinguish VZV from other skin diseases such as smallpox or mpox (monkeypox), which generally show lesions in similar stages of evolution.
Biopsy also can aid in identification. Viruses in the herpes family reveal similar histopathologic characteristics, including acantholysis and vesicle formation, intranuclear inclusions with margination of chromatin, multinucleation, and nuclear molding.7 Immunohistochemistry can be used to differentiate VZV from herpes simplex virus; however, neither microscopic examination nor immunohistochemistry distinguish primary VZV infection from herpes zoster (HZ).8
The mpox rash progresses more slowly than a VZV rash and has a centrifugal rather than central distribution that can involve the palms and soles. Lymphadenopathy is a characteristic finding in mpox.9 Rickettsialpox is distinguished from VZV primarily by the appearance of brown or black eschar after the original papulovesicular lesions dry out.10 Atypical hand, foot, and mouth disease can manifest in adults as widespread papulovesicular lesions. This form is associated with coxsackievirus A6 and may require direct fluorescent antibody assay or polymerase chain reaction of keratinocytes to rule out VZV.11
Herpes zoster occurs in older adults with a history of primary VZV.6 It manifests as vesicular lesions confined to 1 or 2 adjacent dermatomes vs the diffuse spread of VZV over the entire body. However, HZ can become disseminated in immunocompromised individuals, making it difficult to clinically distinguish from VZV.6 Serology can be helpful, as high IgM titers indicate an acute primary VZV infection. Subsequently increased IgG titers steadily wane over time and spike during reactivation.12
Dermatology and infectious disease consultations in our cases yielded a preliminary diagnosis through physical examination that was confirmed by biopsy and subsequent laboratory testing, which allowed for a swift response by the infection control team including isolation precautions to control a potential outbreak. Patients with VZV should remain in respiratory isolation until all lesions have crusted over.6
Individuals who had face-to-face indoor contact for at least 5 minutes or who shared a living space with an infected individual should be assessed for VZV immunity, which is defined as confirmed prior immunization or infection.5,13 Lack of VZV immunity requires postexposure prophylaxis—active immunization for the immunocompetent and passive immunization for the immunocompromised.13 Ultimately, no additional cases were reported in the community where our patients resided.
Immunocompetent children with primary VZV require supportive care only. Oral antiviral therapy is the treatment of choice for immunocompetent adults or anyone at increased risk for complications, while intravenous antivirals are recommended for the immunocompromised or those with VZV-related complications.14 A similar approach is used for HZ. Uncomplicated cases are treated with oral antivirals, and complicated cases (eg, HZ ophthalmicus) are treated with intravenous antivirals.15 Commonly used antivirals include acyclovir, valacyclovir, and famciclovir.14
Our cases highlight the ongoing risk for varicella outbreaks in unvaccinated or undervaccinated communities. Physician vigilance is necessary, and dermatology plays a particularly important role in swift and accurate detection of VZV, as demonstrated in our cases by the recognition of classic physical examination findings of erythematous and vesicular papules in each of the patients. Because primary VZV infection can result in life-threatening complications including hepatitis, encephalitis, and pancreatitis, prompt identification and initiation of therapy is important.6 Similarly, quick notification of public health officials about detected primary VZV cases is vital to containing potential community outbreaks.
- Centers for Disease Control and Prevention. Chickenpox (varicella) for healthcare professionals. Published October 21, 2022. Accessed March 6, 2024. https://www.cdc.gov/chickenpox/hcp/index.html#vaccination-impact
- National Center for Health Statistics. Immunization. Published June 13, 2023. Accessed March 6, 2024. https://www.cdc.gov/nchs/fastats/immunize.htm
- Lee YH, Choe YJ, Lee J, et al. Global varicella vaccination programs. Clin Exp Pediatr. 2022;65:555. doi:10.3345/CEP.2021.01564
- Leung J, Lopez AS, Marin M. Changing epidemiology of varicella outbreaks in the United States during the Varicella Vaccination Program, 1995–2019. J Infect Dis. 2022;226(suppl 4):S400-S406.
- Arkansas Department of Health. Rules Pertaining to Reportable Diseases. Published September 11, 2023. Accessed March 6, 2024. https://www.healthy.arkansas.gov/images/uploads/rules/ReportableDiseaseList.pdf
- Pergam S, Limaye A; The AST Infectious Diseases Community of Practice. Varicella zoster virus (VZV). Am J Transplant. 2009;9(suppl 4):S108-S115. doi:10.1111/J.1600-9143.2009.02901.X
- Hoyt B, Bhawan J. Histological spectrum of cutaneous herpes infections. Am J Dermatopathol. 2014;36:609-619. doi:10.1097/DAD.0000000000000148
- Oumarou Hama H, Aboudharam G, Barbieri R, et al. Immunohistochemical diagnosis of human infectious diseases: a review. Diagn Pathol. 2022;17. doi:10.1186/S13000-022-01197-5
- World Health Organization. Mpox (monkeypox). Published April 18, 2023. Accessed March 7, 2024. https://www.who.int/news-room/fact-sheets/detail/monkeypox
- Akram SM, Jamil RT, Gossman W. Rickettsia akari (Rickettsialpox). StatPearls [Internet]. Updated May 8, 2023. Accessed February 29, 2024. https://www.ncbi.nlm.nih.gov/books/NBK448081/
- Lott JP, Liu K, Landry ML, et al. Atypical hand-foot-mouth disease associated with coxsackievirus A6 infection. J Am Acad Dermatol. 2013;69:736. doi:10.1016/J.JAAD.2013.07.024
- Petrun B, Williams V, Brice S. Disseminated varicella-zoster virus in an immunocompetent adult. Dermatol Online J. 2015;21. doi:10.5070/D3213022343
- Kimberlin D, Barnett E, Lynfield R, et al. Exposure to specific pathogens. In: Red Book: 2021-2024 Report of the Committee of Infectious Disease. 32nd ed. American Academy of Pediatrics; 2021:1007-1009.
- Treatment of varicella (chickenpox) infection. UpToDate [Internet]. Updated February 7, 2024. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-varicella-chickenpox-infection
- Treatment of herpes zoster in the immunocompetent host. UpToDate [Internet]. Updated November 29, 2023. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-herpes-zoster
- Centers for Disease Control and Prevention. Chickenpox (varicella) for healthcare professionals. Published October 21, 2022. Accessed March 6, 2024. https://www.cdc.gov/chickenpox/hcp/index.html#vaccination-impact
- National Center for Health Statistics. Immunization. Published June 13, 2023. Accessed March 6, 2024. https://www.cdc.gov/nchs/fastats/immunize.htm
- Lee YH, Choe YJ, Lee J, et al. Global varicella vaccination programs. Clin Exp Pediatr. 2022;65:555. doi:10.3345/CEP.2021.01564
- Leung J, Lopez AS, Marin M. Changing epidemiology of varicella outbreaks in the United States during the Varicella Vaccination Program, 1995–2019. J Infect Dis. 2022;226(suppl 4):S400-S406.
- Arkansas Department of Health. Rules Pertaining to Reportable Diseases. Published September 11, 2023. Accessed March 6, 2024. https://www.healthy.arkansas.gov/images/uploads/rules/ReportableDiseaseList.pdf
- Pergam S, Limaye A; The AST Infectious Diseases Community of Practice. Varicella zoster virus (VZV). Am J Transplant. 2009;9(suppl 4):S108-S115. doi:10.1111/J.1600-9143.2009.02901.X
- Hoyt B, Bhawan J. Histological spectrum of cutaneous herpes infections. Am J Dermatopathol. 2014;36:609-619. doi:10.1097/DAD.0000000000000148
- Oumarou Hama H, Aboudharam G, Barbieri R, et al. Immunohistochemical diagnosis of human infectious diseases: a review. Diagn Pathol. 2022;17. doi:10.1186/S13000-022-01197-5
- World Health Organization. Mpox (monkeypox). Published April 18, 2023. Accessed March 7, 2024. https://www.who.int/news-room/fact-sheets/detail/monkeypox
- Akram SM, Jamil RT, Gossman W. Rickettsia akari (Rickettsialpox). StatPearls [Internet]. Updated May 8, 2023. Accessed February 29, 2024. https://www.ncbi.nlm.nih.gov/books/NBK448081/
- Lott JP, Liu K, Landry ML, et al. Atypical hand-foot-mouth disease associated with coxsackievirus A6 infection. J Am Acad Dermatol. 2013;69:736. doi:10.1016/J.JAAD.2013.07.024
- Petrun B, Williams V, Brice S. Disseminated varicella-zoster virus in an immunocompetent adult. Dermatol Online J. 2015;21. doi:10.5070/D3213022343
- Kimberlin D, Barnett E, Lynfield R, et al. Exposure to specific pathogens. In: Red Book: 2021-2024 Report of the Committee of Infectious Disease. 32nd ed. American Academy of Pediatrics; 2021:1007-1009.
- Treatment of varicella (chickenpox) infection. UpToDate [Internet]. Updated February 7, 2024. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-varicella-chickenpox-infection
- Treatment of herpes zoster in the immunocompetent host. UpToDate [Internet]. Updated November 29, 2023. Accessed March 6, 2024. https://www.uptodate.com/contents/treatment-of-herpes-zoster
Practice Points
- Primary varicella is a relatively infrequent occurrence since the introduction of vaccination, creating the need for a reminder on the importance of including it in the differential when clinically appropriate.
- When outbreaks do happen, typically among unvaccinated communities, swift identification via physical examination and histology is imperative to allow infection control teams and public health officials to quickly take action.
Getting ready for Boston
A quality educational meeting starts with a great slate of programs tailored to its audience, and CHEST 2024 is on track to offer the highest tier of pulmonary, critical care, and sleep medicine education that attendees have come to expect from the CHEST Annual Meeting.
While planning for the meeting started with the open call for 2024 sessions at the conclusion of the CHEST Annual Meeting 2023, CHEST 2024 began to take shape when the schedule—and the curriculum chairs—came together. In mid-February, members of the Scientific Program Committee gathered in person at CHEST headquarters in Glenview, Illinois, to review submissions and solidify the schedule for the upcoming CHEST 2024 meeting, taking place in Boston, October 6 to 9.
Following CHEST 2023 in Honolulu, those planning for Boston were brimming with excitement to start planning a meeting closer to home. One event in particular that committee members are excited for will be a session dedicated to the “Black Angels,” the nurses who helped cure TB, featuring surviving member, Virginia Allen, and book (The Black Angels: The Untold Story of the Nurses Who Helped Cure Tuberculosis) author, Maria Smilios. Because of the location, both Allen and Smilios will be able to join on-site in Boston and will bring with them, for the first time on public display, a curated selection of papers from Edward Robitzek, MD, courtesy of the Robitzek family. This collection will include records of TB treatment trials that forever changed the course of the disease in 1952.
In addition to this look into the history of chest medicine, the CHEST Annual Meeting 2024 will also feature the latest advancements in the field, including the anticipated hot topic of the meeting, the use of artificial intelligence (AI) in medicine.
“There [are] going to be a lot of hot topics covered at CHEST 2024, like bronchoscopy approaches, treatments for COPD,” said Gabe Bosslet, MD, FCCP, Chair of the Scientific Program Committee. “But if there was one that sort of was the outlier this year, I think it’s artificial intelligence and its use in pulmonary and critical care medicine.”
The sessions covering AI include its presence in medical education, as well as treating interstitial lung disease, chest infections, and more.
Beyond the latest in artificial technology, the CHEST Annual Meeting 2024 will feature more than 200 sessions covering eight curriculum groups with something for everyone in chest medicine:
- Airways Disorders
- Critical Care
- Cardiovascular/Pulmonary Vascular Disease
- Chest Infections/Disaster Medicine/Systemic Disease
- Interstitial Lung Disease/Transplant
- Interdisciplinary/Practice Operations/Education
- Lung Cancer/Interventional Pulmonology/Bronchoscopy/Radiology
- Sleep Medicine
The meeting will host topics for a wide range of experience levels (from those still in training to those who are years or decades into their careers) and welcomes all members of the care team. “These are not physician-centric issues, topics, or sessions. These are sessions that if you’re working around patients with pulmonary or critical care diseases, these are definitively for you,” Dr. Bosslet said.
With something for everyone—and for the first time ever in Boston—CHEST 2024 will not be a meeting to miss. Keep an eye out for registration to open in May, as early bird pricing will be available for a short time.
Dr. Danckers’ social media takeover
For an inside look into what happens during a meeting of the Scientific Program Committee, we invited member of the committee, Mauricio Danckers, MD, FCCP, to take the reins of CHEST social media to share his experience.
Dr. Danckers posted behind-the-scenes pictures of each of the curriculum chairs and teased a picture of the completed session schedule.
To see the takeover posts, visit the CHEST Instagram (@accpCHEST) and view the CHEST 2024 story pinned to the top of the profile.


A quality educational meeting starts with a great slate of programs tailored to its audience, and CHEST 2024 is on track to offer the highest tier of pulmonary, critical care, and sleep medicine education that attendees have come to expect from the CHEST Annual Meeting.
While planning for the meeting started with the open call for 2024 sessions at the conclusion of the CHEST Annual Meeting 2023, CHEST 2024 began to take shape when the schedule—and the curriculum chairs—came together. In mid-February, members of the Scientific Program Committee gathered in person at CHEST headquarters in Glenview, Illinois, to review submissions and solidify the schedule for the upcoming CHEST 2024 meeting, taking place in Boston, October 6 to 9.
Following CHEST 2023 in Honolulu, those planning for Boston were brimming with excitement to start planning a meeting closer to home. One event in particular that committee members are excited for will be a session dedicated to the “Black Angels,” the nurses who helped cure TB, featuring surviving member, Virginia Allen, and book (The Black Angels: The Untold Story of the Nurses Who Helped Cure Tuberculosis) author, Maria Smilios. Because of the location, both Allen and Smilios will be able to join on-site in Boston and will bring with them, for the first time on public display, a curated selection of papers from Edward Robitzek, MD, courtesy of the Robitzek family. This collection will include records of TB treatment trials that forever changed the course of the disease in 1952.
In addition to this look into the history of chest medicine, the CHEST Annual Meeting 2024 will also feature the latest advancements in the field, including the anticipated hot topic of the meeting, the use of artificial intelligence (AI) in medicine.
“There [are] going to be a lot of hot topics covered at CHEST 2024, like bronchoscopy approaches, treatments for COPD,” said Gabe Bosslet, MD, FCCP, Chair of the Scientific Program Committee. “But if there was one that sort of was the outlier this year, I think it’s artificial intelligence and its use in pulmonary and critical care medicine.”
The sessions covering AI include its presence in medical education, as well as treating interstitial lung disease, chest infections, and more.
Beyond the latest in artificial technology, the CHEST Annual Meeting 2024 will feature more than 200 sessions covering eight curriculum groups with something for everyone in chest medicine:
- Airways Disorders
- Critical Care
- Cardiovascular/Pulmonary Vascular Disease
- Chest Infections/Disaster Medicine/Systemic Disease
- Interstitial Lung Disease/Transplant
- Interdisciplinary/Practice Operations/Education
- Lung Cancer/Interventional Pulmonology/Bronchoscopy/Radiology
- Sleep Medicine
The meeting will host topics for a wide range of experience levels (from those still in training to those who are years or decades into their careers) and welcomes all members of the care team. “These are not physician-centric issues, topics, or sessions. These are sessions that if you’re working around patients with pulmonary or critical care diseases, these are definitively for you,” Dr. Bosslet said.
With something for everyone—and for the first time ever in Boston—CHEST 2024 will not be a meeting to miss. Keep an eye out for registration to open in May, as early bird pricing will be available for a short time.
Dr. Danckers’ social media takeover
For an inside look into what happens during a meeting of the Scientific Program Committee, we invited member of the committee, Mauricio Danckers, MD, FCCP, to take the reins of CHEST social media to share his experience.
Dr. Danckers posted behind-the-scenes pictures of each of the curriculum chairs and teased a picture of the completed session schedule.
To see the takeover posts, visit the CHEST Instagram (@accpCHEST) and view the CHEST 2024 story pinned to the top of the profile.


A quality educational meeting starts with a great slate of programs tailored to its audience, and CHEST 2024 is on track to offer the highest tier of pulmonary, critical care, and sleep medicine education that attendees have come to expect from the CHEST Annual Meeting.
While planning for the meeting started with the open call for 2024 sessions at the conclusion of the CHEST Annual Meeting 2023, CHEST 2024 began to take shape when the schedule—and the curriculum chairs—came together. In mid-February, members of the Scientific Program Committee gathered in person at CHEST headquarters in Glenview, Illinois, to review submissions and solidify the schedule for the upcoming CHEST 2024 meeting, taking place in Boston, October 6 to 9.
Following CHEST 2023 in Honolulu, those planning for Boston were brimming with excitement to start planning a meeting closer to home. One event in particular that committee members are excited for will be a session dedicated to the “Black Angels,” the nurses who helped cure TB, featuring surviving member, Virginia Allen, and book (The Black Angels: The Untold Story of the Nurses Who Helped Cure Tuberculosis) author, Maria Smilios. Because of the location, both Allen and Smilios will be able to join on-site in Boston and will bring with them, for the first time on public display, a curated selection of papers from Edward Robitzek, MD, courtesy of the Robitzek family. This collection will include records of TB treatment trials that forever changed the course of the disease in 1952.
In addition to this look into the history of chest medicine, the CHEST Annual Meeting 2024 will also feature the latest advancements in the field, including the anticipated hot topic of the meeting, the use of artificial intelligence (AI) in medicine.
“There [are] going to be a lot of hot topics covered at CHEST 2024, like bronchoscopy approaches, treatments for COPD,” said Gabe Bosslet, MD, FCCP, Chair of the Scientific Program Committee. “But if there was one that sort of was the outlier this year, I think it’s artificial intelligence and its use in pulmonary and critical care medicine.”
The sessions covering AI include its presence in medical education, as well as treating interstitial lung disease, chest infections, and more.
Beyond the latest in artificial technology, the CHEST Annual Meeting 2024 will feature more than 200 sessions covering eight curriculum groups with something for everyone in chest medicine:
- Airways Disorders
- Critical Care
- Cardiovascular/Pulmonary Vascular Disease
- Chest Infections/Disaster Medicine/Systemic Disease
- Interstitial Lung Disease/Transplant
- Interdisciplinary/Practice Operations/Education
- Lung Cancer/Interventional Pulmonology/Bronchoscopy/Radiology
- Sleep Medicine
The meeting will host topics for a wide range of experience levels (from those still in training to those who are years or decades into their careers) and welcomes all members of the care team. “These are not physician-centric issues, topics, or sessions. These are sessions that if you’re working around patients with pulmonary or critical care diseases, these are definitively for you,” Dr. Bosslet said.
With something for everyone—and for the first time ever in Boston—CHEST 2024 will not be a meeting to miss. Keep an eye out for registration to open in May, as early bird pricing will be available for a short time.
Dr. Danckers’ social media takeover
For an inside look into what happens during a meeting of the Scientific Program Committee, we invited member of the committee, Mauricio Danckers, MD, FCCP, to take the reins of CHEST social media to share his experience.
Dr. Danckers posted behind-the-scenes pictures of each of the curriculum chairs and teased a picture of the completed session schedule.
To see the takeover posts, visit the CHEST Instagram (@accpCHEST) and view the CHEST 2024 story pinned to the top of the profile.


Certain Pesticides Linked With Risk for Pancreatic Cancer
One of them, a case-control study, showed an elevated risk in individuals whose adipose tissue contained substances that are now banned.
“The association between pesticides and pancreatic cancer exists. It is of low magnitude but robust, concerning cumulative pesticides and three substances: Mancozeb, glyphosate, and sulfur in spray form,” said Mathias Brugel, MD, hospital practitioner at Basque Coast Hospital Center in Bayonne, France, during his presentation.
Regarding the four other liposoluble substances associated with an increased risk for pancreatic cancer in the second study, “their use has been banned since the 1990s, but they are still present in soils and in the air,” Dr. Brugel told this news organization.
For example, in Reims, France, the assessment of air quality by ATMO Grand Est revealed the presence of banned pesticides in the air, he added. However, Dr. Brugel stressed that a cause-effect relationship between pesticide exposure and the risk for pancreatic cancer cannot be established with these studies.
Incidence Rising Constantly
The incidence of pancreatic adenocarcinoma has been increasing steadily for more than 30 years. In France, nearly 16,000 new cases were reported in 2023, which represented an annual increase of about 2%. According to the National Cancer Institute, “pancreatic adenocarcinoma could become the second leading cause of cancer mortality by 2030.”
“This increase in incidence is particularly strong in France compared with other Western countries. The causes are still poorly understood. One might wonder whether environmental factors like pesticides are involved,” said Dr. Brugel.
Known to have a mechanism of action favoring oncogenesis, pesticides are suspected of being responsible for the rise in certain cancers, especially given their extensive use in France. In total, around 300 substances are authorized, and 65,000 tons are applied each year, making France the largest consumer of pesticides in Europe.
“Contamination is ubiquitous, meaning they are found in soil, water, air, and in individuals,” said Dr. Brugel. According to a study by the Institute for Scientific Expertise Research, pesticide residues were detected in 64% of hair samples taken from French volunteers.
The literature increasingly reported data suggesting a link between pesticide exposure and the development of certain diseases like cancer. A 2021 document by Inserm notably confirmed the strong presumption of a link between occupational pesticide exposure and pathologies such as non-Hodgkin’s lymphoma and prostate cancer.
High-Incidence Zones
To explore the link between pesticide exposure and pancreatic cancer, Dr. Brugel and his colleagues conducted the EcoPESTIPAC and PESTIPAC studies, the results of which were presented at this year’s conference.
In EcoPESTIPAC, researchers conducted a national ecological regression by dividing the entire French territory into 5529 spatial units. The number of pancreatic cancer cases per spatial unit per year (disease-mapping) was determined using the National Health Data System.
Nine chemicals, including glyphosate, were included, thus covering half of pesticide purchases in France. The cumulative quantity of pesticides, regardless of molecule, was also examined. Pesticide exposure was estimated by the median ratio between pesticide purchase and agricultural area per spatial unit over an 11-year period from early 2011 to the end of 2021.
Mor than 134,000 cases of pancreatic cancer were reported during this period. The analysis revealed three high-incidence zones located around Paris, in central France, and in the Mediterranean basin, while spatial units in the western region showed the lowest incidences.
The heterogeneous distribution of the disease suggests the involvement of risk factors, said Dr. Brugel. After adjusting for confounding factors such as smoking, the study showed an increased risk for pancreatic cancer associated with the cumulative quantity of pesticides and three specific substances: Sulfur in spray form, mancozeb, and glyphosate.
Risk Increases
A dose-response relationship was evident. For an increase in pesticide use of 2.5 kg/hectare over 11 years, the risk for pancreatic adenocarcinoma increased from 0.9% to 1.4%. “The increase is relatively small, but one must not forget that this risk applies to all of France,” said Dr. Brugel. Indeed, the risk appeared homogeneous across the entire territory.
This was the first study to explore this link at the national level. Although the association between the four identified factors and pancreatic risk was robust, the study had some limitations. It relied on the quantities of pesticides purchased to estimate the quantities used, Dr. Brugel pointed out.
The second study, PESTIPAC, was a case-control study conducted at the Reims University Hospital to explore the association between pancreatic adenocarcinoma and concentrations of organochlorine pesticides in fat and urine.
The study included 26 patients with pancreatic cancer who had abdominal surgery that allowed for adipose tissue sampling (minimum 10 g). Urine was collected in the morning on an empty stomach.
A control group was formed by including 26 other patients who underwent surgery for a benign abdominal condition such as gallstones or hernia, thus allowing for the same sampling. Individuals in both groups were matched for age and body mass index, two risk factors for pancreatic cancer.
Banned Substances
In total, 345 substances were searched for using chromatography and mass spectrometry. Analyses revealed the presence of five banned substances in all patients, while nine substances were found in half of the samples.
“Contamination is very widespread, both in patients with pancreatic cancer and in the controls,” said Dr. Brugel. Consequently, for this study, between-group comparisons of substances present in all individuals could not be performed.
After adjustment, an association with an increased risk for pancreatic cancer was nonetheless observed with four liposoluble substances: 4,4-DDE, mirex or perchlordecone, trans-nonachlor, and cis-nonachlor. All four substances are herbicides that have been banned for at least 30 years.
The study also aimed to assess the effect of pesticide presence in the body on survival after pancreatic cancer. The results showed no significant difference for overall survival or progression-free survival.
“Pesticides are a credible candidate to explain the increase in the incidence of pancreatic adenocarcinoma,” said Dr. Brugel. However, “if associations between pancreatic cancer and pesticides exist, they remain poorly understood, and it is difficult to establish clear causality.”
Further large-scale studies will be needed to confirm these associations. An evaluation of the general population’s exposure to banned substances also appears justified, according to the researchers.
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
One of them, a case-control study, showed an elevated risk in individuals whose adipose tissue contained substances that are now banned.
“The association between pesticides and pancreatic cancer exists. It is of low magnitude but robust, concerning cumulative pesticides and three substances: Mancozeb, glyphosate, and sulfur in spray form,” said Mathias Brugel, MD, hospital practitioner at Basque Coast Hospital Center in Bayonne, France, during his presentation.
Regarding the four other liposoluble substances associated with an increased risk for pancreatic cancer in the second study, “their use has been banned since the 1990s, but they are still present in soils and in the air,” Dr. Brugel told this news organization.
For example, in Reims, France, the assessment of air quality by ATMO Grand Est revealed the presence of banned pesticides in the air, he added. However, Dr. Brugel stressed that a cause-effect relationship between pesticide exposure and the risk for pancreatic cancer cannot be established with these studies.
Incidence Rising Constantly
The incidence of pancreatic adenocarcinoma has been increasing steadily for more than 30 years. In France, nearly 16,000 new cases were reported in 2023, which represented an annual increase of about 2%. According to the National Cancer Institute, “pancreatic adenocarcinoma could become the second leading cause of cancer mortality by 2030.”
“This increase in incidence is particularly strong in France compared with other Western countries. The causes are still poorly understood. One might wonder whether environmental factors like pesticides are involved,” said Dr. Brugel.
Known to have a mechanism of action favoring oncogenesis, pesticides are suspected of being responsible for the rise in certain cancers, especially given their extensive use in France. In total, around 300 substances are authorized, and 65,000 tons are applied each year, making France the largest consumer of pesticides in Europe.
“Contamination is ubiquitous, meaning they are found in soil, water, air, and in individuals,” said Dr. Brugel. According to a study by the Institute for Scientific Expertise Research, pesticide residues were detected in 64% of hair samples taken from French volunteers.
The literature increasingly reported data suggesting a link between pesticide exposure and the development of certain diseases like cancer. A 2021 document by Inserm notably confirmed the strong presumption of a link between occupational pesticide exposure and pathologies such as non-Hodgkin’s lymphoma and prostate cancer.
High-Incidence Zones
To explore the link between pesticide exposure and pancreatic cancer, Dr. Brugel and his colleagues conducted the EcoPESTIPAC and PESTIPAC studies, the results of which were presented at this year’s conference.
In EcoPESTIPAC, researchers conducted a national ecological regression by dividing the entire French territory into 5529 spatial units. The number of pancreatic cancer cases per spatial unit per year (disease-mapping) was determined using the National Health Data System.
Nine chemicals, including glyphosate, were included, thus covering half of pesticide purchases in France. The cumulative quantity of pesticides, regardless of molecule, was also examined. Pesticide exposure was estimated by the median ratio between pesticide purchase and agricultural area per spatial unit over an 11-year period from early 2011 to the end of 2021.
Mor than 134,000 cases of pancreatic cancer were reported during this period. The analysis revealed three high-incidence zones located around Paris, in central France, and in the Mediterranean basin, while spatial units in the western region showed the lowest incidences.
The heterogeneous distribution of the disease suggests the involvement of risk factors, said Dr. Brugel. After adjusting for confounding factors such as smoking, the study showed an increased risk for pancreatic cancer associated with the cumulative quantity of pesticides and three specific substances: Sulfur in spray form, mancozeb, and glyphosate.
Risk Increases
A dose-response relationship was evident. For an increase in pesticide use of 2.5 kg/hectare over 11 years, the risk for pancreatic adenocarcinoma increased from 0.9% to 1.4%. “The increase is relatively small, but one must not forget that this risk applies to all of France,” said Dr. Brugel. Indeed, the risk appeared homogeneous across the entire territory.
This was the first study to explore this link at the national level. Although the association between the four identified factors and pancreatic risk was robust, the study had some limitations. It relied on the quantities of pesticides purchased to estimate the quantities used, Dr. Brugel pointed out.
The second study, PESTIPAC, was a case-control study conducted at the Reims University Hospital to explore the association between pancreatic adenocarcinoma and concentrations of organochlorine pesticides in fat and urine.
The study included 26 patients with pancreatic cancer who had abdominal surgery that allowed for adipose tissue sampling (minimum 10 g). Urine was collected in the morning on an empty stomach.
A control group was formed by including 26 other patients who underwent surgery for a benign abdominal condition such as gallstones or hernia, thus allowing for the same sampling. Individuals in both groups were matched for age and body mass index, two risk factors for pancreatic cancer.
Banned Substances
In total, 345 substances were searched for using chromatography and mass spectrometry. Analyses revealed the presence of five banned substances in all patients, while nine substances were found in half of the samples.
“Contamination is very widespread, both in patients with pancreatic cancer and in the controls,” said Dr. Brugel. Consequently, for this study, between-group comparisons of substances present in all individuals could not be performed.
After adjustment, an association with an increased risk for pancreatic cancer was nonetheless observed with four liposoluble substances: 4,4-DDE, mirex or perchlordecone, trans-nonachlor, and cis-nonachlor. All four substances are herbicides that have been banned for at least 30 years.
The study also aimed to assess the effect of pesticide presence in the body on survival after pancreatic cancer. The results showed no significant difference for overall survival or progression-free survival.
“Pesticides are a credible candidate to explain the increase in the incidence of pancreatic adenocarcinoma,” said Dr. Brugel. However, “if associations between pancreatic cancer and pesticides exist, they remain poorly understood, and it is difficult to establish clear causality.”
Further large-scale studies will be needed to confirm these associations. An evaluation of the general population’s exposure to banned substances also appears justified, according to the researchers.
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
One of them, a case-control study, showed an elevated risk in individuals whose adipose tissue contained substances that are now banned.
“The association between pesticides and pancreatic cancer exists. It is of low magnitude but robust, concerning cumulative pesticides and three substances: Mancozeb, glyphosate, and sulfur in spray form,” said Mathias Brugel, MD, hospital practitioner at Basque Coast Hospital Center in Bayonne, France, during his presentation.
Regarding the four other liposoluble substances associated with an increased risk for pancreatic cancer in the second study, “their use has been banned since the 1990s, but they are still present in soils and in the air,” Dr. Brugel told this news organization.
For example, in Reims, France, the assessment of air quality by ATMO Grand Est revealed the presence of banned pesticides in the air, he added. However, Dr. Brugel stressed that a cause-effect relationship between pesticide exposure and the risk for pancreatic cancer cannot be established with these studies.
Incidence Rising Constantly
The incidence of pancreatic adenocarcinoma has been increasing steadily for more than 30 years. In France, nearly 16,000 new cases were reported in 2023, which represented an annual increase of about 2%. According to the National Cancer Institute, “pancreatic adenocarcinoma could become the second leading cause of cancer mortality by 2030.”
“This increase in incidence is particularly strong in France compared with other Western countries. The causes are still poorly understood. One might wonder whether environmental factors like pesticides are involved,” said Dr. Brugel.
Known to have a mechanism of action favoring oncogenesis, pesticides are suspected of being responsible for the rise in certain cancers, especially given their extensive use in France. In total, around 300 substances are authorized, and 65,000 tons are applied each year, making France the largest consumer of pesticides in Europe.
“Contamination is ubiquitous, meaning they are found in soil, water, air, and in individuals,” said Dr. Brugel. According to a study by the Institute for Scientific Expertise Research, pesticide residues were detected in 64% of hair samples taken from French volunteers.
The literature increasingly reported data suggesting a link between pesticide exposure and the development of certain diseases like cancer. A 2021 document by Inserm notably confirmed the strong presumption of a link between occupational pesticide exposure and pathologies such as non-Hodgkin’s lymphoma and prostate cancer.
High-Incidence Zones
To explore the link between pesticide exposure and pancreatic cancer, Dr. Brugel and his colleagues conducted the EcoPESTIPAC and PESTIPAC studies, the results of which were presented at this year’s conference.
In EcoPESTIPAC, researchers conducted a national ecological regression by dividing the entire French territory into 5529 spatial units. The number of pancreatic cancer cases per spatial unit per year (disease-mapping) was determined using the National Health Data System.
Nine chemicals, including glyphosate, were included, thus covering half of pesticide purchases in France. The cumulative quantity of pesticides, regardless of molecule, was also examined. Pesticide exposure was estimated by the median ratio between pesticide purchase and agricultural area per spatial unit over an 11-year period from early 2011 to the end of 2021.
Mor than 134,000 cases of pancreatic cancer were reported during this period. The analysis revealed three high-incidence zones located around Paris, in central France, and in the Mediterranean basin, while spatial units in the western region showed the lowest incidences.
The heterogeneous distribution of the disease suggests the involvement of risk factors, said Dr. Brugel. After adjusting for confounding factors such as smoking, the study showed an increased risk for pancreatic cancer associated with the cumulative quantity of pesticides and three specific substances: Sulfur in spray form, mancozeb, and glyphosate.
Risk Increases
A dose-response relationship was evident. For an increase in pesticide use of 2.5 kg/hectare over 11 years, the risk for pancreatic adenocarcinoma increased from 0.9% to 1.4%. “The increase is relatively small, but one must not forget that this risk applies to all of France,” said Dr. Brugel. Indeed, the risk appeared homogeneous across the entire territory.
This was the first study to explore this link at the national level. Although the association between the four identified factors and pancreatic risk was robust, the study had some limitations. It relied on the quantities of pesticides purchased to estimate the quantities used, Dr. Brugel pointed out.
The second study, PESTIPAC, was a case-control study conducted at the Reims University Hospital to explore the association between pancreatic adenocarcinoma and concentrations of organochlorine pesticides in fat and urine.
The study included 26 patients with pancreatic cancer who had abdominal surgery that allowed for adipose tissue sampling (minimum 10 g). Urine was collected in the morning on an empty stomach.
A control group was formed by including 26 other patients who underwent surgery for a benign abdominal condition such as gallstones or hernia, thus allowing for the same sampling. Individuals in both groups were matched for age and body mass index, two risk factors for pancreatic cancer.
Banned Substances
In total, 345 substances were searched for using chromatography and mass spectrometry. Analyses revealed the presence of five banned substances in all patients, while nine substances were found in half of the samples.
“Contamination is very widespread, both in patients with pancreatic cancer and in the controls,” said Dr. Brugel. Consequently, for this study, between-group comparisons of substances present in all individuals could not be performed.
After adjustment, an association with an increased risk for pancreatic cancer was nonetheless observed with four liposoluble substances: 4,4-DDE, mirex or perchlordecone, trans-nonachlor, and cis-nonachlor. All four substances are herbicides that have been banned for at least 30 years.
The study also aimed to assess the effect of pesticide presence in the body on survival after pancreatic cancer. The results showed no significant difference for overall survival or progression-free survival.
“Pesticides are a credible candidate to explain the increase in the incidence of pancreatic adenocarcinoma,” said Dr. Brugel. However, “if associations between pancreatic cancer and pesticides exist, they remain poorly understood, and it is difficult to establish clear causality.”
Further large-scale studies will be needed to confirm these associations. An evaluation of the general population’s exposure to banned substances also appears justified, according to the researchers.
This story was translated from the Medscape French edition using several editorial tools, including AI, as part of the process. Human editors reviewed this content before publication. A version of this article appeared on Medscape.com.
Do New Antiobesity Meds Still Require Lifestyle Management?
Is lifestyle counseling needed with the more effective second-generation nutrient-stimulated, hormone-based medications like semaglutide and tirzepatide?
If so, how intensive does the counseling need to be, and what components should be emphasized?
These are the clinical practice questions at the top of mind for healthcare professionals and researchers who provide care to patients who have overweight and/or obesity.
This is what we know. Lifestyle management is considered foundational in the care of patients with obesity.
Because obesity is fundamentally a disease of energy dysregulation, counseling has traditionally focused on dietary caloric reduction, increased physical activity, and strategies to adapt new cognitive and lifestyle behaviors.
On the basis of trial results from the Diabetes Prevention Program and the Look AHEAD studies, provision of intensive behavioral therapy (IBT) is recommended for treatment of obesity by the Centers for Medicare & Medicaid Services and by the US Preventive Services Task Force (Moyer VA; US Preventive Services Task Force).
IBT is commonly defined as consisting of 12-26 comprehensive and multicomponent sessions over the course of a year.
Reaffirming the primacy of lifestyle management, all antiobesity medications are approved by the US Food and Drug Administration as an adjunct to a reduced-calorie diet and increased physical activity.
The beneficial effect of combining IBT with earlier-generation medications like naltrexone/bupropion or liraglutide demonstrated that more participants in the trials achieved ≥ 10% weight loss with IBT compared with those taking the medication without IBT: 38.4% vs 20% for naltrexone/bupropion and 46% vs 33% for liraglutide.
Although there aren’t trial data for other first-generation medications like phentermine, orlistat, or phentermine/topiramate, it is assumed that patients taking these medications would also achieve greater weight loss when combined with IBT.
The obesity pharmacotherapy landscape was upended, however, with the approval of semaglutide (Wegovy), a glucagon-like peptide-1 (GLP-1) receptor agonist, in 2021; and tirzepatide (Zepbound), a GLP-1 and glucose-dependent insulinotropic polypeptide dual receptor agonist, in 2023.
These highly effective medications harness the effect of naturally occurring incretin hormones that reduce appetite through direct and indirect effects on the brain. Although the study designs differed between the STEP 1 and STEP 3 trials, the addition of IBT to semaglutide increased mean percent weight loss from 15% to 16% after 68 weeks of treatment (Wilding JPH et al; Wadden TA).
Comparable benefits from the STEP 3 and SURMOUNT-1 trials of adding IBT to tirzepatide at the maximal tolerated dose increased mean percent weight loss from 21% to 24% after 72 weeks (Wadden TA; Jastreboff AM). Though multicomponent IBT appears to provide greater weight loss when used with nutrient-stimulated hormone-based therapeutics, the additional benefit may be less when compared with first-generation medications.
So, how should we view the role and importance of lifestyle management when a patient is taking a second-generation medication? We need to shift the focus from prescribing a calorie-reduced diet to counseling for healthy eating patterns.
Because the second-generation drugs are more biologically effective in suppressing appetite (ie, reducing hunger, food noise, and cravings, and increasing satiation and satiety), it is easier for patients to reduce their food intake without a sense of deprivation. Furthermore, many patients express less desire to consume savory, sweet, and other enticing foods.
Patients should be encouraged to optimize the quality of their diet, prioritizing lean protein sources with meals and snacks; increasing fruits, vegetables, fiber, and complex carbohydrates; and keeping well hydrated. Because of the risk of developing micronutrient deficiencies while consuming a low-calorie diet — most notably calcium, iron, and vitamin D — patients may be advised to take a daily multivitamin supplement. Dietary counseling should be introduced when patients start pharmacotherapy, and if needed, referral to a registered dietitian nutritionist may be helpful in making these changes.
Additional counseling tips to mitigate the gastrointestinal side effects of these drugs that most commonly occur during the early dose-escalation phase include eating slowly; choosing smaller portion sizes; stopping eating when full; not skipping meals; and avoiding fatty, fried, and greasy foods. These dietary changes are particularly important over the first days after patients take the injection.
The increased weight loss achieved also raises concerns about the need to maintain lean body mass and the importance of physical activity and exercise counseling. All weight loss interventions, including dietary restriction, pharmacotherapy, or bariatric surgery, result in loss of fat mass and lean body mass.
The goal of lifestyle counseling is to minimize and preserve muscle mass (a component of lean body mass) which is needed for optimal health, mobility, daily function, and quality of life. Counseling should incorporate both aerobic and resistance training. Aerobic exercise (eg, brisk walking, jogging, dancing, elliptical machine, and cycling) improves cardiovascular fitness, metabolic health, and energy expenditure. Resistance (strength) training (eg, weightlifting, resistance bands, and circuit training) lessens the loss of muscle mass, enhances functional strength and mobility, and improves bone density (Gorgojo-Martinez JJ et al; Oppert JM et al).
Robust physical activity has also been shown to be a predictor of weight loss maintenance. A recently published randomized placebo-controlled trial demonstrated the benefit of supervised exercise in maintaining body weight and lean body mass after discontinuing 52 weeks of liraglutide treatment compared with no exercise.
Rather than minimizing the provision of lifestyle management, using highly effective second-generation therapeutics redirects the focus on how patients with obesity can strive to achieve a healthy and productive life.
A version of this article first appeared on Medscape.com.
Is lifestyle counseling needed with the more effective second-generation nutrient-stimulated, hormone-based medications like semaglutide and tirzepatide?
If so, how intensive does the counseling need to be, and what components should be emphasized?
These are the clinical practice questions at the top of mind for healthcare professionals and researchers who provide care to patients who have overweight and/or obesity.
This is what we know. Lifestyle management is considered foundational in the care of patients with obesity.
Because obesity is fundamentally a disease of energy dysregulation, counseling has traditionally focused on dietary caloric reduction, increased physical activity, and strategies to adapt new cognitive and lifestyle behaviors.
On the basis of trial results from the Diabetes Prevention Program and the Look AHEAD studies, provision of intensive behavioral therapy (IBT) is recommended for treatment of obesity by the Centers for Medicare & Medicaid Services and by the US Preventive Services Task Force (Moyer VA; US Preventive Services Task Force).
IBT is commonly defined as consisting of 12-26 comprehensive and multicomponent sessions over the course of a year.
Reaffirming the primacy of lifestyle management, all antiobesity medications are approved by the US Food and Drug Administration as an adjunct to a reduced-calorie diet and increased physical activity.
The beneficial effect of combining IBT with earlier-generation medications like naltrexone/bupropion or liraglutide demonstrated that more participants in the trials achieved ≥ 10% weight loss with IBT compared with those taking the medication without IBT: 38.4% vs 20% for naltrexone/bupropion and 46% vs 33% for liraglutide.
Although there aren’t trial data for other first-generation medications like phentermine, orlistat, or phentermine/topiramate, it is assumed that patients taking these medications would also achieve greater weight loss when combined with IBT.
The obesity pharmacotherapy landscape was upended, however, with the approval of semaglutide (Wegovy), a glucagon-like peptide-1 (GLP-1) receptor agonist, in 2021; and tirzepatide (Zepbound), a GLP-1 and glucose-dependent insulinotropic polypeptide dual receptor agonist, in 2023.
These highly effective medications harness the effect of naturally occurring incretin hormones that reduce appetite through direct and indirect effects on the brain. Although the study designs differed between the STEP 1 and STEP 3 trials, the addition of IBT to semaglutide increased mean percent weight loss from 15% to 16% after 68 weeks of treatment (Wilding JPH et al; Wadden TA).
Comparable benefits from the STEP 3 and SURMOUNT-1 trials of adding IBT to tirzepatide at the maximal tolerated dose increased mean percent weight loss from 21% to 24% after 72 weeks (Wadden TA; Jastreboff AM). Though multicomponent IBT appears to provide greater weight loss when used with nutrient-stimulated hormone-based therapeutics, the additional benefit may be less when compared with first-generation medications.
So, how should we view the role and importance of lifestyle management when a patient is taking a second-generation medication? We need to shift the focus from prescribing a calorie-reduced diet to counseling for healthy eating patterns.
Because the second-generation drugs are more biologically effective in suppressing appetite (ie, reducing hunger, food noise, and cravings, and increasing satiation and satiety), it is easier for patients to reduce their food intake without a sense of deprivation. Furthermore, many patients express less desire to consume savory, sweet, and other enticing foods.
Patients should be encouraged to optimize the quality of their diet, prioritizing lean protein sources with meals and snacks; increasing fruits, vegetables, fiber, and complex carbohydrates; and keeping well hydrated. Because of the risk of developing micronutrient deficiencies while consuming a low-calorie diet — most notably calcium, iron, and vitamin D — patients may be advised to take a daily multivitamin supplement. Dietary counseling should be introduced when patients start pharmacotherapy, and if needed, referral to a registered dietitian nutritionist may be helpful in making these changes.
Additional counseling tips to mitigate the gastrointestinal side effects of these drugs that most commonly occur during the early dose-escalation phase include eating slowly; choosing smaller portion sizes; stopping eating when full; not skipping meals; and avoiding fatty, fried, and greasy foods. These dietary changes are particularly important over the first days after patients take the injection.
The increased weight loss achieved also raises concerns about the need to maintain lean body mass and the importance of physical activity and exercise counseling. All weight loss interventions, including dietary restriction, pharmacotherapy, or bariatric surgery, result in loss of fat mass and lean body mass.
The goal of lifestyle counseling is to minimize and preserve muscle mass (a component of lean body mass) which is needed for optimal health, mobility, daily function, and quality of life. Counseling should incorporate both aerobic and resistance training. Aerobic exercise (eg, brisk walking, jogging, dancing, elliptical machine, and cycling) improves cardiovascular fitness, metabolic health, and energy expenditure. Resistance (strength) training (eg, weightlifting, resistance bands, and circuit training) lessens the loss of muscle mass, enhances functional strength and mobility, and improves bone density (Gorgojo-Martinez JJ et al; Oppert JM et al).
Robust physical activity has also been shown to be a predictor of weight loss maintenance. A recently published randomized placebo-controlled trial demonstrated the benefit of supervised exercise in maintaining body weight and lean body mass after discontinuing 52 weeks of liraglutide treatment compared with no exercise.
Rather than minimizing the provision of lifestyle management, using highly effective second-generation therapeutics redirects the focus on how patients with obesity can strive to achieve a healthy and productive life.
A version of this article first appeared on Medscape.com.
Is lifestyle counseling needed with the more effective second-generation nutrient-stimulated, hormone-based medications like semaglutide and tirzepatide?
If so, how intensive does the counseling need to be, and what components should be emphasized?
These are the clinical practice questions at the top of mind for healthcare professionals and researchers who provide care to patients who have overweight and/or obesity.
This is what we know. Lifestyle management is considered foundational in the care of patients with obesity.
Because obesity is fundamentally a disease of energy dysregulation, counseling has traditionally focused on dietary caloric reduction, increased physical activity, and strategies to adapt new cognitive and lifestyle behaviors.
On the basis of trial results from the Diabetes Prevention Program and the Look AHEAD studies, provision of intensive behavioral therapy (IBT) is recommended for treatment of obesity by the Centers for Medicare & Medicaid Services and by the US Preventive Services Task Force (Moyer VA; US Preventive Services Task Force).
IBT is commonly defined as consisting of 12-26 comprehensive and multicomponent sessions over the course of a year.
Reaffirming the primacy of lifestyle management, all antiobesity medications are approved by the US Food and Drug Administration as an adjunct to a reduced-calorie diet and increased physical activity.
The beneficial effect of combining IBT with earlier-generation medications like naltrexone/bupropion or liraglutide demonstrated that more participants in the trials achieved ≥ 10% weight loss with IBT compared with those taking the medication without IBT: 38.4% vs 20% for naltrexone/bupropion and 46% vs 33% for liraglutide.
Although there aren’t trial data for other first-generation medications like phentermine, orlistat, or phentermine/topiramate, it is assumed that patients taking these medications would also achieve greater weight loss when combined with IBT.
The obesity pharmacotherapy landscape was upended, however, with the approval of semaglutide (Wegovy), a glucagon-like peptide-1 (GLP-1) receptor agonist, in 2021; and tirzepatide (Zepbound), a GLP-1 and glucose-dependent insulinotropic polypeptide dual receptor agonist, in 2023.
These highly effective medications harness the effect of naturally occurring incretin hormones that reduce appetite through direct and indirect effects on the brain. Although the study designs differed between the STEP 1 and STEP 3 trials, the addition of IBT to semaglutide increased mean percent weight loss from 15% to 16% after 68 weeks of treatment (Wilding JPH et al; Wadden TA).
Comparable benefits from the STEP 3 and SURMOUNT-1 trials of adding IBT to tirzepatide at the maximal tolerated dose increased mean percent weight loss from 21% to 24% after 72 weeks (Wadden TA; Jastreboff AM). Though multicomponent IBT appears to provide greater weight loss when used with nutrient-stimulated hormone-based therapeutics, the additional benefit may be less when compared with first-generation medications.
So, how should we view the role and importance of lifestyle management when a patient is taking a second-generation medication? We need to shift the focus from prescribing a calorie-reduced diet to counseling for healthy eating patterns.
Because the second-generation drugs are more biologically effective in suppressing appetite (ie, reducing hunger, food noise, and cravings, and increasing satiation and satiety), it is easier for patients to reduce their food intake without a sense of deprivation. Furthermore, many patients express less desire to consume savory, sweet, and other enticing foods.
Patients should be encouraged to optimize the quality of their diet, prioritizing lean protein sources with meals and snacks; increasing fruits, vegetables, fiber, and complex carbohydrates; and keeping well hydrated. Because of the risk of developing micronutrient deficiencies while consuming a low-calorie diet — most notably calcium, iron, and vitamin D — patients may be advised to take a daily multivitamin supplement. Dietary counseling should be introduced when patients start pharmacotherapy, and if needed, referral to a registered dietitian nutritionist may be helpful in making these changes.
Additional counseling tips to mitigate the gastrointestinal side effects of these drugs that most commonly occur during the early dose-escalation phase include eating slowly; choosing smaller portion sizes; stopping eating when full; not skipping meals; and avoiding fatty, fried, and greasy foods. These dietary changes are particularly important over the first days after patients take the injection.
The increased weight loss achieved also raises concerns about the need to maintain lean body mass and the importance of physical activity and exercise counseling. All weight loss interventions, including dietary restriction, pharmacotherapy, or bariatric surgery, result in loss of fat mass and lean body mass.
The goal of lifestyle counseling is to minimize and preserve muscle mass (a component of lean body mass) which is needed for optimal health, mobility, daily function, and quality of life. Counseling should incorporate both aerobic and resistance training. Aerobic exercise (eg, brisk walking, jogging, dancing, elliptical machine, and cycling) improves cardiovascular fitness, metabolic health, and energy expenditure. Resistance (strength) training (eg, weightlifting, resistance bands, and circuit training) lessens the loss of muscle mass, enhances functional strength and mobility, and improves bone density (Gorgojo-Martinez JJ et al; Oppert JM et al).
Robust physical activity has also been shown to be a predictor of weight loss maintenance. A recently published randomized placebo-controlled trial demonstrated the benefit of supervised exercise in maintaining body weight and lean body mass after discontinuing 52 weeks of liraglutide treatment compared with no exercise.
Rather than minimizing the provision of lifestyle management, using highly effective second-generation therapeutics redirects the focus on how patients with obesity can strive to achieve a healthy and productive life.
A version of this article first appeared on Medscape.com.